Advanced Higher Biology Unit 1 Laboratory Techniques for

Advanced Higher Biology Unit 1 Laboratory Techniques for Biologist

Health and Safety • Identifying and controlling hazards and assessing risk. • The use of physical or biological control measures including the use of personal protective equipment. • Standard laboratory rules and familiarity with risk assessment. • Chemicals or organisms can be intrinsically hazardous. Their use may involve risks to people, to other organisms or to the environment. • The use of control measures, including personal protective equipment as a last resort, to reduce risk. Biological control includes using a more suitable strain of microorganism e. g. less virulent.

What is a Hazard? • anything that poses a potential threat to an individual or the environment. – E. g. physical, chemical, biological, psychological, radiation, ecological, sociological

Risk • The likelihood of a hazard having an impact is the risk • Risk should be considered in terms of; 1. How much harm would exposure to the hazard cause? 2. How much exposure would create impact? • In science labs there will be multiple hazards; some in terms of equipment, some with the chemicals/biologicals involved. Identifying each possible hazard and considering each in terms of need for control is an integral part of designing an experiment.

Methods of Control • Once a hazard has been identified and is considered to have a risk, you should put in place controls. – Eliminate – Substitute – Isolate – Administrate – Personal protective Equipment(PPE) A risk assessment is a full consideration of all the hazards and the potential risk with controls that can be taken to minimise the risk.

• Eliminate – If you can remove the hazard from the procedure then this should be done. • Substitute – be alternatives that do not produce the same level of risk e. g. Biological control includes using a more suitable strain of microorganism • Isolate – isolating particular hazards is a powerful control mechanism e. g. use of fume cupboards, • Administrate – standard operating procedures-SOPs • PPE – This can include dust masks, air filters, suits, gloves, aprons, and footwear.

Health and Safety • Identifying and controlling hazards and assessing risk. • The use of _____ or _____l control measures including the use of personal protective equipment. • Standard laboratory rules and familiarity with risk assessment. • ______ or ______ can be intrinsically hazardous. Their use may involve risks to people, to other organisms or to the environment. • The use of control measures, including personal protective equipment as a last resort, to reduce _______control includes using a more suitable strain of _____ e. g. less virulent.

Health and Safety • Identifying and controlling hazards and assessing risk. • The use of physical or biological control measures including the use of personal protective equipment. • Standard laboratory rules and familiarity with risk assessment. • Chemicals or organisms can be intrinsically hazardous. Their use may involve risks to people, to other organisms or to the environment. • The use of control measures, including personal protective equipment as a last resort, to reduce risk. Biological control includes using a more suitable strain of microorganism e. g. less virulent.

Liquids and Solutions • Dilution series are often linear or log. Standard curves, measurement and determination of an unknown concentration. The use of buffers to maintain and control p. H. • Practise measuring and making solutions and using buffers before embarking upon important experimentation. • Use a colorimeter or spectrophotometer to calibrate a known solution and determine an unknown using eg Bradford reagent. • Use of cylinders, pipettes, burettes, autopipettors and syringes. p. H can be measured using a meter or an indicator. The concentration of a pigmented compound can be quantified using a colorimeter.

