Advanced Spectroscopy 20152016 The chemical shift Professor Dan
















- Slides: 66

Advanced Spectroscopy 2015/2016 The chemical shift Professor Dan Stærk Natural Products Research Department of Drug Design & Pharmacology Faculty of Health and Medical Sciences University of Copenhagen Remember: Open door policy –> you are always welcome to come and ask questions (or send me an e-mail) Give constructive criticism in the black book Dias 1

Course schedule Dias 2 Advanced Spectroscopy 2015/2016

Advanced Spectroscopy 2015/2016 Learning objectives for 1 H chemical shift After this topic you should be able to: ü Identify structural moieties (read chemical environments) influencing chemical shift values of a proton ü Estimate 1 H chemical shift values based on the above observations ü Calculate/predict proton chemical shift values of different molecules AND understand the limitations Dias 3

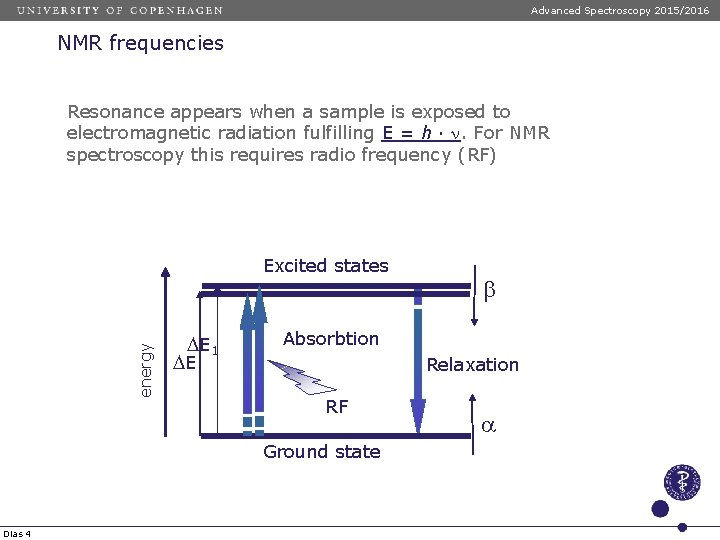
Advanced Spectroscopy 2015/2016 NMR frequencies Resonance appears when a sample is exposed to electromagnetic radiation fulfilling E = h · n. For NMR spectroscopy this requires radio frequency (RF) energy Excited states D E 1 DE Absorbtion Relaxation RF Ground state Dias 4

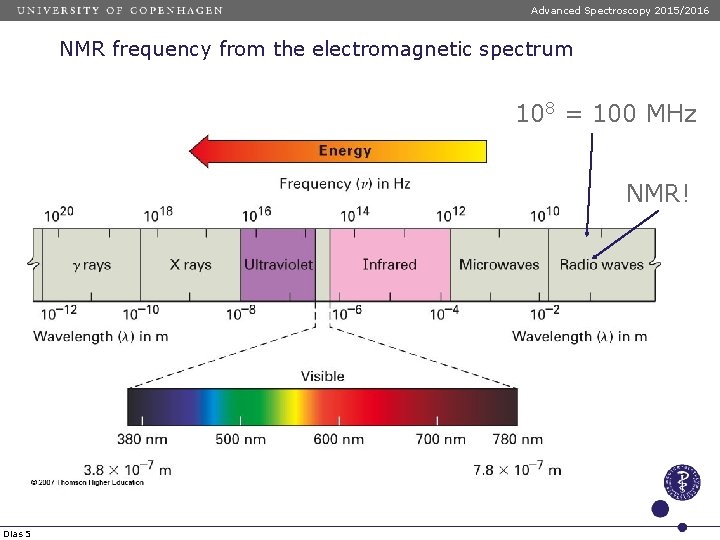
Advanced Spectroscopy 2015/2016 NMR frequency from the electromagnetic spectrum 108 = 100 MHz NMR! Dias 5

Advanced Spectroscopy 2015/2016 NMR frequency of 1 H and 13 C The NMR frequency is given by the below equation, and thus depending on the magnetogyric ratio (g) and the applied field strength (B 0) Dias 6

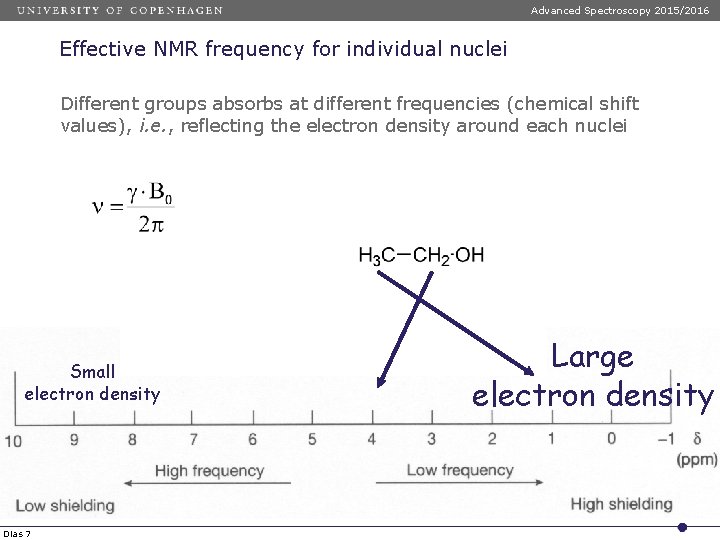
Advanced Spectroscopy 2015/2016 Effective NMR frequency for individual nuclei Different groups absorbs at different frequencies (chemical shift values), i. e. , reflecting the electron density around each nuclei Small electron density Dias 7 Large electron density

Advanced Spectroscopy 2015/2016 Effective NMR frequency for individual nuclei Different groups absorbs at different frequencies (chemical shift values), i. e. , reflecting the electron density around each nuclei Shielding factor: describes the electron density of each nuclei Small electron density Dias 8 Large electron density

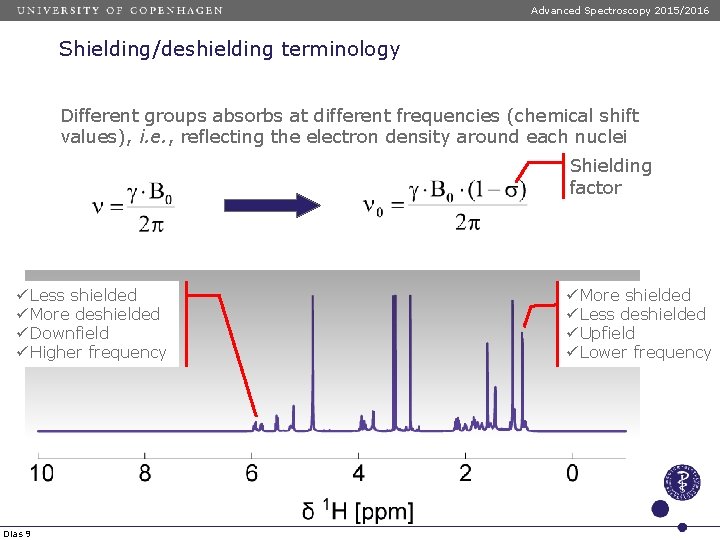
Advanced Spectroscopy 2015/2016 Shielding/deshielding terminology Different groups absorbs at different frequencies (chemical shift values), i. e. , reflecting the electron density around each nuclei Shielding factor ü Less shielded ü More deshielded ü Downfield ü Higher frequency Dias 9 ü More shielded ü Less deshielded ü Upfield ü Lower frequency

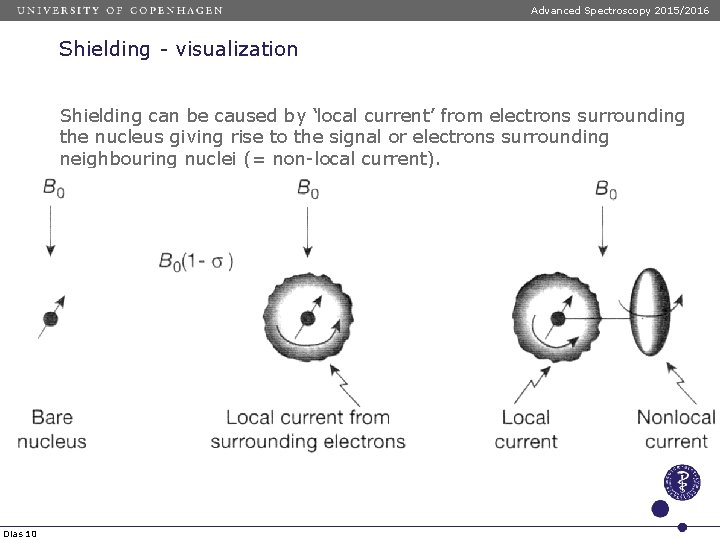
Advanced Spectroscopy 2015/2016 Shielding - visualization Shielding can be caused by ‘local current’ from electrons surrounding the nucleus giving rise to the signal or electrons surrounding neighbouring nuclei (= non-local current). Small electron density Dias 10

Advanced Spectroscopy 2015/2016 Chemical shift – calculation and ranges NMR frequencies (n) are measured in Hz, but transformed to chemical shift values (d, ppm) to allow comparison of spectra acquired at different field strengths. 1 H: 13 C: 0 -12 ppm 0 -240 ppm Reference: TMS = Si(CH 3)4; by definition 0 ppm Dias 11

Advanced Spectroscopy 2015/2016 Exercise 1 – chemical shift vs frequency The methyl group in acetophenone resonates at d 2. 59 relative to TMS in a spectrum acquired using a 600 MHz NMR spectrometer. ① What is the resonance frequency n in Hz in the same spectrum? ② What is the resonance frequency n in Hz in a spectrum acquired using a 400 MHz spectrometer? ③ What is the chemical shift value d in a spectrum acquired using a 400 MHz spectrometer? Dias 12

