Advances in Endometrial Cancer Therapy Exploring the Role













- Slides: 40

Advances in Endometrial Cancer Therapy: Exploring the Role of Immunotherapy Supported by an educational grant from Glaxo. Smith. Kline.

About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details

Faculty Ana Oaknin, MD, Ph. D Head of the Gynaecologic Cancer Programme Vall d’Hebron Institute of Oncology (VHIO) Hospital Universitari Vall d’Hebron Barcelona Hospital Campus Barcelona, Spain Ana Oaknin, MD, has disclosed that she has received consulting fees from Astra. Zeneca, Clovis Oncology, Genmab, Immunogen, Pharm. Mar, Roche, and Tesaro.

Recurrent/Metastatic Endometrial Cancer: Overview § EC is the most common gynaecologic malignancy in women in the developed world with an estimated 382, 069 new cases diagnosed in 2018 ‒ Highest incidence in Eastern Europe and North America ‒ US: estimated 65, 620 new cases and 12, 590 deaths in 2020 § The incidence of EC is increasing annually by an estimated 1% to 2%; it is predicted to double by 2030 in the US § Prognosis: 5 -yr survival rates ‒ Stage I: 95% ‒ Stage IV: metastatic: 17. 3% Zhang. Front Oncol. 2019; 9: 1440. Bray. CA Cancer J Clin. 2018; 68: 394. Siegel 2020. CA Cancer J Clin. 2020; 70: 7. Slide credit: clinicaloptions. com

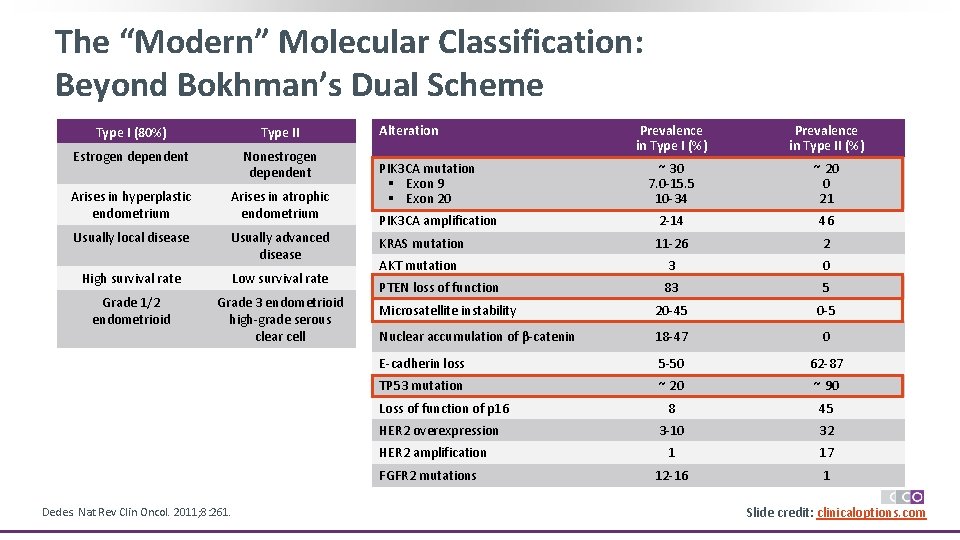
The “Modern” Molecular Classification: Beyond Bokhman’s Dual Scheme Type I (80%) Type II Estrogen dependent Nonestrogen dependent Arises in hyperplastic endometrium Arises in atrophic endometrium Usually local disease Usually advanced disease High survival rate Low survival rate Grade 1/2 endometrioid Grade 3 endometrioid high-grade serous clear cell Alteration Prevalence in Type I (%) Prevalence in Type II (%) ~ 30 7. 0 -15. 5 10 -34 ~ 20 0 21 PIK 3 CA amplification 2 -14 46 KRAS mutation 11 -26 2 AKT mutation 3 0 PTEN loss of function 83 5 Microsatellite instability 20 -45 0 -5 Nuclear accumulation of β-catenin 18 -47 0 E-cadherin loss 5 -50 62 -87 TP 53 mutation ~ 20 ~ 90 8 45 3 -10 32 1 17 12 -16 1 PIK 3 CA mutation § Exon 9 § Exon 20 Loss of function of p 16 HER 2 overexpression HER 2 amplification FGFR 2 mutations Dedes. Nat Rev Clin Oncol. 2011; 8: 261. Slide credit: clinicaloptions. com

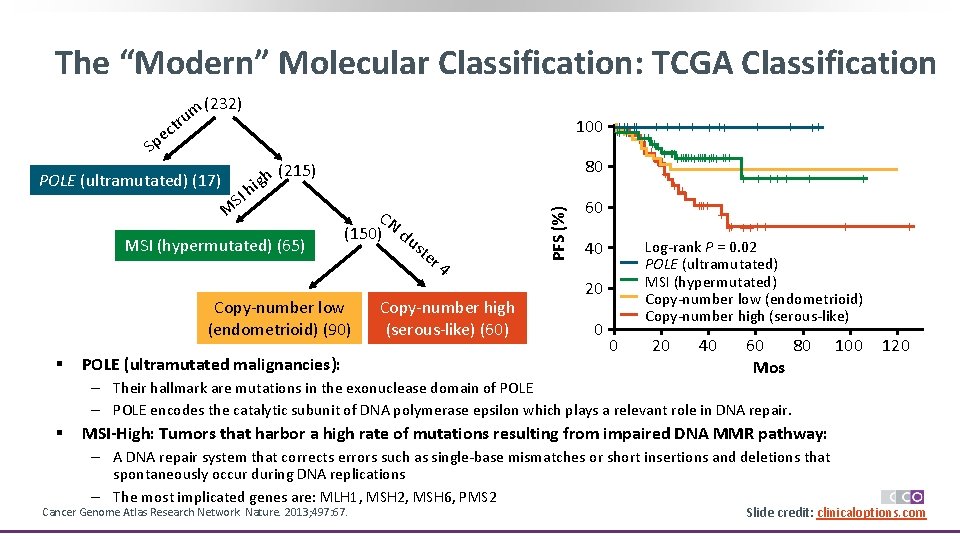
The “Modern” Molecular Classification: TCGA Classification m (232) u tr ec p S M SI h g i h (215) MSI (hypermutated) (65) CN (150) clu Copy-number low (endometrioid) (90) § POLE (ultramutated malignancies): ste r 4 Copy-number high (serous-like) (60) PFS (%) POLE (ultramutated) (17) +++ + + ++ 100 +++ ++++++++++++ ++ ++ 80 + + +++ ++ ++ 60 + + ++ ++++ Log-rank P = 0. 02 40 POLE (ultramutated) MSI (hypermutated) Copy-number low (endometrioid) Copy-number high (serous-like) 20 0 + 0 20 40 60 80 Mos 100 120 ‒ Their hallmark are mutations in the exonuclease domain of POLE ‒ POLE encodes the catalytic subunit of DNA polymerase epsilon which plays a relevant role in DNA repair. § MSI-High: Tumors that harbor a high rate of mutations resulting from impaired DNA MMR pathway: ‒ A DNA repair system that corrects errors such as single-base mismatches or short insertions and deletions that spontaneously occur during DNA replications ‒ The most implicated genes are: MLH 1, MSH 2, MSH 6, PMS 2 Cancer Genome Atlas Research Network. Nature. 2013; 497: 67. Slide credit: clinicaloptions. com

MSI/d. MMR: Concept and Incidence § DNA MMR: Highly conserved mechanism used to restore DNA integrity after the occurrence of mismatching errors, including single-base mismatches or short insertions and deletions ‒ 4 genes that play a critical role in this process include: MLH 1 , MSH 2, MSH 6 and PMS 2 § MSI: Condition of genetic hypermutability resulting from defective DNA MMR § MSI/d. MMR tumor: A tumor that accumulates thousands of mutations, particularly clustered in microsatellites and consisting in repeat length alterations, resulting in MSI Tumor Type* MSI-High, % Uterine corpus endometrial 28. 3 Stomach adeno Colon adeno EC can be classified as microsatellite stable (MSS/70%-75%) or microsatellite 21. 9 instability-high (MSI/25%-30%) 16. 6 Rectal adeno 9. 2 Adrenal cortical 5. 4 Esophageal 3. 3 Ovarian 3. 2 Hepatocellular 2. 9 Cervical squamous 2. 3 *At least 2% MSI-High incidence Luchini. Annals Oncol. 2019; 30: 1232. Cortes-Ciriano. Nat Commun. 2017; 8: 15180. Slide credit: clinicaloptions. com

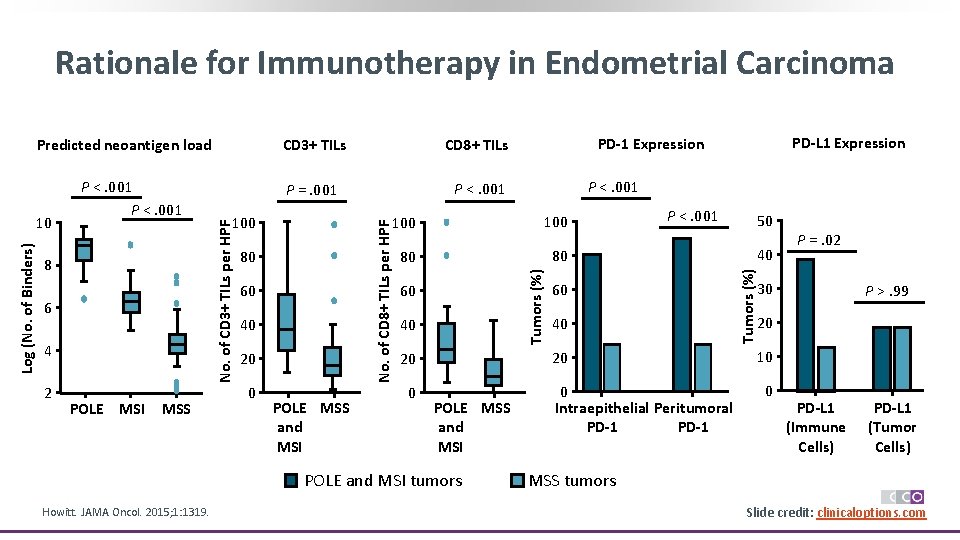
Rationale for Immunotherapy in Endometrial Carcinoma 4 2 POLE MSI MSS P <. 001 80 60 40 20 0 POLE MSS and MSI 60 40 20 20 0 P <. 001 50 40 80 80 POLE MSS and MSI POLE and MSI tumors Howitt. JAMA Oncol. 2015; 1: 1319. P <. 001 100 PD-L 1 Expression PD-1 Expression 0 Intraepithelial Peritumoral PD-1 Tumors (%) 6 P =. 001 No. of CD 8+ TILs per HPF 8 CD 8+ TILs 100 No. of CD 3+ TILs per HPF Log (No. of Binders) 10 P <. 001 CD 3+ TILs Tumors (%) Predicted neoantigen load P =. 02 30 P >. 99 20 10 0 PD-L 1 (Immune Cells) PD-L 1 (Tumor Cells) MSS tumors Slide credit: clinicaloptions. com

