ADVANCES IN CHEMICAL REACTIVITY EVALUATION AT DOW MARABETH

ADVANCES IN CHEMICAL REACTIVITY EVALUATION AT DOW MARABETH HOLSINGER, PH. D GLOBAL TECHNOLOGY LEADER, REACTVE CHEMICALS / ANALYTICAL SCIENCES Our intent is that by sharing enhanced practices for identifying and managing uncontrolled chemical reactions, the chemical process industries can realize benefits from improved reliability of operations, fewer incidents over the life of a process, and reduced consequences of any incidents that occur. P 2 SAC - Purdue Process Safety and Assurance Center – Fall Conference West Lafayette, IN Nov 10, 2016

Dow Business and Market Focus 2 Delivering Customer Solutions Agricultural Sciences § Seeds § Crop Protection Consumer Solutions Infrastructure Solutions § Dow Electronic § Dow Coating § § Materials Dow Automotive Systems Consumer Care § Materials Performance Mon. Dow Building & Construction Energy & Water Soln. Performance Materials & Chemicals § Chlor Alkali and Vinyl § Polyurethanes § Industrial Solutions VORALUX™ Specialty Flex Foams AR™ Fast-Etch Organic BARC BETAMATE™ Structural Adhesives POLYOX™ Polymers Revenue: $6. 4 B Revenue: $4. 4 B P 2 SAC – Purdue Process Safety Conference Performance Plastics § Dow Packaging and § § Specialty Plastics Dow Elastomers Dow Electrical and Telecommunications Energy Hydrocarbons FR 63 Polymeric Flame Retardant FILMTEC™ RO Revenue: $7. 4 B ECOSURF™ Nonionic Surfactants Revenue: $12. 0 B MOLYKOTE® Lubricants & Silastic® Elastomers Dow Corning® 3901 Liquid Satin FORMASHIELD™ Technology ENFORCER™ Polyurethane for Artificial Turf Performance Silicones 100% PE Stand-up Pouch Pac. Xpert™ Packaging Technology INFUSE™ Olefin Block Copolymers for Athletic Shoe Midsoles Revenue: $18. 4 B Dow Corning® Façade Solutions Burj Khalifa Emaar Properties Syl-Off® Release Coatings Revenue: $4. 7 B‡ 9 -Nov

Preventing Reactive Chemicals Incidents 3 Dow has a wide variety of chemical processes ― ― ― Different types of chemistries and operations in different businesses The scope of hazard scenarios is wide and complicated From R&D labs to pilot plants and full-scale manufacturing To effectively eliminate potential incidents in our processes at all scales, Dow relies on its strong Reactive Chemicals Program: ― Based on three key principles: To know how much inherent energy is present in every process step or unit operation ü To know the conditions under which this energy can be released such that adequate controls can be put in place to manage the risks ü To hold the owner (of a process, lab, or plant) responsible to ensure that reactive chemicals hazards are identified, adequate layers of protection are in place, and formalized and documented periodic reviews are conducted Evolved into a recognized industry model leveraged through CCPS 1 ü ― 1. Frurip, D. J. ; The Essential Elements of a Successful Reactive Chemicals Program, Chemical Engineering Transactions, 26, (2012), pp. 1 -6 P 2 SAC – Purdue Process Safety Conference ― 9 -Nov

Dow’s Reactive Chemicals Program 4 Dow’s Reactive Chemicals Program was established after an incident happened in Dow in 1966. The purpose of Dow’s Reactive Chemicals Program is to prevent uncontrolled or unexpected chemical reactions that have the potential to result in injury, property damage or environmental harm. Propargyl bromide + Chloropicrin Dec 1966 - Three craters left by explosions caused by mixing of materials that formed a shock sensitive material using 2 railcars and a tank truck (3 fatalities). P 2 SAC – Purdue Process Safety Conference from The Dow Chemical Company 9 -Nov

Formalized and Documented Hazard Reviews 5 An essential aspect of a strong Reactive Chemicals Program involves periodic Reactive Chemicals/Process Hazard Analysis (RC/PHA) and Laboratory Hazard Analysis (RC/LHA) reviews for new and existing Dow facilities, new leaders, and capital projects. ― ― ― Multi-disciplinary review teams are formed from employees with broad technical expertise and backgrounds. Typically includes representatives from the facility, the Technology Center, EH&S, and Process Safety and Reactive Chemicals (RC) functions, along with “outside-in” expertise from R&D, engineering, and manufacturing. The review team’s goal - to provide a company-wide, consistent, and as accurate as currently available perspective on hazard identification and the evaluation of adequacy of hazard controls, reaching consensus on whether additional controls are needed, or if additional hazard or risk evaluation is warranted. Using provided documentation and Dow’s RC/PHA Questionnaire as a guide, they ü ü ü Identify and document potential hazards for every unit operation, including conditions leading to such hazards Capture follow-up actions for items not resolved at the meeting May perform a walk through inspection of the facility P 2 SAC – Purdue Process Safety Conference 9 -Nov
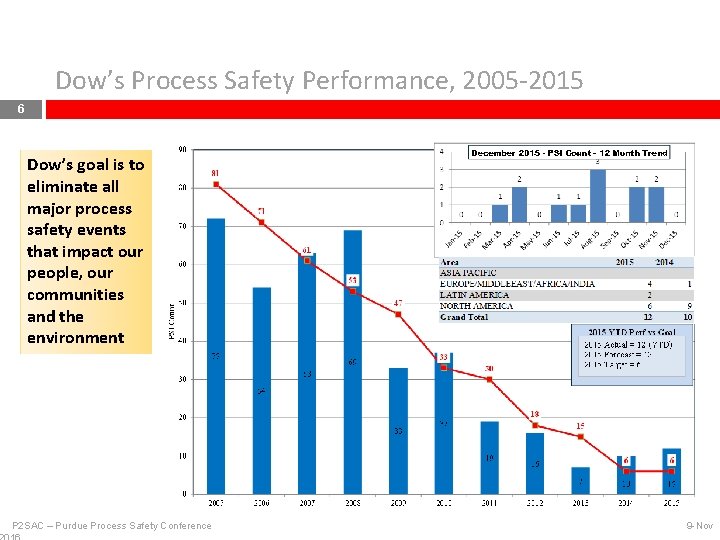
Dow’s Process Safety Performance, 2005 -2015 6 Dow’s goal is to eliminate all major process safety events that impact our people, our communities and the environment P 2 SAC – Purdue Process Safety Conference 9 -Nov
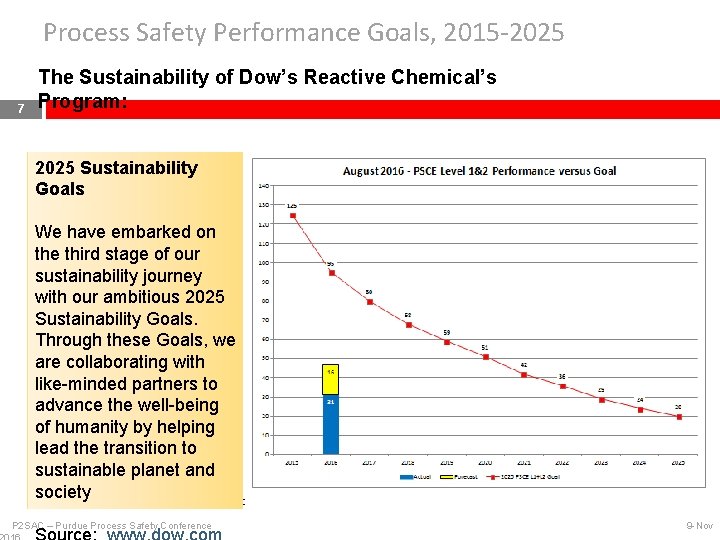
Process Safety Performance Goals, 2015 -2025 7 The Sustainability of Dow’s Reactive Chemical’s Program: 2025 Sustainability Goals We have embarked on the third stage of our sustainability journey with our ambitious 2025 Sustainability Goals. Through these Goals, we are collaborating with like-minded partners to advance the well-being of humanity by helping lead the transition to sustainable planet and society PSCE: Process Safety & Containment Event P 2 SAC – Purdue Process Safety Conference 9 -Nov
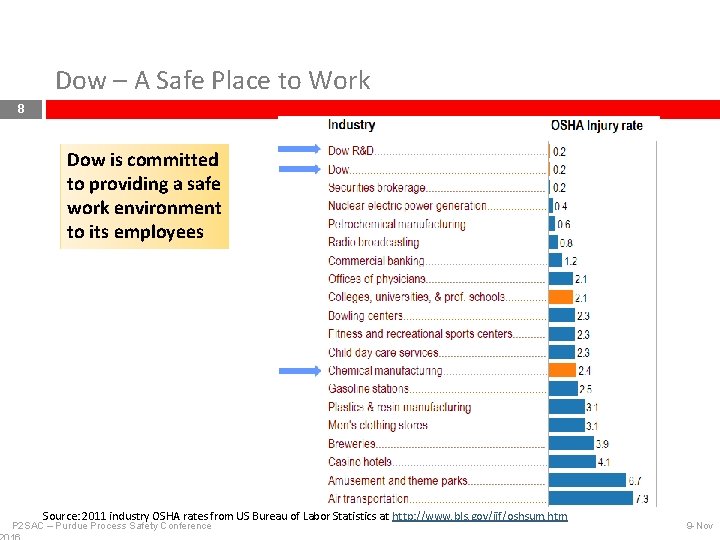
Dow – A Safe Place to Work 8 Dow is committed to providing a safe work environment to its employees Source: 2011 industry OSHA rates from US Bureau of Labor Statistics at http: //www. bls. gov/iif/oshsum. htm P 2 SAC – Purdue Process Safety Conference 9 -Nov