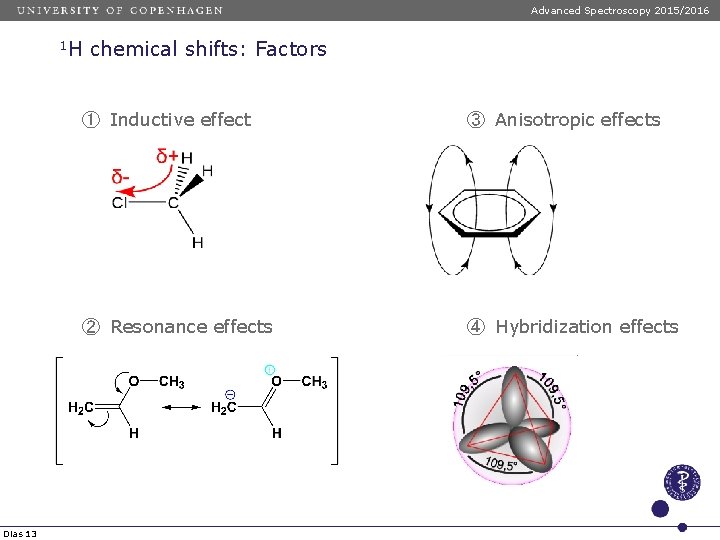
Advanced Spectroscopy 2015/2016 1 H Dias 13 chemical shifts: Factors ① Inductive effect ③ Anisotropic effects ② Resonance effects ④ Hybridization effects

Advanced Spectroscopy 2015/2016 Inductive effect Electronegative atoms/electron-withdrawing groups decrease the shielding of a bonded or close proton. Deshielding effect increases with increasing number of electron-withdrawing groups (additive effect). CH-Cl 3 CH 2 -Cl 2 CH 3 -Cl CH 3 -H 7. 27 ppm 5. 30 ppm 3. 05 ppm 0. 23 ppm Electron density Deshielding Dias 14 Shielding

Advanced Spectroscopy 2015/2016 Inductive effect Electronegative atoms/electron-withdrawing groups decrease the shielding of a bonded or close proton. Electropositive atoms/electron-donating groups increase the shielding of a bonded or close proton. CH-F CH 2 -OH CH 3 -NH 2 4. 26 3. 38 2. 47 CH 3 -H CH 3 -Si. Me 3 CH 3 -Li 0. 23 0. 00 -0. 4 Electron density Deshielding Dias 15 Shielding

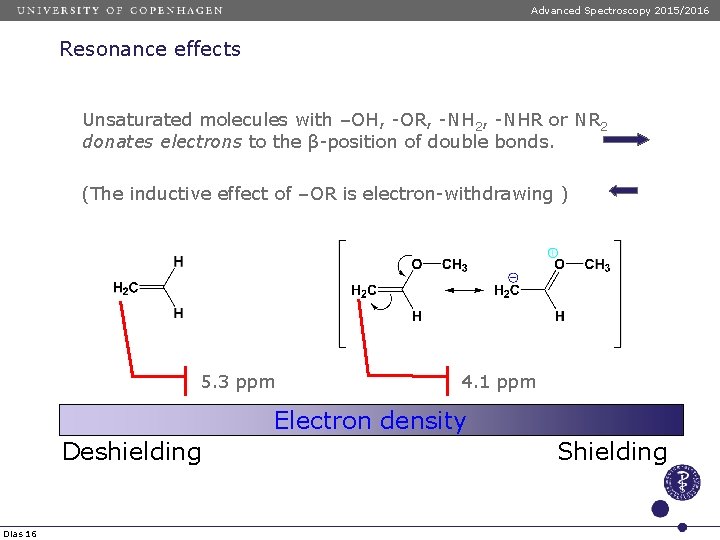
Advanced Spectroscopy 2015/2016 Resonance effects Unsaturated molecules with –OH, -OR, -NH 2, -NHR or NR 2 donates electrons to the β-position of double bonds. (The inductive effect of –OR is electron-withdrawing ) 5. 3 ppm 4. 1 ppm Electron density Deshielding Dias 16 Shielding

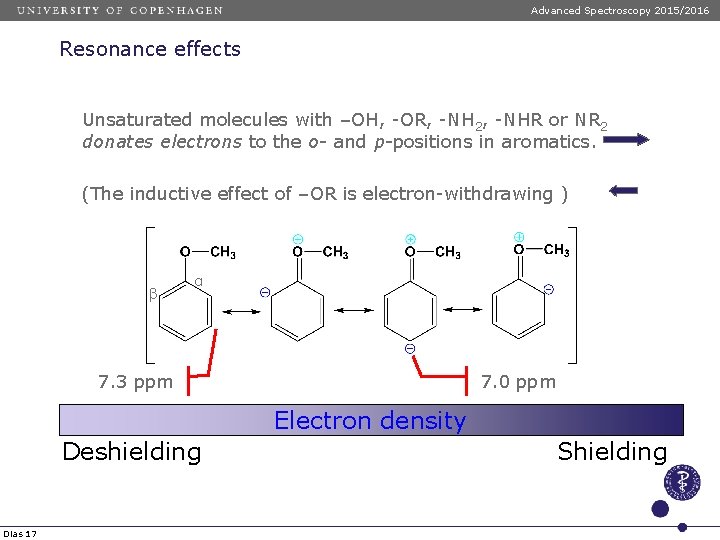
Advanced Spectroscopy 2015/2016 Resonance effects Unsaturated molecules with –OH, -OR, -NH 2, -NHR or NR 2 donates electrons to the o- and p-positions in aromatics. (The inductive effect of –OR is electron-withdrawing ) β α 7. 3 ppm 7. 0 ppm Electron density Deshielding Dias 17 Shielding

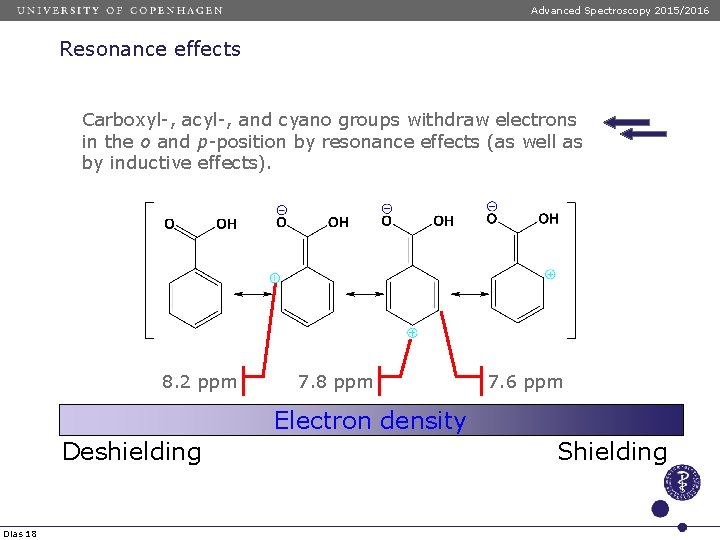
Advanced Spectroscopy 2015/2016 Resonance effects Carboxyl-, acyl-, and cyano groups withdraw electrons in the o and p-position by resonance effects (as well as by inductive effects). 8. 2 ppm 7. 8 ppm 7. 6 ppm Electron density Deshielding Dias 18 Shielding

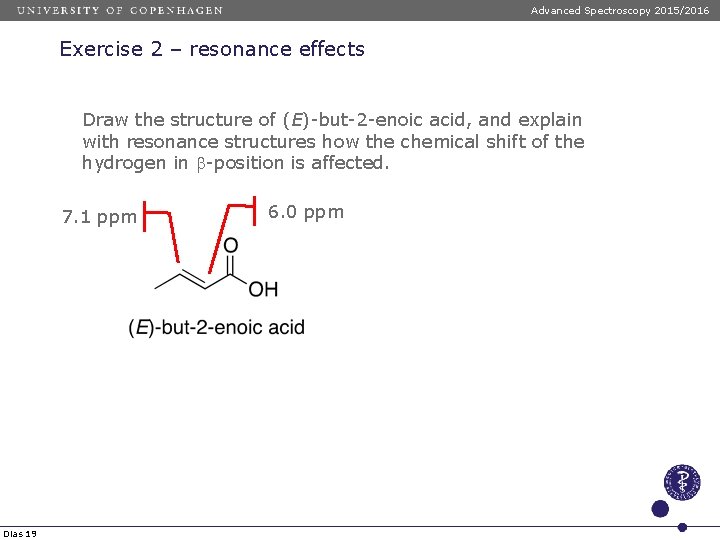
Advanced Spectroscopy 2015/2016 Exercise 2 – resonance effects Draw the structure of (E)-but-2 -enoic acid, and explain with resonance structures how the chemical shift of the hydrogen in -position is affected. 7. 1 ppm Dias 19 6. 0 ppm

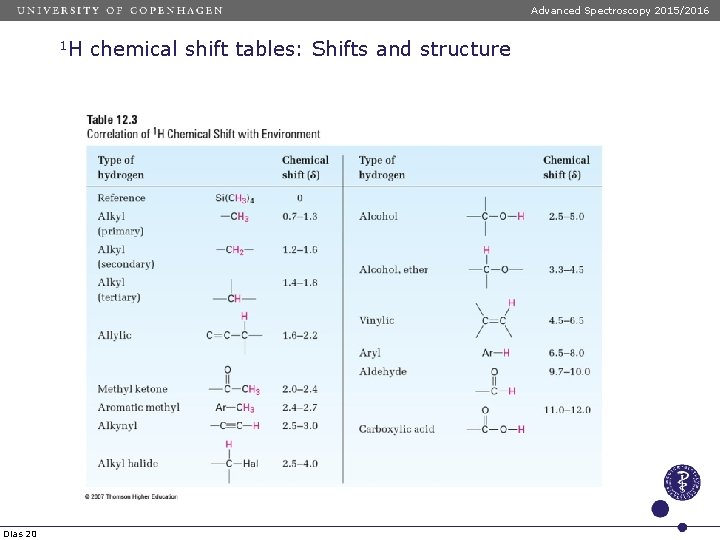
Advanced Spectroscopy 2015/2016 1 H Dias 20 chemical shift tables: Shifts and structure

