Advanced Chemical Reactions Reaction rates Equilibrium AcidsBases Redox

Advanced Chemical Reactions Reaction rates, Equilibrium, Acids/Bases, Redox Reactions

Entropy (S) Measure of disorder or randomness in a system Natural tendency for system to increase entropy (more random) EXAMPLE – Diffusion ◦ As molecules are dispersed, entropy increases ◦ Continued dispersal leads to a uniform solution

Spontaneous reaction Remember, things tend towards an increase in entropy Spontaneous reaction releases energy ◦ Exothermic ◦ Solid gas increases entropy Gibbs free energy – max amt of E that can be used in another process

2 nd law of thermodynamics Entropy never decreases in a system and instead will increase over time UNLESS you change the surroundings ◦ Spraying air freshener ◦ Spray it into a collapsible box

Kinetics Study of reaction rates (rate at which a chemical reaction takes place) Measured by: ◦ Rate of formation of products ◦ Rate of disappearance of reactants ◦ Changes in concentration of reactants or products

Factors that Affect Rxn Rate Concentration Pressure Temperature Surface Area All of the above have a DIRECT relationship

When do chemical reactions occur? When reactants collide Normally, molecules bounce off each other b/c of electron clouds repulsion BUT, if those molecules have a LARGE amount of energy, they can overcome the repulsion and react Molecules also must collide in the right orientation
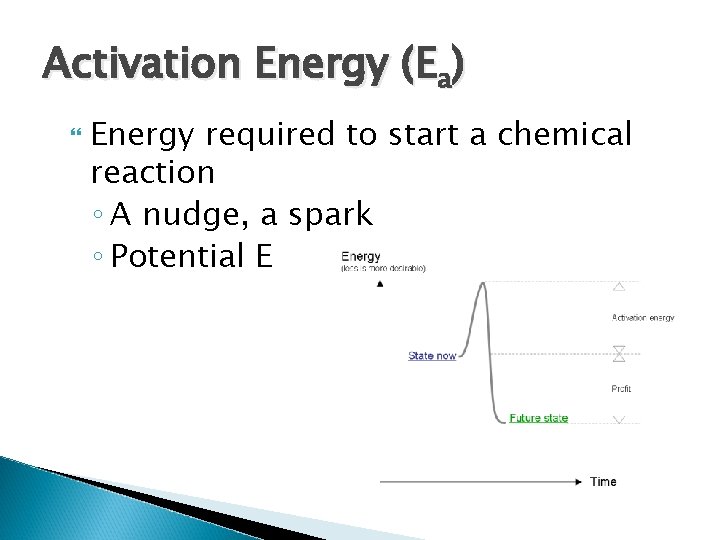
Activation Energy (Ea) Energy required to start a chemical reaction ◦ A nudge, a spark ◦ Potential E
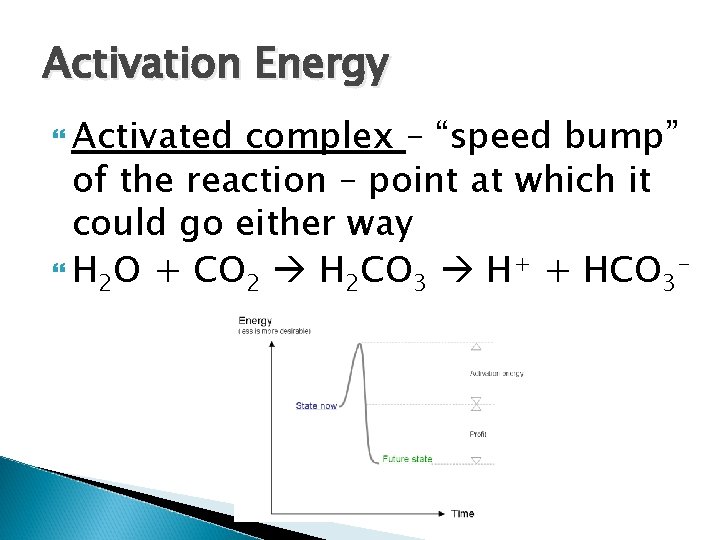
Activation Energy Activated complex – “speed bump” of the reaction – point at which it could go either way H 2 O + CO 2 H 2 CO 3 H+ + HCO 3 -
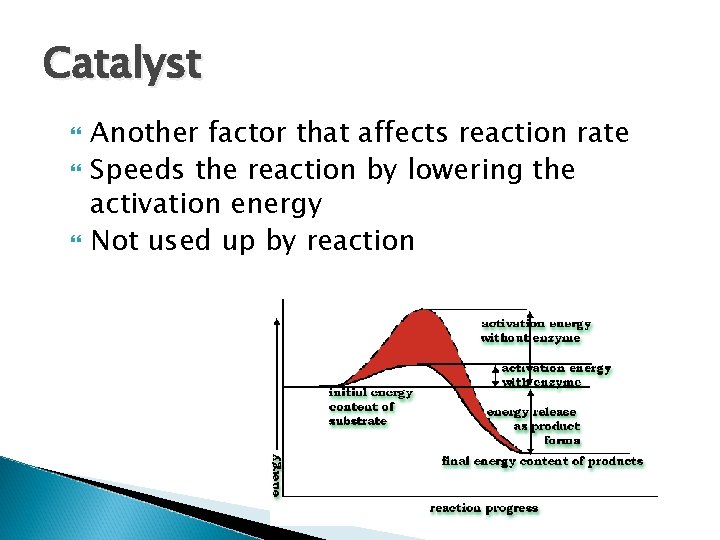
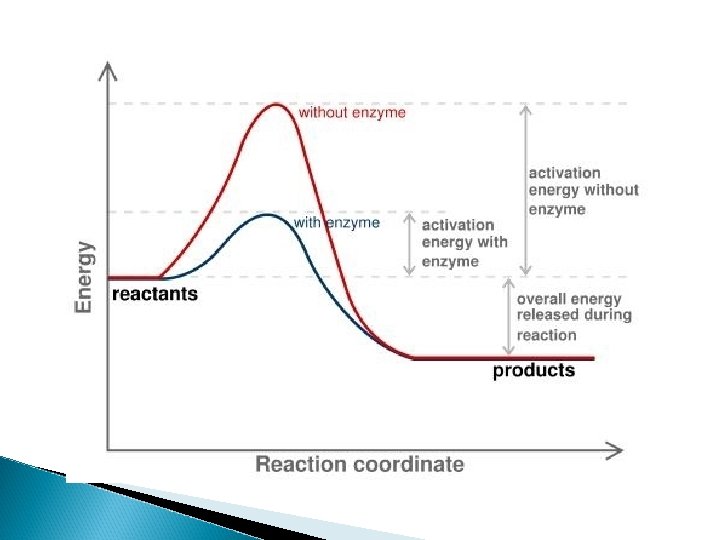
Catalyst Another factor that affects reaction rate Speeds the reaction by lowering the activation energy Not used up by reaction