Clinical Data of Immunotherapy in Advanced/Recurrent Endometrial Cancer

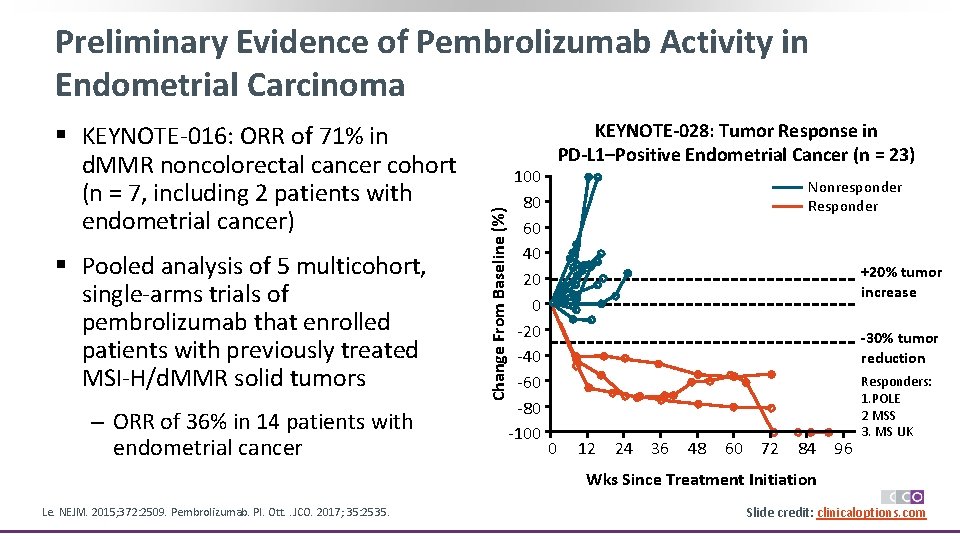
Preliminary Evidence of Pembrolizumab Activity in Endometrial Carcinoma § Pooled analysis of 5 multicohort, single-arms trials of pembrolizumab that enrolled patients with previously treated MSI-H/d. MMR solid tumors ‒ ORR of 36% in 14 patients with endometrial cancer 100 80 60 40 20 0 -20 -40 -60 -80 -100 KEYNOTE-028: Tumor Response in PD-L 1–Positive Endometrial Cancer (n = 23) Nonresponder Responder Change From Baseline (%) § KEYNOTE-016: ORR of 71% in d. MMR noncolorectal cancer cohort (n = 7, including 2 patients with endometrial cancer) +20% tumor increase -30% tumor reduction 0 12 24 36 48 60 72 84 96 Responders: 1. POLE 2 MSS 3. MS UK Wks Since Treatment Initiation Le. NEJM. 2015; 372: 2509. Pembrolizumab. PI. Ott. . JCO. 2017; 35: 2535. Slide credit: clinicaloptions. com

KEYNOTE-158: Pembrolizumab for Advanced Endometrial Cancer Patients with unresectable or metastatic endometrial cancer with progression on or intolerance to standard therapy; ECOG PS 0 or 1; evaluable tumor for biomarker assay; no autoimmune disease or noninfectious pneumonitis Pembrolizumab 200 mg IV Q 3 W Up to 35 cycles or until PD, unacceptable toxicity, consent withdrawal Cohort D: endometrial cancer Cohort K: non-CRC, MSI-H solid tumor § Primary endpoint: ORR by central review using RECIST v 1. 1 criteria ‒ Response assessed every 9 wks in Yr 1; every 12 wks thereafter § Secondary endpoints: PFS, OS, Do. R, safety O’Malley ESMO 2019. Abstr 3394. Slide credit: clinicaloptions. com

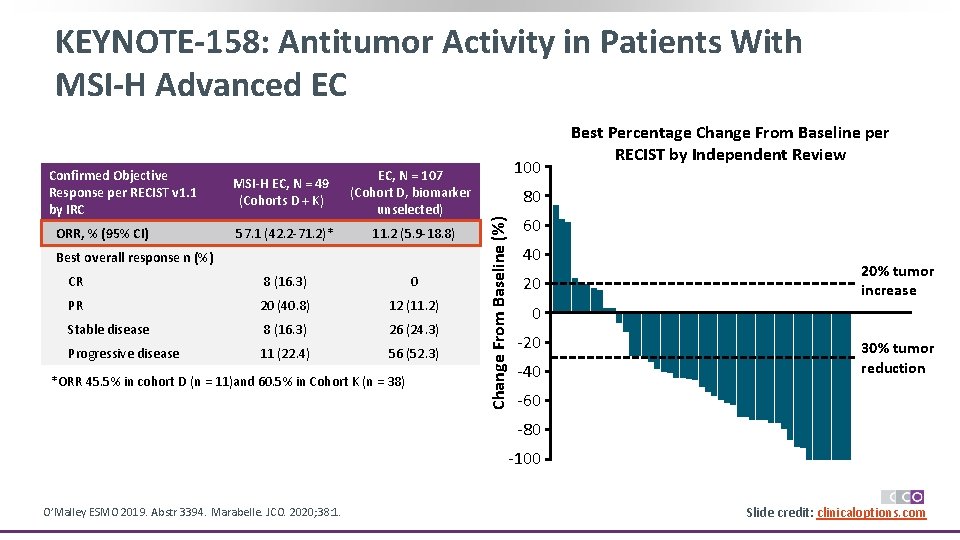
KEYNOTE-158: Antitumor Activity in Patients With MSI-H Advanced EC ORR, % (95% CI) MSI-H EC, N = 49 (Cohorts D + K) EC, N = 107 (Cohort D, biomarker unselected) 57. 1 (42. 2 -71. 2)* 11. 2 (5. 9 -18. 8) Best overall response n (%) CR 8 (16. 3) 0 PR 20 (40. 8) 12 (11. 2) Stable disease 8 (16. 3) 26 (24. 3) Progressive disease 11 (22. 4) 56 (52. 3) *ORR 45. 5% in cohort D (n = 11)and 60. 5% in Cohort K (n = 38) 100 80 Change From Baseline (%) Confirmed Objective Response per RECIST v 1. 1 by IRC Best Percentage Change From Baseline per RECIST by Independent Review 60 40 20 20% tumor increase 0 -20 -40 30% tumor reduction -60 -80 -100 O’Malley ESMO 2019. Abstr 3394. Marabelle. JCO. 2020; 38: 1. Slide credit: clinicaloptions. com

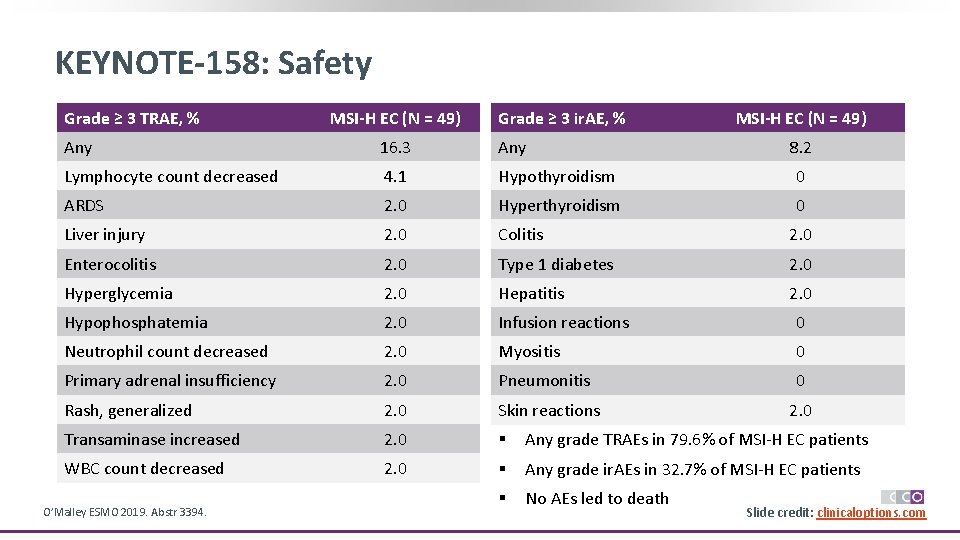
KEYNOTE-158: Safety Grade ≥ 3 TRAE, % MSI-H EC (N = 49) Grade ≥ 3 ir. AE, % MSI-H EC (N = 49) Any 16. 3 Any Lymphocyte count decreased 4. 1 Hypothyroidism 0 ARDS 2. 0 Hyperthyroidism 0 Liver injury 2. 0 Colitis 2. 0 Enterocolitis 2. 0 Type 1 diabetes 2. 0 Hyperglycemia 2. 0 Hepatitis 2. 0 Hypophosphatemia 2. 0 Infusion reactions 0 Neutrophil count decreased 2. 0 Myositis 0 Primary adrenal insufficiency 2. 0 Pneumonitis 0 Rash, generalized 2. 0 Skin reactions 2. 0 Transaminase increased 2. 0 § Any grade TRAEs in 79. 6% of MSI-H EC patients WBC count decreased 2. 0 § Any grade ir. AEs in 32. 7% of MSI-H EC patients § No AEs led to death O’Malley ESMO 2019. Abstr 3394. 8. 2 Slide credit: clinicaloptions. com