Equipment • Biological systems are often liquid or in solution, even if not originally, they are frequently put into solution for analysis. • Apparatus when working with liquids should be considered in terms of; accuracy, precision and the volumes involved. Beakers (care with the approx. ), measuring cylinders and pasteur pipettes are common basic pieces of apparatus.
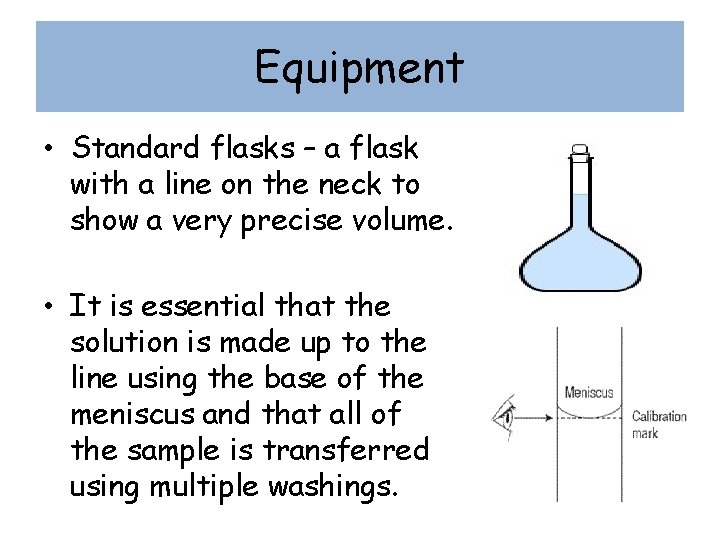
Equipment • Standard flasks – a flask with a line on the neck to show a very precise volume. • It is essential that the solution is made up to the line using the base of the meniscus and that all of the sample is transferred using multiple washings.

Equipment • Pipettes- graduated or with one line for the set volume. Care must be taken when reading to the base of meniscus, and when moving from drawing liquid to the air • Burettes – range in size and graduation, used for titrations and accurate measurements of small volumes. • Micropipettes/ Autopipettes – a dial on the handle can be used to set a specific volume

Dilutions • Solutions are diluted for many reasons, depending on what the solution is to be used for the method of dilution can vary.

Linear Dilutions • Used if you need a sample over a reasonably small range. Taking a proportion from the stock and diluting using the solvent (normally distilled water) to bring up to volume. • e. g. making up samples at 1, 2, 3, 4 and 5% or even 1, 2, 5, 15 and 25 % - both would still be plotted over a continuous even scale

Serial Dilutions Where dilution is needed across a wide range a serial dilution produces samples which differ from the next by an order of magnitude each time http: //www. wikihow. com/Do-Serial 1 ml 1 ml Dilutions

Log Scales • Serial dilutions give you a scale that can be represented in different ways. • Often the numbers are given with no indication that the scale is a log scale and you must be very careful when reading off the values.
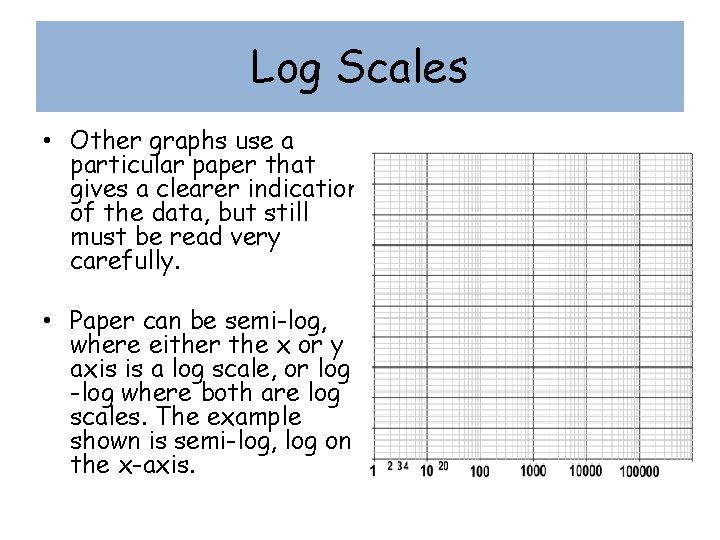
Log Scales • Other graphs use a particular paper that gives a clearer indication of the data, but still must be read very carefully. • Paper can be semi-log, where either the x or y axis is a log scale, or log -log where both are log scales. The example shown is semi-log, log on the x-axis.