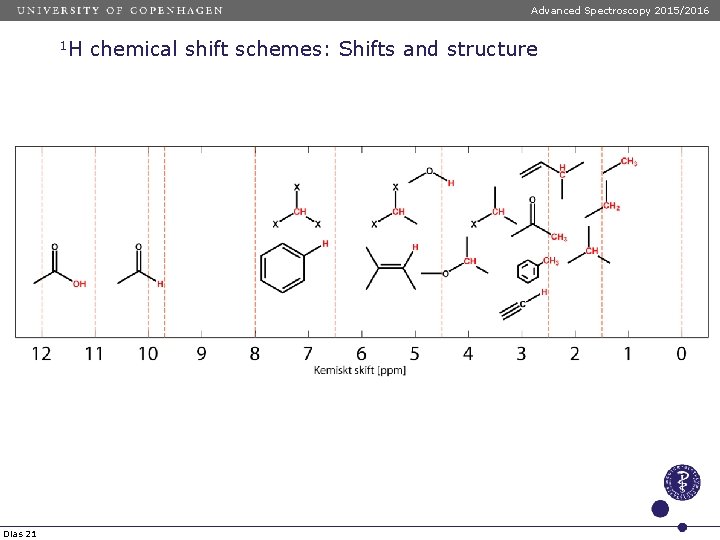
Advanced Spectroscopy 2015/2016 1 H Dias 21 chemical shift schemes: Shifts and structure

Advanced Spectroscopy 2015/2016 1 H shift calculations – Schoolery’s rule Empirical correlations between structure and chemical shifts are collected in the form tables of additive terms. δ = 0. 23 + ΔX + ΔY Dias 22

Advanced Spectroscopy 2015/2016 1 H shift calculations – double bonds Empirical correlations between structure and chemical shifts are collected in the form tables of additive terms. δ = 5. 28 + Zgem + Zcis + Ztrans Dias 23

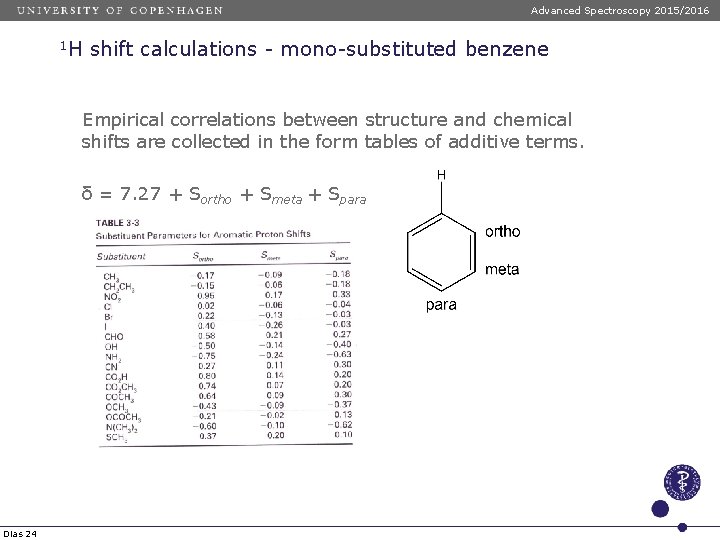
Advanced Spectroscopy 2015/2016 1 H shift calculations - mono-substituted benzene Empirical correlations between structure and chemical shifts are collected in the form tables of additive terms. δ = 7. 27 + Sortho + Smeta + Spara Dias 24

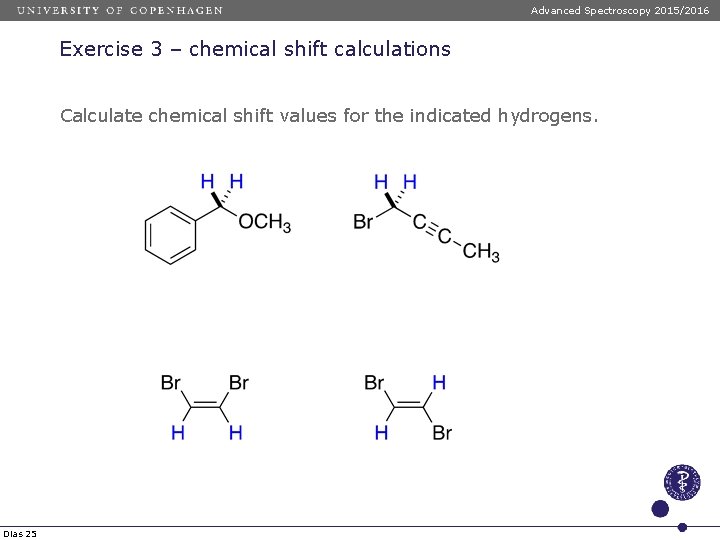
Advanced Spectroscopy 2015/2016 Exercise 3 – chemical shift calculations Calculate chemical shift values for the indicated hydrogens. Dias 25

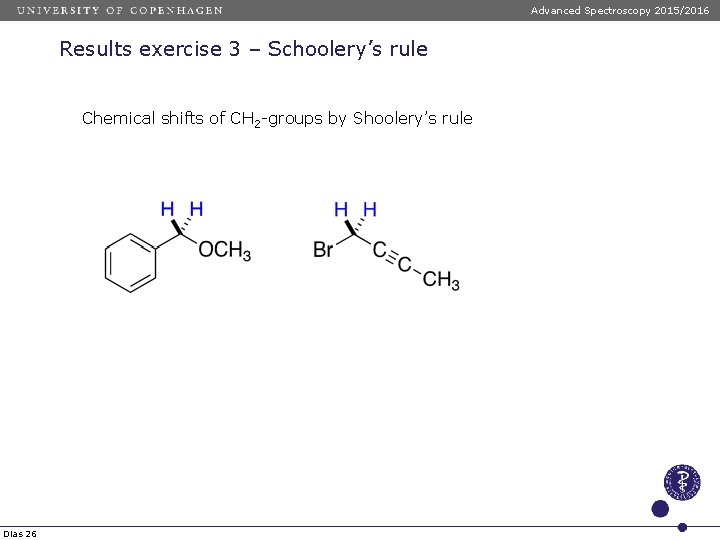
Advanced Spectroscopy 2015/2016 Results exercise 3 – Schoolery’s rule Chemical shifts of CH 2 -groups by Shoolery’s rule δ = 0. 23 + ΔX + ΔY δ = 0. 23 + 2. 36 (OR) + 1. 83 (Ph) = 4. 42 δ = 0. 23 + 1. 44 (C=CR) + 2. 33 (Br) = 4. 00 Dias 26

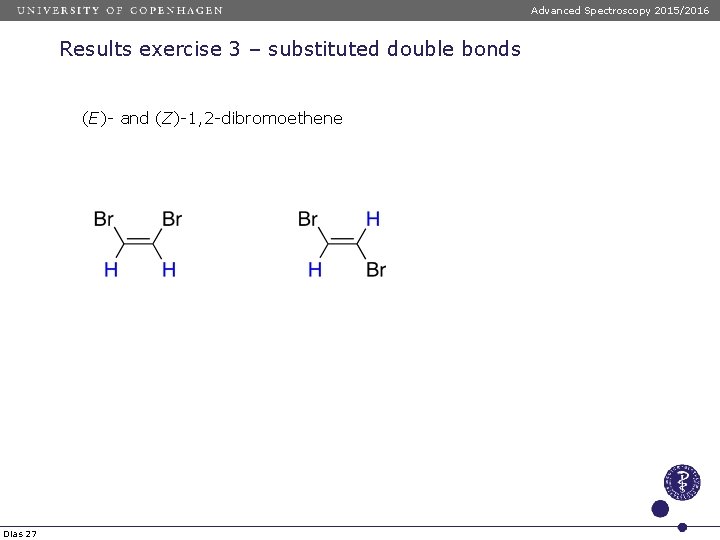
Advanced Spectroscopy 2015/2016 Results exercise 3 – substituted double bonds (E)- and (Z)-1, 2 -dibromoethene δ = 5. 28 + Zgem + Zcis + Ztrans δ = 5. 28 + 1. 04 + 0. 55 = 6. 87 δ = 5. 28 + 1. 04 + 0. 40 + 0 = 6. 72 Dias 27

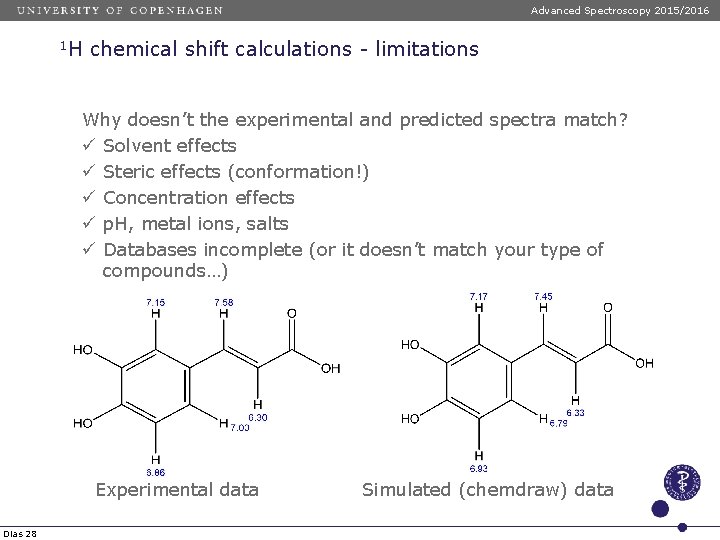
Advanced Spectroscopy 2015/2016 1 H chemical shift calculations - limitations Why doesn’t the experimental and predicted spectra match? ü Solvent effects ü Steric effects (conformation!) ü Concentration effects ü p. H, metal ions, salts ü Databases incomplete (or it doesn’t match your type of compounds…) Experimental data Dias 28 Simulated (chemdraw) data