Dow’s Reactive Chemicals Capability 9 Member of Dow’s Analytical Sciences Organization Strategic Focus: • Technical problem solving utilizing reactive chemicals (RC) technologies for complex issues at all scales • New technology and application development • Advanced solutions ensuring safe operating limits, operational reliability, process development and optimization. • 24/7 Immediate Response and Incident Investigations • Reactive Chemicals Process Hazard Analysis (RC/PHA, RC/LHA) and New Leader Reviews (NLR) • Compliance testing for REACH, MSDS, NFPA, DOT etc. • Understanding reactive chemicals issues through deep knowledge of business chemistries and process technologies (RC Business focal points) • Increase awareness of RC hazards through training to labs, plants, Tech Centers and business/functional stakeholders P 2 SAC – Purdue Process Safety Conference 9 -Nov

Reactive Chemicals Hazard Evaluation Tools 10 • The types of testing and calculations, and the amount of effort we spend, depends on the type of operation, the scale of the process, the conditions of the process, the amount of energy release, and the potential for gas and pressure generation. P 2 SAC – Purdue Process Safety Conference 9 -Nov

Thermal Screening Tools 11 Desktop methods or literature • • Heat of reaction and adiabatic temperature rise calculations Dow’s internal database CHETAH calculations Bretherick’s Handbook of Reactive Chemical Hazards Chemical Reactivity Worksheet (CRW) High energy functional groups MSDS, hazard statements (R and H phrases) TA Instruments HEL Heat of mixing calorimetry (HOM) Differential Scanning Calorimetry (DSC) Thermal Screening Unit (TSU) Temperature ramp or isothermal mode • Accurate measurement of pressure and detected onset • Can detect exotherm and endotherm • Disadvantage – Heat data is not quantitative • Process Disadvantage – Data cannot be modeled P 2 SAC – Purdue Safety Conference • 9 -Nov

Heat of Reaction Calculations 12 For early stage of process development, small scale Start from a balanced equation for the reaction Using simplified analog reactions if needed Find or estimate enthalpies formation for the reactants and products involved in the reaction • Phase correction, concentration effect § • Data from literature, compilations and handbooks § NBS Tables, NIST Web. Book, Pedley, DIPPR, Cox and Pilcher… Dow’s internal database and tools Adiabatic temperature rise calculations • Liquid, gas, solid, aqueous solutions Need heat capacity data CHETAH calculations • Most recent version: CHETAH 10. 0 was released in January 2016 P 2 SAC – Purdue Process Safety Conference 9 -Nov

Chemical Reactivity Evaluation – Desktop Methods 13 CHETAH 10. 0 (Chemical Thermodynamic and Energy Release) Program: A unique tool for predicting both thermochemical properties and certain ”reactive chemicals” hazards associated with a pure chemical, a mixture of chemicals, or a chemical reaction. Requires only knowledge of the molecular structure(s) of the components involved, then implements Benson's method of group additivity. Useful for classifying materials for their ability to decompose with violence, for estimating heats of reaction (Britton and Harrison, 2014) or combustion, and for predicting lower flammable limits. P 2 SAC – Purdue Process Safety Conference 9 -Nov

