Advanced Theories of Covalent Bonding Valence Bond Theory

Advanced Theories of Covalent Bonding • • Valence Bond Theory Hybridization of Atomic Orbitals Multiple Bonds Molecular Orbital Model

Valence Shell Electron Pair Repulsion (VSEPR) Theory • In molecules or polyatomic ions valence electrons exist in pairs; • Electron pairs repel each other: they are arranged in an orientation that gives minimum electron-pair repulsions; • Relative strength of electron-pair repulsions: Nonbonding pair repulsion > Nonbonding-bonding repulsion > bonding pair repulsion;

Determination of Molecular Shapes by the VSEPR Model • Molecular shapes determined by number and orientation of electron-pairs around central atom; • Shapes with minimum electron-pairs repulsion are preferred: Nonbonding pairs placed farthest apart from each other.
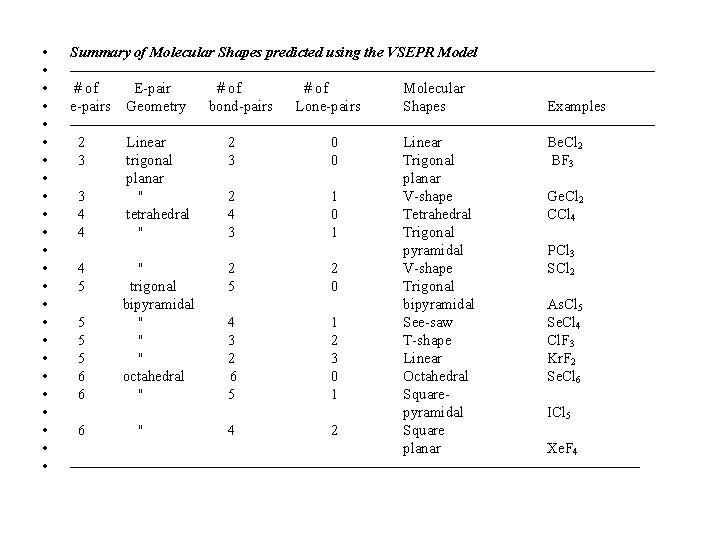
• • • • • • Summary of Molecular Shapes predicted using the VSEPR Model ———————————————————— # of E-pair # of Molecular e-pairs Geometry bond-pairs Lone-pairs Shapes Examples ———————————————————— 2 Linear 2 0 Linear Be. Cl 2 3 trigonal 3 0 Trigonal BF 3 planar 3 '' 2 1 V-shape Ge. Cl 2 4 tetrahedral 4 0 Tetrahedral CCl 4 4 '' 3 1 Trigonal pyramidal PCl 3 4 '' 2 2 V-shape SCl 2 5 trigonal 5 0 Trigonal bipyramidal As. Cl 5 5 '' 4 1 See-saw Se. Cl 4 5 '' 3 2 T-shape Cl. F 3 5 '' 2 3 Linear Kr. F 2 6 octahedral 6 0 Octahedral Se. Cl 6 6 '' 5 1 Squarepyramidal ICl 5 6 '' 4 2 Square planar Xe. F 4 ———————————————————
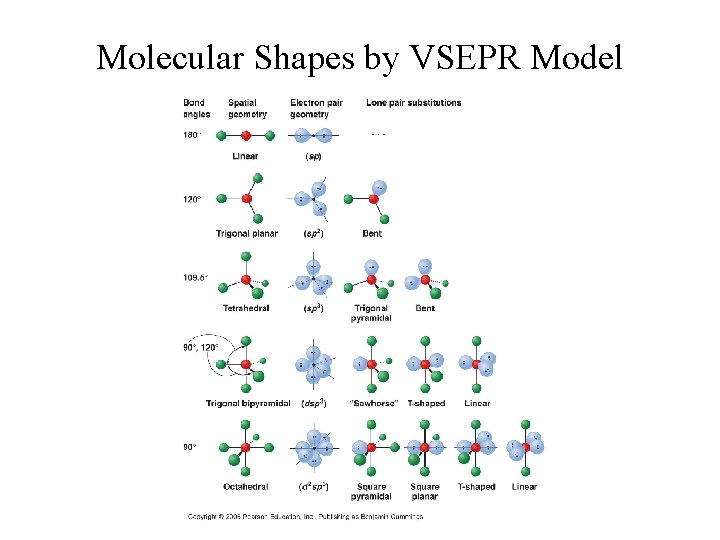
Molecular Shapes by VSEPR Model

How to Predict Polarity • Polarity of molecule depends on molecular shape and bond polarity. 1. Molecules (or polyatomic ions) with symmetrical shapes are non-polar; 2. Molecules with single lone-pairs on central atoms are generally polar;

Models for Chemical Bonding • Localized electron model (Valence bond method and orbital hybridization) • Molecular orbital Theory (Delocalized electron model)

Localized Electron Model • • Good for explaining molecular shapes; Two singly occupied orbitals from separate atoms form -overlaps. • Electron pairs occupy overlap area to form -bonds; • Two types of covalent bonds may be formed: 1. s -bond: orbitals overlap along internuclear axis 2. p -bond: side-to-side overlap of orbitals perpendicular to the internuclear axis

Orbital Hybridization 1. Hybridization describes the mixing of atomic orbitals on the central atom to account for the bond length, bond energy, and bond angle.
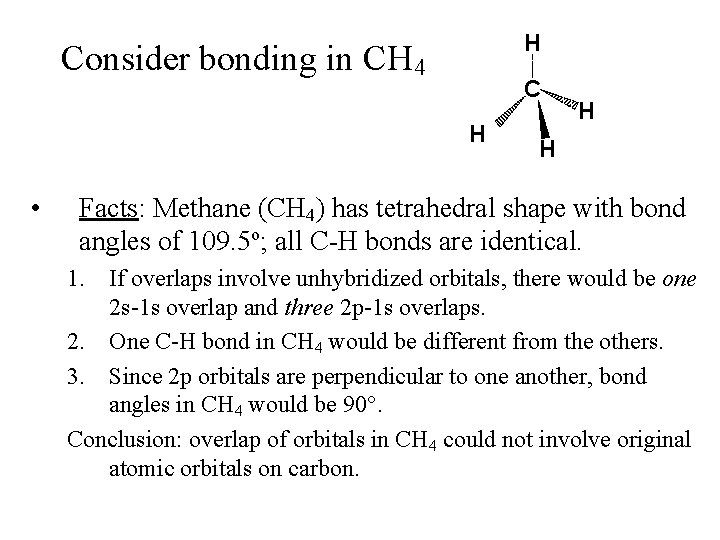
H Consider bonding in CH 4 C H • H H Facts: Methane (CH 4) has tetrahedral shape with bond angles of 109. 5 o; all C-H bonds are identical. 1. If overlaps involve unhybridized orbitals, there would be one 2 s-1 s overlap and three 2 p-1 s overlaps. 2. One C-H bond in CH 4 would be different from the others. 3. Since 2 p orbitals are perpendicular to one another, bond angles in CH 4 would be 90°. Conclusion: overlap of orbitals in CH 4 could not involve original atomic orbitals on carbon.
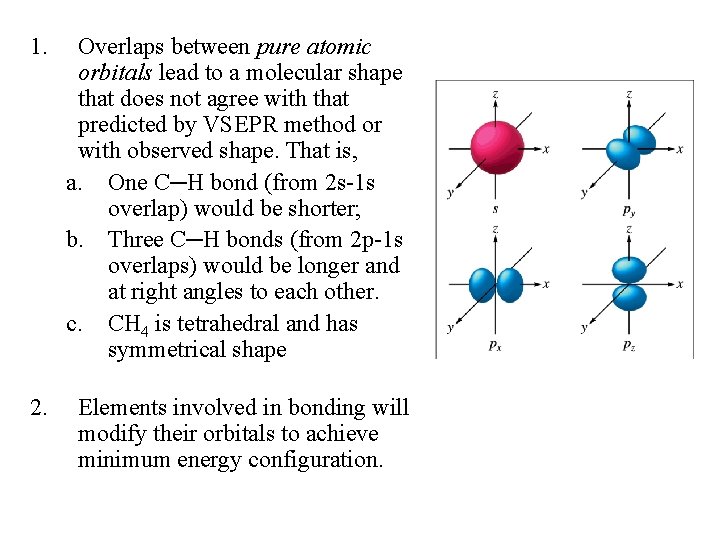
1. Overlaps between pure atomic orbitals lead to a molecular shape that does not agree with that predicted by VSEPR method or with observed shape. That is, a. One C─H bond (from 2 s-1 s overlap) would be shorter; b. Three C─H bonds (from 2 p-1 s overlaps) would be longer and at right angles to each other. c. CH 4 is tetrahedral and has symmetrical shape 2. Elements involved in bonding will modify their orbitals to achieve minimum energy configuration.

