Advanced Inorganic Chemistry By Dr MAHMOUD NAJIM Angular

Advanced Inorganic Chemistry By Dr. MAHMOUD NAJIM Angular Overlap Method

Molecular orbital diagram (Fig. 1)does not provide a complete understanding of theory of complexes or a method to quantify the energies involved and one way to approach this problem is AOM which can illustrate a qualitative discussion of the physical rationale for theory. Fig. 2 showed that the interaction of two atomic orbitals having symmetry appropriate for positive overlap to give a bonding and antibonding orbital with energy determined by the overlap and the energies of the original orbitals. The degree of the overlap (SML) will depend upon the angle between the orbitals ( Fig. 3) which can be used as a basis of AOM to treat the coordination compounds by MOT assumed to the following equation: SML = Sσ cosѲ …………. 1 The energy of interaction Ε is taken as: Ε = βS 2 ……………. . 2 ML Were β is constant inversely proportional to the deference in energy between the original orbitals. Because it is constant for given metal ion and set of ligands, so only the angular dependence of the overlap integral need to be considered.
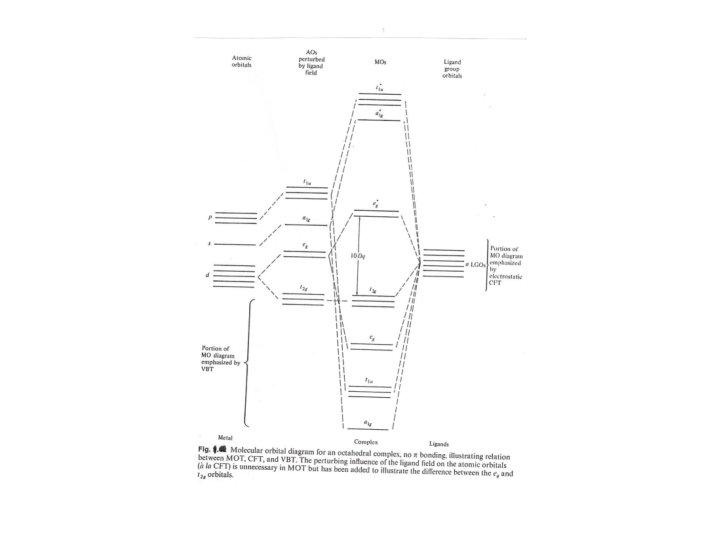
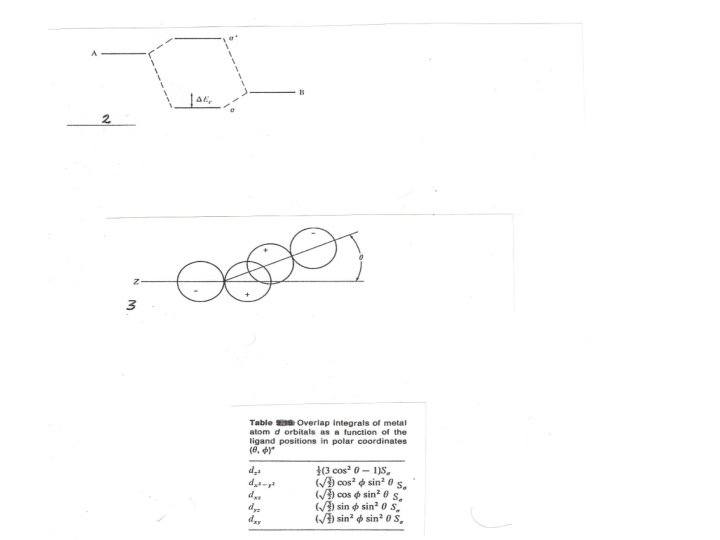