Chemical Reactivity Evaluation – Desktop Methods 14 CHETAH 10. 0 (Chemical Thermodynamic and Energy Release) Program: What’s new in 10. 0 ? • New interface • Expanded ideal gas Benson groups database (now 1060 groups) • Updated thermodynamic values provided for 20 Benson groups based on latest best values in the literature. • Entropy values were predicted and added to the database for many missing ideal gas Benson groups • Various property databases reviewed for consistency and accuracy • Improved accuracy of mixture flammability calculations (Britton, 2002; Britton and Frurip, 2003) • Change in programming language for CHETAH makes the program more compatible and seamless with the current versions of the Microsoft Windows OS • Output reports have been improved for clarity of presentation. • Maintains a clear record of use with past sessions - easily be modified and P 2 SAC – Purdue Process Safety Conference 9 -Nov
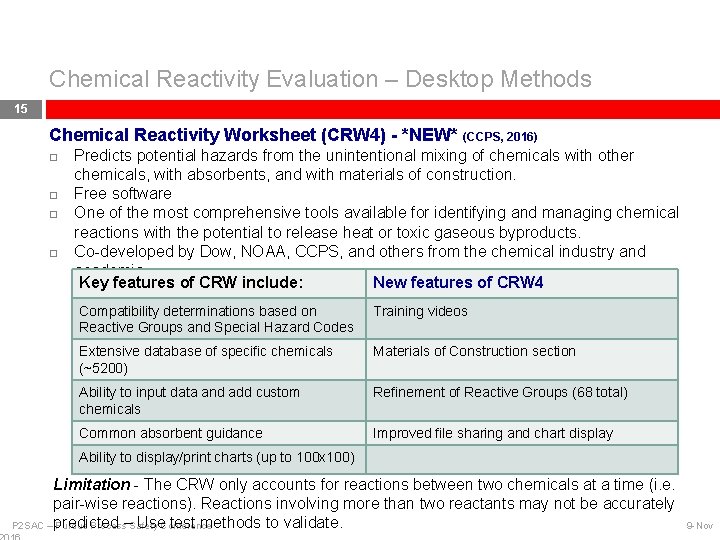
Chemical Reactivity Evaluation – Desktop Methods 15 Chemical Reactivity Worksheet (CRW 4) - *NEW* (CCPS, 2016) Predicts potential hazards from the unintentional mixing of chemicals with other chemicals, with absorbents, and with materials of construction. Free software One of the most comprehensive tools available for identifying and managing chemical reactions with the potential to release heat or toxic gaseous byproducts. Co-developed by Dow, NOAA, CCPS, and others from the chemical industry and academia Key features of CRW include: New features of CRW 4 Compatibility determinations based on Reactive Groups and Special Hazard Codes Training videos Extensive database of specific chemicals (~5200) Materials of Construction section Ability to input data and add custom chemicals Refinement of Reactive Groups (68 total) Common absorbent guidance Improved file sharing and chart display Ability to display/print charts (up to 100 x 100) Limitation - The CRW only accounts for reactions between two chemicals at a time (i. e. pair-wise reactions). Reactions involving more than two reactants may not be accurately Use. Conference test methods to validate. P 2 SAC – predicted Purdue Process–Safety 9 -Nov
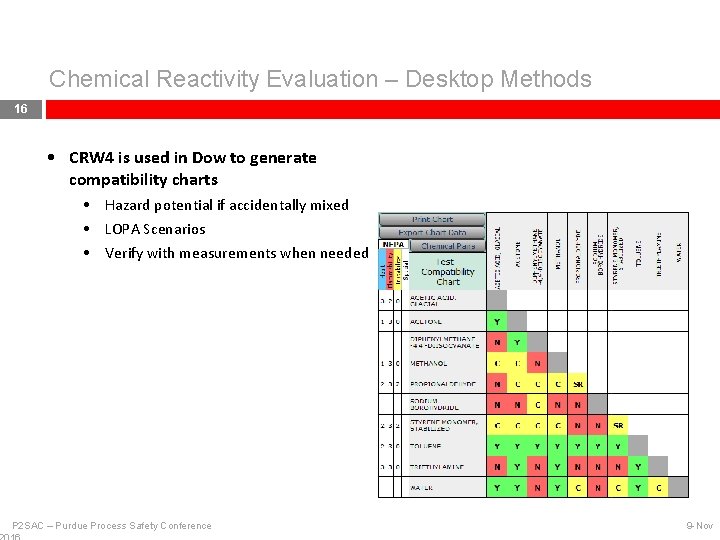
Chemical Reactivity Evaluation – Desktop Methods 16 • CRW 4 is used in Dow to generate compatibility charts • Hazard potential if accidentally mixed • LOPA Scenarios • Verify with measurements when needed P 2 SAC – Purdue Process Safety Conference 9 -Nov

Chemical Reactivity Evaluation – Desktop Methods 17 Yoshida’s Correlation - to screen for potential impact sensitivity hazards by DSC Strictly empirical method derived from a series of ~35 sealed-cell DSC (Differential Scanning Calorimetry) tests on known high energy materials in the powder form, mainly nitro compounds (Yoshida, 1987). • Predicts a material’s hazard potential when subjected to low-to-moderate energy forces (e. g. crushing, scraping with a spatula, dropping containers, hitting with a hammer, puncturing with a fork truck tine) • Conservative, since the original test data materials were all classified for their ability to propagate an explosion under high energy stimulus. Some of these did not propagate an explosion at BAM (German Republic Materials Testing Lab. ) • DSC test data are plotted as the log 10 of the integrated energy of the exothermic decomposition versus the log 10 (Tonset, extrapolated°C - 25°C). • Typically, materials having a DSC decomposition energy below -250 cal/g (-1046 J/g) will not be an explosion hazard when subjected to low-to- P 2 SAC – Purdue Process Safety Conference 9 -Nov

Chemical Reactivity Evaluation – Desktop Methods 18 Example: DSC of an unknown material in a sealed. N 2 capillary. A large exotherm is detected at 205 o. C with a reported enthalpy of -450 cal/g (-1883 J/g). Note: Within Dow, we enter the DSC detected onset temperature instead of the extrapolated onset temperature (easy to obtain, more conservative) Limitations: Use caution when applying to liquids and non nitro-containing compounds Work to improve this correlation not incorporated here (Oxley et al. , 1999, 2010; Chervin and Bodman, 2003; and Bodman and Chervin, 2004). If result is “above the line”, consider testing, especially if material contains known energetic functional groups. P 2 SAC – Purdue Process Safety Conference 9 -Nov