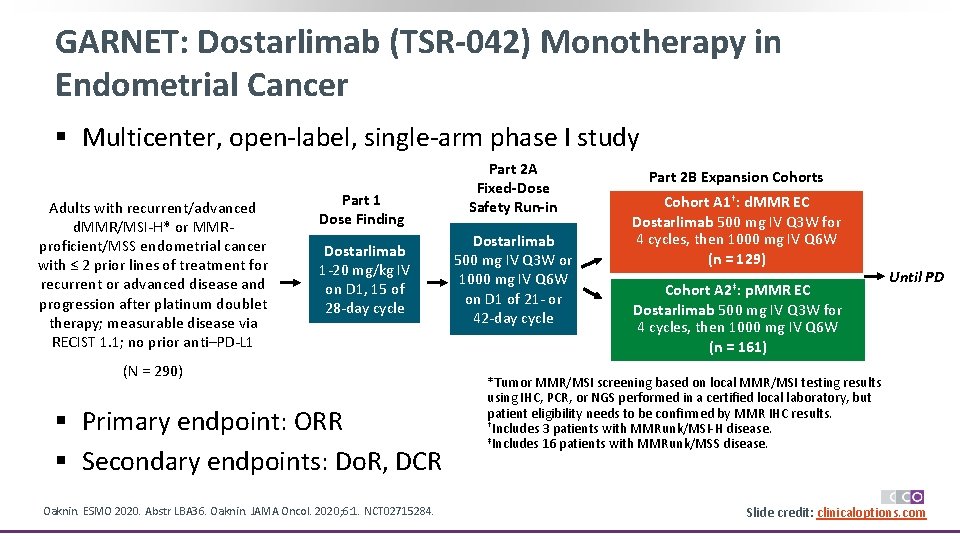
GARNET: Dostarlimab (TSR-042) Monotherapy in Endometrial Cancer § Multicenter, open-label, single-arm phase I study Adults with recurrent/advanced d. MMR/MSI-H* or MMRproficient/MSS endometrial cancer with ≤ 2 prior lines of treatment for recurrent or advanced disease and progression after platinum doublet therapy; measurable disease via RECIST 1. 1; no prior anti–PD-L 1 Part 1 Dose Finding Dostarlimab 1 -20 mg/kg IV on D 1, 15 of 28 -day cycle (N = 290) § Primary endpoint: ORR § Secondary endpoints: Do. R, DCR Oaknin. ESMO 2020. Abstr LBA 36. Oaknin. JAMA Oncol. 2020; 6: 1. NCT 02715284. Part 2 A Fixed-Dose Safety Run-in Dostarlimab 500 mg IV Q 3 W or 1000 mg IV Q 6 W on D 1 of 21 - or 42 -day cycle Part 2 B Expansion Cohorts Cohort A 1†: d. MMR EC Dostarlimab 500 mg IV Q 3 W for 4 cycles, then 1000 mg IV Q 6 W (n = 129) Cohort A 2‡: p. MMR EC Dostarlimab 500 mg IV Q 3 W for 4 cycles, then 1000 mg IV Q 6 W (n = 161) Until PD *Tumor MMR/MSI screening based on local MMR/MSI testing results using IHC, PCR, or NGS performed in a certified local laboratory, but patient eligibility needs to be confirmed by MMR IHC results. †Includes 3 patients with MMRunk/MSI-H disease. ‡Includes 16 patients with MMRunk/MSS disease. Slide credit: clinicaloptions. com

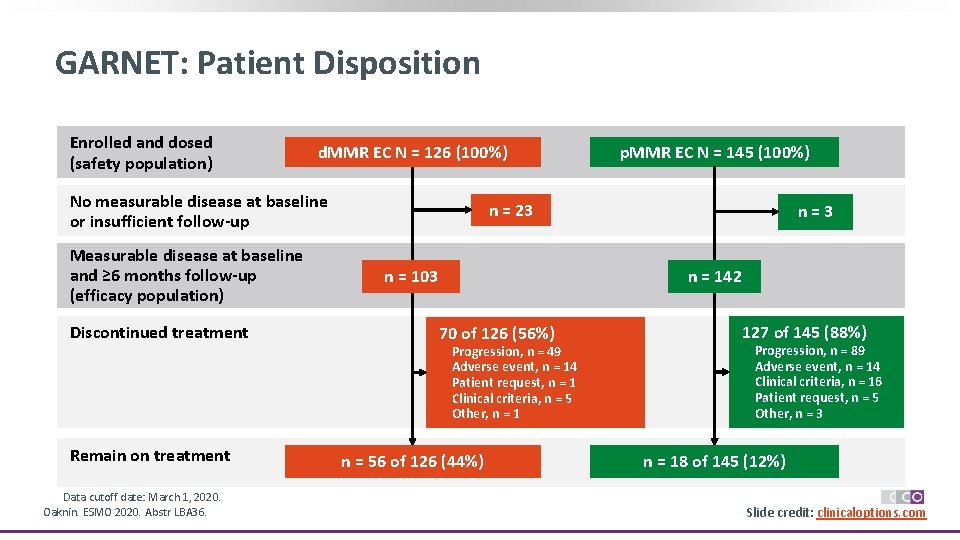
GARNET: Patient Disposition Enrolled and dosed (safety population) d. MMR EC N = 126 (100%) No measurable disease at baseline or insufficient follow-up Measurable disease at baseline and ≥ 6 months follow-up (efficacy population) Discontinued treatment Remain on treatment Data cutoff date: March 1, 2020. Oaknin. ESMO 2020. Abstr LBA 36. p. MMR EC N = 145 (100%) n = 23 n = 103 n=3 n = 142 70 of 126 (56%) Progression, n = 49 Adverse event, n = 14 Patient request, n = 1 Clinical criteria, n = 5 Other, n = 1 n = 56 of 126 (44%) 127 of 145 (88%) Progression, n = 89 Adverse event, n = 14 Clinical criteria, n = 16 Patient request, n = 5 Other, n = 3 n = 18 of 145 (12%) Slide credit: clinicaloptions. com

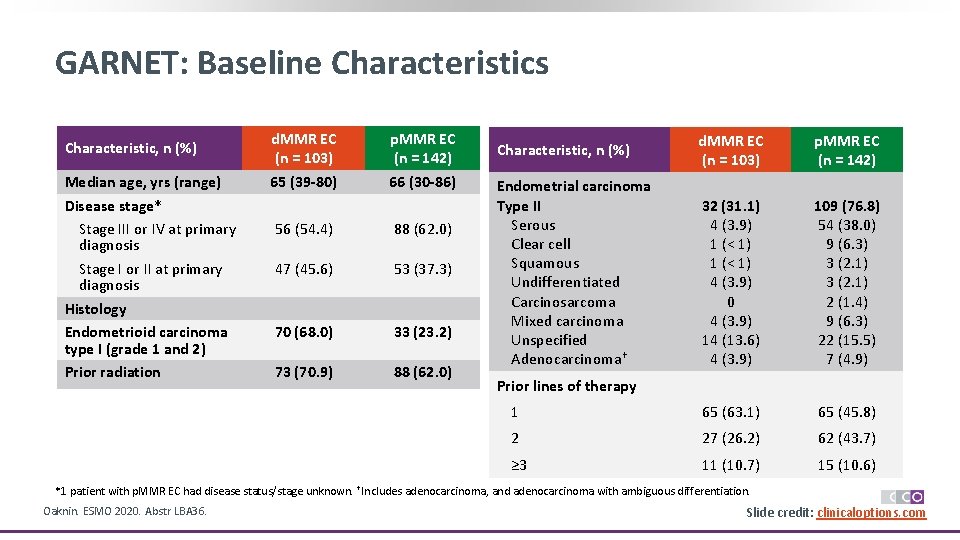
GARNET: Baseline Characteristics Characteristic, n (%) Median age, yrs (range) Disease stage* Stage III or IV at primary diagnosis Stage I or II at primary diagnosis Histology Endometrioid carcinoma type I (grade 1 and 2) Prior radiation d. MMR EC (n = 103) p. MMR EC (n = 142) 65 (39 -80) 66 (30 -86) 56 (54. 4) 88 (62. 0) 47 (45. 6) 53 (37. 3) 70 (68. 0) 33 (23. 2) 73 (70. 9) 88 (62. 0) d. MMR EC (n = 103) p. MMR EC (n = 142) 32 (31. 1) 4 (3. 9) 1 (< 1) 4 (3. 9) 0 4 (3. 9) 14 (13. 6) 4 (3. 9) 109 (76. 8) 54 (38. 0) 9 (6. 3) 3 (2. 1) 2 (1. 4) 9 (6. 3) 22 (15. 5) 7 (4. 9) 1 65 (63. 1) 65 (45. 8) 2 27 (26. 2) 62 (43. 7) ≥ 3 11 (10. 7) 15 (10. 6) Characteristic, n (%) Endometrial carcinoma Type II Serous Clear cell Squamous Undifferentiated Carcinosarcoma Mixed carcinoma Unspecified Adenocarcinoma† Prior lines of therapy *1 patient with p. MMR EC had disease status/stage unknown. †Includes adenocarcinoma, and adenocarcinoma with ambiguous differentiation. Oaknin. ESMO 2020. Abstr LBA 36. Slide credit: clinicaloptions. com