Hybridization of atomic orbitals in CH 4 • The 2 s and 2 p atomic orbitals in carbon mix to form 4 identical hybrid orbitals for bonding. Mixing of atomic orbitals is called hybridization. • Hybridization of one 2 s and three 2 p orbitals yields four sp 3 hybrid orbitals on the carbon atom. • Four sp 3 hybrid orbitals around a central atom yield a tetrahedral arrangement. • Each sp 3 hybrid orbital overlap with 1 s orbital of Hatom yields a tetrahedral molecule of CH 4.
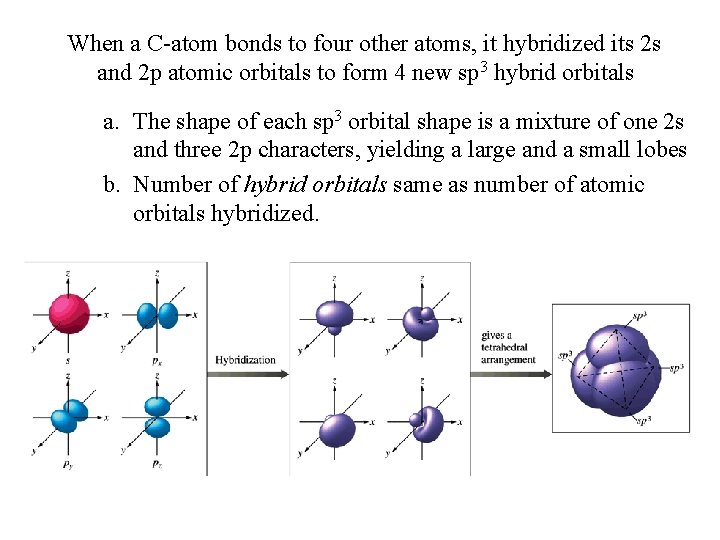
When a C-atom bonds to four other atoms, it hybridized its 2 s and 2 p atomic orbitals to form 4 new sp 3 hybrid orbitals a. The shape of each sp 3 orbital shape is a mixture of one 2 s and three 2 p characters, yielding a large and a small lobes b. Number of hybrid orbitals same as number of atomic orbitals hybridized.
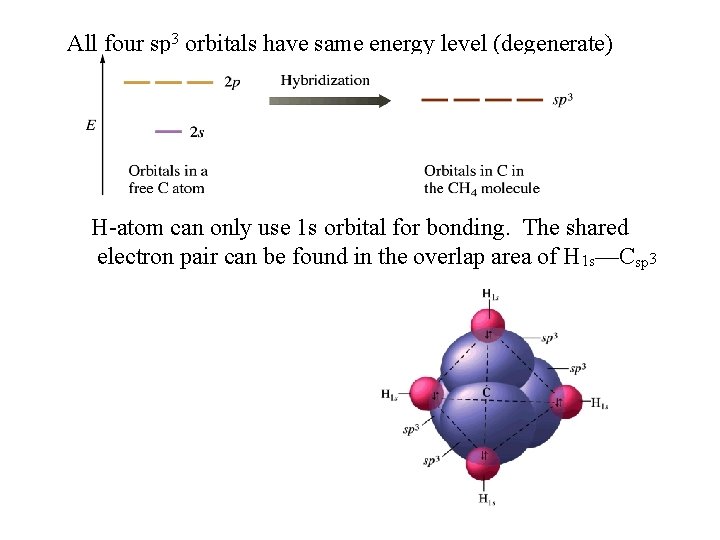
All four sp 3 orbitals have same energy level (degenerate) H-atom can only use 1 s orbital for bonding. The shared electron pair can be found in the overlap area of H 1 s—Csp 3
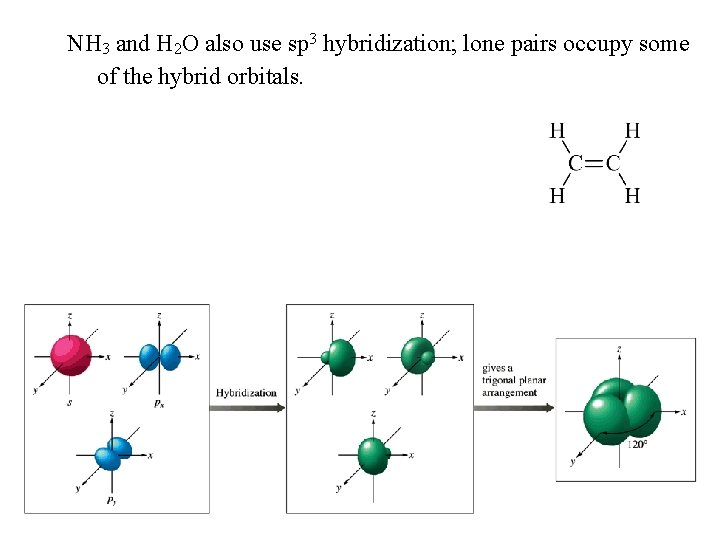
NH 3 and H 2 O also use sp 3 hybridization; lone pairs occupy some of the hybrid orbitals.

Orbitals Hybridization C. sp 2 Hybridization 1. C 2 H 4, ethylene is one example 2. Lewis and VSEPR structures tell us what to expect 3. H atoms still can only use 1 s orbitals 4. C atom hybridizes 2 s and two 2 p orbitals into 3 sp 2 hybrid orbitals

Formation of C=C Double Bond in Ethylene
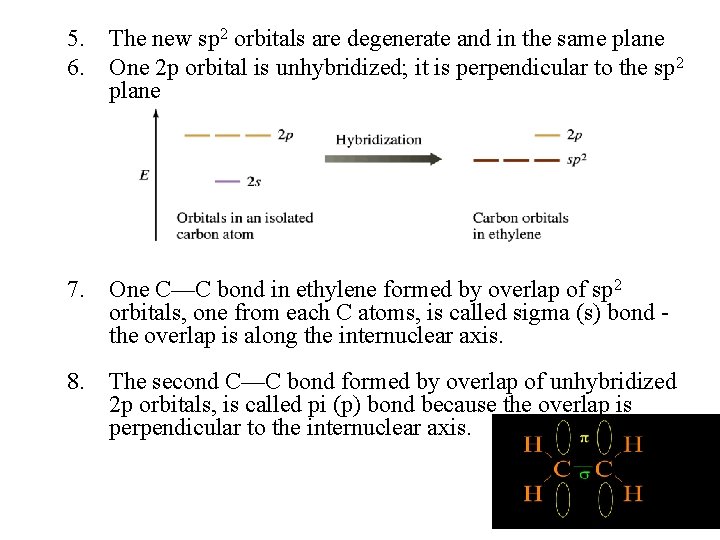
5. The new sp 2 orbitals are degenerate and in the same plane 6. One 2 p orbital is unhybridized; it is perpendicular to the sp 2 plane 7. One C—C bond in ethylene formed by overlap of sp 2 orbitals, one from each C atoms, is called sigma (s) bond the overlap is along the internuclear axis. 8. The second C—C bond formed by overlap of unhybridized 2 p orbitals, is called pi (p) bond because the overlap is perpendicular to the internuclear axis.
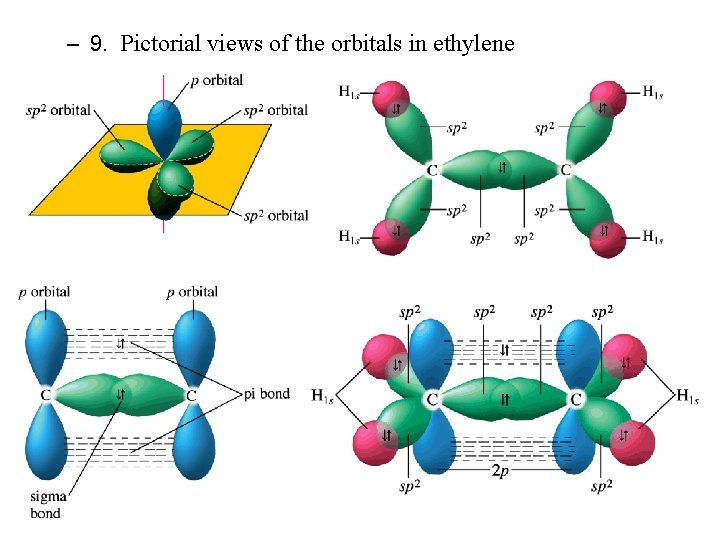
– 9. Pictorial views of the orbitals in ethylene
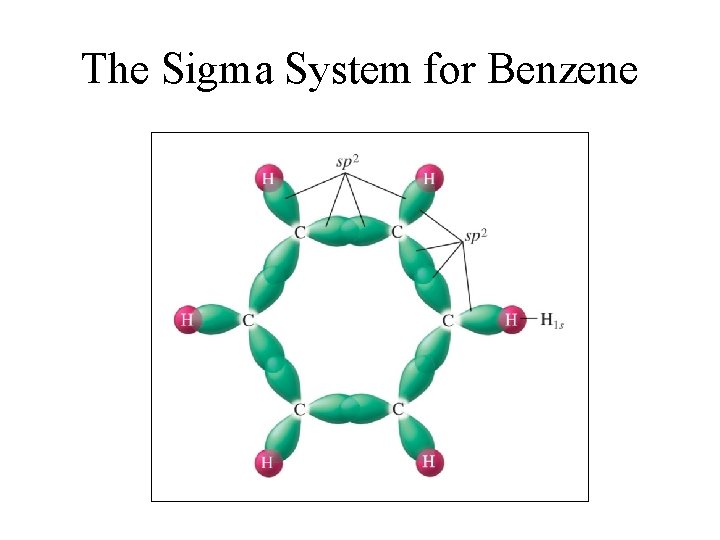
The Sigma System for Benzene
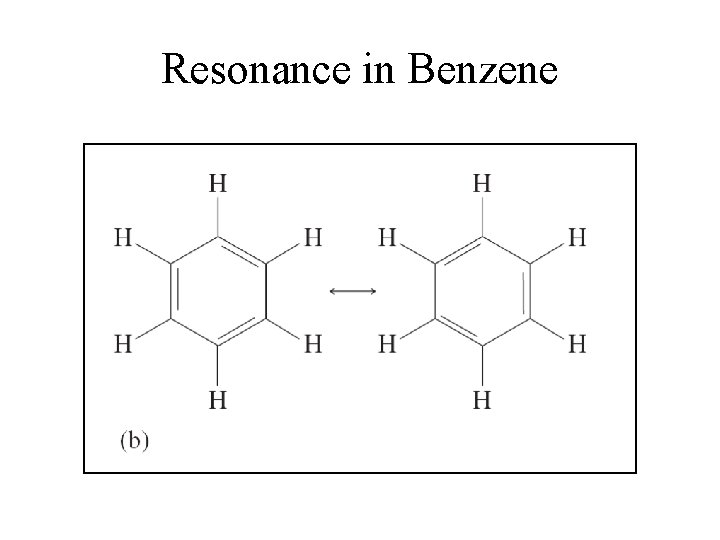
Resonance in Benzene

