Advanced Transport Phenomena Energy Transport Module 5 Lecture


























- Slides: 28

Advanced Transport Phenomena Energy Transport Module 5 - Lecture 18 Dr. R. Nagarajan Professor Dept of Chemical Engineering IIT Madras

ENERGY TRANSPORT

MECHANISMS Ø Convection Ø Diffusion (heat conduction) Ø Radiation Ø First 2 analogous to momentum & mass transport Ø Third is unique Ø All participate in most high-temperature, chemically reacting systems Ø Radiation especially important in large systems (e. g. , forest fires) and at high pressures (rocket propulsion)

CONVECTION Ø A material in motion transports energy by convection Ø Moving solids transport energy by convection Ø Conventional view: solids at rest, convection occurs in fluids Ø Local enthalpy flux by convection = Ø local mass-flux Ø h enthalpy per unit mass

FORCED AND FREE CONVECTION Ø When fluid motion is mechanically produced forced convection Ø When energy or mass transfer lead to local density non- uniformities free (natural) convection Both can operate simultaneously

DIFFUSION Ø In solids & liquids by propagation of molecular vibrations Ø In gases by motion of constituent molecules Ø Governed by Fourier heat flux law: Ø k local thermal conductivity of material Ø Time-averaged effect of turbulent eddy convection net energy flux, “eddy diffusion” Ø aeff = a + at

RADIATION Ø Energy transport through empty space Ø Propagation of electromagnetic waves, or flow of photons Ø Latter view useful: analogies between behavior of photons & material molecules Ø “photon phase” interacting with “material phase” Ø Photons require matter for emission, scattering & absorption

RADIATION FEATURES Ø All matter at T > 0 K emits a distribution of photons. Ø Rate & distribution strongly T-dependent Ø Distance a photon travels before absorption/ scattering in normal engineering applications can be comparable to system dimensions Ø Radiation cannot be treated as a diffusion process Ø Photons often in free-molecular regime even in continuum flows Ø In the simplest cases, can be entirely decoupled

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Temperature profiles & isotherms in a flow field can be mapped by: Ø Immersion methods (e. g. , small thermocouples) Ø Remote or non-intrusive methods (e. g. , spectroscopy) Ø Energy fluxes ( ) are measured only at important boundary surfaces using local heat-flux gages Ø Overall heat transfer rate for total surface area Aw can be measured as:

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Reference heat flux that would diffuse across a quiescent fluid gap of dimension L subjected to same overall temperature difference (Tw – T∞ ) Ø Ratio of prevailing to reference energy flux (dimensionless heat transfer coefficient, Nusselt number for heat transfer)

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Average heat flux per unit temperature difference: Ø Sometimes written as Ø Units must be stated

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø If actual average energy flux is viewed as result of pure diffusion across a quiescent fluid film, thickness of this “equivalent stagnant film” Ø Hence Ø Ratio of resistance to energy diffusion across a fluid layer of thickness L to actual resistance to energy diffusion across prevailing thermal boundary layer

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Local dimensional heat transfer coefficient = Ø Local dimensionless heat transfer coefficient, Nuh:

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø For a solid surface in contact with a non-reacting gas, local normal component of energy flux in solid is same as that in adjacent gas; hence: n coordinate measured normal to surface fluid side

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Thermal Boundary layer near a curved solid surface

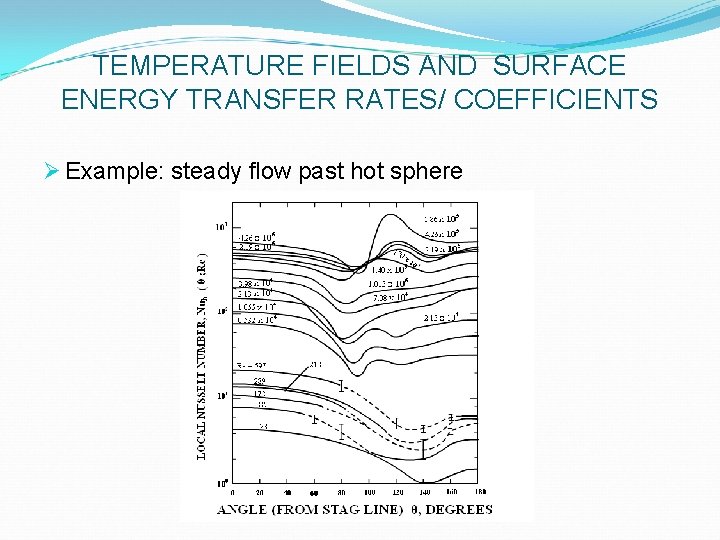
TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Axially symmetric flow of fluid of temperature T∞ Ø Isothermal hot sphere of diameter dw Ø Temperature field T(r, q) established as a result of conduction & convection in fluid-filled space r > (dw /2) Ø Local radial heat flux (in absence of radiation)