Buffers • p. H is a continuous scale indicating the hydrogen ion concentration in a solution. Specifically the inverse log of the hydrogen ion concentration (p. H=-log[H+]). • A buffer is something which resists p. H change, it is a mixture of compounds, one of which removes ‘extra’ hydrogen ions, another which removes ‘extra’ hydroxides from its original set point. • Buffers can be used to set and maintain a particular p. H, since biological cells and systems often require a specific p. H range this is very useful.

Practical techniques • http: //www. ruf. rice. edu/~bioslabs/methods/s olutions/stocks. htm

Liquids and Solutions • Dilution series are often ______ or _____. Standard curves, measurement and determination of an unknown concentration. The use of _____ to maintain and control p. H. • Practise measuring and making solutions and using buffers before embarking upon important experimentation. • Use a _____ or spectrophotometer to _____ a known solution and determine an unknown using eg Bradford reagent. • Use of cylinders, pipettes, burettes, autopipettors and syringes. _______ can be measured using a meter or an indicator. The concentration of a pigmented compound can be quantified using a _____.

Liquids and Solutions • Dilution series are often linear or log. Standard curves, measurement and determination of an unknown concentration. The use of buffers to maintain and control p. H. • Practise measuring and making solutions and using buffers before embarking upon important experimentation. • Use a colorimeter or spectrophotometer to calibrate a known solution and determine an unknown using eg Bradford reagent. • Use of cylinders, pipettes, burettes, autopipettors and syringes. p. H can be measured using a meter or an indicator. The concentration of a pigmented compound can be quantified using a colorimeter.

Separation Techniques • Separation techniques use solubility, size, shape and charge • Examples of separation techniques are centrifugation, paper, thin layer and affinity chromatography, protein electrophoresis, separation of proteins and iso-electric points.

Separation Techniques • Biological systems often involve mixtures, these could be of; cells, DNA fragments, proteins and other compounds. • To analyse systems properly you must be able to separate the component parts and to do this you need something that can differentiate between the parts. • This can be differences in; solubility, size, shape or charge, or indeed any combination of these attributes.

Centrifugation • A centrifuge is a piece of apparatus that spins tubes very fast, as it spins the heavier components will move further down the tube. • The solid found at the base of the tube is called the pellet and the liquid the supernatant. • https: //www. youtube. com/watch? v=ncr-9 i. MEzw. U
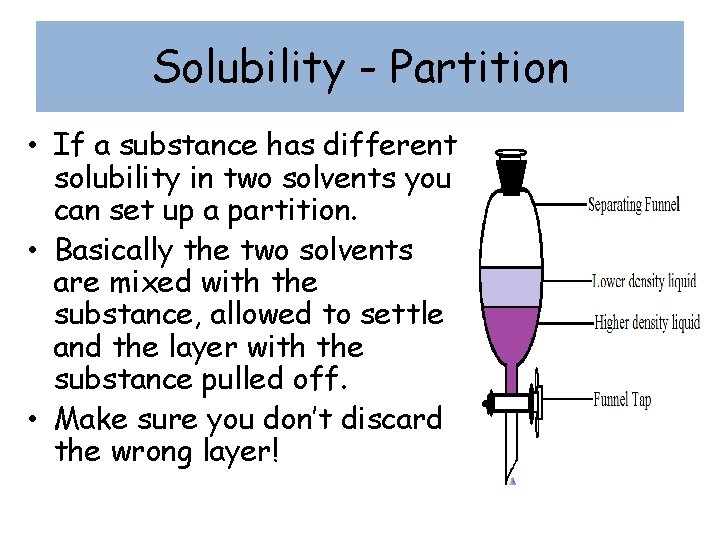
Solubility - Partition • If a substance has different solubility in two solvents you can set up a partition. • Basically the two solvents are mixed with the substance, allowed to settle and the layer with the substance pulled off. • Make sure you don’t discard the wrong layer!

Solubility -Chromatography • uses two phases; a stationary phase and a mobile phase. • The substance is carried along the stationary phase by the mobile phase, when it reaches the limit of its solubility it is ‘dropped’ and deposited on the stationary phase.