Advancements in Heat of Mixing Calorimetry 19 Heat of Mixing Calorimetry – • The “ 2 -drop” was invented at Dow to measure heats of solution, absorption, reaction, gas evolution; excellent for assessing chemical compatibility • To obtain DHrxn, must measure % conversion Dow evaluated and selected Thermal Hazard Technology’s (THT) micro reaction calorimeter (m. RC) • Higher temperature range -5 to 170ºC • Rapid equilibration, easy to use, portable, and small sample sizes • More operating modes: titration calorimetry, isothermal calorimetry and differential scanning calorimetry for applications in hazard analysis, thermal stability, Cp determination, rxn kinetics P 2 SAC – Purdue Process Safety Conference 9 -Nov
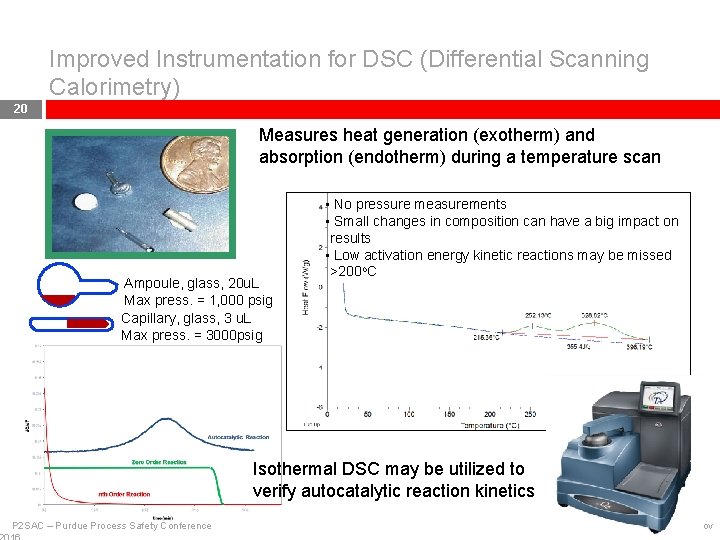
Improved Instrumentation for DSC (Differential Scanning Calorimetry) 20 Measures heat generation (exotherm) and absorption (endotherm) during a temperature scan Ampoule, glass, 20 u. L Max press. = 1, 000 psig Capillary, glass, 3 u. L Max press. = 3000 psig • No pressure measurements • Small changes in composition can have a big impact on results • Low activation energy kinetic reactions may be missed >200 o. C Isothermal DSC may be utilized to verify autocatalytic reaction kinetics P 2 SAC – Purdue Process Safety Conference 9 -Nov
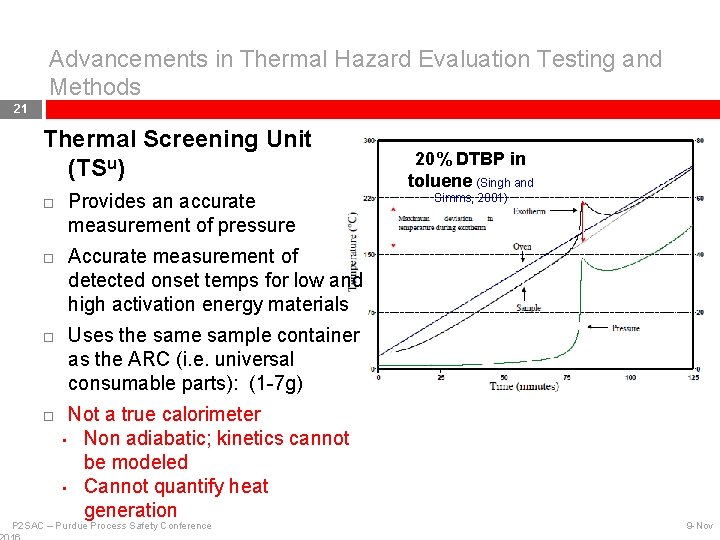
Advancements in Thermal Hazard Evaluation Testing and Methods 21 Thermal Screening Unit (TSu) Provides an accurate measurement of pressure 20% DTBP in toluene (Singh and Simms, 2001) Accurate measurement of detected onset temps for low and high activation energy materials Uses the sample container as the ARC (i. e. universal consumable parts): (1 -7 g) Not a true calorimeter • Non adiabatic; kinetics cannot be modeled • Cannot quantify heat generation P 2 SAC – Purdue Process Safety Conference 9 -Nov

Advancements in Thermal Hazard Evaluation Testing and Methods 22 Within Dow, we utilize the TSu: • For screening worst-case pressure scenarios for relief design (i. e. VSP test pre-screen) • For quantifying non-condensable gases under isothermal conditions • In combination with DSC to obtain both thermokinetics (from DSC) and pressure data (from Tsu) • To generate results fast and economically. If no significant exothermic and pressure generation events, then no need to get more tests. Advantages over the ARC: • A test in the ARC may take up to three days longer to complete. • DSC and TSu are cheaper and less costly to maintain (than an ARC) • Using these instruments can save wear and tear on the ARC. • Can be set up for injection runs • Easier to recover after a ruptured sample sphere in TSU than ARC Returning an ARC to service can take up to two weeks longer than returning a P 2 SAC – Purdue Process Conference TSU to. Safety service (considering repair, calibration, and performance validation). 9 -Nov •

More Advanced Calorimetric Testing Tools 23 Accelerating Rate Calorimetry (ARC) • • • Measures heat (exotherm only) and pressure under adiabatic conditions More sensitive than DSC Data suitable for modeling (with Phi correction) NETZSCH Microcalorimeters C 80, TAM III… • Very sensitive • Can confirm ARC kinetic extrapolation to lower temperatures • Long-term storage stability testing Self accelerating P 2 SAC – • Purdue Process Safety Conference decomposition temperature (SADT) • TA Instruments 9 -Nov
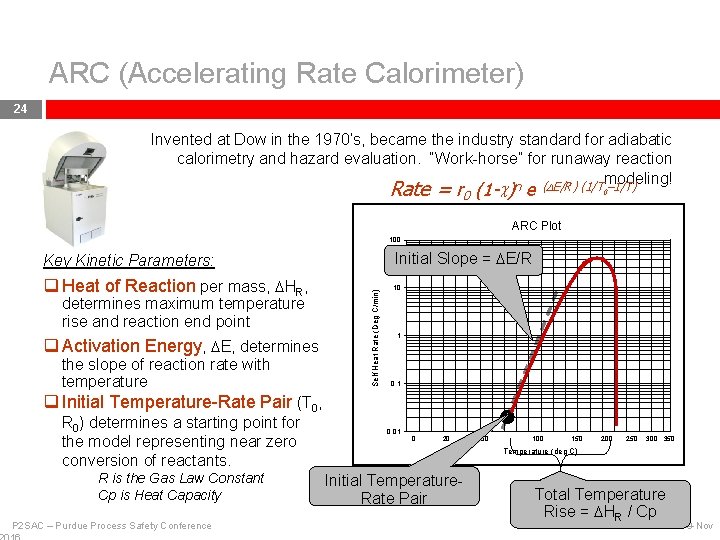
ARC (Accelerating Rate Calorimeter) 24 Invented at Dow in the 1970’s, became the industry standard for adiabatic calorimetry and hazard evaluation. “Work-horse” for runaway reaction modeling! Rate = r 0 (1 -c)n e (DE/R) (1/T 0– 1/T) ARC Plot 100 Initial Slope = DE/R q Heat of Reaction per mass, DHR, 10 determines maximum temperature rise and reaction end point q Activation Energy, DE, determines the slope of reaction rate with temperature Self Heat Rate (Deg C/min) Key Kinetic Parameters: 1 0. 1 q Initial Temperature-Rate Pair (T 0, R 0) determines a starting point for the model representing near zero conversion of reactants. R is the Gas Law Constant Cp is Heat Capacity P 2 SAC – Purdue Process Safety Conference 0. 01 0 20 50 100 150 200 250 300 350 Temperature (deg C) Initial Temperature. Rate Pair Total Temperature Rise = DHR / Cp 9 -Nov