Equilibrium Two basic categories for reactions 1. Completion reactions – 1 -way (combustion, decomp, rusting) 2. Reversible reactions – products can re-form original reactants Reversible reactions often use 2 arrows b/c reactions occur at the same time

Equilibrium Chemical equilibrium is DYNAMIC, not STATIC

Equilibrium Chemical equilibrium – reactions in which the forward and reverse reaction rates are equal

Equilibrium Constant (Keq) Every reaction has a condition of equilibrium at a given temperature That means that 2 reactants will react to form products until a state is reached where the amounts of products and reactants no longer change Things will stay that way until the system is somehow altered

Le Chatelier’s Principle When a system at equilibrium is disturbed, the system adjusts in a way to reduce the change. Chemical equilibria responds to 3 kinds of stress or change 1. Change in concentration 2. Change in temperature 3. Change in pressure

Changes in Concentration Increasing concentration of reactant will make the rate of the forward reaction faster than the reverse ◦ Called a shift right ◦ Continues until new equilibrium H 3 O+ + HCO 3 2 H 2 O + CO 2 Increasing concentration of product leads to shift left

Changes in Pressure Only affects gases Imagine volume has been decreased, increasing the pressure Immediate effect is increase in concentration of both product & reactant According to principle, system will adjust to decrease the pressure