Delocalization • Describes molecules that require resonance. • In molecules that require resonance, it is the bonding that is most clearly delocalized, the bonds are localized. • p orbitals perpendicular to the plane of the molecule are used to form molecular orbitals. • The electrons in the molecular orbitals are delocalized above and below the plane of the molecule.
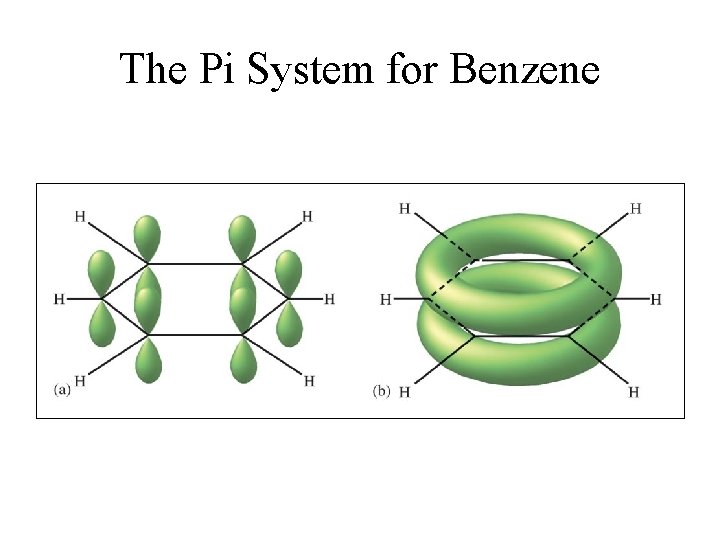
The Pi System for Benzene
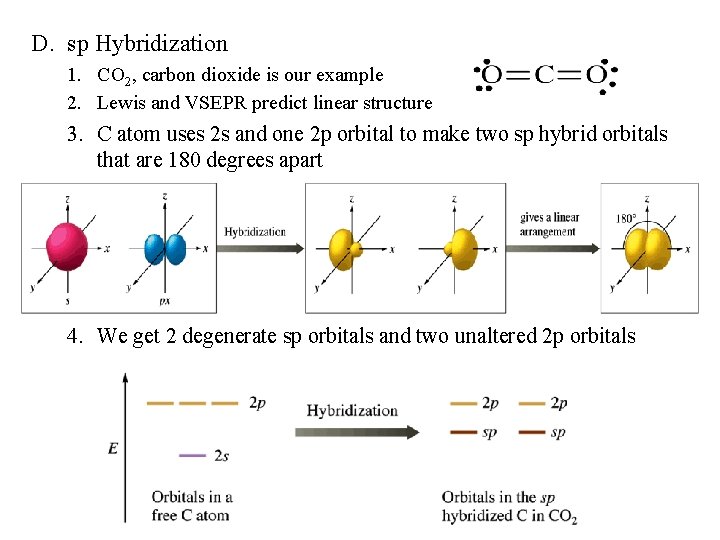
D. sp Hybridization 1. CO 2, carbon dioxide is our example 2. Lewis and VSEPR predict linear structure 3. C atom uses 2 s and one 2 p orbital to make two sp hybrid orbitals that are 180 degrees apart 4. We get 2 degenerate sp orbitals and two unaltered 2 p orbitals
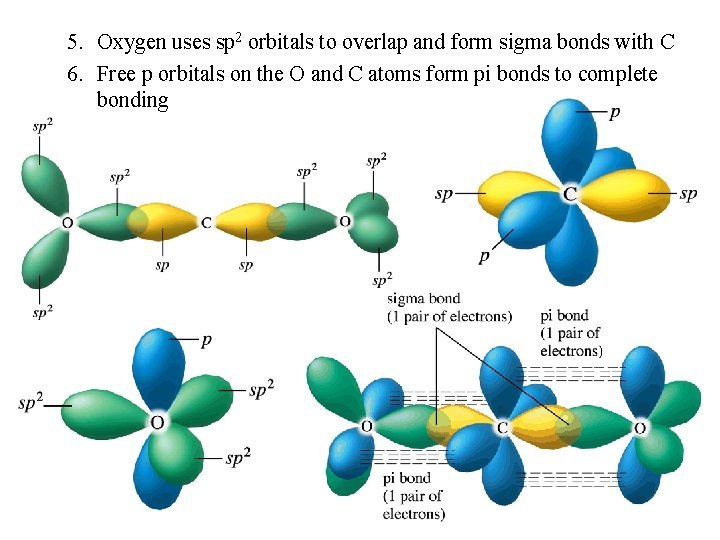
5. Oxygen uses sp 2 orbitals to overlap and form sigma bonds with C 6. Free p orbitals on the O and C atoms form pi bonds to complete bonding
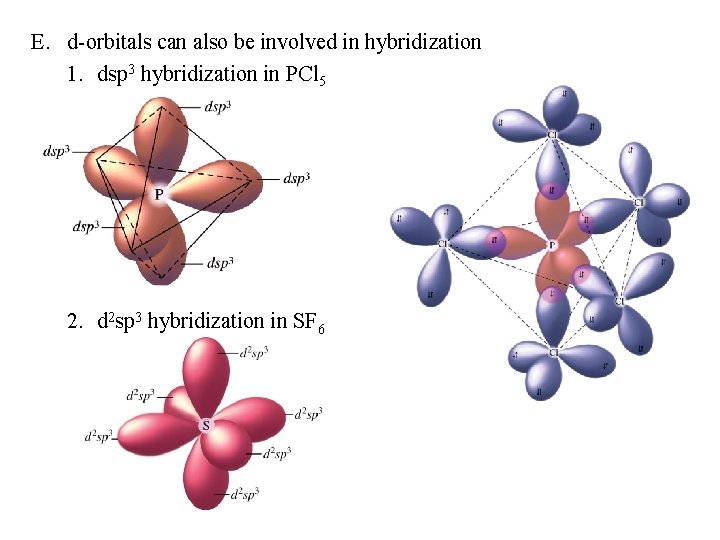
E. d-orbitals can also be involved in hybridization 1. dsp 3 hybridization in PCl 5 2. d 2 sp 3 hybridization in SF 6

Types of Hybridization • There are five different types of hybridization: 1. 2. 3. 4. 5. • sp - required by two pairs of electrons around an atom. sp 2 - required by three pairs of electrons around an atom. sp 3 - required by four pairs of electrons around an atom. dsp 3 - required by five pairs of electrons around an atom. d 2 sp 3 - required by six pairs of electrons around an atom. A bond acts as one effective pair of electrons whether it is a single, double, or triple bond.

Hybridization and the Localized Electron Model # Electron-pairs on Central Atom Arrangement Hybridization 2 linear sp 3 trigonal planar sp 2 4 tetrahedral sp 3 5 trigonal bipyramid dsp 3 or sp 3 d 6 octahedral d 2 sp 3 or sp 3 d 2
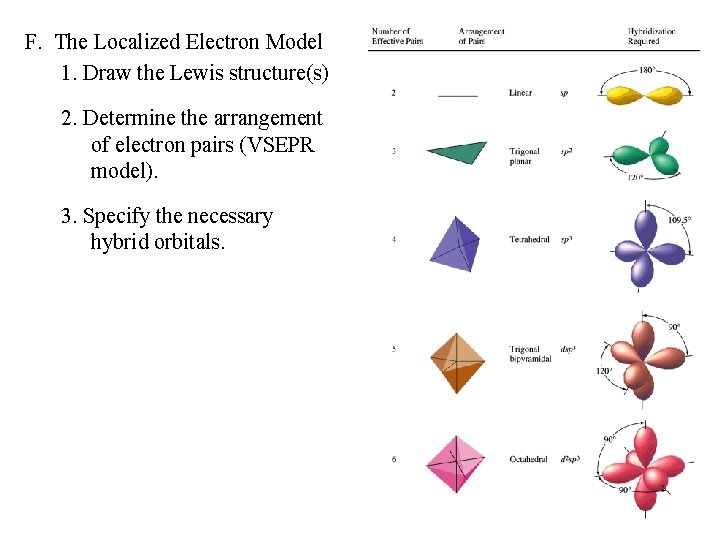
F. The Localized Electron Model 1. Draw the Lewis structure(s) 2. Determine the arrangement of electron pairs (VSEPR model). 3. Specify the necessary hybrid orbitals.