Open Cup ARC Testing 25 Air is circulated through a preheater and purged through a sample (powder, granules, liquid, or liquid soaked insulation etc. ) through a dip tube A thermocouple is placed in the sample adjacent to the air outlet Heat-Wait-Search, and detection of exotherm Excellent for quantifying oxidative runaway potential of high surface area materials soaked with organic fluids to determine conditions resulting in a fire…. P 2 SAC – Purdue Process Safety Conference 9 -Nov
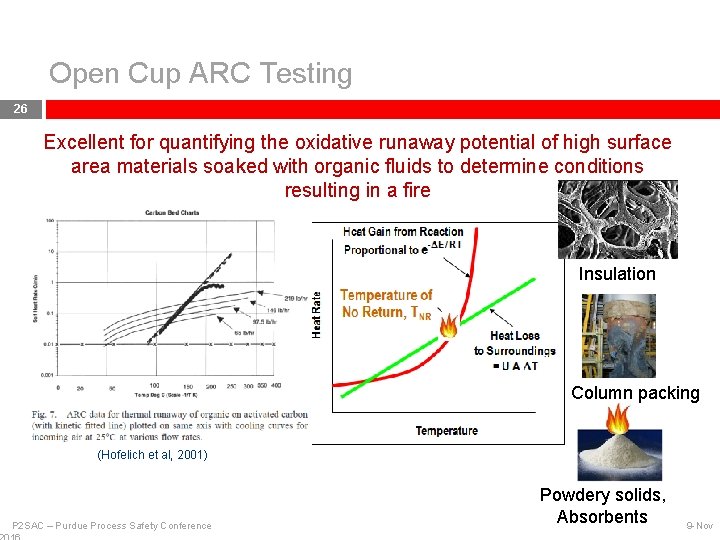
Open Cup ARC Testing 26 Excellent for quantifying the oxidative runaway potential of high surface area materials soaked with organic fluids to determine conditions resulting in a fire Insulation Column packing (Hofelich et al, 2001) P 2 SAC – Purdue Process Safety Conference Powdery solids, Absorbents 9 -Nov

Advancements in Thermal Hazard Evaluation Testing and Methods 27 Microcalorimetry At Dow, many types of micro-calorimeters (e. g. TAM III, C-80) are utilized for validating extrapolated kinetic models from ARC data to lower temperatures. Example: Using ARC and micro-calorimetric data with known ambient heat losses from piles of powdery solids (Frank-Kamenetskii Method), estimate how thick a layer of powdery product “X” must be before the heat gains will outpace the heat losses At 100°C we predict that “X” will run away at thicknesses greater than 4 inches, and as small as 1 inch at 110°C P 2 SAC – Purdue Process Safety Conference 9 -Nov

Scalable Testing Tools and Modeling 28 Vent Sizing Package (VSP) • • Reaction Calorimetry (RC 1) • • Phi factor is close to 1 Primary tool for relief sizing Can simulate process conditions Can get kinetics of desired reactions Fauske Modeling • • • Development of detailed kinetic models for the process/chemistry Time to Maximum Rate (TMR) Heat gain and heat loss evaluation for determination of Temperature of No Return (TNR) P 2 SAC – Purdue Process Safety Conference Mettler Toledo 9 -Nov
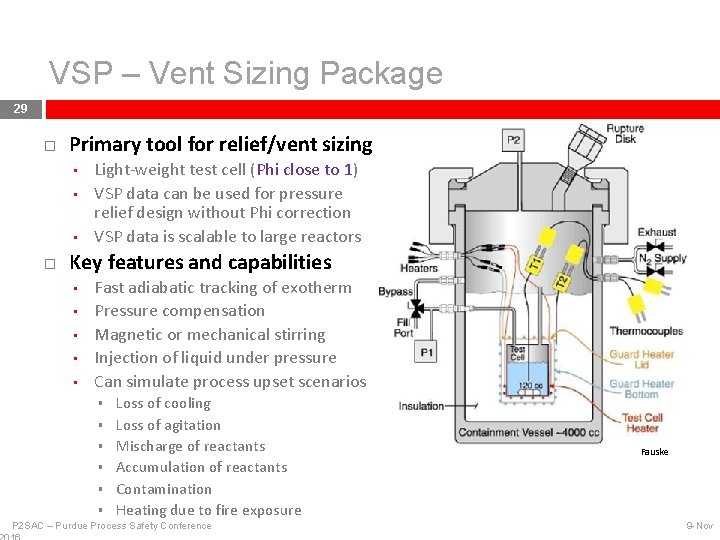
VSP – Vent Sizing Package 29 Primary tool for relief/vent sizing • • • Light-weight test cell (Phi close to 1) VSP data can be used for pressure relief design without Phi correction VSP data is scalable to large reactors Key features and capabilities • • • Fast adiabatic tracking of exotherm Pressure compensation Magnetic or mechanical stirring Injection of liquid under pressure Can simulate process upset scenarios § § § Loss of cooling Loss of agitation Mischarge of reactants Accumulation of reactants Contamination Heating due to fire exposure P 2 SAC – Purdue Process Safety Conference Fauske 9 -Nov
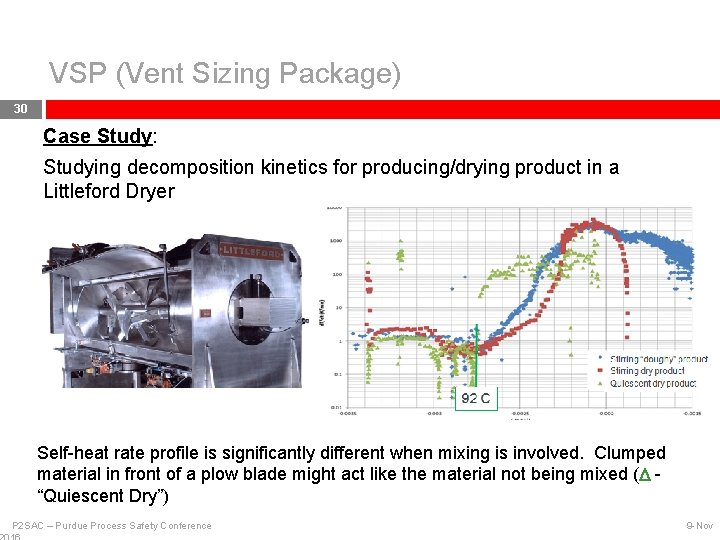
VSP (Vent Sizing Package) 30 Case Study: Studying decomposition kinetics for producing/drying product in a Littleford Dryer Self-heat rate profile is significantly different when mixing is involved. Clumped material in front of a plow blade might act like the material not being mixed (D “Quiescent Dry”) P 2 SAC – Purdue Process Safety Conference 9 -Nov