For the overlap of dz 2 orbital with a ligand on the z axis(Ѳ =0 ) and according to the functions for d orbitals given in Table we may rewrite the above equation as follows: 2 Ε = β Sσ f(Ѳ, Φ ) ………………… 3 By using of the last equation and the Table considering the two eg orbitals of an octahedral set we can evaluate SML = ½. 4 √ 3/2 Sσ …………. . 4 Substitution of eq. 4 on eq. 2 we can have Ε = 3βS σ2 ……………. 5 The same result should be determined for the second eg orbital Fig. 4 showed a comparison of the results from the AOM and CFT , these methods defined that 10 Dq = 3β S σ2
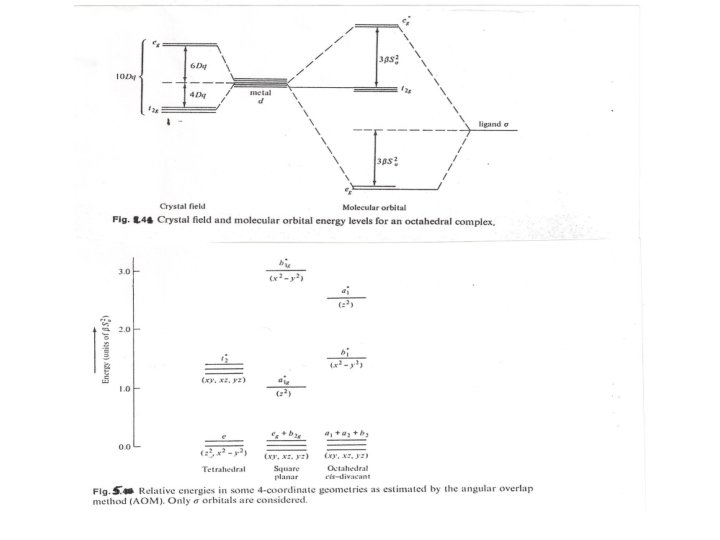

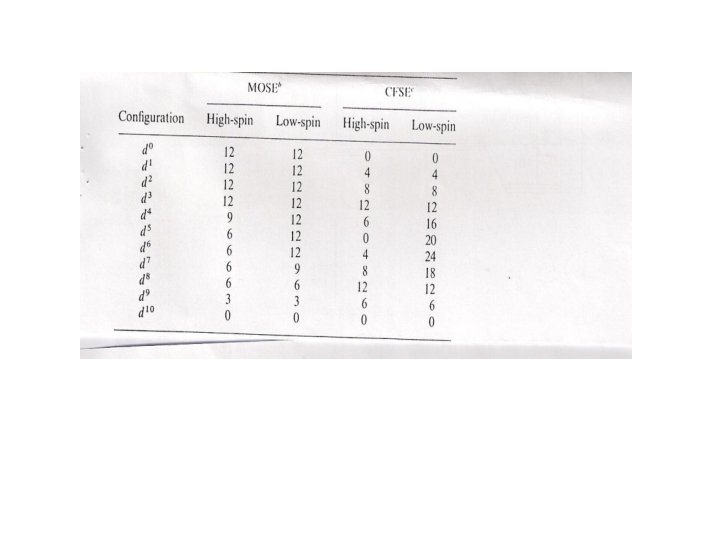
The source of the splitting energy of the t 2 g and eg(eg*) for AOM and CFT was referred to different causes. For CFT is the barycentre of the unpaired d orbitals while it is the barycentre for the unpaired d orbitals and the ligand σ donor orbitals for AOM, so it becomes 12βS σ2 of MOSE immediately after four ligand electrons occupying the eg level for the metal ( 3βS 2 σfor each e) without regard to d electrons. Changing from d 1 - d 3 cause no change in the MOSE because t 2 g level strictly non bonding with σ-only system, although high spin d 4 will be less stabilized (12 - 3)βS σ2 similar to weak field case (Table). For various geometry it is easy to determine SML according to the Table and by repeating the previous calculation we can obtain: 2 EZ 2 = (1/4+ ¼+ ¼ +1/4 )βSσ …………… 6 2 2 =( Ex - y ¾+ 3/4 +3/4) βS 2 σ …………. . 7 For square planar the destabilizations of βS σ2 for occupation of the σ*Z 2 antibonding orbital and of 3βS σ2 for the occupation of σ* 2 x – y 2 orbital