Exercise Draw the Lewis structure for PCl 5. § What is the shape of a phosphorus pentachloride molecule? trigonal bipyramidal § What are the bond angles? 90 o and 120 o

sp 3 d or dsp 3 Hybridization • Hybridization of a 3 s, three 3 p, and a 3 d orbitals yields five sp 3 d hybrid orbitals. • Five hybrid orbitals yield a trigonal bipyramidal arrangement. • If all five orbitals form covalent bonds, a molecule with trigonal bipyramidal shape is obtained.
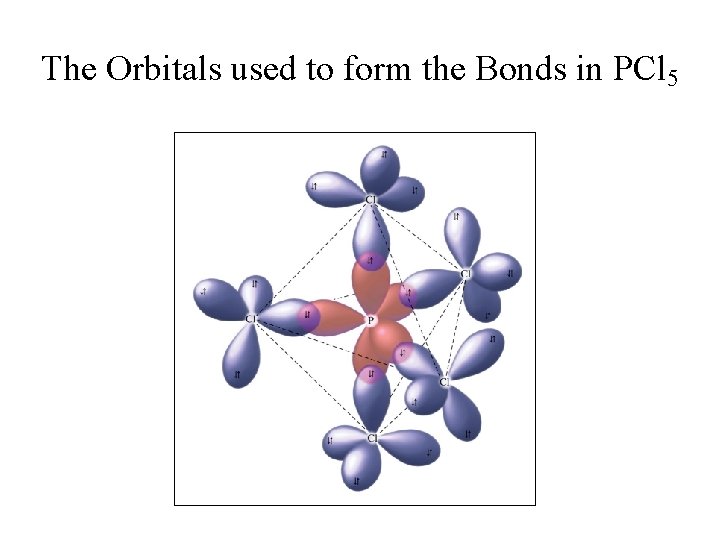
The Orbitals used to form the Bonds in PCl 5

Exercise Draw the Lewis structure for Xe. F 4. § What is the shape of a xenon tetrafluoride molecule? octahedral § What are the bond angles? 90 o and 180 o

sp 3 d 2 or d 2 sp 3 Hybridization • Combination of one s, three p, and two d orbitals yields six sp 3 d 2 hybrid orbitals; • Six equivalent sp 3 d 2 hybrid orbitals around a central tom gives an octahedral arrangement. • If all six orbitals form covalent bonds, an octahedral molecule is obtained.
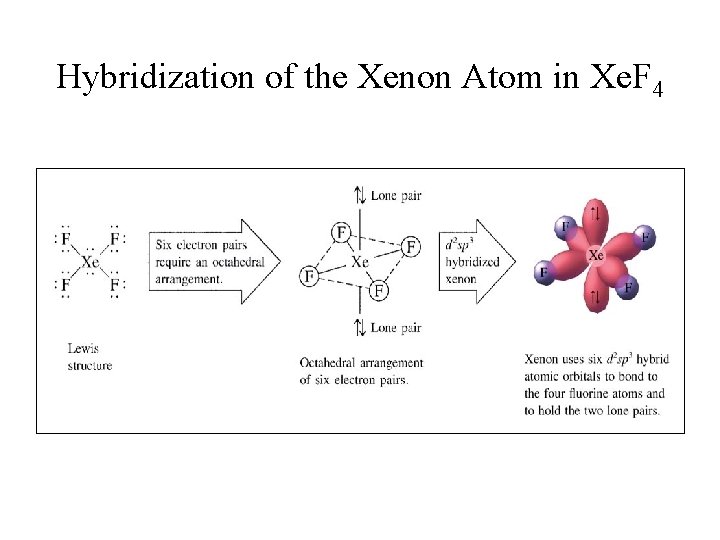
Hybridization of the Xenon Atom in Xe. F 4

Concept Check Draw the Lewis structure for HCN. Which hybrid orbitals are used? Draw HCN: § Showing all bonds between atoms. § Labeling each bond as or .

Concept Check Determine the bond angle and expected hybridization of the central atom for each of the following molecules: NH 3 SO 2 Kr. F 2 NH 3 – 109. 5 o, sp 3 SO 2 – 120 o, sp 2 Kr. F 2 – 90 o, 120 o, dsp 3 CO 2 – 180 o, sp ICl 5 – 90 o, 180 o, d 2 sp 3 CO 2 ICl 5

Using the Localized Electron Model • Draw the Lewis structure(s). • Determine the arrangement of electron pairs using the VSEPR model. • Specify the hybrid orbitals needed to accommodate the electron pairs.

Exercise #1 on Hybridization Describe the bonding in the water molecule using the localized electron model. O Solution H H • Since there are four electron pairs around oxygen atom, the oxygen is sp 3 hybridized. The water molecule has a bent structure (or V-shape)

Sigma ( ) and Pi ( ) Bonds • Bonds formed by end-to-end overlaps of orbitals along the orbital axes are called sigma (σ) bonds. • Bonds formed by sidewise overlaps between two parallel unhybridized p-orbitals are called pi (π) bonds. The overlaps of orbitals occur above and below the bond axis • All single bonds are σ-bonds. • A double bond consists of one σ bond and one π bond. • A triple bond is made up of one σ and two π bonds.

Exercise #2 on Sigma and Pi Bonds How many σ bonds are there in the commercial insecticide, “Sevin, ” shown below? How many π bonds? Solution There are 27 σ bonds. There are 6 π bonds.

Practice with Hybrid Orbitals Give the hybridization and predict the geometry of the central atom in the ion IF 2+. Lewis structure: F I F There are 4 effective electron pairs around the central atom making it sp 3 hybridized. The VSEPR structure has a tetrahedral basis but since it has two bonding pairs, it will take on a bent shape with a bond angle smaller than 109. 5°.
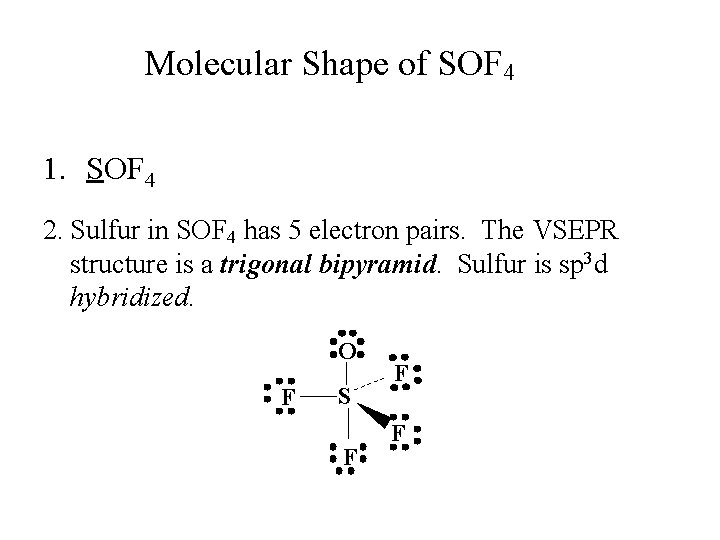
Molecular Shape of SOF 4 1. SOF 4 2. Sulfur in SOF 4 has 5 electron pairs. The VSEPR structure is a trigonal bipyramid. Sulfur is sp 3 d hybridized. O F S F F F

Hybridization and Structure of C 2 H 2 3. HC≡CH Each carbon has 2 effective electron pairs. The VSEPR structure is linear. Each carbon is sp hybridized. H C C H

The Molecular Orbital Model • The localized electron model does an excellent job in predicting and justifying molecular shapes. But it does not deal with molecules with unpaired electrons. It also neglects bond energies. • The molecular orbital (MO) model views electrons as belonging to the entire molecule rather than to individual atoms in the molecule. • MO theory allows us to get a clearer understanding of resonance.

Molecular Orbital Theory • Regards a molecule as a collection of nuclei and electrons, where the electrons are assumed to occupy orbitals that extend over the entire molecule. • Electrons fill molecular orbitals in the same manner they do the atomic orbitals. • The electrons are assumed to be delocalized rather than always located between a given pair of atoms.