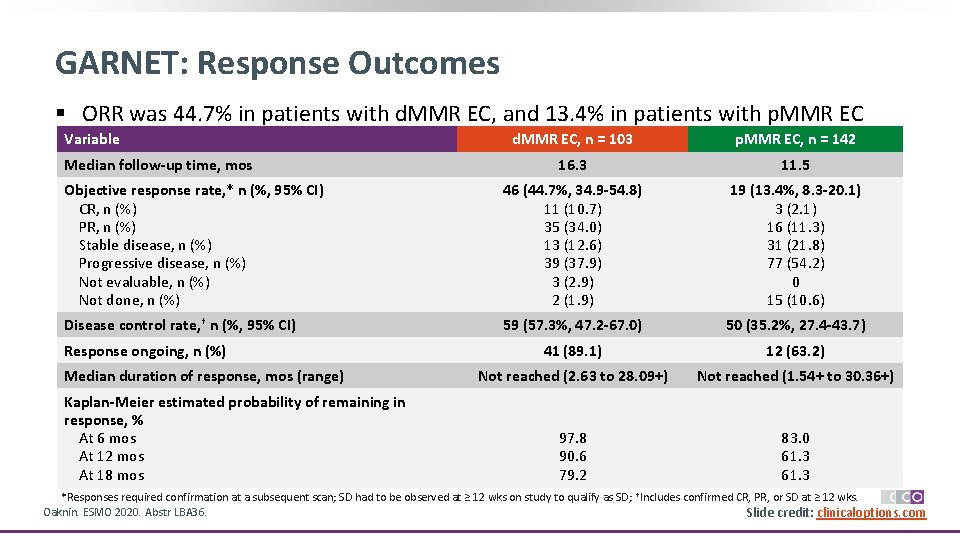
GARNET: Response Outcomes § ORR was 44. 7% in patients with d. MMR EC, and 13. 4% in patients with p. MMR EC Variable d. MMR EC, n = 103 p. MMR EC, n = 142 16. 3 11. 5 Objective response rate, * n (%, 95% CI) CR, n (%) PR, n (%) Stable disease, n (%) Progressive disease, n (%) Not evaluable, n (%) Not done, n (%) 46 (44. 7%, 34. 9 -54. 8) 11 (10. 7) 35 (34. 0) 13 (12. 6) 39 (37. 9) 3 (2. 9) 2 (1. 9) 19 (13. 4%, 8. 3 -20. 1) 3 (2. 1) 16 (11. 3) 31 (21. 8) 77 (54. 2) 0 15 (10. 6) Disease control rate, † n (%, 95% CI) 59 (57. 3%, 47. 2 -67. 0) 50 (35. 2%, 27. 4 -43. 7) 41 (89. 1) 12 (63. 2) Not reached (2. 63 to 28. 09+) Not reached (1. 54+ to 30. 36+) 97. 8 90. 6 79. 2 83. 0 61. 3 Median follow-up time, mos Response ongoing, n (%) Median duration of response, mos (range) Kaplan-Meier estimated probability of remaining in response, % At 6 mos At 12 mos At 18 mos *Responses required confirmation at a subsequent scan; SD had to be observed at ≥ 12 wks on study to qualify as SD; †Includes confirmed CR, PR, or SD at ≥ 12 wks. Oaknin. ESMO 2020. Abstr LBA 36. Slide credit: clinicaloptions. com

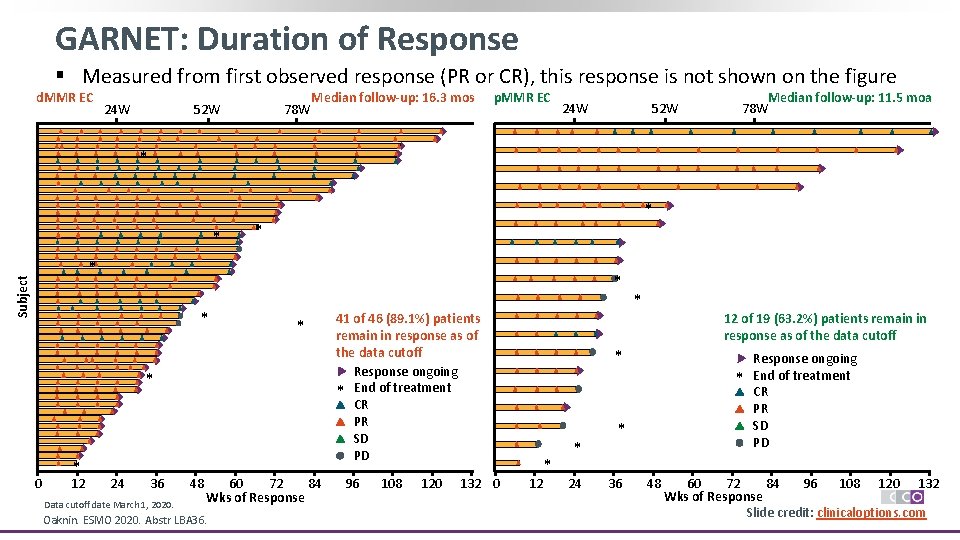
GARNET: Duration of Response § Measured from first observed response (PR or CR), this response is not shown on the figure d. MMR EC 24 W 52 W 78 W Median follow-up: 16. 3 mos p. MMR EC 24 W 52 W Median follow-up: 11. 5 moa 78 W * * Subject * * * 0 * 12 24 36 Data cutoff date March 1, 2020. 48 60 72 84 Wks of Response Oaknin. ESMO 2020. Abstr LBA 36. 41 of 46 (89. 1%) patients remain in response as of the data cutoff Response ongoing * End of treatment CR PR SD PD 96 108 120 132 0 * 12 of 19 (63. 2%) patients remain in response as of the data cutoff * 12 * * 24 * * 36 48 Response ongoing End of treatment CR PR SD PD 60 72 84 96 108 120 132 Wks of Response Slide credit: clinicaloptions. com

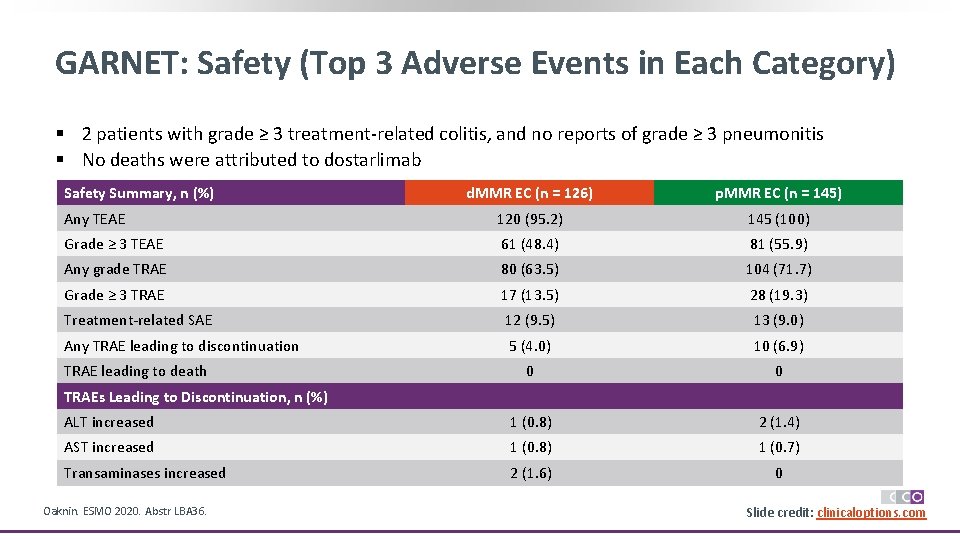
GARNET: Safety (Top 3 Adverse Events in Each Category) § 2 patients with grade ≥ 3 treatment-related colitis, and no reports of grade ≥ 3 pneumonitis § No deaths were attributed to dostarlimab Safety Summary, n (%) d. MMR EC (n = 126) p. MMR EC (n = 145) Any TEAE 120 (95. 2) 145 (100) Grade ≥ 3 TEAE 61 (48. 4) 81 (55. 9) Any grade TRAE 80 (63. 5) 104 (71. 7) Grade ≥ 3 TRAE 17 (13. 5) 28 (19. 3) Treatment-related SAE 12 (9. 5) 13 (9. 0) Any TRAE leading to discontinuation 5 (4. 0) 10 (6. 9) 0 0 ALT increased 1 (0. 8) 2 (1. 4) AST increased 1 (0. 8) 1 (0. 7) Transaminases increased 2 (1. 6) 0 TRAE leading to death TRAEs Leading to Discontinuation, n (%) Oaknin. ESMO 2020. Abstr LBA 36. Slide credit: clinicaloptions. com

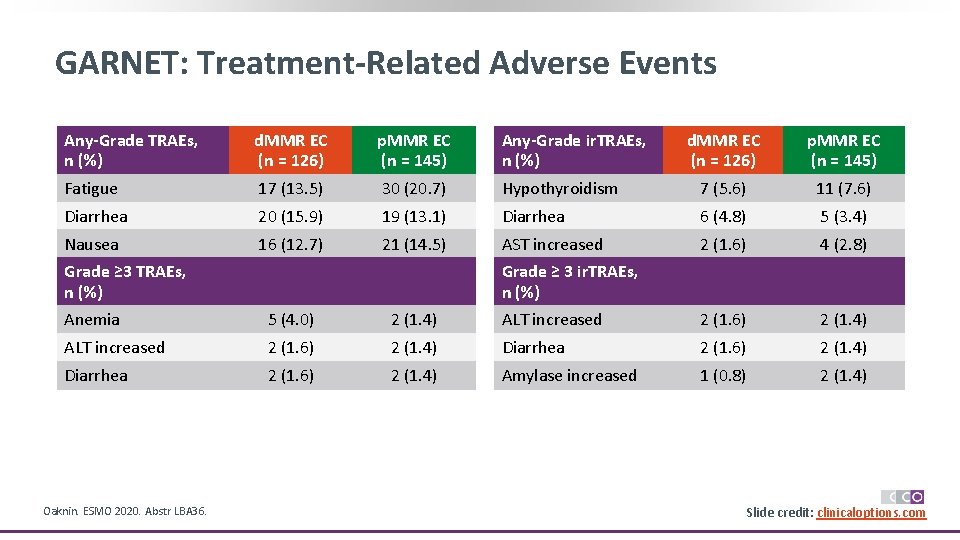
GARNET: Treatment-Related Adverse Events Any-Grade TRAEs, n (%) d. MMR EC (n = 126) p. MMR EC (n = 145) Any-Grade ir. TRAEs, n (%) d. MMR EC (n = 126) p. MMR EC (n = 145) Fatigue 17 (13. 5) 30 (20. 7) Hypothyroidism 7 (5. 6) 11 (7. 6) Diarrhea 20 (15. 9) 19 (13. 1) Diarrhea 6 (4. 8) 5 (3. 4) Nausea 16 (12. 7) 21 (14. 5) AST increased 2 (1. 6) 4 (2. 8) Grade ≥ 3 TRAEs, n (%) Grade ≥ 3 ir. TRAEs, n (%) Anemia 5 (4. 0) 2 (1. 4) ALT increased 2 (1. 6) 2 (1. 4) Diarrhea 2 (1. 6) 2 (1. 4) Amylase increased 1 (0. 8) 2 (1. 4) Oaknin. ESMO 2020. Abstr LBA 36. Slide credit: clinicaloptions. com