Changes in Pressure A pressure increase favors the reaction that produces fewer molecules (stoichiometry) 2 NOCl 2 NO + Cl 2 H 2 O + CO H 2 + CO 2

Changes in Temperature Remember that endothermic & exothermic are opposites Increasing the temp adds E so the endothermic will go faster to use it If it is exothermic forward, increasing the temp favors the reactants If it is endothermic forward, increasing the temp favors the products

Acids & Bases

Characteristics Arrhenius definitions Acids – sour taste, p. H less than 7, turn litmus paper red – example = vinegar ◦ Generate hydronium ions (H 3 O+) or H+ Bases – bitter taste, slippery feel, p. H higher than 7, turn litmus paper blue – example = ammonia ◦ Generate hydroxide ions (OH-) Outdated definitions because not always true

STRONG vs WEAK Strong acids & bases COMPLETELY dissociate or ionize in water (one way reaction) ◦ HNO 3 + H 2 O H 3 O+ + NO 3◦ Na. OH Na+ + OHWeak acids & bases only partially dissociate (reversible reaction) ◦ HOCl + H 2 O H 3 O+ + Cl. O◦ NH 3 + H 2 O NH 4+ + OH-

Bronsted-Lowry Acid – proton donor (H+) Base – proton acceptor (H+) u. NH 3 + H 2 O NH 4+ + OHu. H 2 O is the Bronsted-Lowry acid & NH 3 is the Bronsted-Lowry base u. Always reactants

Conjugate Acid – Formed when a proton is gained (H+) Conjugate Base – Formed when an proton is lost (H+) u. NH 3 + H 2 O NH 4+ + OH NH 4+ is the conjugate acid & OHis the conjugate base Always products

Amphoteric Can act as an acid or a base depending on what it is combined with

Water Can act as a Bronsted-Lowry acid or base H 2 O + H 2 O H 3 O+ + OH Called the self-ionization of water Results in equal concentrations of H 3 O+ and OH- in pure water [H 3 O+] = [OH-] = 1. 00 x 10 -7 M
![Water [H 3 O+] x [OH-] = 1. 00 x 10 -7 x 1. Water [H 3 O+] x [OH-] = 1. 00 x 10 -7 x 1.](http://slidetodoc.com/presentation_image_h/24d19801eb9b7dd3445db327396e1fa8/image-28.jpg)
Water [H 3 O+] x [OH-] = 1. 00 x 10 -7 x 1. 00 x 10 -7 = 1. 00 x 10 -14 Found to be true for other aqueous solutions at equilibrium [H 3 O+] x [OH-] = 1. 00 x 10 -14 Also abbreviated as Kw
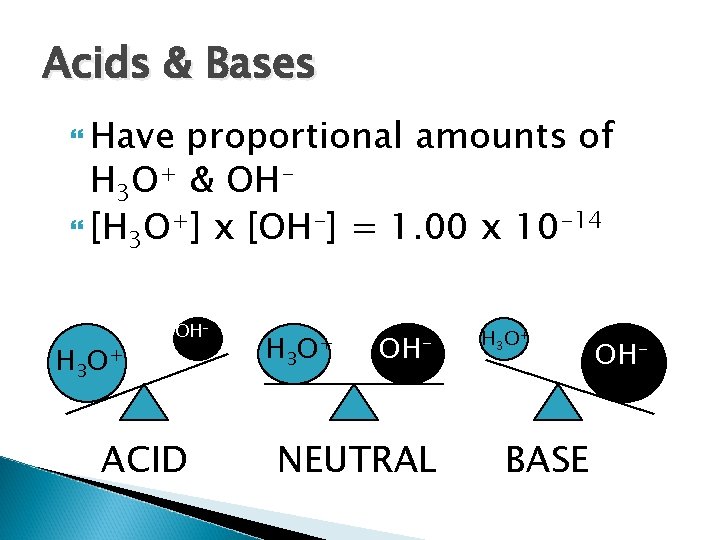
Acids & Bases Have proportional amounts of H 3 O+ & OH [H 3 O+] x [OH-] = 1. 00 x 10 -14 H 3 O+ OH- ACID H 3 O+ OH- NEUTRAL H 3 O+ BASE OH-
![Calculating [H 3 O+] & [OH-] using Kw [H 3 O+] x [OH-] = Calculating [H 3 O+] & [OH-] using Kw [H 3 O+] x [OH-] =](http://slidetodoc.com/presentation_image_h/24d19801eb9b7dd3445db327396e1fa8/image-30.jpg)
Calculating [H 3 O+] & [OH-] using Kw [H 3 O+] x [OH-] = 1. 00 x 10 -14 If [H 3 O+] = 1. 00 x 10 -2, what is [OH-]? [OH-] = 1. 00 x 10 -12 If [H 3 O+] = 1. 00 x 10 -5, what is [OH-]? [OH-] = 1. 00 x 10 -9