Paper Chromatography • Uses paper as the stationary phase and a solvent as the mobile phase. • The mobile phase could be just water, or a more complex mix of solvents to help distinguish between the bands. The solvent moves up (or down depending on how the tank is set up) and compounds are dropped out of solution at particular points in the chromatogram. • The Rf value is calculated 1. 2. 3. 4. 5. 6. Solvent Paper suspended in tank Reference sample Unknown sample Solvent front Origin line

Thin Layer chromatography • TLC (not just tender loving care) • Uses a different stationary phase, commonly cellulose or silica gel spread in a thin layer on top of glass, plastic or metal. • https: //www. youtube. com/watch? v=qdm. KGs k. Cyh 8

Affinity chromatography • This time the stationary phase is chosen to have the ability to bind to the required substance. This could be through chemical bonding, charge or even antibody association. • The mixture is poured through (if gel) or over the stationary phase and the rest discarded. The ‘entrapped’ substance is then removed from the stationary phases using a second process, called ‘elution’.

Affinity chromatography https: //www. youtube. com/watch? v=pn. T 5 87 w. UGy. Y • http: //www. bio. davidson. edu/genomics/m ethod/Affinity. html

Gel Electrophoresis • It is often used for nucleic acids and proteins. • The gel is made of agarose, placed in a buffer and the samples introduced to wells formed when the gel was set in its mould. • The tank is set up with a positive and negative terminal, smaller fragments move more quickly (do not get tangled on the agarose protein strands) than the larger fragments. • This technique can be used in combination with blotting techniques to further isolate the components wanted

Iso-electric point (IEP) • The IEP is the p. H at which a protein has no net charge, this is important for conformational folding in proteins and can be used as a defining value for proteins. • It can be calculated using information on all of the amino acids acid/basic characteristics and their number in the protein or it can be determined using a gel set up with a p. H gradient and measuring the p. H that the protein migrates to (since where it has no net charge it will drop out of solution). • https: //www. youtube. com/watch? v=3 Uo. V 5 mv. NOr 4

Separation Techniques • Separation techniques use _____, size, _______and charge • Examples of separation techniques are _____, paper, thin layer and affinity _____, protein electrophoresis, separation of ____ and iso-electric points.

Separation Techniques • Separation techniques use solubility, size, shape and charge • Examples of separation techniques are centrifugation, paper, thin layer and affinity chromatography, protein electrophoresis, separation of proteins and iso-electric points.

Antibody Techniques • Detection and identification of specific proteins. Immunoassay techniques use antibodies linked to reporter enzymes. Use of labelled antibodies in protein blotting and immunohistochemical staining of tissue. • Creation of monoclonal antibodies. • Monoclonal antibodies used in the diagnosis and detection of disease. • ELISA technique used to identify the presence of specific antigens.

Antibody Techniques • Antibodies are an essential part of the immune system, but have become a very important part of techniques that require specific recognition. • Using an antibody that is tagged with a fluorescent dye or attached to a reporter enzyme, which will produce a visible signal when given the enzyme substrate can be used to identify and/or quantify the presence of a particular antigen.
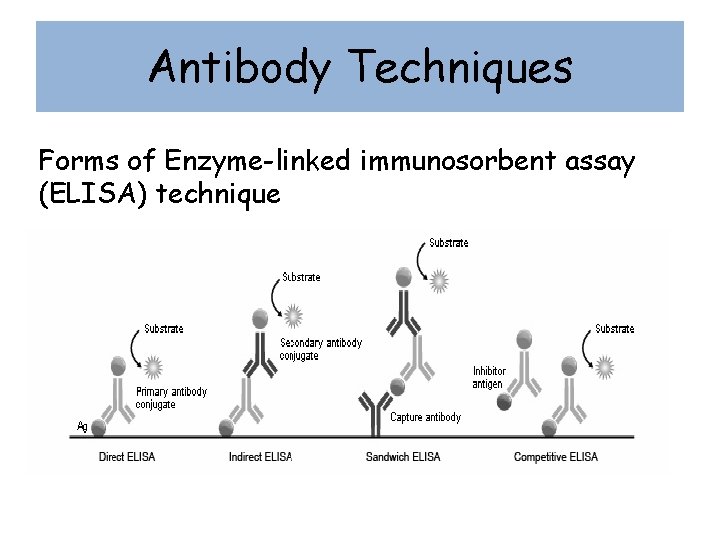
Antibody Techniques Forms of Enzyme-linked immunosorbent assay (ELISA) technique