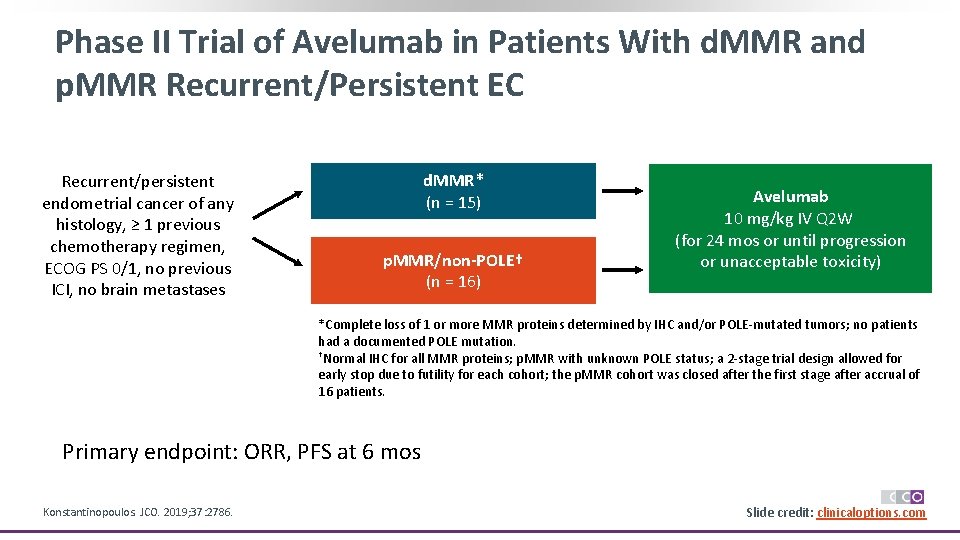
Phase II Trial of Avelumab in Patients With d. MMR and p. MMR Recurrent/Persistent EC Recurrent/persistent endometrial cancer of any histology, ≥ 1 previous chemotherapy regimen, ECOG PS 0/1, no previous ICI, no brain metastases d. MMR* (n = 15) p. MMR/non-POLE† (n = 16) Avelumab 10 mg/kg IV Q 2 W (for 24 mos or until progression or unacceptable toxicity) *Complete loss of 1 or more MMR proteins determined by IHC and/or POLE-mutated tumors; no patients had a documented POLE mutation. †Normal IHC for all MMR proteins; p. MMR with unknown POLE status; a 2 -stage trial design allowed for early stop due to futility for each cohort; the p. MMR cohort was closed after the first stage after accrual of 16 patients. Primary endpoint: ORR, PFS at 6 mos Konstantinopoulos. JCO. 2019; 37: 2786. Slide credit: clinicaloptions. com

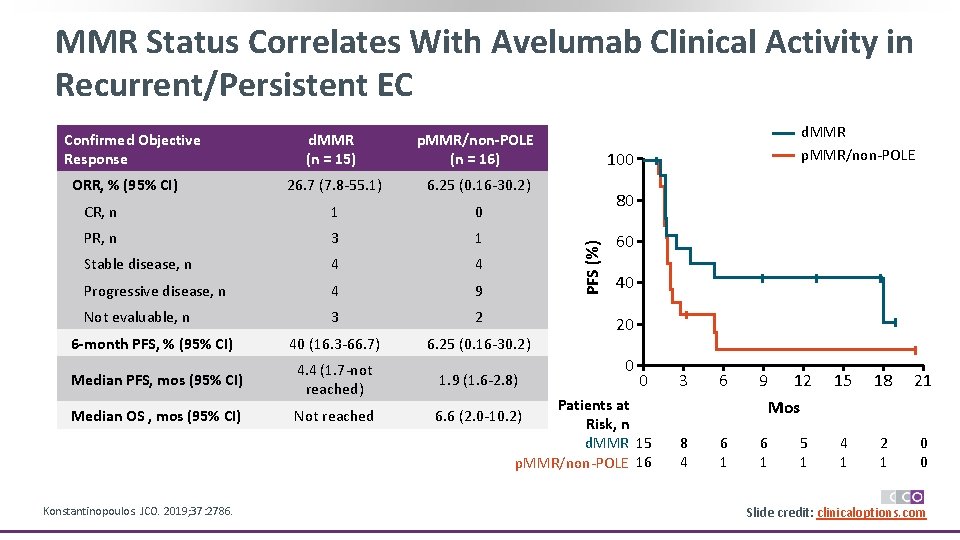
MMR Status Correlates With Avelumab Clinical Activity in Recurrent/Persistent EC d. MMR (n = 15) p. MMR/non-POLE (n = 16) 26. 7 (7. 8 -55. 1) 6. 25 (0. 16 -30. 2) CR, n 1 0 PR, n 3 1 Stable disease, n 4 4 Progressive disease, n 4 9 Not evaluable, n 3 2 40 (16. 3 -66. 7) 6. 25 (0. 16 -30. 2) ORR, % (95% CI) 6 -month PFS, % (95% CI) Median PFS, mos (95% CI) 4. 4 (1. 7 -not reached) 1. 9 (1. 6 -2. 8) Median OS , mos (95% CI) Not reached 6. 6 (2. 0 -10. 2) Konstantinopoulos. JCO. 2019; 37: 2786. d. MMR p. MMR/non-POLE 100 80 PFS (%) Confirmed Objective Response 60 40 20 0 0 3 6 9 12 Mos Patients at Risk, n d. MMR 15 p. MMR/non-POLE 16 8 4 6 1 5 1 15 18 21 4 1 2 1 0 0 Slide credit: clinicaloptions. com

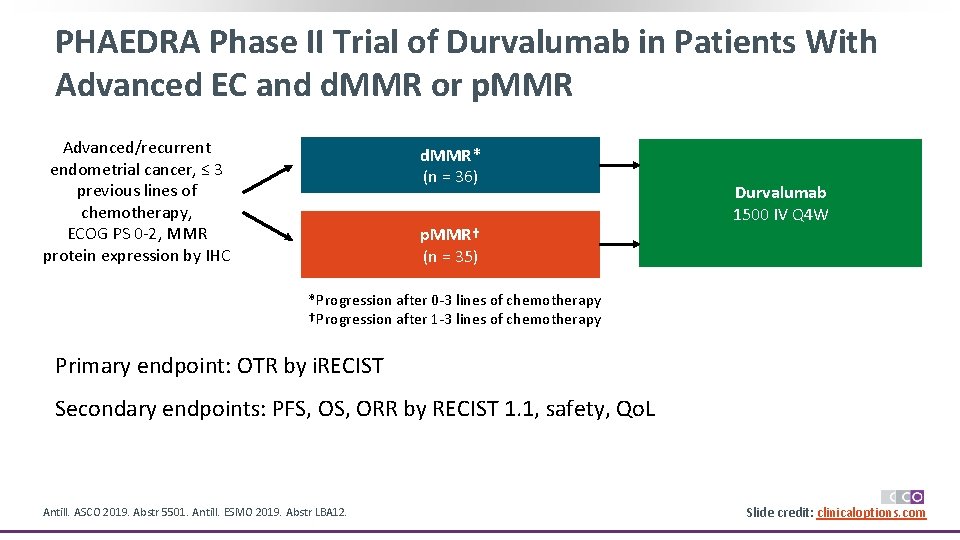
PHAEDRA Phase II Trial of Durvalumab in Patients With Advanced EC and d. MMR or p. MMR Advanced/recurrent endometrial cancer, ≤ 3 previous lines of chemotherapy, ECOG PS 0 -2, MMR protein expression by IHC d. MMR* (n = 36) p. MMR† (n = 35) Durvalumab 1500 IV Q 4 W *Progression after 0 -3 lines of chemotherapy †Progression after 1 -3 lines of chemotherapy Primary endpoint: OTR by i. RECIST Secondary endpoints: PFS, ORR by RECIST 1. 1, safety, Qo. L Antill. ASCO 2019. Abstr 5501. Antill. ESMO 2019. Abstr LBA 12. Slide credit: clinicaloptions. com

PHAEDRA: Antitumor Activity in Patients With d. MMR Advanced EC Endpoint d. MMR (n = 36) p. MMR (n = 35) 17 (47, 32– 63) 1 (3, 1– 15) CR, n 6 0 PR, n 11 1 Stable disease, n 6 10 PFS, median, mos 5. 5 1. 8 OS, median, mos Not reached 11. 5 71 51 OTR, n (%, 95% CI) 12 -mo OS, % Antill. ESMO 2019. Abstr LBA 12. § Durvalumab was the first, second, and subsequent line of nonhormonal therapy in 58%, 39%, and 3% of patients with d. MMR EC and 9%, 63%, and 29% of patients with p. MMR EC § Among patients with d. MMR, OTR rate was 57% as first line and 38% as second line Slide credit: clinicaloptions. com

Combination Checkpoint Inhibitor Studies in Advanced/Recurrent Endometrial Cancer Checkpoint Inhibitors Plus Antiangiogenic Agents Checkpoint Inhibitors Plus Chemotherapy KEYNOTE-146[1] NRG-GY 018[5]: Pembrolizumab + Paclitaxel/Carboplatin KEYNOTE-775 (phase III)[2] ENGOT-en 9/LEAP-001 (phase III)[3] Pembrolizumab + Lenvatinib NCT 03367741[4]: Nivolumab + Cabozantinib At. TEnd/ENGOT-en 7[6]: Atezolizumab + Paclitaxel/Carboplatin RUBY (ENGOT-EN 6; GOG-3031)[7]: Dostarlimab + Chemotherapy 1. Makker. JCO. 2020; 38: 2981. 2. NCT 03517449. 3. NCT 03884101. 4. NCT 03367741. 5. NCT 03914612. 6. NCT 03603184. 7. NCT 03981796. Slide credit: clinicaloptions. com