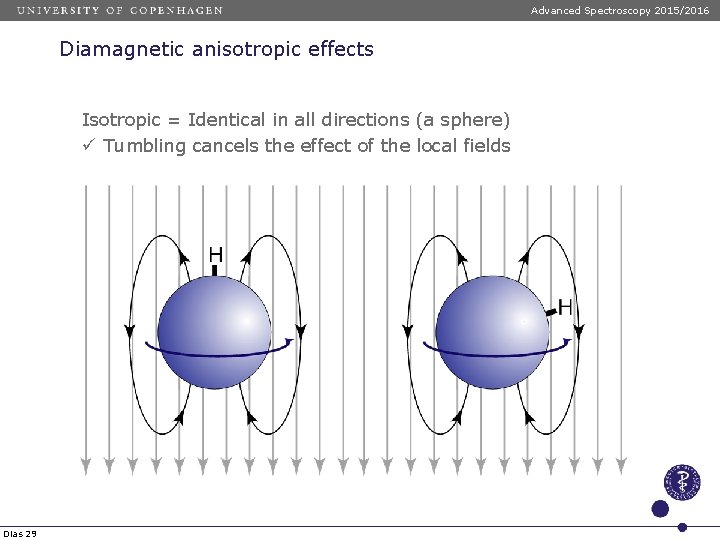
Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects Isotropic = Identical in all directions (a sphere) ü Tumbling cancels the effect of the local fields Dias 29

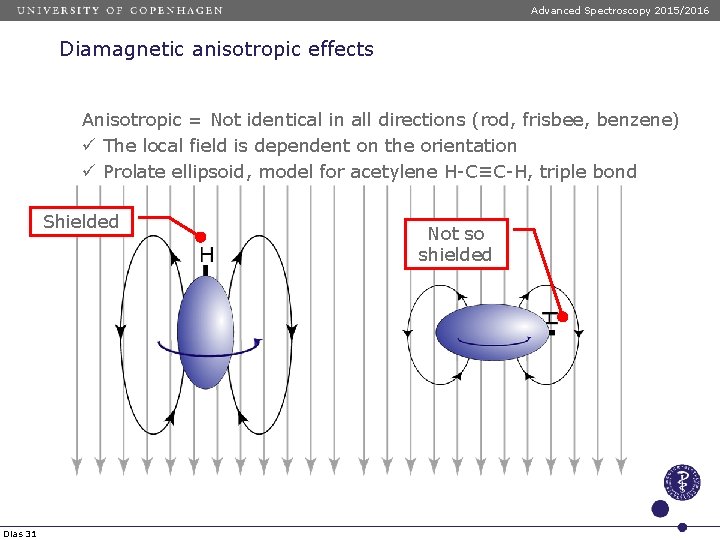
Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects Anisotropic = Not identical in all directions (rod, frisbee, benzene) ü The local field is dependent on the orientation ü Oblate ellipsoid = model for benzene Shielded Deshielded Dias 30

Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects Anisotropic = Not identical in all directions (rod, frisbee, benzene) ü The local field is dependent on the orientation ü Prolate ellipsoid, model for acetylene H-C≡C-H, triple bond Shielded Dias 31 Not so shielded

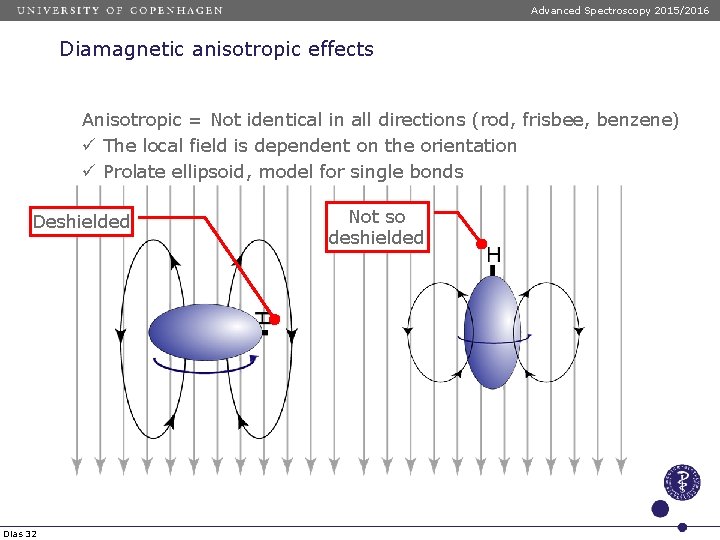
Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects Anisotropic = Not identical in all directions (rod, frisbee, benzene) ü The local field is dependent on the orientation ü Prolate ellipsoid, model for single bonds Deshielded Dias 32 Not so deshielded

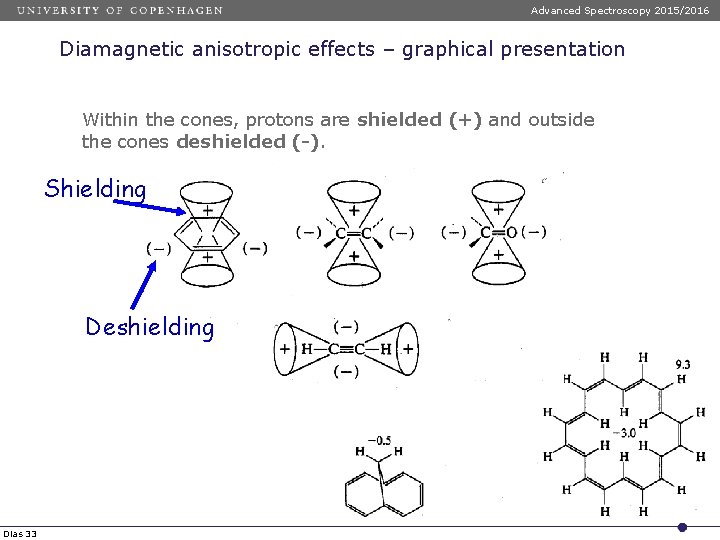
Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects – graphical presentation Within the cones, protons are shielded (+) and outside the cones deshielded (-). Shielding Deshielding Dias 33

Advanced Spectroscopy 2015/2016 Exercise 4 - diamagnetic anisotropic effects ü Which hydrogen of the Hsyn/Hanti pair is observed at lowest chemical shift value? ü Which hydrogen of the Hexo/Hendo pair is observed at lowest chemical shift value? Dias 34

Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects in aliphatics (I) Axial hydrogens are shielded Equatorial hydrogens typical app. 0. 5 ppm higher than axial Dias 35 Equatorial hydrogens are deshielded

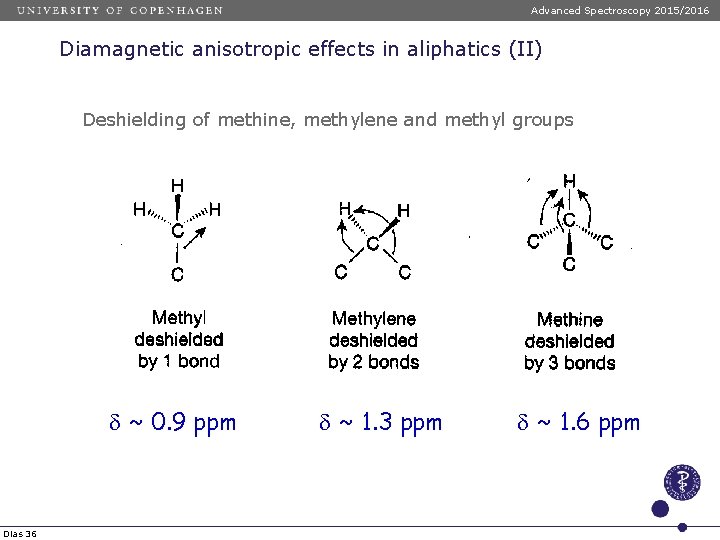
Advanced Spectroscopy 2015/2016 Diamagnetic anisotropic effects in aliphatics (II) Deshielding of methine, methylene and methyl groups d ~ 0. 9 ppm Dias 36 d ~ 1. 3 ppm d ~ 1. 6 ppm

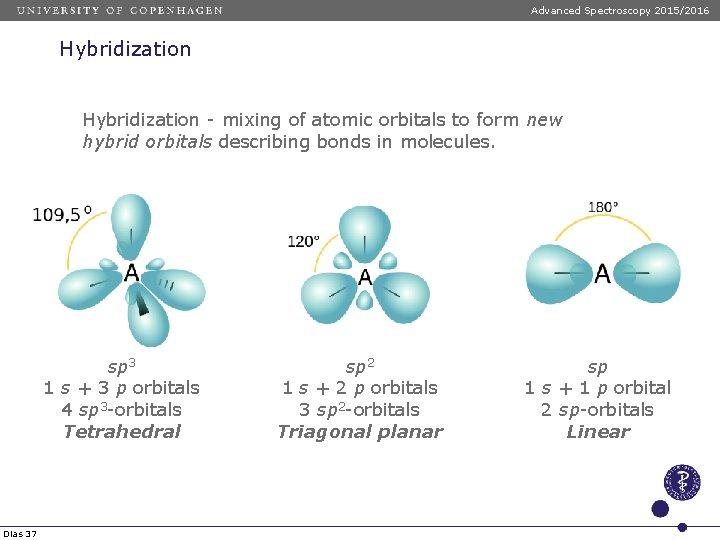
Advanced Spectroscopy 2015/2016 Hybridization - mixing of atomic orbitals to form new hybrid orbitals describing bonds in molecules. sp 3 1 s + 3 p orbitals 4 sp 3 -orbitals Tetrahedral Dias 37 sp 2 1 s + 2 p orbitals 3 sp 2 -orbitals Triagonal planar sp 1 s + 1 p orbital 2 sp-orbitals Linear

Advanced Spectroscopy 2015/2016 Hybridization - mixing of atomic orbitals to form new hybrid orbitals describing bonds in molecules. The less s-character (sp < sp 2 < sp 3) the lower shielding of the hydrogen attached to the hybridized carbon atom Strongly affected by diamagnetic anisotropy! Alkyne range 1. 8 -2. 9 ppm Alkene/aromatic range 4. 5 -8 ppm Alifatic range 0. 5 -4. 5 ppm sp sp 2 sp 3 Electron density Deshielding Dias 38 Shielding