For the occupation of σ*x 2 – y 2 orbital t 2 and e levels in tetrahedral symmetry …………. 8 Exz = Exy = Eyz = 4/3 βS σ2 Ex 2 – y 2 = Ez 2 = 0 ……………. . 9 This last equation is of considerable interest which can show that Dq(tet)=4/9 Dq(oct) as below 2 2 4/3 βSσ ( tetr. ) / 3βSσ ( oct. ) = 4/9 …………. 10 As a comparison between VBT, CFT and MOT we can refer to the following: 1 Both CFT and MOT described coordination complexes by the existence of two sets of 2 orbitals separated by an energy gaps, 10 Dq = 3βSσ , if the energy to pair the electrons is greater than this , high spin complexes will form otherwise low spin will formed. 2 Both CFT and MOT showed that the visible spectra of the complexes are attributed to electronic transitions such as t 2 g → eg* , but the fundamental assumptions were different. 3 Both CFT and MOT describe complexes in terms of interactions metal orbitals and the ligands and with the greater the interactions the greater of the amount of 10 Dq, and then the greater metal- ligand bond not from the amounts of the electrostatic effects. 4 VBT concentrate only on the formation of d 2 sp 3 hybridization which agree with the MOT description ( a 1 g, t 1 u and eg molecular orbitals).
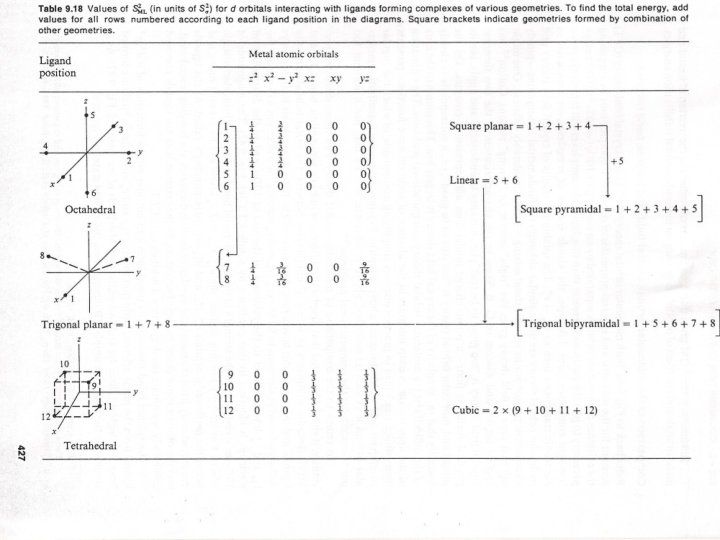

π- Bonding in metal complexes π - Bonding is very useful in explaining the stability of many complexes , in addition to σ-bonding formed between M-L Some ligands can formed π - Bonds when it consists appropriate orbitals for π - Bonding with a metal d-orbitals (t 2 g) Three types of ligands can do that: 1 Ligands with p-orbitals perpindicular to the σ-bond 2 Ligands with a d-orbital lying in a plane that include the metal atom. 3 Ligands with a π* orbital lying in a plane that include the metal atom ( Fig. 6 a, b and c respectively) The effect of the π bonding on the value of ∆o depends on whether the π ligand orbitals act as electron donors or acceptors There are two principles to be described , the first when atomic orbitals overlap strongly , they mix strongly , and the resulting bonding molecular orbitals are significantly lower in energy and the antibonding molecular orbitals are significantly higher in energy than the atomic orbitals. Second when the two atomic orbitals have similar energies they interact strongly , but with large different energies they mix only slightly even if there overlap is large.
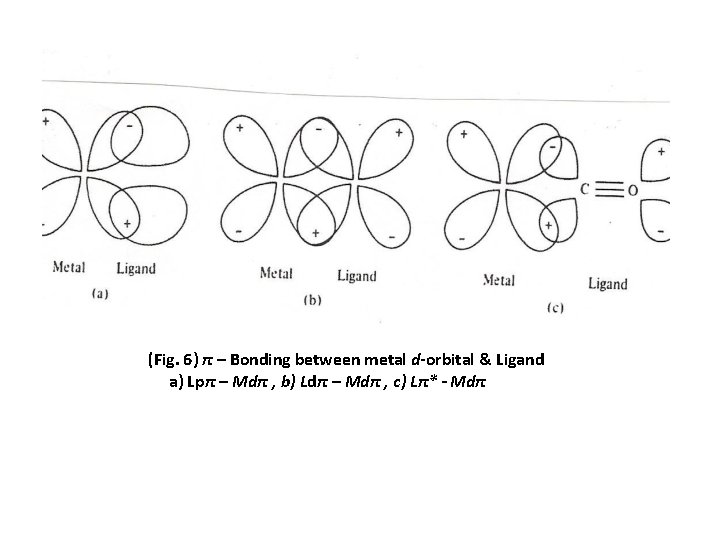
(Fig. 6) π – Bonding between metal d-orbital & Ligand a) Lpπ – Mdπ , b) Ldπ – Mdπ , c) Lπ* - Mdπ

