ADVANCED CERAMICS Assist Prof Dr Bugra Cicek Yldz

ADVANCED CERAMICS Assist. Prof. Dr. Bugra Cicek Yıldız Technical University

The Aim of the Course The aim of this lecture is to educate graduate students in mineralogy, chemistry, materials science and related disciplines on the subject of advanced ceramics, in comparison with traditional ceramics

• Introdcution to ceramics • Classification of Ceramics • Mineralogy, Structure, and Green Processing of Clay Minerals • Important Ceramic Phase Systems • Mineralogy and Chemistry of the Ceramic Firing Process • Introduction to Advanced Ceramics • Oxide Ceramics: Structure, Technology, and Applications • Non-Oxide Ceramics: Structure, Technology, and Applications • Insulating Ceramics/High Thermal Conductive • Ceramics • Semiconductive Ceramics • Piezoelectric Ceramics • Magnetic Ceramics • Superconductive Ceramics • Ceramics for Biomedical Applications • Ceramic-Matrix Composites • Decorative Ceramics What we will learn?

1 Final Exam 1 Midterm Exam 1 Homework Attendance Course Frame
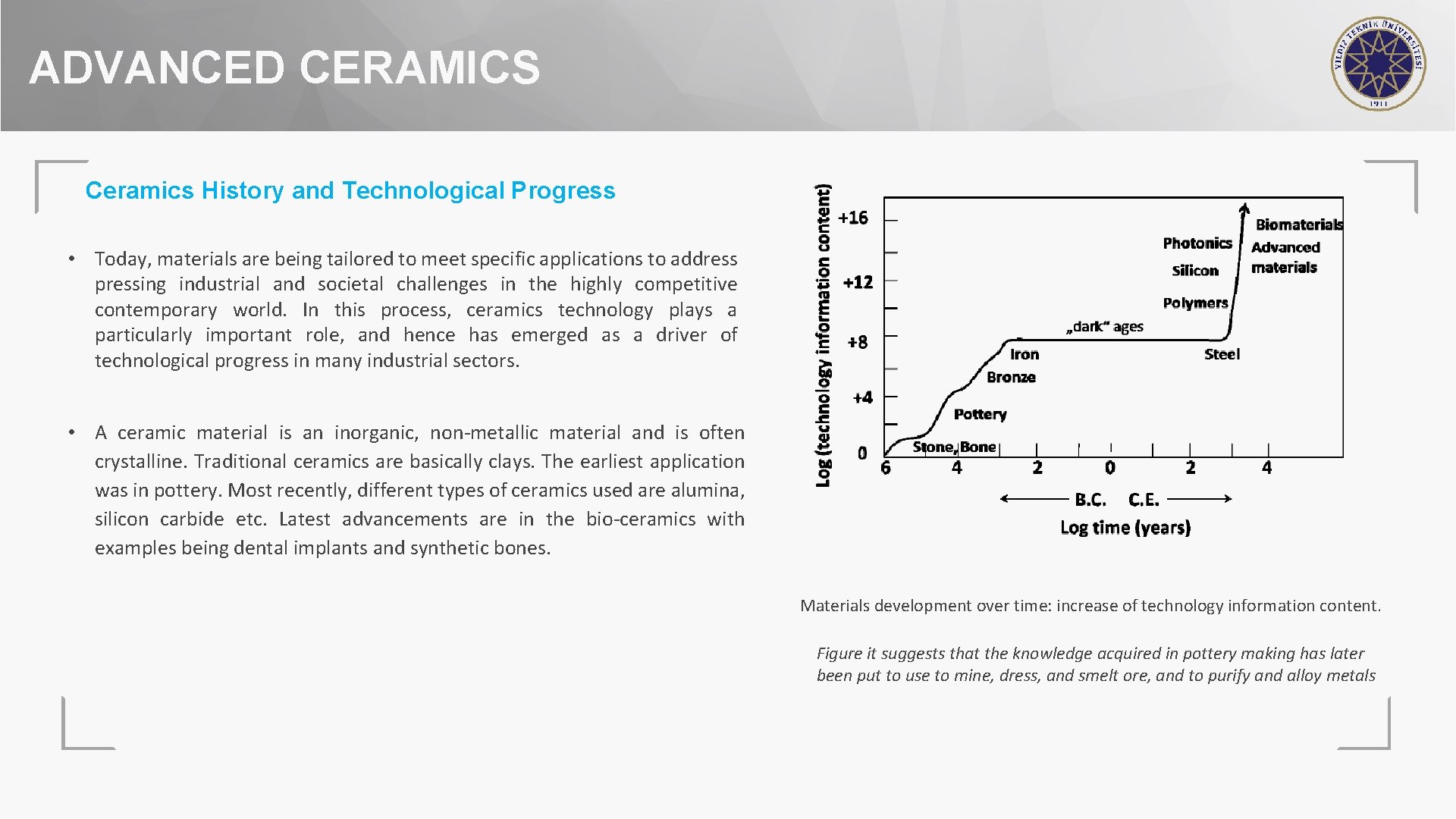
ADVANCED CERAMICS Ceramics History and Technological Progress • Today, materials are being tailored to meet specific applications to address pressing industrial and societal challenges in the highly competitive contemporary world. In this process, ceramics technology plays a particularly important role, and hence has emerged as a driver of technological progress in many industrial sectors. • A ceramic material is an inorganic, non-metallic material and is often crystalline. Traditional ceramics are basically clays. The earliest application was in pottery. Most recently, different types of ceramics used are alumina, silicon carbide etc. Latest advancements are in the bio-ceramics with examples being dental implants and synthetic bones. Materials development over time: increase of technology information content. Figure it suggests that the knowledge acquired in pottery making has later been put to use to mine, dress, and smelt ore, and to purify and alloy metals