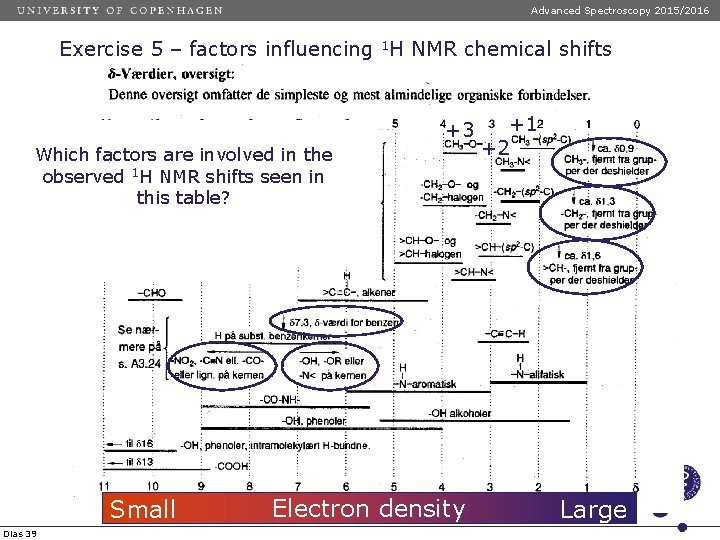
Advanced Spectroscopy 2015/2016 Exercise 5 – factors influencing 1 H NMR chemical shifts +3 Which factors are involved in the observed 1 H NMR shifts seen in this table? Small Dias 39 Electron density +1 +2 Large

Advanced Spectroscopy 2015/2016 Learning objectives for ü Differences between ü Appearance of the spectra 13 C 13 C Dias 40 NMR and 1 H NMR spectroscopy NMR spectra and information obtainable from ü Chemical shift ranges in ü Calculate/predict chemical shift 13 C NMR spectroscopy chemical shift values of different molecules

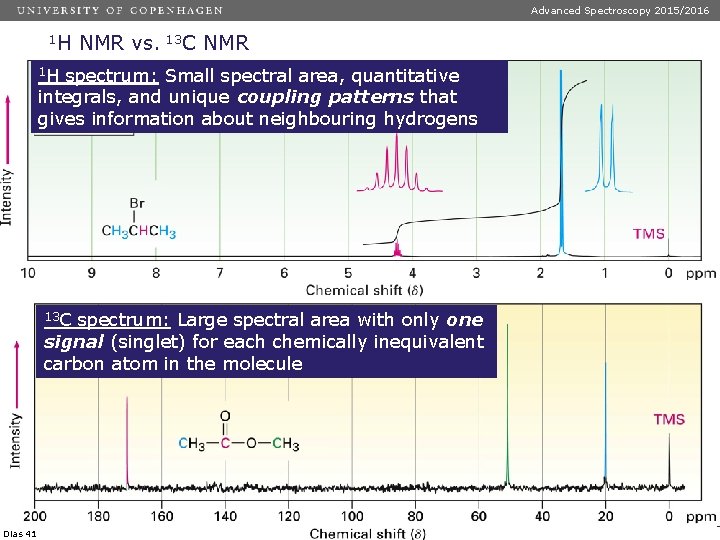
Advanced Spectroscopy 2015/2016 1 H NMR vs. 13 C NMR 1 H spectrum: Small spectral area, quantitative integrals, and unique coupling patterns that gives information about neighbouring hydrogens 13 C spectrum: Large spectral area with only one signal (singlet) for each chemically inequivalent carbon atom in the molecule Dias 41

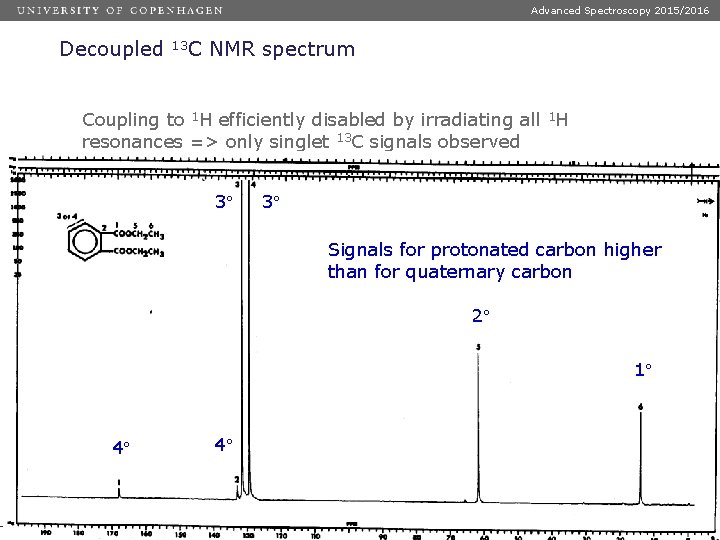
Advanced Spectroscopy 2015/2016 Decoupled 13 C NMR spectrum Coupling to 1 H efficiently disabled by irradiating all 1 H resonances => only singlet 13 C signals observed 3 3 Signals for protonated carbon higher than for quaternary carbon 2 1 4 Dias 42 4

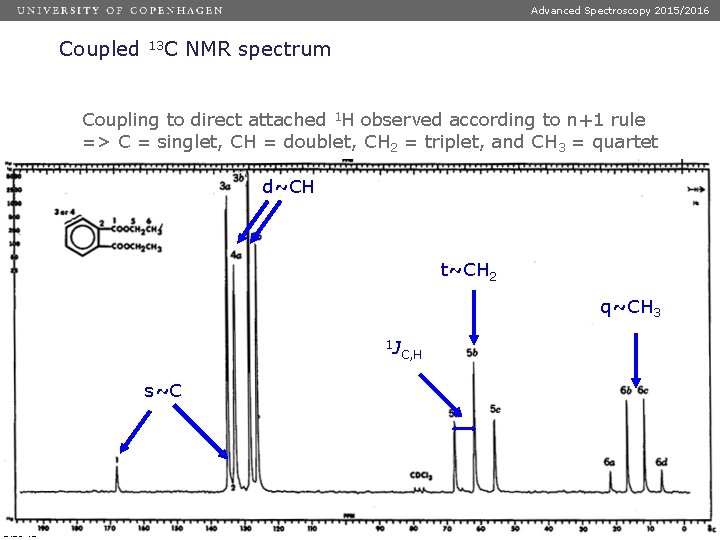
Advanced Spectroscopy 2015/2016 Coupled 13 C NMR spectrum Coupling to direct attached 1 H observed according to n+1 rule => C = singlet, CH = doublet, CH 2 = triplet, and CH 3 = quartet d~CH t~CH 2 q~CH 3 1 J s~C Dias 43 C, H

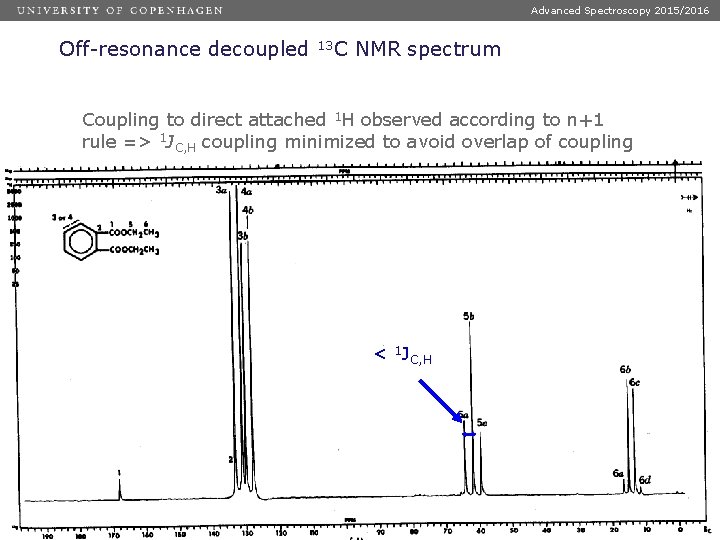
Advanced Spectroscopy 2015/2016 Off-resonance decoupled 13 C NMR spectrum Coupling to direct attached 1 H observed according to n+1 rule => 1 JC, H coupling minimized to avoid overlap of coupling patterns < 1 JC, H Dias 44

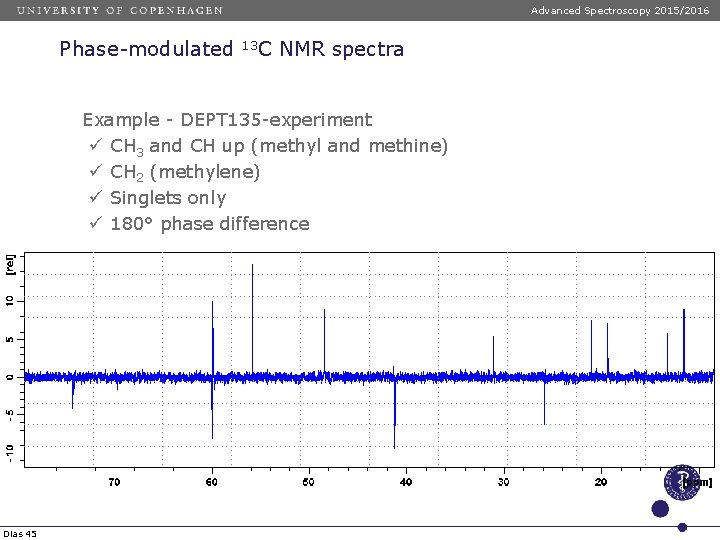
Advanced Spectroscopy 2015/2016 Phase-modulated 13 C NMR spectra Example - DEPT 135 -experiment ü CH 3 and CH up (methyl and methine) ü CH 2 (methylene) ü Singlets only ü 180° phase difference Dias 45