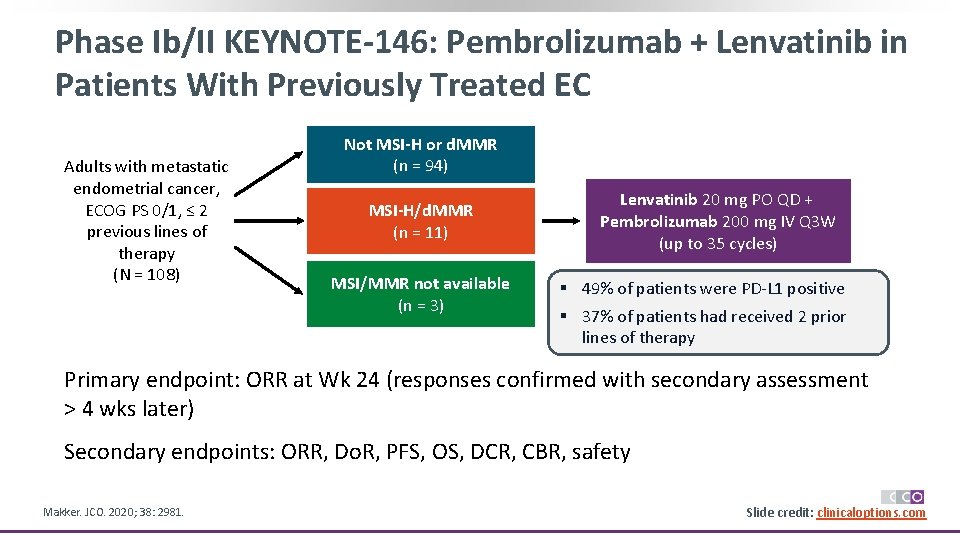
Phase Ib/II KEYNOTE-146: Pembrolizumab + Lenvatinib in Patients With Previously Treated EC Adults with metastatic endometrial cancer, ECOG PS 0/1, ≤ 2 previous lines of therapy (N = 108) Not MSI-H or d. MMR (n = 94) MSI-H/d. MMR (n = 11) MSI/MMR not available (n = 3) Lenvatinib 20 mg PO QD + Pembrolizumab 200 mg IV Q 3 W (up to 35 cycles) § 49% of patients were PD-L 1 positive § 37% of patients had received 2 prior lines of therapy Primary endpoint: ORR at Wk 24 (responses confirmed with secondary assessment > 4 wks later) Secondary endpoints: ORR, Do. R, PFS, OS, DCR, CBR, safety Makker. JCO. 2020; 38: 2981. Slide credit: clinicaloptions. com

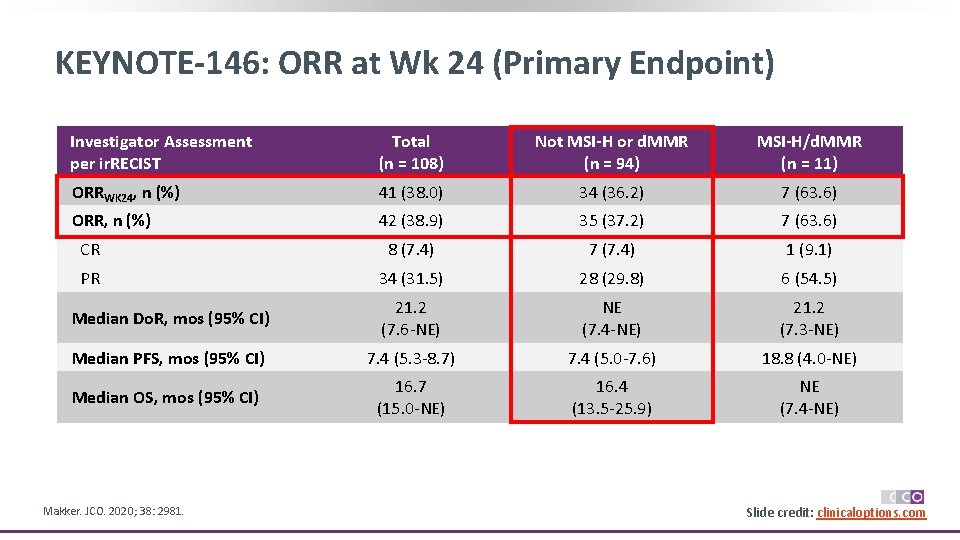
KEYNOTE-146: ORR at Wk 24 (Primary Endpoint) Investigator Assessment per ir. RECIST Total (n = 108) Not MSI-H or d. MMR (n = 94) MSI-H/d. MMR (n = 11) ORRWK 24, n (%) 41 (38. 0) 34 (36. 2) 7 (63. 6) ORR, n (%) 42 (38. 9) 35 (37. 2) 7 (63. 6) CR 8 (7. 4) 7 (7. 4) 1 (9. 1) PR 34 (31. 5) 28 (29. 8) 6 (54. 5) Median Do. R, mos (95% CI) 21. 2 (7. 6 -NE) NE (7. 4 -NE) 21. 2 (7. 3 -NE) Median PFS, mos (95% CI) 7. 4 (5. 3 -8. 7) 7. 4 (5. 0 -7. 6) 18. 8 (4. 0 -NE) Median OS, mos (95% CI) 16. 7 (15. 0 -NE) 16. 4 (13. 5 -25. 9) NE (7. 4 -NE) Makker. JCO. 2020; 38: 2981. Slide credit: clinicaloptions. com

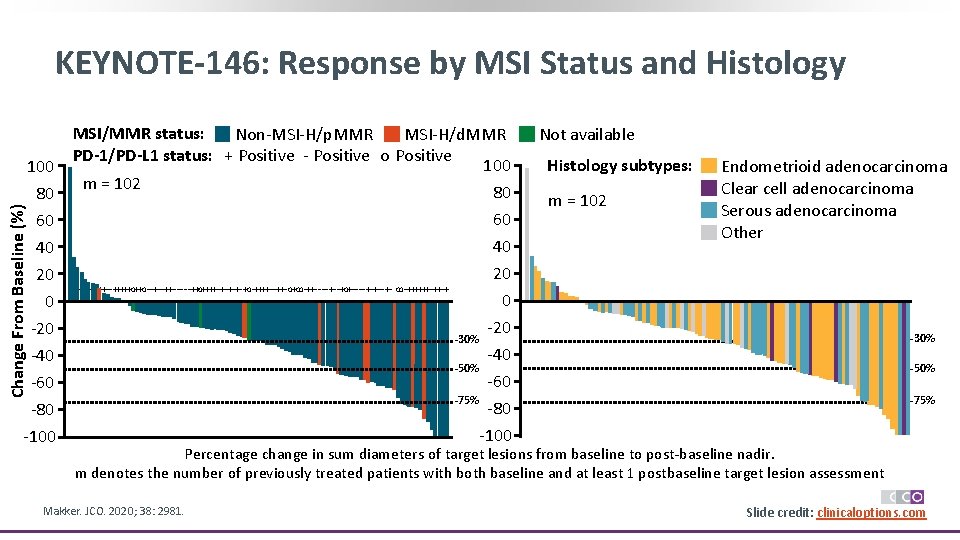
KEYNOTE-146: Response by MSI Status and Histology 80 MSI/MMR status: Non-MSI-H/p. MMR MSI-H/d. MMR PD-1/PD-L 1 status: + Positive - Positive o Positive 100 m = 102 80 60 60 40 40 Change From Baseline (%) 100 20 0 -20 -50% -60 -75% -80 m = 102 Endometrioid adenocarcinoma Clear cell adenocarcinoma Serous adenocarcinoma Other 0 - -30% -40 -100 - Histology subtypes: 20 ---+++++++--+++++o++o--+--++-----++o++++-+-+o-++++--++-o+oo-++----+--+o+----+-+--+- oo-++++++-++-+ - Not available -20 -30% -40 -50% -60 -75% -80 -100 Percentage change in sum diameters of target lesions from baseline to post-baseline nadir. m denotes the number of previously treated patients with both baseline and at least 1 postbaseline target lesion assessment Makker. JCO. 2020; 38: 2981. Slide credit: clinicaloptions. com

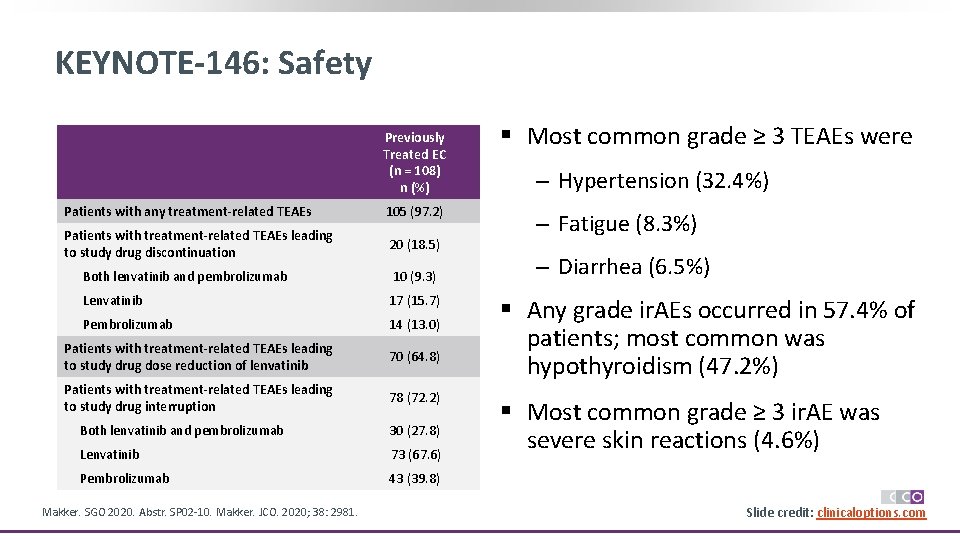
KEYNOTE-146: Safety Previously Treated EC (n = 108) n (%) Patients with any treatment-related TEAEs 105 (97. 2) Patients with treatment-related TEAEs leading to study drug discontinuation 20 (18. 5) Both lenvatinib and pembrolizumab 10 (9. 3) Lenvatinib 17 (15. 7) Pembrolizumab 14 (13. 0) Patients with treatment-related TEAEs leading to study drug dose reduction of lenvatinib 70 (64. 8) Patients with treatment-related TEAEs leading to study drug interruption 78 (72. 2) Both lenvatinib and pembrolizumab 30 (27. 8) Lenvatinib 73 (67. 6) Pembrolizumab 43 (39. 8) Makker. SGO 2020. Abstr. SP 02 -10. Makker. JCO. 2020; 38: 2981. § Most common grade ≥ 3 TEAEs were ‒ Hypertension (32. 4%) ‒ Fatigue (8. 3%) ‒ Diarrhea (6. 5%) § Any grade ir. AEs occurred in 57. 4% of patients; most common was hypothyroidism (47. 2%) § Most common grade ≥ 3 ir. AE was severe skin reactions (4. 6%) Slide credit: clinicaloptions. com