The p. H Scale 1909 – Sorenson – negative exponents are annoying… So let’s just look at the exponents! Logarithm – power to which 10 must be raised to equal that number log 100 = 2 because 100 = 102 log 0. 001 = -3 because 0. 001 = 103

Logarithm Practice log log 10, 000 = 0. 01 = 10 = 0. 000001 = 1=

The p. H Scale Represents the “power” of “Hydrogen” p. H = - log [H 3 O+] What is the p. H of a HNO 3 solution with a [H 3 O+] of 0. 00010? p. H = - log [0. 00010] = -(-4) =4

p. H Practice What is the p. H of a solution of HCl, a strong acid, with a hydronium ion concentration of [0. 2]? p. H = - log [. 2] p. H = 0. 70
![What if it’s a base? [H 3 O+] p. H x [OH-] = 1. What if it’s a base? [H 3 O+] p. H x [OH-] = 1.](http://slidetodoc.com/presentation_image_h/24d19801eb9b7dd3445db327396e1fa8/image-35.jpg)
What if it’s a base? [H 3 O+] p. H x [OH-] = 1. 00 x 10 -14 + p. OH = 14 p. OH = -log [OH-] What’s the p. OH of a base with the [OH-] = 2. 3 x 10 -4? Finding the [OH-] of a strong base is just like finding the [H 3 O+] of a strong acid

Example What is the p. H of a solution of KOH, a strong base, with a hydroxide concentration of 0. 0136 M? p. OH= -log [OH-] = -log [0. 0136] p. OH= 1. 87 p. H + p. OH = 14 So p. H = 12. 13

Neutralization Reaction of H 3 O+ & OH- to form water molecules and often a salt H 3 O+ & OH- 2 H 2 O ◦ Neutral means [H 3 O+] = [OH-] HCl + Na. OH H 2 O + Na. Cl Common way to deal with acid & base spills Baking soda = Na. HCO 3, Ammonia = NH 3

Indicators Change color at a certain p. H level Red cabbage juice – changes to blue between 3 & 4 and to green at 8/9 Litmus paper – red or blue Phenolphthalein – turns bright pink in the presence of a base UNIVERSAL INDICATOR WAND

Titration Used to determine the unknown concentration of a known reactant Uses an indicator to show the equivalence point For strong acid/strong base… Equivalence point is where [H 3 O+] = [OH-] or where moles of acid = moles of base Often uses phenolphthalein
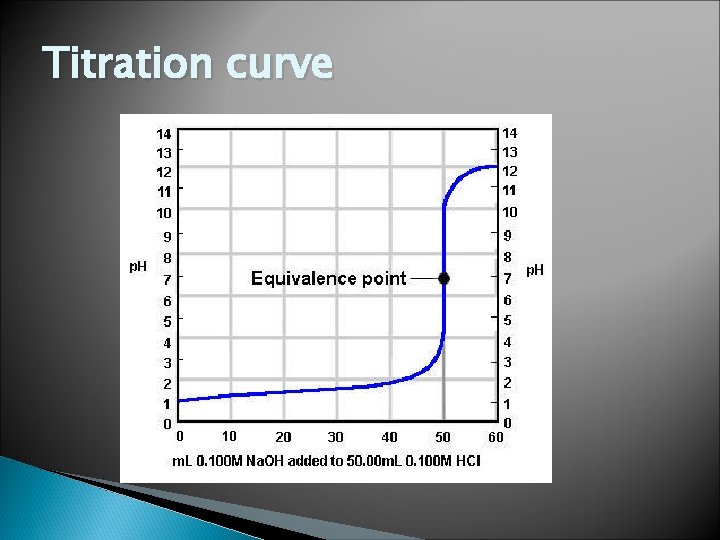
Titration curve