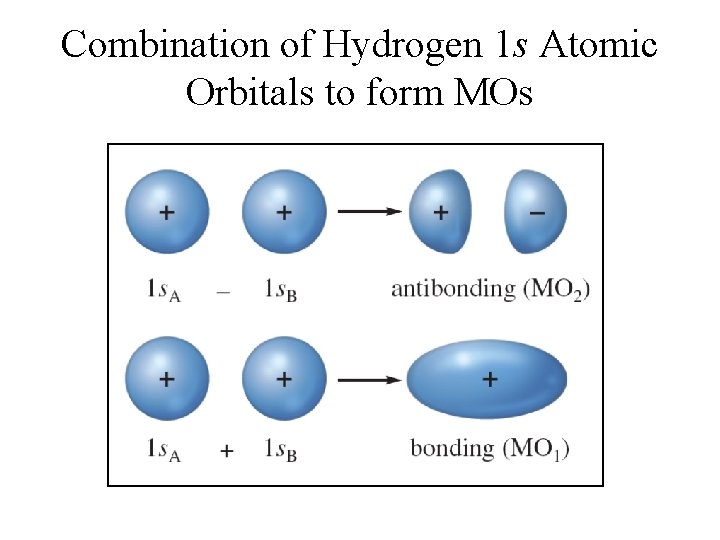
Electron Probability in MO • The electron probability of both molecular orbitals is centered along the line passing through the two nuclei. § Sigma (σ) molecular orbitals (MOs) • In the molecule only the molecular orbitals are available for occupation by electrons.

Key Ideas of the MO Model 1. All valence electrons in a molecule exist in a set of molecular orbitals of a given energy. 2. Valence orbitals of each atom are not acting independently, but rather interact as a whole to form a set of molecular orbitals. 3. Each pair of atomic orbitals interacts to form a set of bonding and antibonding molecular orbitals. 4. Energy for bonding molecular orbitals is lower than their atomic orbitals. 5. Antibonding molecular orbitals have higher energy that their atomic orbitals.
Combination of Hydrogen 1 s Atomic Orbitals to form MOs
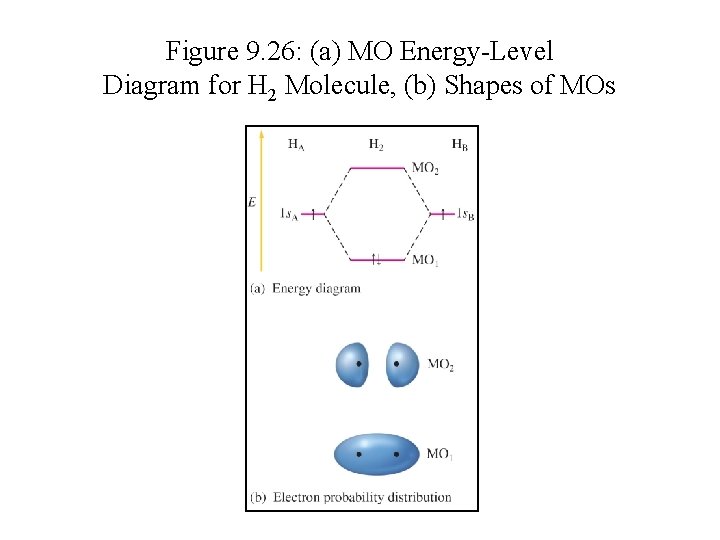
Figure 9. 26: (a) MO Energy-Level Diagram for H 2 Molecule, (b) Shapes of MOs
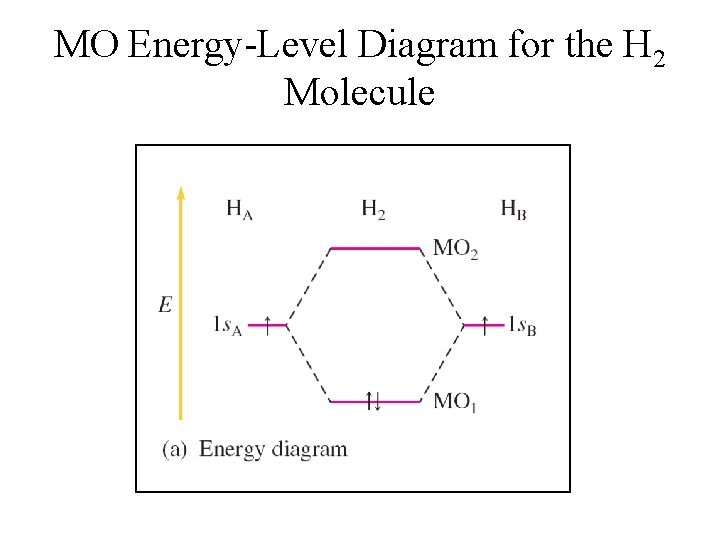
MO Energy-Level Diagram for the H 2 Molecule

Bond Order in MO Model BO = # bonding electrons - # antibonding electrons 2 1. Bond order (BO) is a measure of net bonding interactions. 2. BO must be greater than 0 for a stable molecule to form. 3. The higher the BO, the stronger the bond.
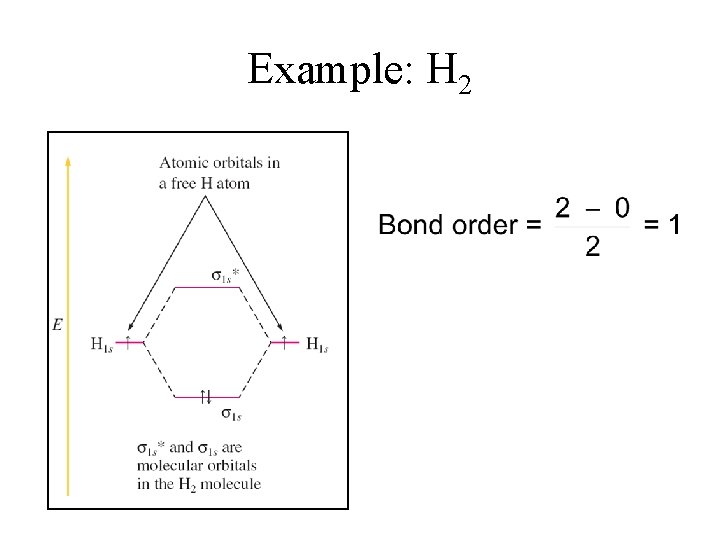
Example: H 2
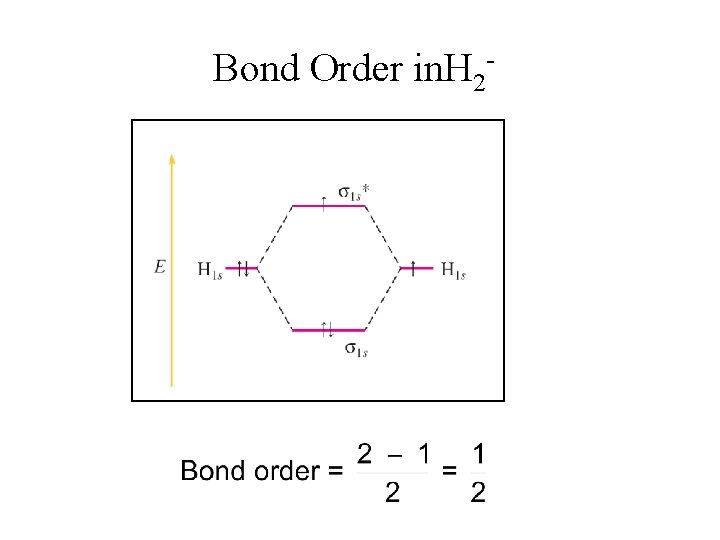
Bond Order in. H 2

Bond Order • Larger bond order means greater bond strength.