FDA Approval of Pembrolizumab + Lenvatinib for Advanced EC That Is Not MSI-H or d. MMR § Pembrolizumab plus lenvatinib is indicated for the treatment of advanced endometrial carcinoma that is not MSI-high or d. MMR 1 § FDA, Australian Therapeutic Goods Administration, and Health Canada collaborated on review, allowing simultaneous decision in all 3 countries[1] § Approval based on data from KEYNOTE-146[1] ‒ 94 (87%) patients had tumors that were not MSI-high or d. MMR 2 N = 108[2] Objective Response Rate ORR (95% CI) 40. 3% (31. 6 -49. 5) CR rate 8 (6. 5) PR rate 43 (33. 9) Response duration Median in mos (range) no. with duration ≥ 6 mos NE (8. 5 -NE) 36 Treatment is associated with any-grade AEs (>50%): hypertension (59. 7%), diarrhea (52. 4%) ‒ Of these, most patients were aged ≥ 65 yrs and 49% were PD-L 1 positive[2] 1. US Food and Drug Administration. Press Release. September 17, 2019. 2. Makker. JCO. 2020; 38: 2981. Slide credit: clinicaloptions. com

Phase III KEYNOTE-775: Second-line Pembrolizumab + Lenvatinib vs Chemotherapy in Advanced EC Stratified by MMR status (d. MMR vs p. MMR); p. MMR by ECOG PS, geographic region, prior pelvic radiation Advanced, recurrent or metastatic endometrial cancer, 1 previous platinum-based chemotherapy regimen for advanced disease, ECOG PS 0/1 (N = 827) Pembrolizumab 200 mg IV Q 3 W + Lenvatinib 20 mg PO QD Physician’s choice chemotherapy (doxorubicin or paclitaxel) Primary endpoints: PFS, OS Secondary endpoints: ORR, HRQo. L, safety and tolerability, PK NCT 03517449. Slide credit: clinicaloptions. com

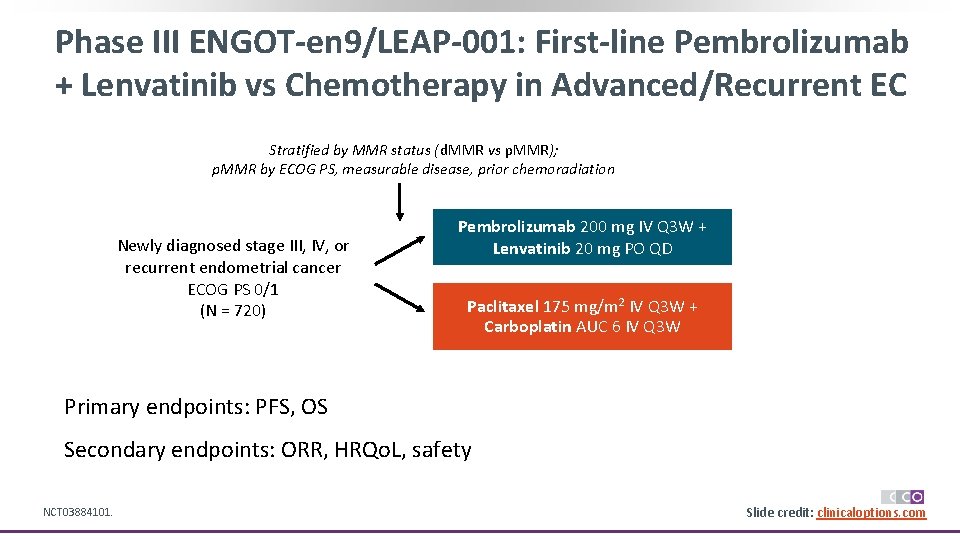
Phase III ENGOT-en 9/LEAP-001: First-line Pembrolizumab + Lenvatinib vs Chemotherapy in Advanced/Recurrent EC Stratified by MMR status (d. MMR vs p. MMR); p. MMR by ECOG PS, measurable disease, prior chemoradiation Newly diagnosed stage III, IV, or recurrent endometrial cancer ECOG PS 0/1 (N = 720) Pembrolizumab 200 mg IV Q 3 W + Lenvatinib 20 mg PO QD Paclitaxel 175 mg/m 2 IV Q 3 W + Carboplatin AUC 6 IV Q 3 W Primary endpoints: PFS, OS Secondary endpoints: ORR, HRQo. L, safety NCT 03884101. Slide credit: clinicaloptions. com

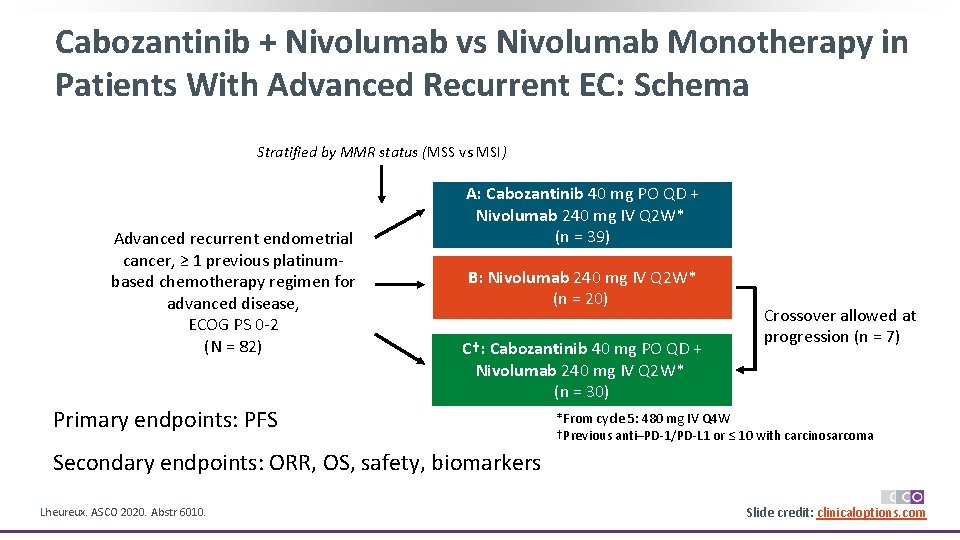
Cabozantinib + Nivolumab vs Nivolumab Monotherapy in Patients With Advanced Recurrent EC: Schema Stratified by MMR status (MSS vs MSI) Advanced recurrent endometrial cancer, ≥ 1 previous platinumbased chemotherapy regimen for advanced disease, ECOG PS 0 -2 (N = 82) A: Cabozantinib 40 mg PO QD + Nivolumab 240 mg IV Q 2 W* (n = 39) B: Nivolumab 240 mg IV Q 2 W* (n = 20) C†: Cabozantinib 40 mg PO QD + Nivolumab 240 mg IV Q 2 W* (n = 30) Primary endpoints: PFS Crossover allowed at progression (n = 7) *From cycle 5: 480 mg IV Q 4 W †Previous anti–PD-1/PD-L 1 or ≤ 10 with carcinosarcoma Secondary endpoints: ORR, OS, safety, biomarkers Lheureux. ASCO 2020. Abstr 6010. Slide credit: clinicaloptions. com

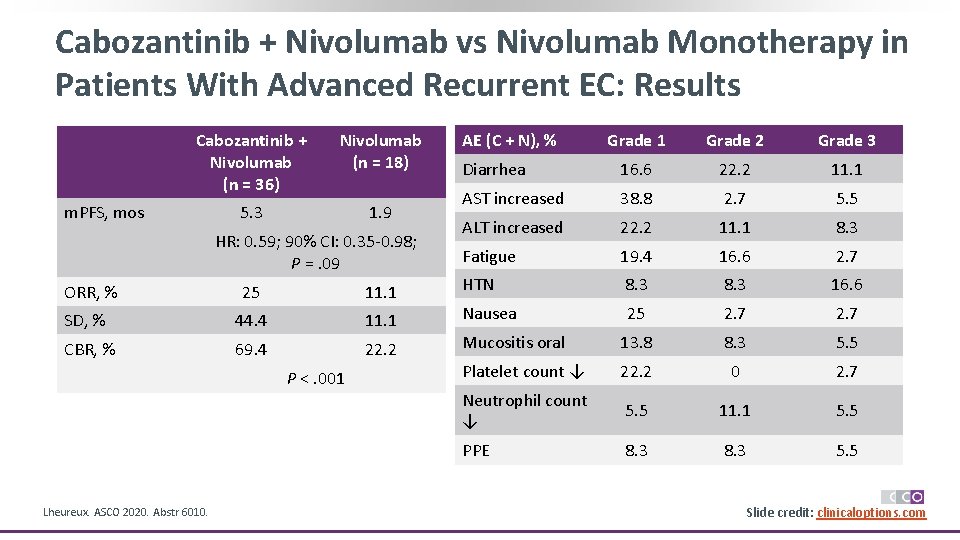
Cabozantinib + Nivolumab vs Nivolumab Monotherapy in Patients With Advanced Recurrent EC: Results Cabozantinib + Nivolumab (n = 36) Nivolumab (n = 18) 5. 3 1. 9 m. PFS, mos HR: 0. 59; 90% CI: 0. 35 -0. 98; P =. 09 ORR, % 25 11. 1 SD, % 44. 4 11. 1 CBR, % 69. 4 22. 2 P <. 001 Lheureux. ASCO 2020. Abstr 6010. AE (C + N), % Grade 1 Grade 2 Grade 3 Diarrhea 16. 6 22. 2 11. 1 AST increased 38. 8 2. 7 5. 5 ALT increased 22. 2 11. 1 8. 3 Fatigue 19. 4 16. 6 2. 7 HTN 8. 3 16. 6 Nausea 25 2. 7 Mucositis oral 13. 8 8. 3 5. 5 Platelet count ↓ 22. 2 0 2. 7 Neutrophil count ↓ 5. 5 11. 1 5. 5 PPE 8. 3 5. 5 Slide credit: clinicaloptions. com