Titration Set-up

Oxidation-Reduction Reactions Remember that electronegativity is a measure of how tightly atoms hold on to their electrons Atoms with large electronegativity differences form ionic bonds by electron transfers 2 Na + Cl 2 2 Na. Cl Can be written 2 Na + Cl 2 2 Na+Cl-

Oxidation-Reduction Reactions 2 Na + Cl 2 2 Na+Cl Oxidation = Loss of electrons (OIL) ◦ 2 Na+ Reduction = Gain of electrons (RIG) ◦ Cl 2 2 Cl These 2 reactions happen together Oxidation-Reduction or REDOX OIL RIG & Leo the lion!


How do we know if an atom is oxidized or reduced? Use “oxidation” numbers The number of electrons that were added or removed in a REDOX reaction Often, it’s the charge the atom would have if it were an ion

Assigning known oxidation #s Atoms/ions by themselves Uncombined = 0 Monatomic ion = ion charge Practice O 2 Zn 2+

Assigning oxidation #s Compounds Neutral molecules add up to 0 Polyatomic ions add up to the charge Assign the most electronegative atom its oxidation number first Hydrogen is USUALLY +1, Oxygen -2 but not always! Practice

Identifying Redox Reactions From the given equation, assign oxidation numbers H 3 O+ + Zn H 2 + H 2 O + Zn 2+ Since Zn changes from 0 to +2 and some of the H changes from +1 to 0, it is a redox reaction

How do we know if an atom is oxidized or reduced? If the oxidation number goes UP during a reaction, it is oxidized since electrons are LOST H 3 O+ + Zn H 2 + H 2 O + Zn 2+ If the oxidation number goes DOWN during a reaction, it is a reduction since electrons are GAINED

Connecting oxidation #s to electron configuration 1 s 2 2 p 3 – 5 valence electrons, -3 oxidation number 1 s 2 2 p 6 3 s 1 – 1 valence electron, +1 oxidation number 1 s 2 2 p 5 1 s 2 2 p 6 3 s 2 3 p 1

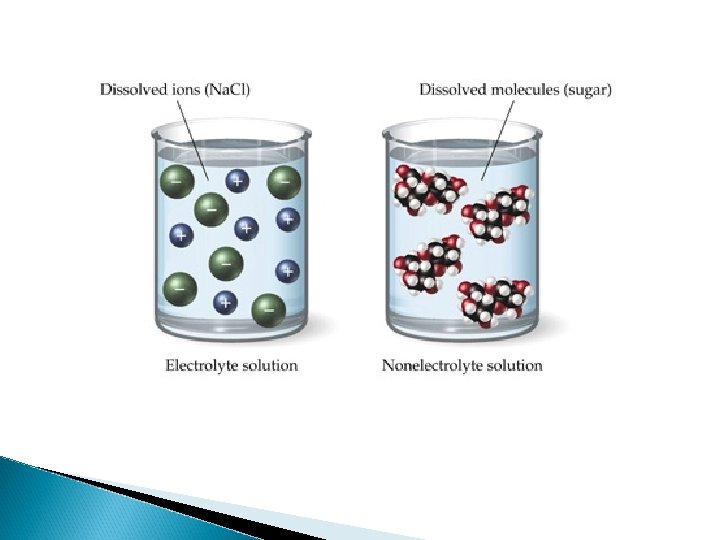
Conductivity Ability to conduct electricity Depends on whether it contains charged particles that can move ◦ Metals – electrons move freely ◦ Na. Cl (s) – has ions but no movement ◦ Na. Cl (aq) – ions can move ◦ Pure water

Electrolytes Substance that dissolves in a liquid solvent & produces ions that conduct electricity STRONG – completely dissociate into ions (ionic compounds) WEAK – partially dissociate to provide a few ions NON – don’t dissociate at all
- Slides: 53