1 The ligand with filled orbitals has no low energy vacant π orbitals ( like Cl-, H 2 O), and they lie lower in energy than the partially filled d orbitals of the metals. They form a molecular orbitals, bonding lower than the ligand orbitals and antibonding lie above the energy of the d orbitals of the free metal ion. (Fig. 7). As a result a strong π- donor ligand interaction decrease ∆o. 2 A ligands that can accept electrons into their π orbitals , we will see that such ligand lie higher in spectrachemical series and give rise to a large ligand field splitting parameter. These ligands has filled π orbitals at lower energies than metal t 2 g orbitals and low energy empty π orbitals that are available for occupation. The π-acceptor orbitals are vacant antibonding orbitals on the ligand, as in CO and N 2, and these orbitals lie above the metal d orbital in energy. When these vacant π* ligand orbitals are close in energy to the metal t 2 g orbitals and the metal-ligand π overlap is strong, a little e density will be delocalized from the metal to the ligand. As example the π* orbital of CO has its larger amplititude on the C atom, while the full bonding π orbitals of CO is low in energy and is largely localized on the O atom( as O is more electronegative than C), and will make CO behave as a net π acceptor. Most ligands with π acceptor orbitals are higher in energy than the metal d orbitals, they form molecular orbitals with the metal in which d orbitals character are lowered in energy, and the net result is that ∆o is increased by the π-acceptor interaction
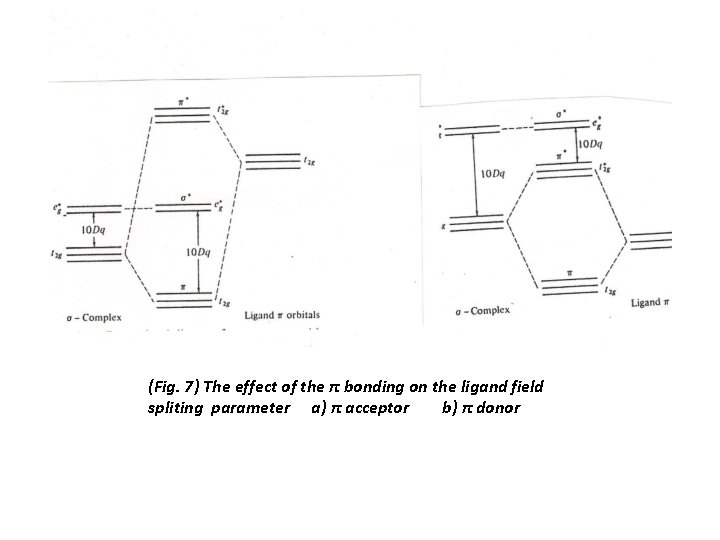
(Fig. 7) The effect of the π bonding on the ligand field spliting parameter a) π acceptor b) π donor

The important rule on π bonding is clear on the order of the ligands in the spectrochemical series which can be divided as below: π -donor < weak π -donor < no π effects< π –acceptor I- < Br- < Cl- < F- < H 2 O < NH 2 < PR 3 < CO Tetrahedral species can be divided roughly into two broad clases: 1 Oxo species in which the metal ion is high in oxidation state ( > or =6)and be very extensive π bonding(Mn. O 4 -, Cr. O 4 -) 2 Complexes with lower oxidation state ( +2, +3) and the ligands are halide ions, amine or RO-
- Slides: 15