Flammability and Dust Explosion Testing 31 Flammability testing • Flash point (FP) • Flammable limits (LFL and UFL) • Limiting oxygen concentration (LOC) • Autoignition temperature (AIT) • Electrical conductivity Dust explosion testing* • Particle size distribution (PSD) • Minimum ignition energy (MIE) • Dust explosion severity (Kst/Pmax) • Minimum ignition temperature of dust cloud (MAIT) and dust layer (MIT) • Minimum explosive concentration (MEC) *Internal and external labs P 2 SAC – Purdue Process Safety Conference MIKE 3 20 Liter Sphere 9 -Nov

Energetic Materials Testing 32 Explosivity, UN/DOT classification • Internal testing capabilities • • At Dow, we occasionally encounter chemicals in our processes that contain high energy functional groups and/or may have explosive properties Thermal stability (DSC) Impact sensitivity (drop weight test) Work with external labs on UN/DOT explosivity testing for transportation classification • • Friction sensitivity Card gap tests Koenen tube tests Time/pressure tests BAM Friction Apparatus P 2 SAC – Purdue Process Safety Conference UN: United Nations; DOT: US Department of Transportation BAM Fallhammer 9 -Nov
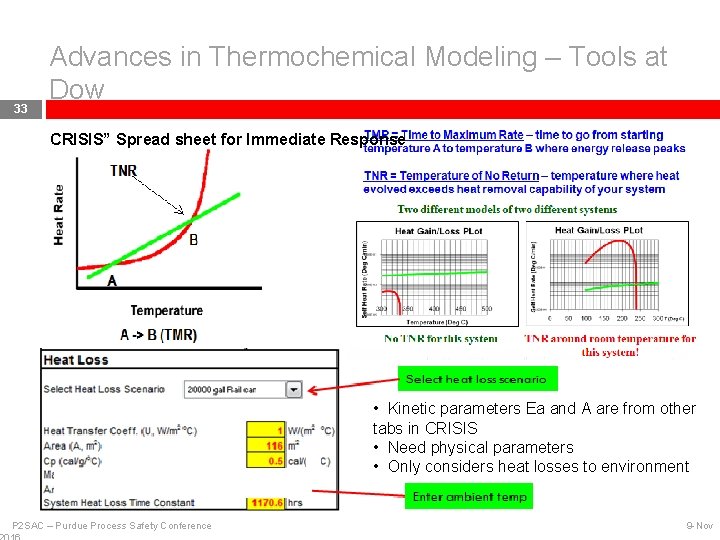
33 Advances in Thermochemical Modeling – Tools at Dow “CRISIS” Spread sheet for Immediate Response • Kinetic parameters Ea and A are from other tabs in CRISIS • Need physical parameters • Only considers heat losses to environment P 2 SAC – Purdue Process Safety Conference 9 -Nov
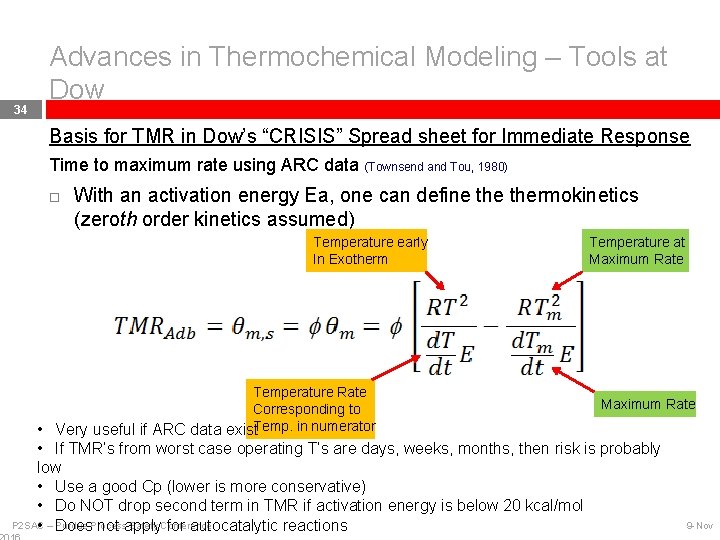
34 Advances in Thermochemical Modeling – Tools at Dow Basis for TMR in Dow’s “CRISIS” Spread sheet for Immediate Response Time to maximum rate using ARC data (Townsend and Tou, 1980) With an activation energy Ea, one can define thermokinetics (zeroth order kinetics assumed) Temperature early In Exotherm Temperature Rate Corresponding to • Very useful if ARC data exist. Temp. in numerator Temperature at Maximum Rate • If TMR’s from worst case operating T’s are days, weeks, months, then risk is probably low • Use a good Cp (lower is more conservative) • Do NOT drop second term in TMR if activation energy is below 20 kcal/mol P 2 SAC Safety Conference • – Purdue Does. Process not apply for autocatalytic reactions 9 -Nov

35 Advances in Thermochemical Modeling – Tools at Dow “CRISIS” Spread sheet for Immediate Response: Temperature Limit from D Estimates the adiabatic Time to Maximum Rate (TMR) using dynamic DSC experimental data Detected onset temperature (T) defines a data point of heat rate based on the detection limit of the DSC (45 µW/g) Assuming a reasonable activation energy (Ea) and heat capacity (Cp), one can estimate the temperature limit (T) for a desired TMR, or for a TMR at T, assuming zeroth order kinetics. Very useful at early stages (R&D) Use a good Cp (lower is more conservative) Use a conservative Ea (50 k. J/mol) If TMR’s from worst case operating T’s are days, weeks, months, then risk is probably low P 2 SAC – Purdue Process Safety Conference 9 -Nov

Example: Temperature Limit from ARC 36 At what temperature can a product be safely stored during a turnaround ? Normal residence times in the tank are <3 days. The turnaround will take about 7 days. Because the product is viscous, it is normally stored at a temperature of 450 C. • Available data: ARC Detected onset Temp: 124 o. C @ 0. 03 o. C/min • PHI factor: 1. 91 Temp Limit is 670 C at TMR(ad) = 10 days (240 hrs) P 2 SAC – Purdue Process Safety Conference 9 -Nov