Monoclonal Antibodies • monoclonal (made from one parent cell) antibodies required the technique of somatic cell fusion is used. • Taking a cell that is immortal (a myeloma cancer cell) and one that produces antibodies (B-lymphocyte) and fusing them together produces a hybridoma – an immortal cell that produces antibodies to specific antigens. • The fusion is most commonly brought about using a particular fusion agent called polyethylene glycol (PEG).

Labelled Antibodies • used for immunohistological staining, where an antibody to a particular antigen can be used to identify particular tissue proteins e. g. used in tissue typing. • Used in conjunction with gel electrophoresis and blotting the antibodies can be used to identify particular proteins e. g. western blotting.

Antibody Techniques • Immunoassay techniques use antibodies linked to ______enzymes. Use of labelled antibodies in protein blotting and immunohistochemical ____ of tissue. • ______ antibodies used in the diagnosis and detection of disease. • ELISA technique used to identify the presence of specific ____.

Antibody Techniques • Immunoassay techniques use antibodies linked to reporter enzymes. Use of labelled antibodies in protein blotting and immunohistochemical staining of tissue. • Monoclonal antibodies used in the diagnosis and detection of disease. • ELISA technique used to identify the presence of specific antigens.

Microscopy • Haemocytometers and flow cytometry are methods of cell counting. • Fluorescence microscopy allows particular protein structures to be visualised. • Bright field microscopy is used to examine whole organisms, parts of organisms or thin sections of dissected tissue.

Microscopy • Resolving power – Measure of how well you can distinguish between two points • Light microscopy – Limited by wavelength of light – Allows you to see to ~0. 2 nm • Electron Microscope – Electron microscope allows you to see ~0. 2 nm

Microscopy • Bright Field Uses a bright light source and the light that passes through gives your image (what you generally consider as a light microscope). • It allows you to look at whole organisms, parts or thin sections of tissue. – For beautiful images of cyclosis in Elodea see here https: //www. youtube. com/watch? v=KOr. F 9 DMCp. AE – For beautiful images of Amoeba see here https: //www. youtube. com/watch? v=rt. D 7 XRha. Ayo • Dark field uses the contrasted areas i. e where the light is blocked as your image

Microscopy • Fluorescence by tagging a particular part of the sample or using a chemical reaction that produces fluorescence even specific proteins can be shown up. • Phase contrast using specific light wavelengths or spin to create the image.
Fluorescent Microscopy https: //www. youtube. com/watch? v=u 0 CRa 0 gi. S 4 For a step-by step introduction see here https: //www. youtube. com/watch? v=f. Zn 6 Fk ixi. HI&list=PLelr. OCut. Xam. N 0 Gbj. BTKGGCAl q. L 7 n. WLt 0 The first few minutes of this is worth watching https: //www. youtube. com/watch? v=Ahzh. O zg. Yoqw&list=PLelr. OCut. Xam. N 0 Gbj. BTKGGC Alq. L 7 n. WLt 0 -&index=2

Cell Counting and Sorting • A basic use of microscopy is to sample the number of cells found in a particular area. • This could be for microorganisms or from blood or tissue samples in multicellular organisms. • This could be done using haemocytometer or flow cytometry