MO Energy-Level Diagram for He 2 Molecule
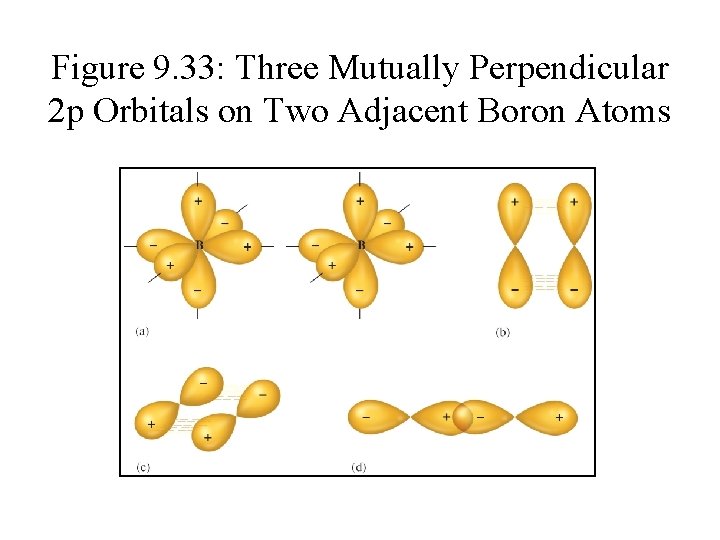
Figure 9. 33: Three Mutually Perpendicular 2 p Orbitals on Two Adjacent Boron Atoms
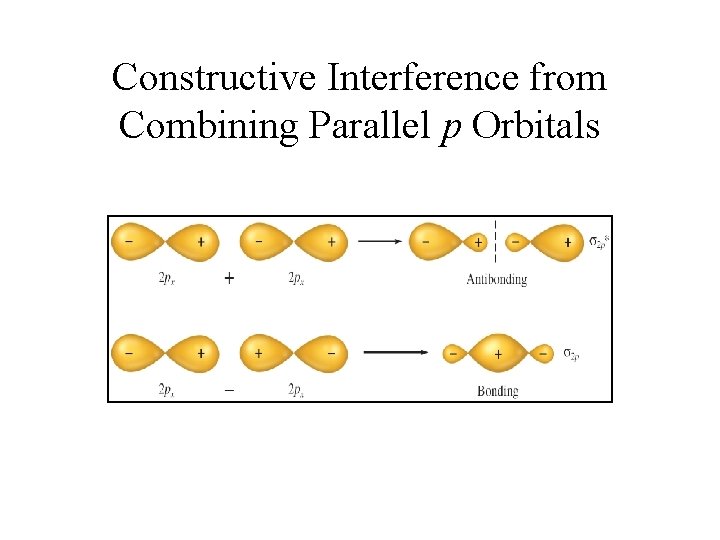
Constructive Interference from Combining Parallel p Orbitals
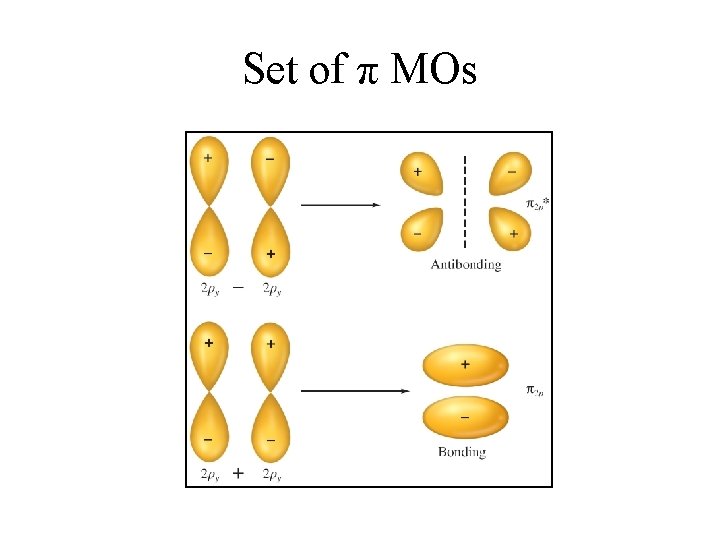
Set of π MOs
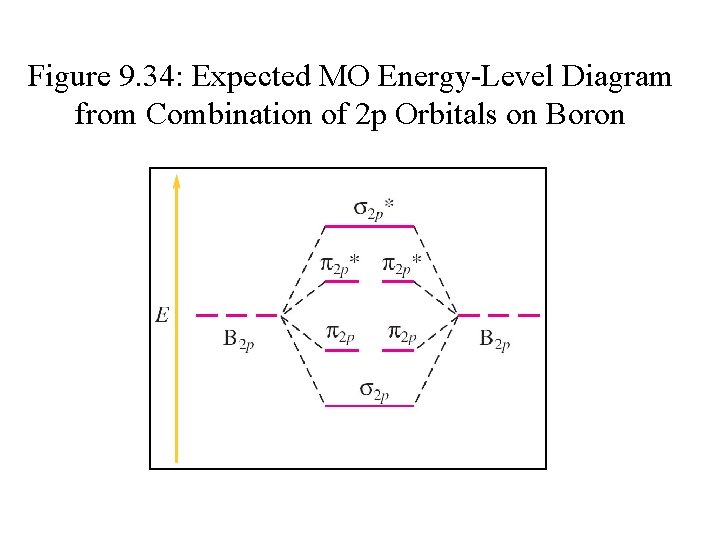
Figure 9. 34: Expected MO Energy-Level Diagram from Combination of 2 p Orbitals on Boron

Expected MO Energy-Level Diagram for B 2 Molecule
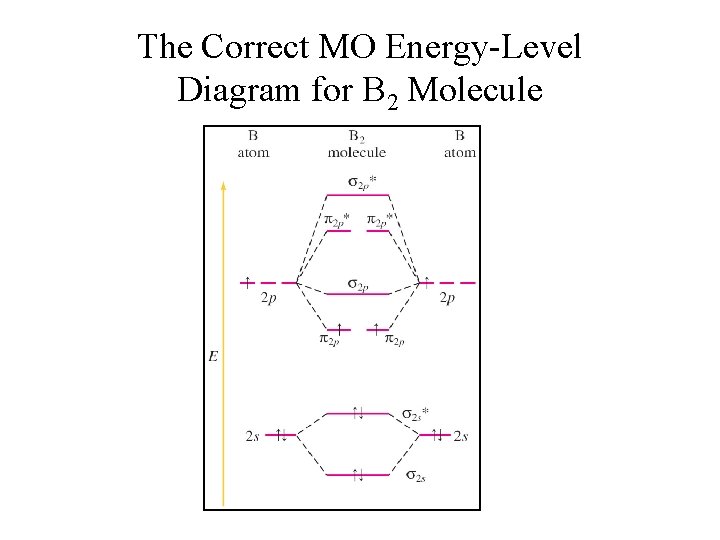
The Correct MO Energy-Level Diagram for B 2 Molecule
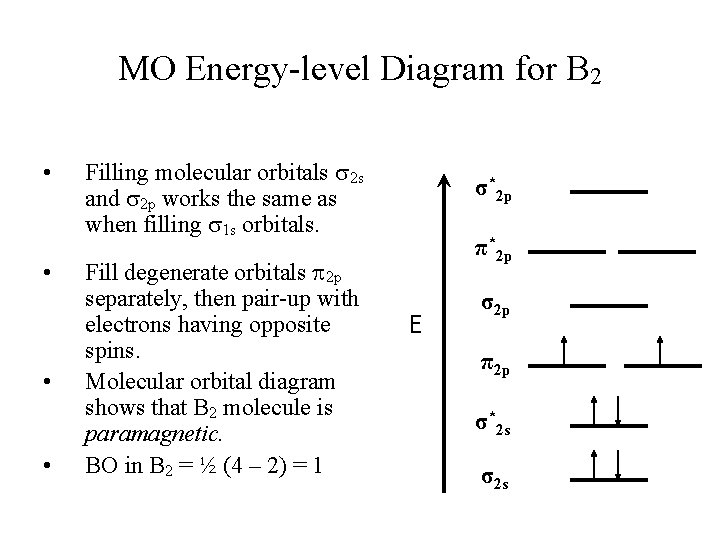
MO Energy-level Diagram for B 2 • • Filling molecular orbitals 2 s and 2 p works the same as when filling 1 s orbitals. Fill degenerate orbitals 2 p separately, then pair-up with electrons having opposite spins. Molecular orbital diagram shows that B 2 molecule is paramagnetic. BO in B 2 = ½ (4 – 2) = 1 σ*2 p π*2 p E σ2 p π2 p σ*2 s σ2 s

MO Energy-level Diagram for C 2 • • Molecular orbital diagram for C 2 shows that the molecule is diamagnetic. Bond order in C 2 = ½ (6 – 2) = 2 σ*2 p π*2 p E σ2 p π2 p σ*2 s σ2 s
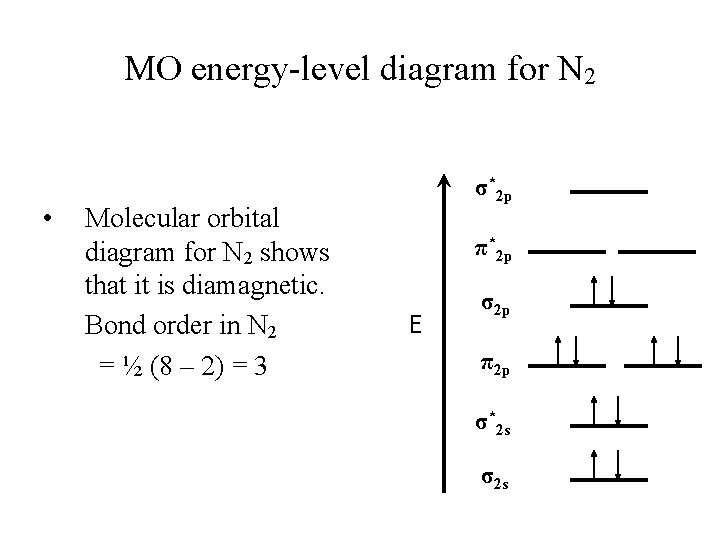
MO energy-level diagram for N 2 • Molecular orbital diagram for N 2 shows that it is diamagnetic. Bond order in N 2 = ½ (8 – 2) = 3 σ*2 p π*2 p E σ2 p π2 p σ*2 s σ2 s
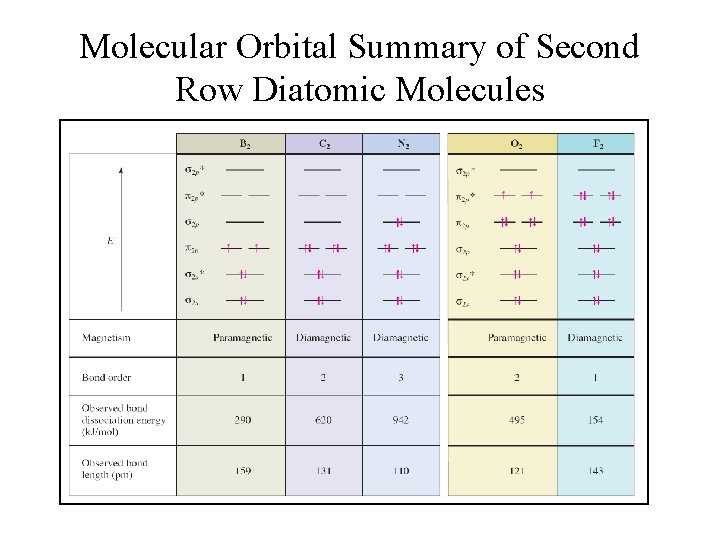
Molecular Orbital Summary of Second Row Diatomic Molecules
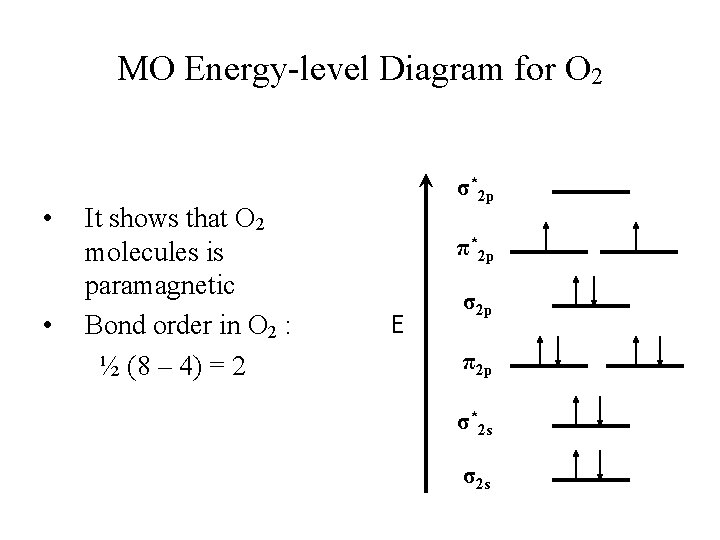
MO Energy-level Diagram for O 2 • • It shows that O 2 molecules is paramagnetic Bond order in O 2 : ½ (8 – 4) = 2 σ*2 p π*2 p E σ2 p π2 p σ*2 s σ2 s