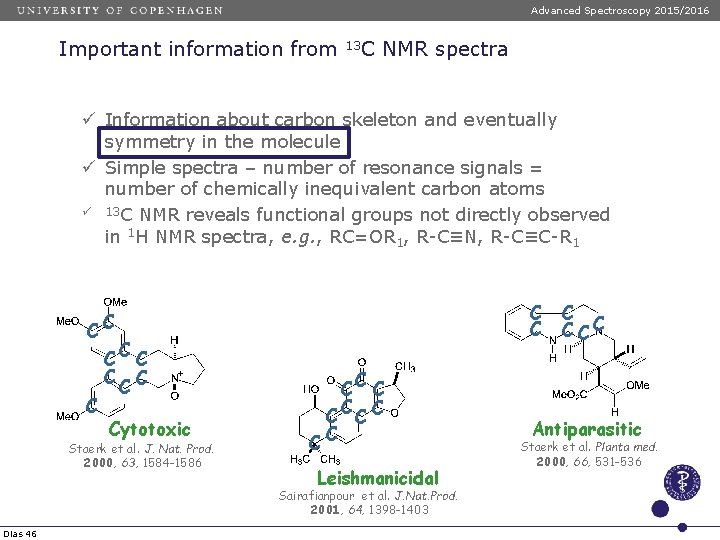
Advanced Spectroscopy 2015/2016 Important information from 13 C NMR spectra ü Information about carbon skeleton and eventually symmetry in the molecule ü Simple spectra – number of resonance signals = number of chemically inequivalent carbon atoms ü 13 C NMR reveals functional groups not directly observed in 1 H NMR spectra, e. g. , RC=OR 1, R-C≡N, R-C≡C-R 1 C C CC C C Cytotoxic Staerk et al. J. Nat. Prod. 2000, 63, 1584 -1586 C CC C Leishmanicidal Sairafianpour et al. J. Nat. Prod. 2001, 64, 1398 -1403 Dias 46 C CCC Antiparasitic Staerk et al. Planta med. 2000, 66, 531 -536

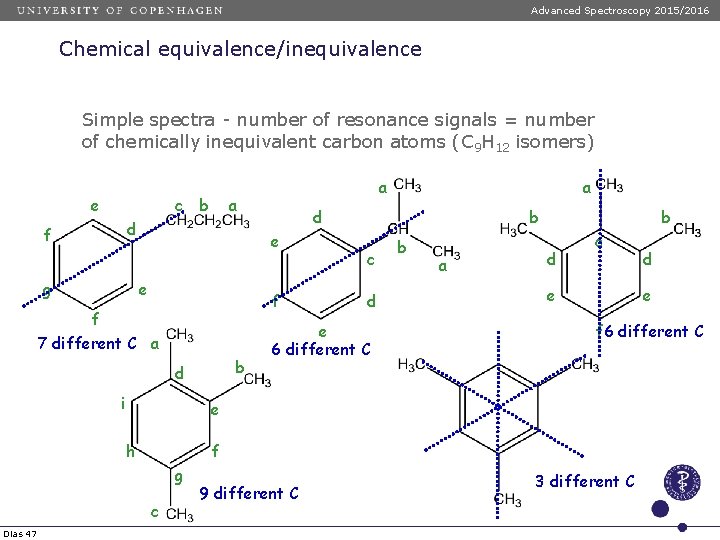
Advanced Spectroscopy 2015/2016 Chemical equivalence/inequivalence Simple spectra - number of resonance signals = number of chemically inequivalent carbon atoms (C 9 H 12 isomers) e c b a d f g e f b d i a d e f 7 different C a b c d e 6 different C b a b d c e f 6 different C f g c 9 different C d e e h Dias 47 a 3 different C

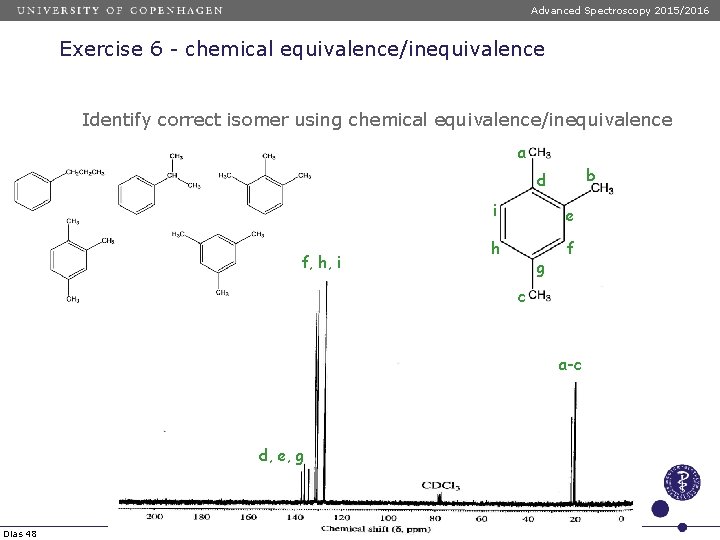
Advanced Spectroscopy 2015/2016 Exercise 6 - chemical equivalence/inequivalence Identify correct isomer using chemical equivalence/inequivalence a b d f, h, i i e h f g c a-c d, e, g Dias 48

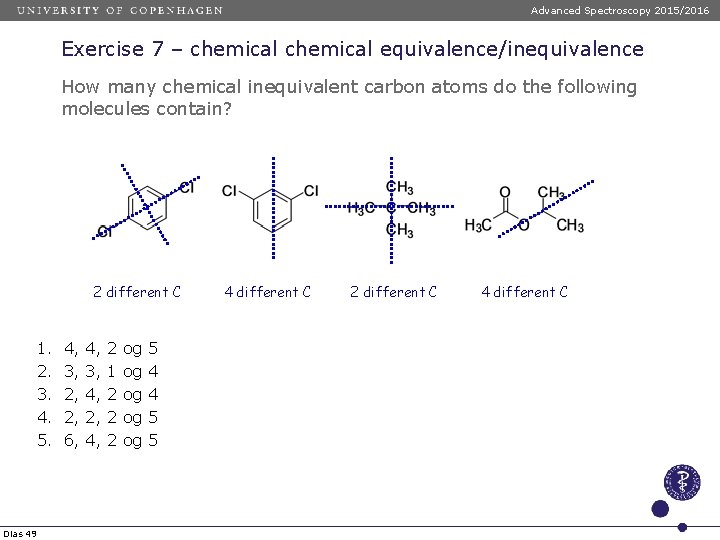
Advanced Spectroscopy 2015/2016 Exercise 7 – chemical equivalence/inequivalence How many chemical inequivalent carbon atoms do the following molecules contain? 2 different C 1. 2. 3. 4. 5. Dias 49 4, 3, 2, 2, 6, 4, 3, 4, 2, 4, 2 1 2 2 2 og og og 5 4 4 5 5 4 different C 2 different C 4 different C

Advanced Spectroscopy 2015/2016 13 C chemical shift – calculation and ranges NMR frequencies (n) are measured in Hz, but transformed to chemical shift values (d, ppm) to allow comparison of spectra acquired at different field strengths. 1 H: 13 C: 0 -12 ppm 0 -240 ppm Reference: TMS = Si(CH 3)4; by definition 0 ppm Dias 50

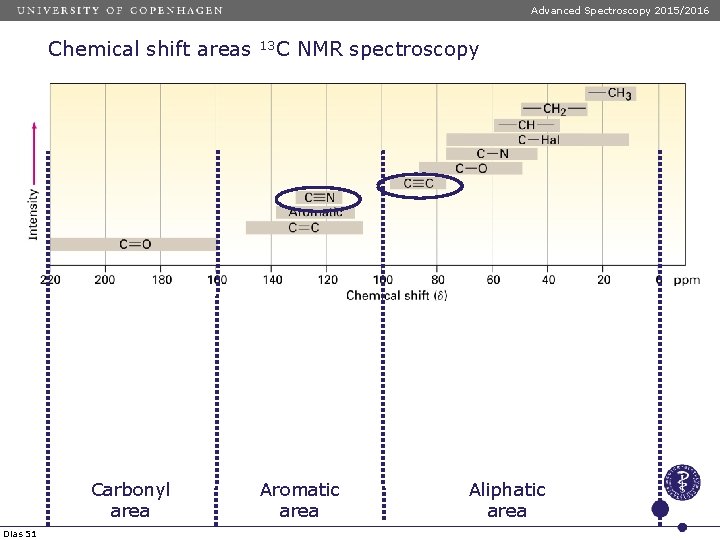
Advanced Spectroscopy 2015/2016 Chemical shift areas Carbonyl area Dias 51 13 C NMR spectroscopy Aromatic area Aliphatic area

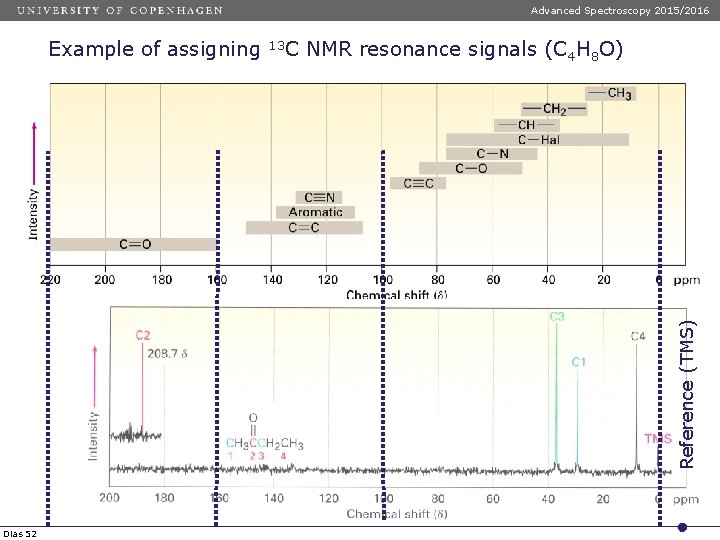
Advanced Spectroscopy 2015/2016 13 C NMR resonance signals (C 4 H 8 O) Reference (TMS) Example of assigning Dias 52