Haemocytometer • special slide that was originally used in blood cell counting. • slide is etched with a very fine grid, of known dimensions. • The slide is thicker than normal, and allows a small volume of liquid to be trapped between the slide and the cover slip (also thicker than normal). • Since the distance from slide to cover slip (0. 1 mm normally) and the widths are all known the volume of liquid can be calculated. • A direct count of the cells can then be used to calculate the cell density. • Some grids have multiple sizes to allow you to pick the most appropriate for the cell type (think spacing on quadrats) and also to allow multiple counts in one sample for averaging.
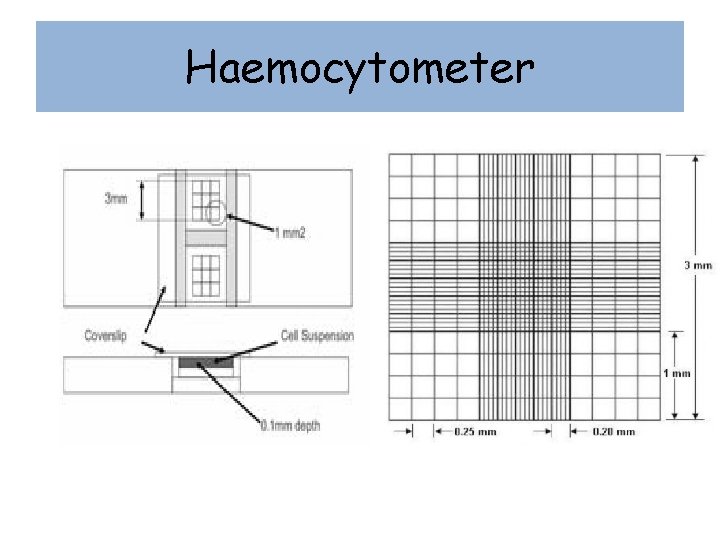
Haemocytometer

Flow Cytometry • Flow cytometry uses a system of light detection to count and sort cells. • Cells are made into single cells suspension, since mammalian cells are contact dependent and attach to each other this is normally done with a protease digestion. • Once in suspension the cells are feed into a funnel connected to an ultrasonic transducer which forces the liquid to form droplets. • The cells are surrounded by a liquid buffer called the sheath fluid and passed in droplets only big enough to contain one cell down the funnel. • A laser is used to count the droplets that contain cells as they fall through.
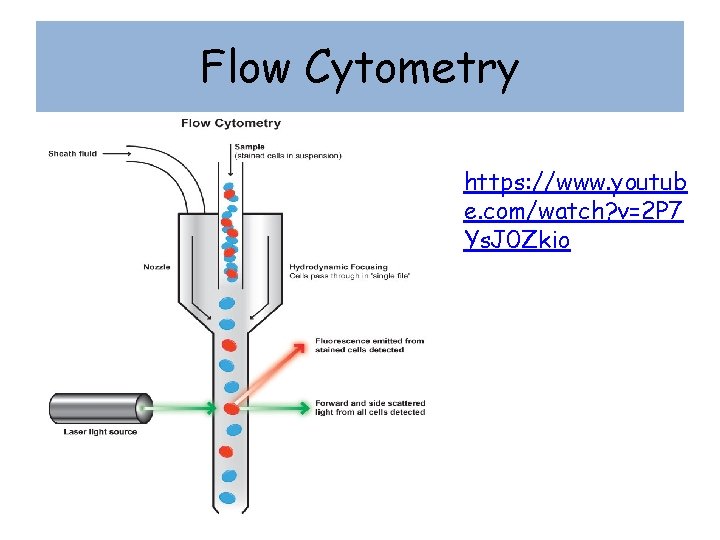
Flow Cytometry https: //www. youtub e. com/watch? v=2 P 7 Ys. J 0 Zkio