Paramagnetism • Paramagnetism – substance is attracted into the inducing magnetic field. § Unpaired electrons (O 2) • Diamagnetism – substance is repelled from the inducing magnetic field. § Paired electrons (N 2)
Apparatus Used to Measure the Paramagnetism of a Sample

Fig 9. 39: Liquid Oxygen Poured into Space Between Poles of Magnet Donald W. Clegg

Paramagnetism vs. Diamagnetism • Paramagnetism implies the presence of unpaired electrons, whereas diamagnetism implies that all electrons are paired. • Atoms or molecules that have paramagnetic property are attracted to a magnetic field. • Atoms or molecules that are diamagnetic indicates are repelled by magnetic field. • Paramagnetic effect is generally much stronger than diamagnetic effect.
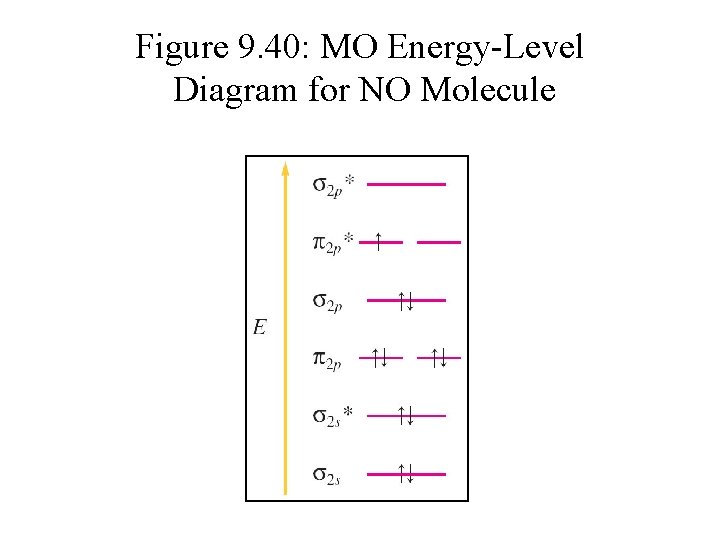
Figure 9. 40: MO Energy-Level Diagram for NO Molecule

MO Energy-Level Diagram for NO+ and CN- Ions

Homonuclear Diatomic Molecules • Composed of 2 identical atoms. • Only the valence orbitals of the atoms contribute significantly to the molecular orbitals of a particular molecule.

Heteronuclear Molecules • Heteronuclear molecules are molecules made up of different atoms. • Molecules that are made up of atoms close together on the periodic table have similar molecular orbital diagrams as homonuclear molecules. • When two atoms of a diatomic molecule are very different, the energy-level diagram is more complicated.

Heteronuclear Diatomic Molecule: HF • The σ molecular orbital containing the bonding electron pair shows greater electron probability close to the fluorine. • The electron pair is not shared equally. • This causes the fluorine atom to have a slight excess of negative charge and leaves the hydrogen atom partially positive. • This is exactly the bond polarity observed for HF.
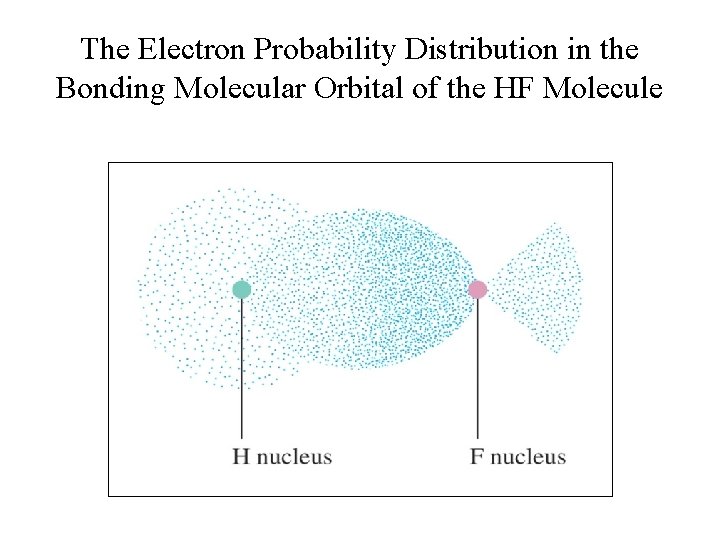
The Electron Probability Distribution in the Bonding Molecular Orbital of the HF Molecule
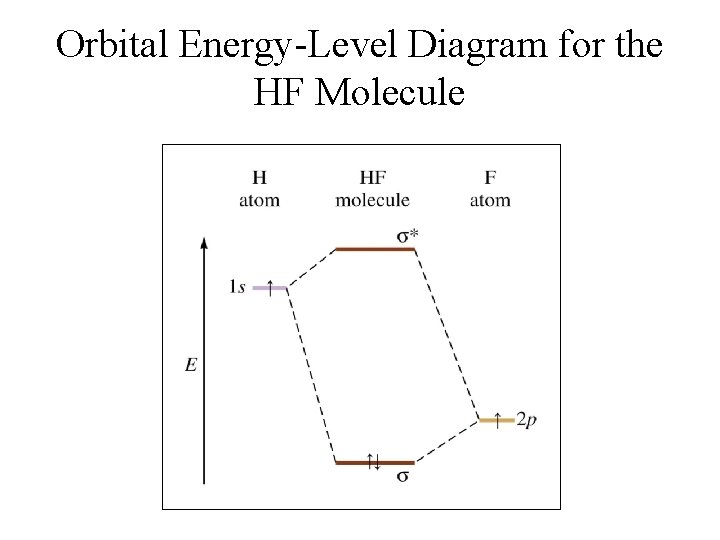
Orbital Energy-Level Diagram for the HF Molecule

Heteronuclear Diatomic Molecule: HF • The 2 p orbital of fluorine is at a lower energy than the 1 s orbital of hydrogen because fluorine binds its valence electrons more tightly. § Electrons prefer to be closer to the fluorine atom. • Thus the 2 p electron on a free fluorine atom is at a lower energy than the 1 s electron on a free hydrogen atom.
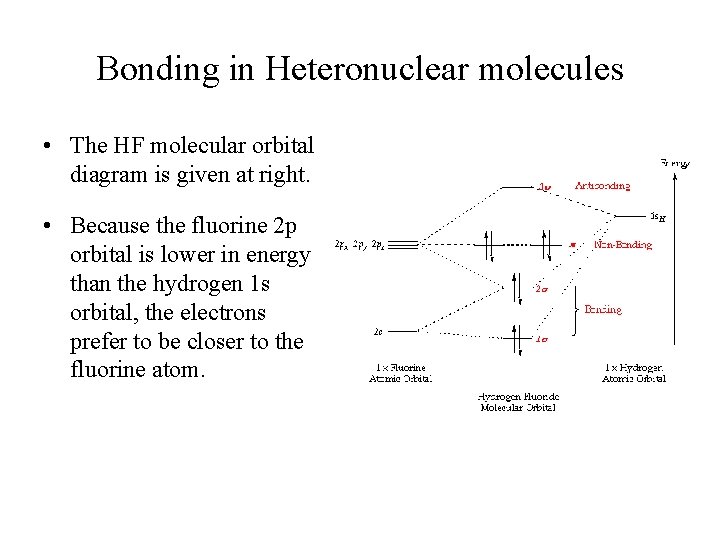
Bonding in Heteronuclear molecules • The HF molecular orbital diagram is given at right. • Because the fluorine 2 p orbital is lower in energy than the hydrogen 1 s orbital, the electrons prefer to be closer to the fluorine atom.