Summary and Conclusions 37 Dow continues to be a strong leader in Process Safety and Reactive Chemicals to advance best practices and develop new technologies for identifying and managing process hazard risks. This is demonstrated by the diversity of innovative test methods and modeling approaches which Dow employs to gather critical information to quantity potential hazards for our operations and help to determine that we have adequate protection to prevent incidents and to mitigate the consequences. As part of our commitment to Responsible Care, we have also continued to leverage key aspects of Dow’s Reactive Chemicals Program, including hazard review protocols. Our intent is that by sharing Dow’s practices for identification, assessment and management of reactive chemicals hazards, the chemical process industries can realize benefits from improved reliability of operations, fewer incidents over the life of a process, and reduced consequences of any incidents that may occur. Thank you! Questions ? P 2 SAC – Purdue Process Safety Conference 9 -Nov
References ASME, 2009 ASME Code UG-140, Pressure Vessels with Overpressure Protection By System Design, 2007 ASME Boiler and Pressure Vessel Code, Section VIII Division 1. 2009 b Addenda (June 2009). CCPS, 1995 “Guidelines for Chemical Reactivity Evaluation and Application to Process Design”, Center for Chemical Process Safety. New York, NY: American Institution of Chemical Engineers (1995). Domalski et al. , 1993 E. S. Domalski and E. D. Hearing, J. Phys. Chem. Ref. Data, 22(4) (1993), pp. 816 -829 Bodman and Chervin, 2004 G. T. Bodman, S. Chervin, “Use of ARC in Screening for Explosive Properties “, J. Haz. Mat. , 115(1 -3) (2004), pp. 101105. CCPS, 2008 “Guidelines for Hazard Evaluation Procedures”, 3 rd ed. , Center for Chemical Process Safety, New Jersey: John Wiley & Sons, Inc. (2008). Dow’s Reactive Chemicals Program is provided on CD. Duh et al. , 1996 Yih-Shing Duh, Chang-Chia Hsu, Chen-Shan Kao, Shuh Woei Yu, “Applications of reaction calorimetry in reaction kinetics and thermal hazard evaluation” Therm. Acta, 285(1) (1996) pp. 67 -79 Britton, 2002 L. G. Britton, “Using Heats of Oxidation to Evaluate Flammability Hazards “, Proc. Saf. Prog. , 21 (2002), pp. 31– 54. CCPS, 2010 “Guidelines for Safe Handling of Powders and Bulk Solids”, Center for Chemical Process Safety, New Jersey: John Wiley & Sons (2010). See section 4. 3. 3 – Laboratory Test Methods for Chemical Incompatibility Hazards. FAI, 2002 FAI VSP 2 Manual and Methodology , Fauske & Associates, Inc. , Burr Ridge, Illinois, USA (2002) Britton and Frurip, 2003 L. G. Britton, D. J. Frurip “Further Uses of the Heat of Oxidation in Chemical Hazard Assessment “, Proc. Saf. Prog. 22 (2003), pp. 1– 19 CCPS, 2016 D. Gorman, J. Farr, A. Hielscher, T. Nguyen, G. Baran, B. Drake, E. Ford, D. Frurip, K. Mulligan, J. Ryan, D. Sliva, D. Viveros, “Chemical Reactivity Worksheet Version 4 (CRW 4)“, Center for Chemical Process Safety at http: //www. aiche. org/ccps/resources/chemicalreactivity-worksheet-40 Fisher et al. , 1995 H. G. Fisher (1992). Emergency relief system design using DIERS technology: The Design Institute for Emergency Relief Systems (DIERS) project manual. New York, NY: Design Inst. for Emergency Relief Systems Britton and Harrison, 2014 L. G. Britton, B. K. Harrison, “Using CHETAH to Estimate Lower Flammable Limit, Minimum Ignition Energy, and other Flammability Parameters”, Proc. Saf. Prog. , 33 (2014), pp. 314– 328 CEFIC, 2012 Bulk Storage Guideline of Hydrogen Peroxide, CEFIC, March 2012 Frurip et al. , 1995 D. J. Frurip, A. Chakrabarti, J. R. Dowey, H. D. Ferguson, S. K. Gupta, T. C. Hofelich, M. S. La. Barge, A. J. Pasztor Jr. , L. M. Peerey, S. M. Eveland, R. A. Suckow (1995). “Determination of Chemical Process Heats by Experiment and Prediction. ” United States: American Institute of Chemical Engineers, New York, NY United States). Chervin and Bodman, 2003 S. Chervin and G. T. Bodman , “Method for Estimating Decomposition Characteristics of Energetic Chemicals “, Proc. Saf. Prog. , 22 (2003), pp. 241– 243 Frurip et al, 1997 D. J. Frurip, T. C. Hofelich, D. Leggett, J. Kurland, J. Niemeir, “A Review of Chemical Compatibility Issues”, Proceedings AICh. E Prevention Symposium (1997) Chakrabarti, 1995 A. Chakrabarti, “International symposium on runaway reactions and pressure relief design, ” Boston, MA (United States), 2 -4 Aug 1995. (1995). Experimental approaches to testing in VSP for Process different types of Conference reactions for P 2 SACthe – Purdue Safety emergency relief design by simplified equations. 9 -Nov
References Frurip, 2004 D. J. Frurip, “The role of ASTEM E 27 methods in hazard assessment: Part I”, Proc. Saf. Prog. , 23 (2004), p. 266 Grossel, 2008 S. S. Grossel, “Guidelines for Hazard Evaluation Procedures, 3 rd ed. , ” AICh. E Center for Chemical Process Safety, Hoboken, NJ: John Wiley & Sons, Inc. (2008) Keller et al. , 1997 Keller, A. ; Stark, D. ; Fierz, H. ; Heinzle, E. ; Hungerbuhler, K. , “Estimation of the Time To Maximum Rate Using Dynamic DSC Experiments “, J. Loss Prev. Proc. Ind. , 10(1) (1997), pp. 3141. Frurip, 2008 D. J. Frurip, “Selection of the Proper Calorimetric Test Strategy in Reactive Chemicals Hazard Evaluation”, Org. Proc. Res. & Dev. , 12(6) (2008) pp. 1287 -1292. Harrison, 2009 B. K. Harrison , “CHETAH 9. 