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Total heat-flow rate from solid to fluid = area-weighted sum of fluxes: Ø total rate of heat transfer needed to maintain a sphere at constant temperature Tw; hence:

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Local heat transfer coefficients Since: the two dimensionless htc’s are interrelated by:

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere

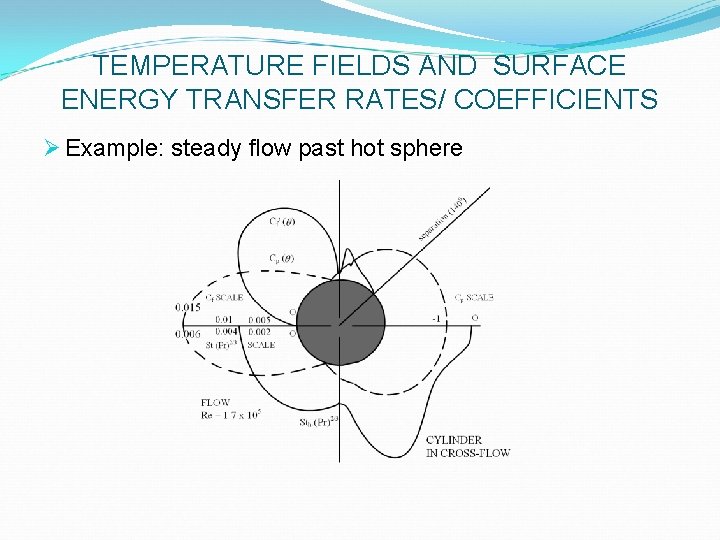
TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Measurements show increasing importance of rear (p/2 < q < p) of cylinder as Re increases Ø Surface-averaged htc’s, Nuh (Re, Pr), can be obtained by q–integration of the data Ø Or by direct measurements (e. g. , transient calorimetry) Ø Can be predicted (in simple cases) by analytical or numerical solution of conservation equations Ø More commonly, measured by full-scale or model experiments

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Transient Calorimetry: Ø Fluid-to-solid heat-transfer rate measured by following rate of change of temperature of a nearlyisothermal calorimeter suddenly immersed in a fluid Ø Macroscopic energy balance enveloping entire calorimeter): (applied to CV

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Vs volume of calorimeter Aw wetted area of calorimeter L its reference length (rc)s its heat capacity per unit volume Ts(t) recorded calorimeter temperature

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Transient Calorimetry: Ø Integrating: where Ø (effective time-constant for exponential decay of the fraction of initial temperature difference still remaining at time t)

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Transient Calorimetry: Ø (Vs/Aw)/L = shape-dependent pure number (1/6 for a sphere) Ø fluid thermal diffusivity, Ø th-1 can be estimated as slope of and can then be determined.

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Transient Calorimetry: Ø Representing entire calorimeter by a single temperature => temperature nonuniformities within calorimeter material (Cu or Al) negligible compared to those within wetting fluid film BL Ø Valid only if Biot number for energy transfer, Bih << 1, where: Ø Bih ratio of internal (calorimeter) resistance to external (fluid film) resistance to energy diffusion

TEMPERATURE FIELDS AND SURFACE ENERGY TRANSFER RATES/ COEFFICIENTS Ø Example: steady flow past hot sphere Ø Nuh can be considered quasi-steady if th >> fluid-film characteristic adjustment time Ø So, transient calorimetric measurements must be made under conditions such that Ø Easiest to satisfy in flowing gases since (rc)gas values at STP are smaller than for solids by 3 OM