Bonding in Homonuclear Diatomic Molecules • Bonds are formed when atomic orbitals overlap in space. • This is not possible with 1 s orbitals in period two elements because each of the orbitals is too close to its own nucleus. • Only the 2 s and 2 p orbitals (containing the valence electrons) can participate in bond formation. • Sigma (σ) bonds are formed along internuclear axis. Sigma type molecular orbitals can be formed using 2 s orbitals or 2 px orbitals (assuming bonding along the x-axis) • In addition, pi (π) type molecular orbitals are formed by the combination of 2 py or 2 pz orbitals. • The combination of atomic orbitals yields bonding ( and ) and antibonding (σ* and π*) molecular orbitals.

Exercise #2 on Molecular Orbitals Determine the electron configuration and bond order for the following. If it exists, discuss its magnetism. 1. F 2σ*2 p How many valence electrons does F 2 - have? π*2 p 15 valence electrons BO = 8 b. e. - 7 a. e. 2 E =½ F 2 - would be paramagnetic. σ2 p π2 p σ*2 s σ2 s
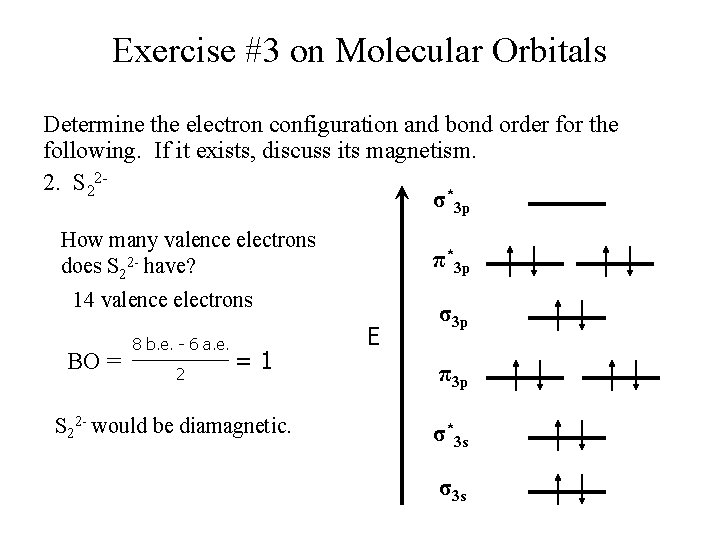
Exercise #3 on Molecular Orbitals Determine the electron configuration and bond order for the following. If it exists, discuss its magnetism. 2. S 22σ*3 p How many valence electrons does S 22 - have? π*3 p 14 valence electrons BO = 8 b. e. - 6 a. e. 2 =1 S 22 - would be diamagnetic. E σ3 p π3 p σ*3 s σ3 s
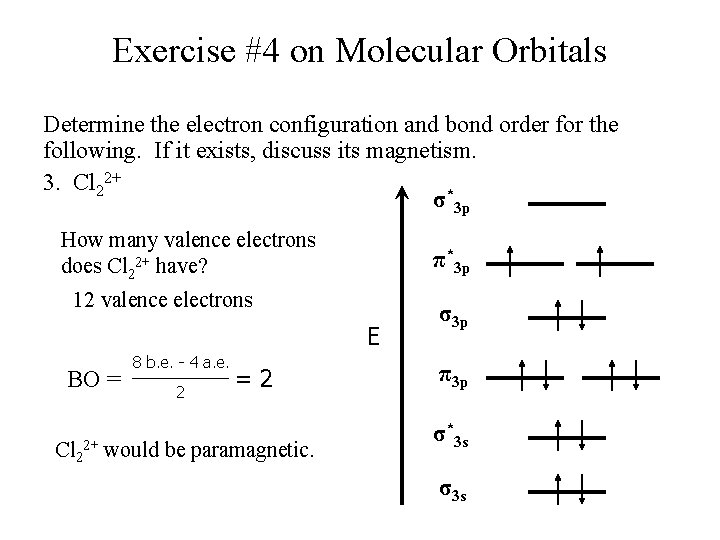
Exercise #4 on Molecular Orbitals Determine the electron configuration and bond order for the following. If it exists, discuss its magnetism. 3. Cl 22+ σ*3 p How many valence electrons does Cl 22+ have? π*3 p 12 valence electrons E BO = 8 b. e. - 4 a. e. 2 =2 Cl 22+ would be paramagnetic. σ3 p π3 p σ*3 s σ3 s

Combining the Localized Electron and Molecular Orbital Models • Advantage of molecular orbital model - correctly predicts relative bond strength and magnetism of simple diatomic molecules. • It can account for bond polarity and correctly portrays electrons as being localized or delocalized in polyatomic molecules. • Major disadvantage - too complex for polyatomic molecules. An approximation is used.

Combining the Localized Electron and Molecular Orbital Models • The localized electron model assumes that electrons are confined between a given pair of atoms in a molecule. • Combining the localized electron and molecular orbital models gives us a more accurate description, such that: σ-bonds can be regarded as localized, while π-bonding is best described as delocalized. • Since a double bond consists of one σ-bond and one π-bond: Electrons in σ-bonds are considered localized. Electrons involved in -bonds are said to be delocalized.
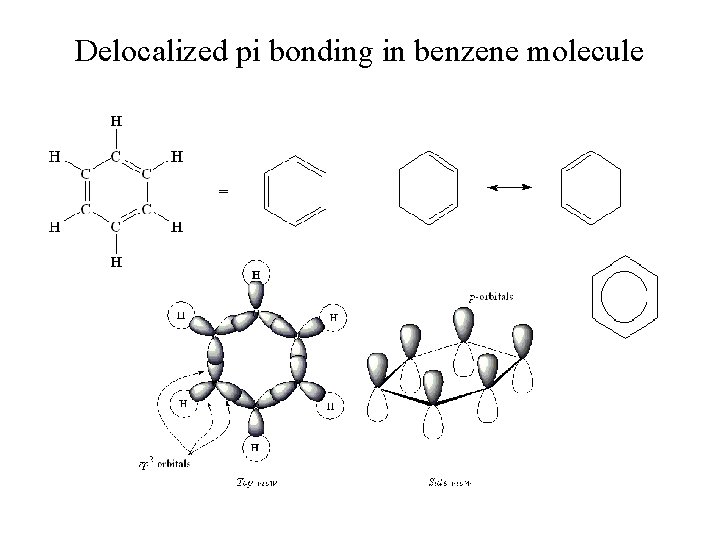
Delocalized pi bonding in benzene molecule

Molecular Orbital Energies • MO 1 is lower in energy than the s orbitals of free atoms, while MO 2 is higher in energy than the s orbitals. § Bonding molecular orbital – lower in energy § Antibonding molecular orbital – higher in energy

Electron Configurations in MO • Molecular electron configurations can be written similar to atomic electron configurations. • Each molecular orbital can hold 2 electrons with opposite spins. • The number of orbitals are conserved.

Bonding in H 2

Sigma Bonding and Antibonding Molecular Orbitals
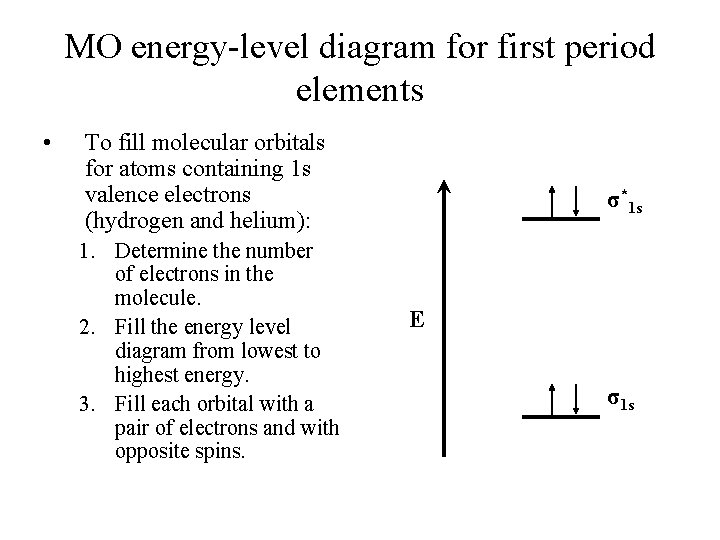
MO energy-level diagram for first period elements • To fill molecular orbitals for atoms containing 1 s valence electrons (hydrogen and helium): 1. Determine the number of electrons in the molecule. 2. Fill the energy level diagram from lowest to highest energy. 3. Fill each orbital with a pair of electrons and with opposite spins. σ*1 s E σ1 s

Exercises on Molecular Orbitals Use MO theory to describe the bonding and stability of a. H 22 - b. H 2+ H 22 - has 4 electrons. BO = σ*1 s σ1 s 2 b. e. - 2 a. e. 2 =0 H 2 ion will not be formed. 2 - H 2+ has 1 electron. BO = 1 b. e. - 0 a. e. 2 =½ (H 2+ may be formed, but it would not be much more stable than two separate hydrogen atoms)

Pi Bonding and Antibonding Orbitals
- Slides: 94