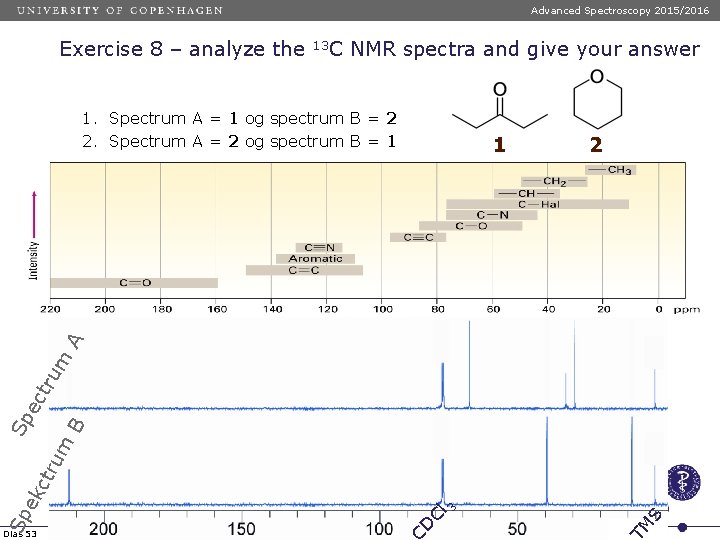
Advanced Spectroscopy 2015/2016 Exercise 8 – analyze the 13 C NMR spectra and give your answer 1. Spectrum A = 1 og spectrum B = 2 2. Spectrum A = 2 og spectrum B = 1 2 S Cl 3 M Dias 53 D Sp ek ctr um B Sp ec tru m A 1

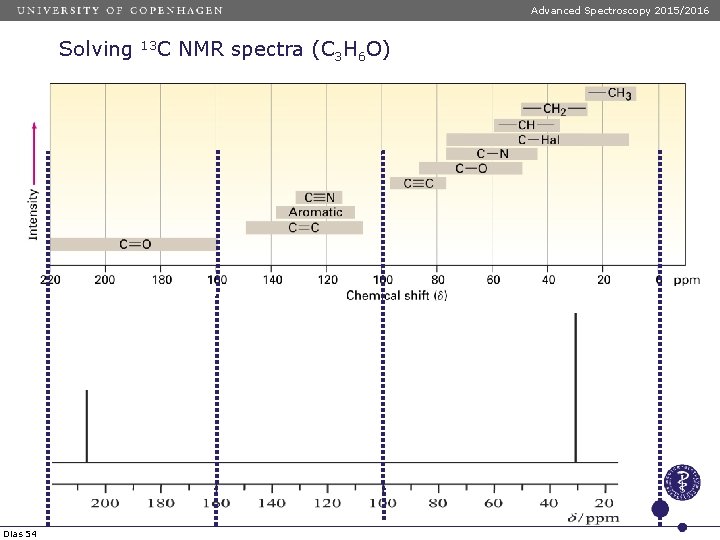
Advanced Spectroscopy 2015/2016 Solving Dias 54 13 C NMR spectra (C 3 H 6 O)

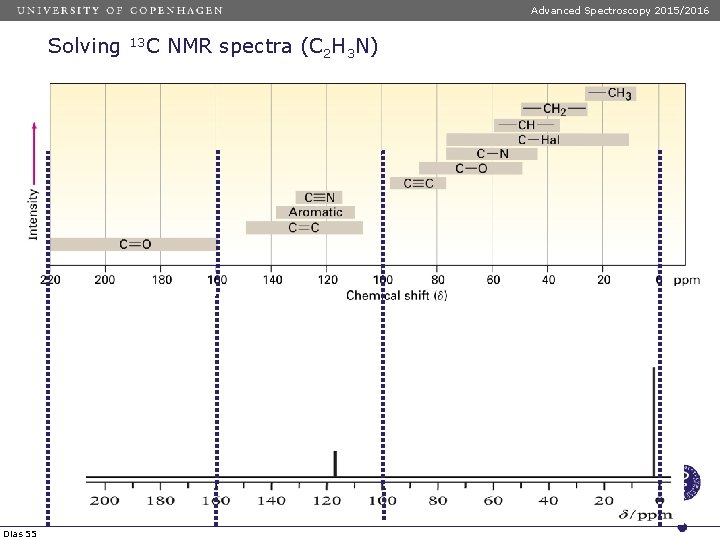
Advanced Spectroscopy 2015/2016 Solving Dias 55 13 C NMR spectra (C 2 H 3 N)

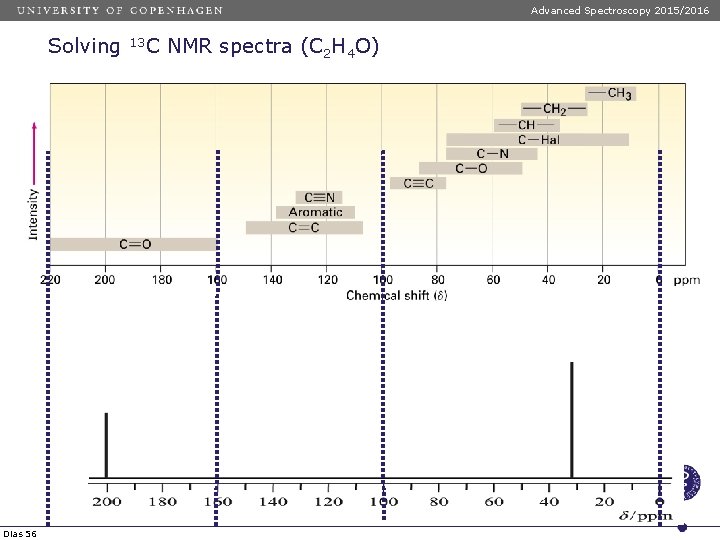
Advanced Spectroscopy 2015/2016 Solving Dias 56 13 C NMR spectra (C 2 H 4 O)

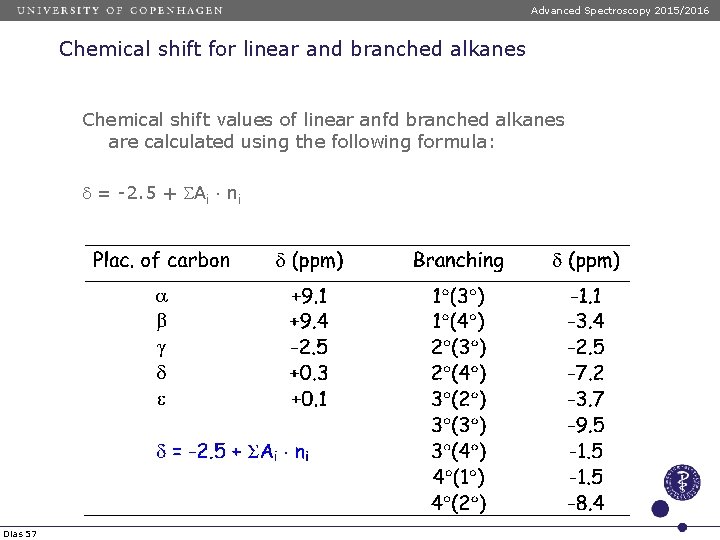
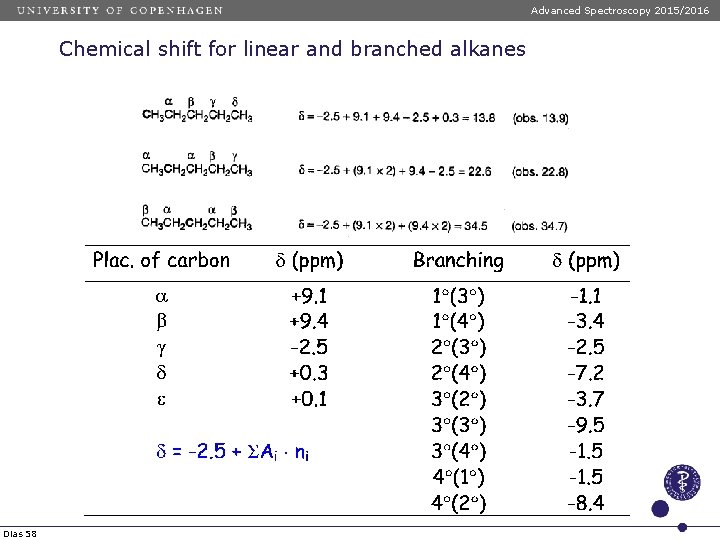
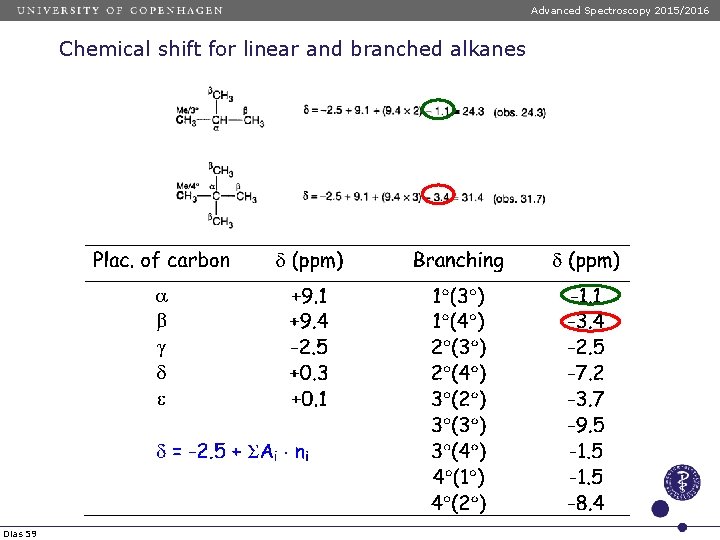
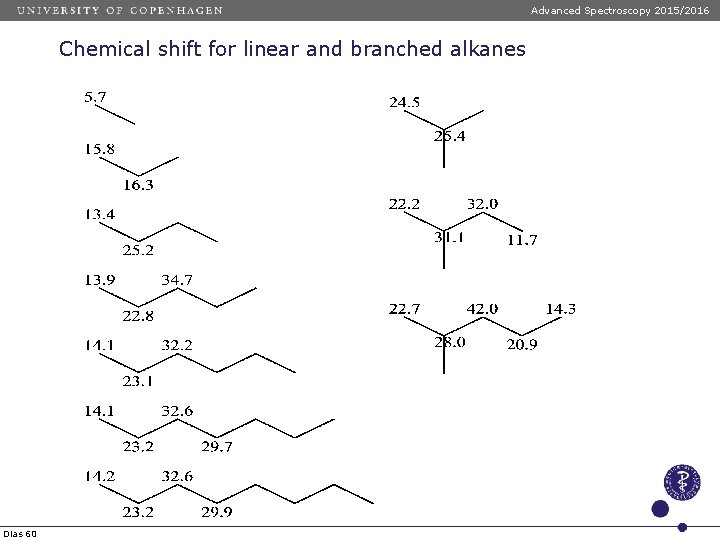
Advanced Spectroscopy 2015/2016 Chemical shift for linear and branched alkanes Chemical shift values of linear anfd branched alkanes are calculated using the following formula: d = -2. 5 + SAi ni Dias 57