Combination Checkpoint Inhibitor Studies in Advanced/Recurrent Endometrial Cancer Checkpoint Inhibitors Plus Antiangiogenic Agents Checkpoint Inhibitors Plus Chemotherapy KEYNOTE-146[1] NRG-GY 018[5]: Pembrolizumab + Paclitaxel/Carboplatin KEYNOTE-775 (phase III)[2] ENGOT-en 9/LEAP-001 (phase III)[3] Pembrolizumab + Lenvatinib NCT 0336774[4]: Nivolumab + Cabozantinib At. TEnd/ENGOT-en 7[6]: Atezolizumab + Paclitaxel/Carboplatin RUBY (ENGOT-EN 6; GOG-3031)[7]: Dostarlimab + Chemotherapy 1. Makker. JCO. 2020; 38: 2981. 2. NCT 03517449. 3. NCT 03884101. 4. NCT 03367741. 5. NCT 03914612. 6. NCT 03603184. 7. NCT 03981796. Slide credit: clinicaloptions. com

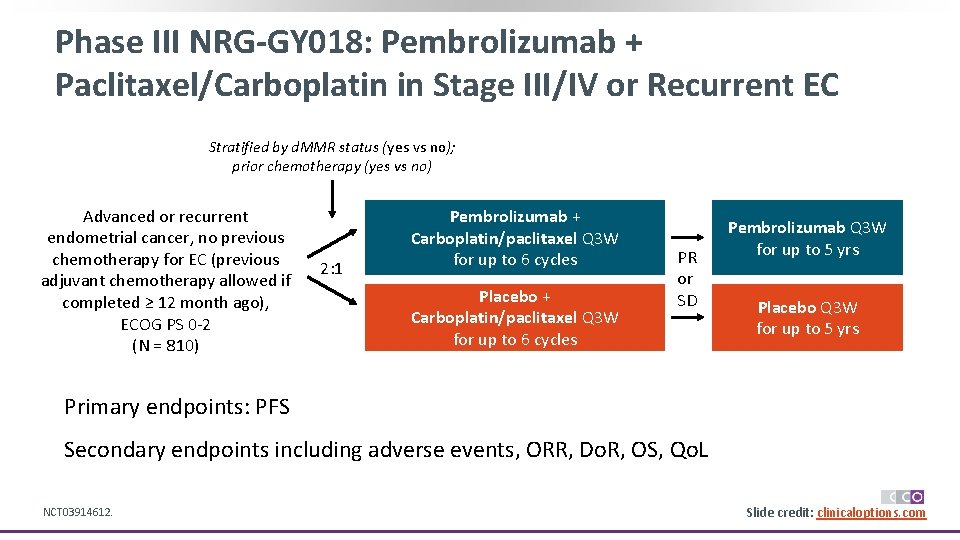
Phase III NRG-GY 018: Pembrolizumab + Paclitaxel/Carboplatin in Stage III/IV or Recurrent EC Stratified by d. MMR status (yes vs no); prior chemotherapy (yes vs no) Advanced or recurrent endometrial cancer, no previous chemotherapy for EC (previous adjuvant chemotherapy allowed if completed ≥ 12 month ago), ECOG PS 0 -2 (N = 810) 2: 1 Pembrolizumab + Carboplatin/paclitaxel Q 3 W for up to 6 cycles Placebo + Carboplatin/paclitaxel Q 3 W for up to 6 cycles PR or SD Pembrolizumab Q 3 W for up to 5 yrs Placebo Q 3 W for up to 5 yrs Primary endpoints: PFS Secondary endpoints including adverse events, ORR, Do. R, OS, Qo. L NCT 03914612. Slide credit: clinicaloptions. com

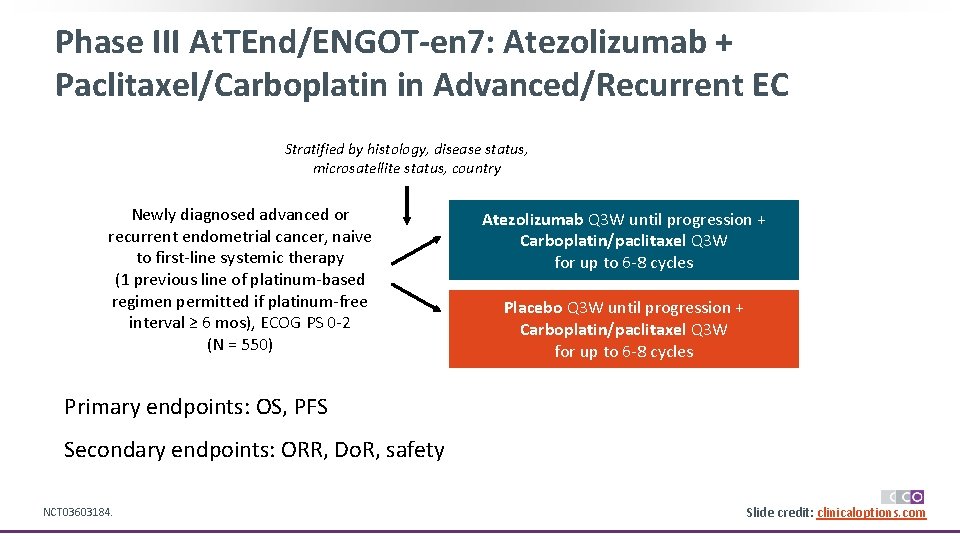
Phase III At. TEnd/ENGOT-en 7: Atezolizumab + Paclitaxel/Carboplatin in Advanced/Recurrent EC Stratified by histology, disease status, microsatellite status, country Newly diagnosed advanced or recurrent endometrial cancer, naive to first-line systemic therapy (1 previous line of platinum-based regimen permitted if platinum-free interval ≥ 6 mos), ECOG PS 0 -2 (N = 550) Atezolizumab Q 3 W until progression + Carboplatin/paclitaxel Q 3 W for up to 6 -8 cycles Placebo Q 3 W until progression + Carboplatin/paclitaxel Q 3 W for up to 6 -8 cycles Primary endpoints: OS, PFS Secondary endpoints: ORR, Do. R, safety NCT 03603184. Slide credit: clinicaloptions. com

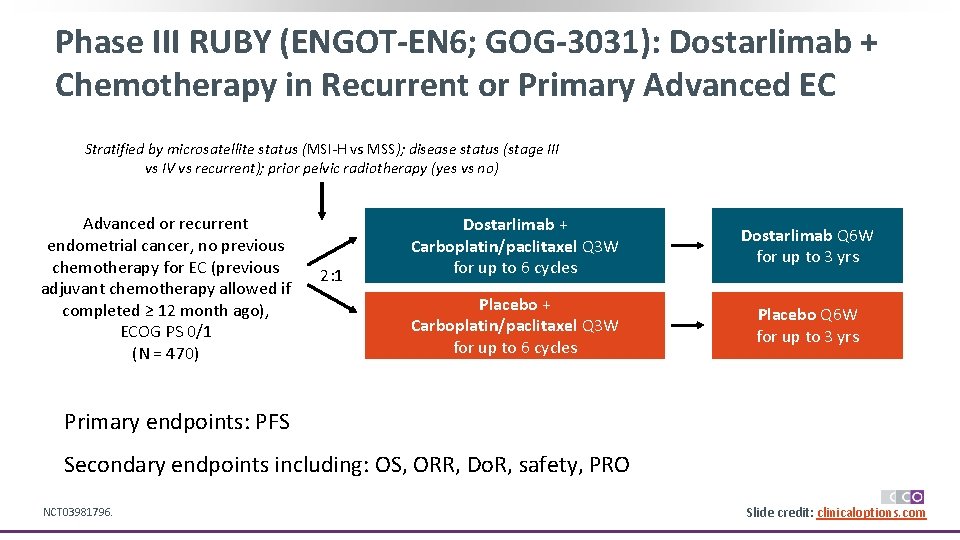
Phase III RUBY (ENGOT-EN 6; GOG-3031): Dostarlimab + Chemotherapy in Recurrent or Primary Advanced EC Stratified by microsatellite status (MSI-H vs MSS); disease status (stage III vs IV vs recurrent); prior pelvic radiotherapy (yes vs no) Advanced or recurrent endometrial cancer, no previous chemotherapy for EC (previous adjuvant chemotherapy allowed if completed ≥ 12 month ago), ECOG PS 0/1 (N = 470) 2: 1 Dostarlimab + Carboplatin/paclitaxel Q 3 W for up to 6 cycles Dostarlimab Q 6 W for up to 3 yrs Placebo + Carboplatin/paclitaxel Q 3 W for up to 6 cycles Placebo Q 6 W for up to 3 yrs Primary endpoints: PFS Secondary endpoints including: OS, ORR, Do. R, safety, PRO NCT 03981796. Slide credit: clinicaloptions. com

General Principles for Managing ir. AEs § Consult promptly with relevant specialists for affected organ systems (eg, gastroenterology, dermatology) § Management generally based on severity of symptoms ‒ Mild (grade 1): supportive care, consider holding drug ‒ Moderate (grade 2): hold drug, redose if toxicity improves, consider low-dose steroids (prednisone 0. 5 -1 mg/kg/day) ‒ Severe (grade 3): discontinue drug, monitor closely (likely inpatient), start high-dose steroids (prednisone 1 -2 mg/kg/day) with a slow taper (≥ 1 mo) ‒ If not improving in 1 -3 days, increase immunosuppression § Dose reduction is not a recommended strategy § Avoid delays in recognition and intervention Brahmer. JCO. 2018; 36: 1714. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of Gynecologic Cancers! Capsule Summaries of all the key data from recent oncology conferences Podcasts on endometrial cancer with expert faculty commentary on recent advances in this topic clinicaloptions. com/oncology