0 Users Manual, ” The ASTM Computer Program for Chemical Thermodynamics and Energy Release Evaluation. West Conshohocken, PA: ASTM Kossoy and Koludarova, 1995 A. Kossoy, E. Koludarova, “Specific Features of Kinetics Evaluation in Calorimetric Studies of Runaway Reactions”, J. Loss Prev. Proc. Ind. , 8(4) (1995), pp. 229– 235 Frurip et al. , 2009 D. J. Frurip, K. Bakkum, H. Johnstone, A. Tesolin-Gee, A. ; B. Vallieu, K. van Gelder, “Reactive Chemicals Emergency Response and Post-event Calorimetric Testing”, Proc. Saf. Prog. , 29(1) (2009), pp. 2 -10 Hofelich et al. , 1994 T. C. Hofelich, J. B. Powers, D. J. Frurip, “The Determination of Compatibility via Thermal Analysis and Mathematical Modeling”, Proc. Saf. Prog. , 13(4) (1994), pp. 227 -233 Kossoy and Benin, 1997. A. Kossoy, A. Benin Developments in the field of methodology and software for thermal hazards assessment, Proceedings of 21 st Meeting of DIERS Users Group of the AICh. E, Pittsburgh, Pennsylvania, USA (1997). Frurip, 2012 D. J. Frurip, “The Essential Elements of a Successful Reactive Chemicals Program”, Chem. Eng. Trans. 26, (2012), pp. 1 -6. DOI: 10. 3303/CET 1226001 Hofelich et al. , 2001 T. C. Hofelich, M. S. La. Barge, D. A. Drott, “Prevention of thermal runaways in carbon beds”, J. Loss Prev. in the Proc. Ind. , 12(6) (1999), pp. 517523 Kossoy et al. , 2001 A. Kossoy, A. Benin, Yu Akhmetshin, From Experimental Data via Kinetic Model to Predicting Reactivity and Assessing Reaction Hazards (methodology and software), Proceedings of Mary Kay O’Connor Process Safety Center 2001 Annual Symposium, Beyond Regulatory Compliance, Making Safety Second Nature, College Station, Texas, USA, Oct. 2001, pp. 321– 335. Frurip and Elwell, 2007 D. J. Frurip and T. I. Elwell, “Effective use of Differential Scanning Calorimetry in Reactive Chemicals Hazard Evaluation, ” Proc. Saf. Prog, 26(1), (2007) pp. 51 Hofelich and La. Barge, 2002 T. C. Hofelich and M. S. La. Barge, “On the Use and Misuse of Detected Onsets Temperature of Calorimetric Experiments for Reactive Chemicals”, J. Loss. Prev. Proc. Ind. , 15(3) (2002) pp 163 -168 Kossoy and Benin, 2003 A. Kossoy, A. Benin, “Further Development of the CISP Thermal Safety Software: New Features”, Proceedings of the Annual Meeting of European DIERS User's Group (EDUG), April 7 -8, Sent Etienne (2003) Johnson et al. , 2003 R. W. Johnson, S. W. Rudy and S. D. Unwin, “Essential Practices for Managing Chemical Reactivity Hazards. “ New York, NY: American Institute of Chemical Engineers (2003). See section 4. 4 and table 4. 8. Kossoy and Hofelich, 2003 A. Kossoy, T. Hofelich, “Methodology and software for reactivity rating”, Proc. Saf. Prog. , 22 (4) (2003), pp. 235 -240 Gorman et al. , 2014 D. Gorman, J. Farr, R. Bellair, W. Freeman, D. Frurip, A. Hielscher, H. Johnstone, M. Linke, P. Murphy, M. Sheng, K. van Gelder, D. P 2 SACViveros – Purdue Process Safety Conference 9 -Nov
References Kossoy and Sheinman, 2004 A. A. Kossoy, I. Y. Sheinman, “Evaluating Thermal Explosion Hazard by Using Kinetics-Based Simulation Approach”, Process Safety and Environmental Protection, 82(6) (2004), pp. 421 -430 Oxley et al. , 1999 J. C. Oxley, J. L. Smith, E. Rogers, M. A. Hamad, E. Resende, “Small Scale Explosivity Testing”, Journal of Energetic Materials, 17(4) (1999), pp. 331343. Wagman et al. , 1982 D. D. Wagman, W. H. Evans, V. B. Parker, R. H. Schumm, I. Halow, S. M. Bailey, K. L. Churney, and R. L. Nuttall, “The NBS Tables of Chemical and Thermodynamic Properties”, J. Phys. Chem. Ref. Data, Supplement No. 2, 11 (1982) Kossoy and Akhmetshin, 2007 A. Kossoy, Yu Akhmetshin, “Identification of Kinetic Models for the Assessment of Reaction Hazards”, Proc. Saf. Prog. , 26 (3) (2007), pp. 209– 220 Oxley et al. , 2010 J. C. Oxley, J. L. Smith, K. Marimaganti, “Developing Small-Scale Tests to Predict Explosivity”, J Therm. Analysis and Calorimetry, 102(2) (2010) pp. 597 -603. Whiting, 1988 L. F. Whiting, “Evaluation of a Capillary Tube Sample Container for Differential Scanning Calorimetry”, Thermochim. Acta, 136 (1988), p. 231 Leggett, 2001 David J Leggett, “Safe Process Development from Reaction Hazards Testing”, Thermochimica Acta, 367 -368, (2001), pp. 351 -365 Singh and Simms, 2001 J. Singh, C. Simms , “The Thermal Screening Unit (Tsu) - A Tool for Reactive Chemical Screening. In Symposium Series No. 148, Hazards XVI: Analysing the Past, Planning the Future”, Symposium Proceeding (pp. 67 -79). Melbourne, Australia: Institution of Chemical Engineers Whiting, 1997 L. Whiting, M. La. Barge, J. Steinmetzer, “Global Technical Information Management in thermal Hazard Evaluation”, J. Thermal Analysis, 49 (1997), pp. 1617 -1624 Mannan et al. , 2001 M. S. Mannan, W. J. Rogers, A. Aldeeb, “A Systematic Approach to Reactive Chemicals Analysis In Symposium Series No. 148, Hazards XVI: Analysing the Past, Planning the Future”, Symposium Proceeding (pp. 41 -58). Melbourne, Australia: Institution of Chemical Engineers (2001) Stack, 2010 Robert J. Stack, “High Integrity Protection System Design Using a Risk Base Approach”, 2010 Spring Meeting and 6 th Global Congress on Process Safety Conference Proceedings, San Antonio, TX: AICh. E (2010) Yoshida et al. , 1987 T. Yoshida, F. Yoshizawa, M. Itoh, T. Matsunaga, M. Watanabe, M. Tamura, “Prediction of Fire and Explosion Hazards of Reactive Chemicals (I). Estimation of Explosive Properties of Self-Reactive Chemicals from SC-DSC Data”, Kogyo Kayaku, 48(5) (1987), pp. 311 -316. Mc. Intosh and Nolan, 2001 R. D. Mc. Intosh, P. F. Nolan, “Review of the selection and design of mitigation systems for runaway chemical reactions “, J. Loss Prev. Proc. Ind. 14 (2001), pp. 27– 42 Tipler et al. , 2013 S. Tipler, D. Eure, K. First, R. J. Stack, T. J. Wagner, J. W. Champion, “Practical Examples of Alternate Overpressure Protection Systems: An Owner’s Perspective”, 2013 Spring Meeting and 9 th Global Congress on Process Safety Conference Proceedings, San Antonio, TX: AICh. E (2013) Melhem et al. , 1995 G. A. Melhem, H. G. Fisher, D. A. Shaw, “An Advanced Method for the Estimation P 2 SAC Purdue. Kinetics, Process. Scaleup, Safety and Conference of – Reaction Pressure Relief Townsend and Tou, 1980 D. I. Townsend, J. C. Tou , “Thermal Hazard Evaluation by an Accelerating Rate Calorimeter”, Thermochim. Acta, 37 (1980), pp. 1– 30. 9 -Nov
- Slides: 40