Advanced Spectroscopy 2015/2016 Chemical shift for linear and branched alkanes Dias 58

Advanced Spectroscopy 2015/2016 Chemical shift for linear and branched alkanes Dias 59

Advanced Spectroscopy 2015/2016 Chemical shift for linear and branched alkanes Dias 60

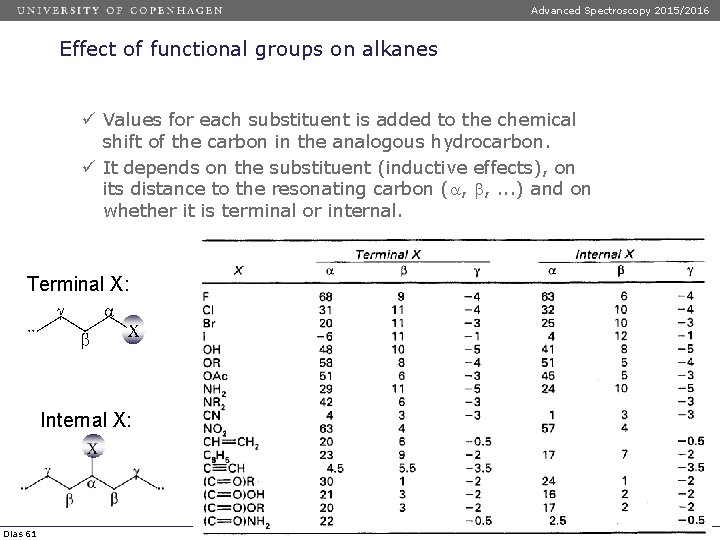
Advanced Spectroscopy 2015/2016 Effect of functional groups on alkanes ü Values for each substituent is added to the chemical shift of the carbon in the analogous hydrocarbon. ü It depends on the substituent (inductive effects), on its distance to the resonating carbon ( , , . . . ) and on whether it is terminal or internal. Terminal X: Internal X: Dias 61

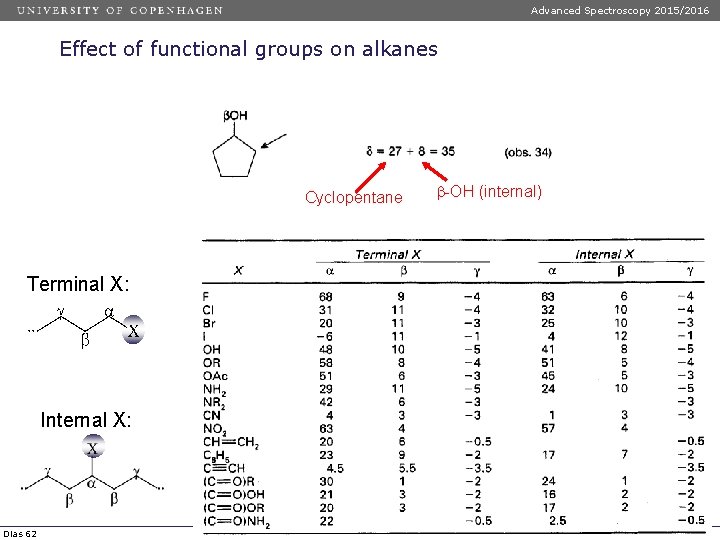
Advanced Spectroscopy 2015/2016 Effect of functional groups on alkanes Cyclopentane Terminal X: Internal X: Dias 62 -OH (internal)

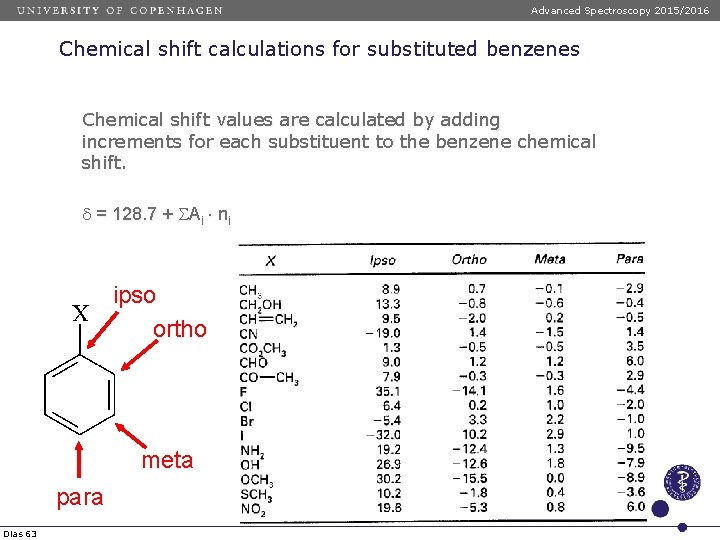
Advanced Spectroscopy 2015/2016 Chemical shift calculations for substituted benzenes Chemical shift values are calculated by adding increments for each substituent to the benzene chemical shift. d = 128. 7 + SAi ni ipso ortho meta para Dias 63

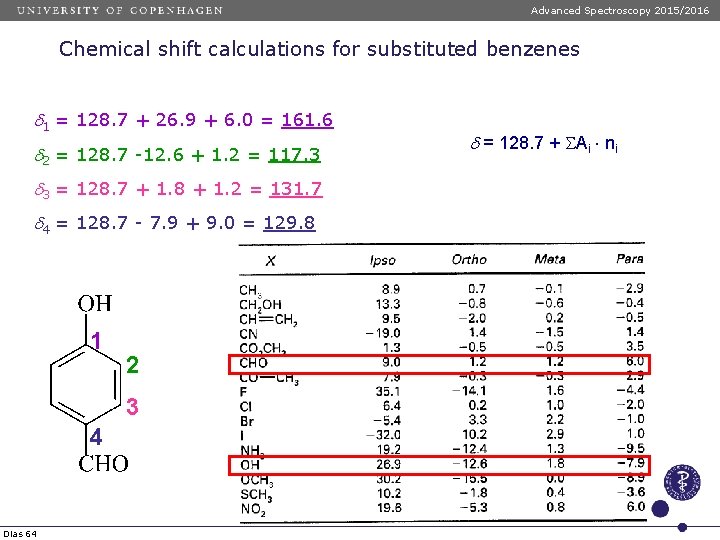
Advanced Spectroscopy 2015/2016 Chemical shift calculations for substituted benzenes d 1 = 128. 7 + 26. 9 + 6. 0 = 161. 6 d 2 = 128. 7 -12. 6 + 1. 2 = 117. 3 d 3 = 128. 7 + 1. 8 + 1. 2 = 131. 7 d 4 = 128. 7 - 7. 9 + 9. 0 = 129. 8 1 2 3 4 Dias 64 d = 128. 7 + SAi ni

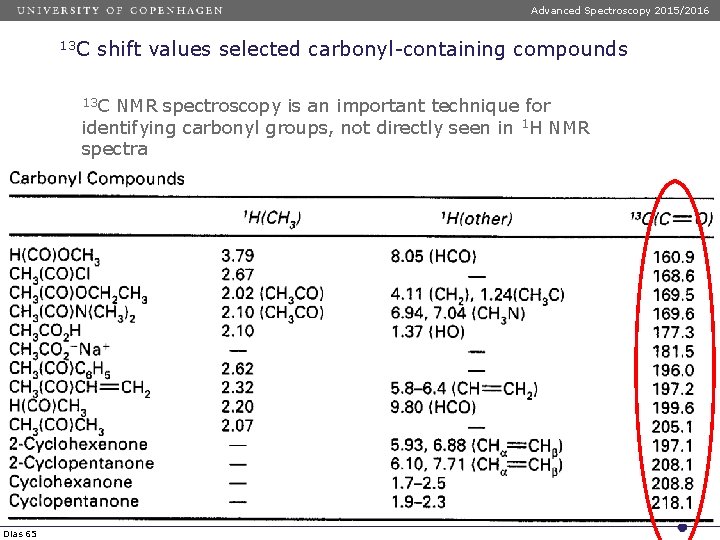
Advanced Spectroscopy 2015/2016 13 C shift values selected carbonyl-containing compounds 13 C NMR spectroscopy is an important technique for identifying carbonyl groups, not directly seen in 1 H NMR spectra Dias 65

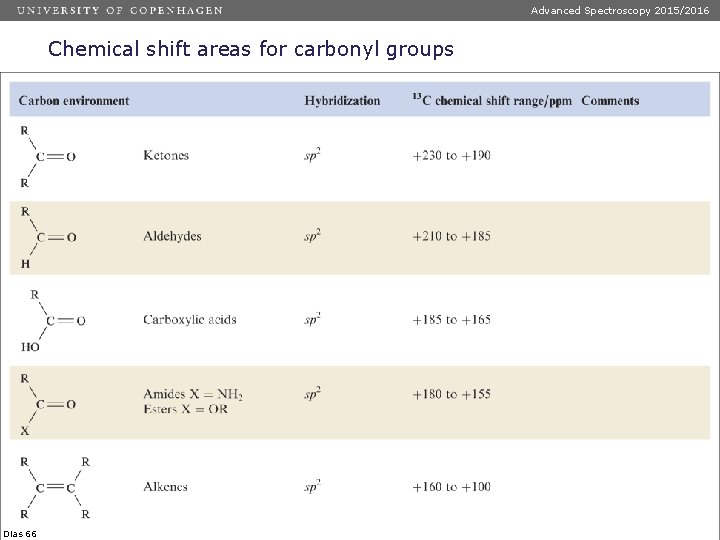
Advanced Spectroscopy 2015/2016 Chemical shift areas for carbonyl groups Dias 66