FACS (fluorescence-activated cell counter) • Cells are tagged using antibodies connected to fluorescent dyes. • As the light hits the cell, the fluorescing cells scatter the light differently and this information is used to charge the droplet containing the cell, the charged droplet can then be deflected and counted. • The droplets can also be sorted according to the dye level. • This can be adapted to measure the mass of DNA or RNA in a cell, helping to define cells according to their point in the cell cycle.
Microscopy • _____ and flow cytometry are methods of cell _____. • _____ microscopy allows particular protein structures to be visualised. • Bright field microscopy is used to examine whole organisms, parts of organisms or thin sections of _____ tissue.

Microscopy • Haemocytometers and flow cytometry are methods of cell counting. • Fluorescence microscopy allows particular protein structures to be visualised. • Bright field microscopy is used to examine whole organisms, parts of organisms or thin sections of dissected tissue.

Cell Culture and Aseptic Technique • Aseptic technique includes sterilisation of containers, equipment and materials and disinfection of working area. • Explants or cells in inoculum are used for cell culturing • Complex media containing growth factors can be extracted from serum for animal cell lines. • Estimation of viable (active cells) and total cell counts. • Lifetime of primary cell lines are limited unlike cancer cell lines.

Aseptic Technique • Systems that are used to prevent contamination. • This includes – sterilising all containers, equipment and materials used and disinfecting work surfaces prior to and after working • Sterilising process examples could be by use of – heat (dry, flame or steam) or chemical.

Cell Culture • Cells grown in culture are much more easily manipulated an essential part of cell and molecular biology. • Some cells are fairly easy to grow in culture, but many have specific requirements

Cell Culture 1. Inoculum • the inoculum is the starting material you use to grow a culture from. • Plant cells require a minimum inoculum density this may be due to cell signalling molecules required for growth. • Taken from plant or animal tissue this is called an explant. • Other cell types just need one cell and the correct medium e. g. bacterial or fungal cultures.

Cell Culture 2. Nutrient requirements • some cells require particular growth medium, salts, sugars or other substrate molecules in specific combinations 3. Complex Media - Growth factors • plant and animal cells also often have specific growth factors (complexes of proteins) for particular tissue types e. g. EGF – epidermal growth factor for skin cells. • These growth factors are often extracted from the tissue fluid or serum of the animal or plant you are trying to culture.

Cell Culture 4. Anchorage dependence • Mammalian cells, require a surface they can attach to and spread out in a single layer to a confluent layer. • This cuts down the number of cells that can be grown in one vessel so protease digestion is often used to separate cells.

Health of Culture 1. Total cell count – basic measures of cell number can be done using absorption/ transmission. 2. Viable cell count – the number of active cells in your culture, normally uses a form of metabolic tracking. Haemocytometer is used to calculate total and viable cell counts • https: //www. youtube. com/watch? v=p. P 0 x. ERLUh yc

Health of Culture 3. Lifetime of cell lines • primary cell lines are limited in the number of divisions that they can go through before they die. • In human cells this is called the hayflick number which is linked to the number of telomere repeats found at the end of chromosomes. • Immortal cell lines formed from cancer cells do not have this limit and so are often used for mammalian cell culture work.
Cell Culture and Aseptic Technique • _____ technique includes _____ of containers, equipment and materials and disinfection of working area. • _____ or cells in inoculum are used for cell culturing • Complex media containing ______factors can be extracted from serum for animal cell lines. • Estimation of _____ (active cells) and total cell counts. • Lifetime of primary cell lines are ____unlike cancer cell lines.

Cell Culture and Aseptic Technique • Aseptic technique includes sterilisation of containers, equipment and materials and disinfection of working area. • Explants or cells in inoculum are used for cell culturing • Complex media containing growth factors can be extracted from serum for animal cell lines. • Estimation of viable (active cells) and total cell counts. • Lifetime of primary cell lines are limited unlike cancer cell lines.
- Slides: 64