ADVANCED CERAMICS Ceramics History and Technological Progress • Ceramics are the oldest man - made materials. By definition, they are inorganic, nonmetallic, silicate - based materials, insoluble in water and many acids and alkalis, and contain at least 30% crystalline compounds. In general, ceramics are shaped at ambient temperature from a specific raw materials mix by a large variety of forming techniques and tools and obtain their typical properties by fi ring beyond 800 ° C • A comparative analysis of ceramics with other engineering materials is shown in table. The purpose of presenting this comparative analysis is to show importance ofceramics among different engineering metals and polymers. This comparison would enable to justify application areas of ceramics.
ADVANCED CERAMICS Ceramics Applications • Pottery products, sanitary ware, floor and roof tiles • Crucibles, kiln linings, other refractories • High end applications such as in ceramic matrix composites, tiles in space shuttle, bullet proof jackets, disk brakes, ball bearing applications, bio-ceramics

ADVANCED CERAMICS Classification of Ceramics Four levels of hierarchical triangles relating different groups of materials Level 1 (materials supergroups): metals, polymers, ceramics Level 2: glasses, hydraulic ceramics, ceramics Level 3 (ceramic subgroups): advanced oxide ceramics, advanced non - oxide ceramics, classic silicate ceramics; Level 4 (phase diagrams): Si. O 2 , Ca. O + Mg. O, Al 2 O 3 + Fe 2 O 3. The three main groups of silicatic ceramic materials
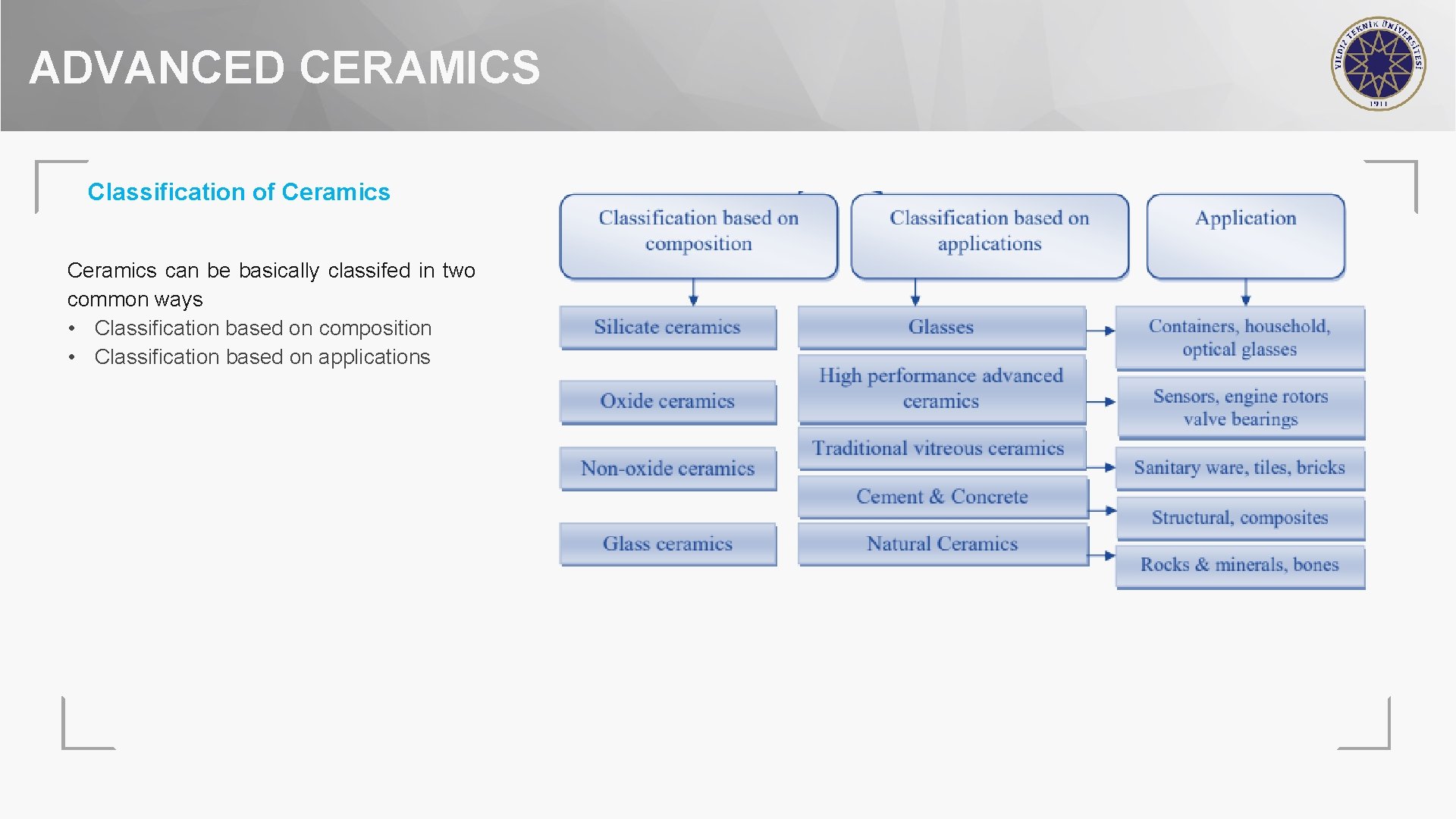
ADVANCED CERAMICS Classification of Ceramics can be basically classifed in two common ways • Classification based on composition • Classification based on applications
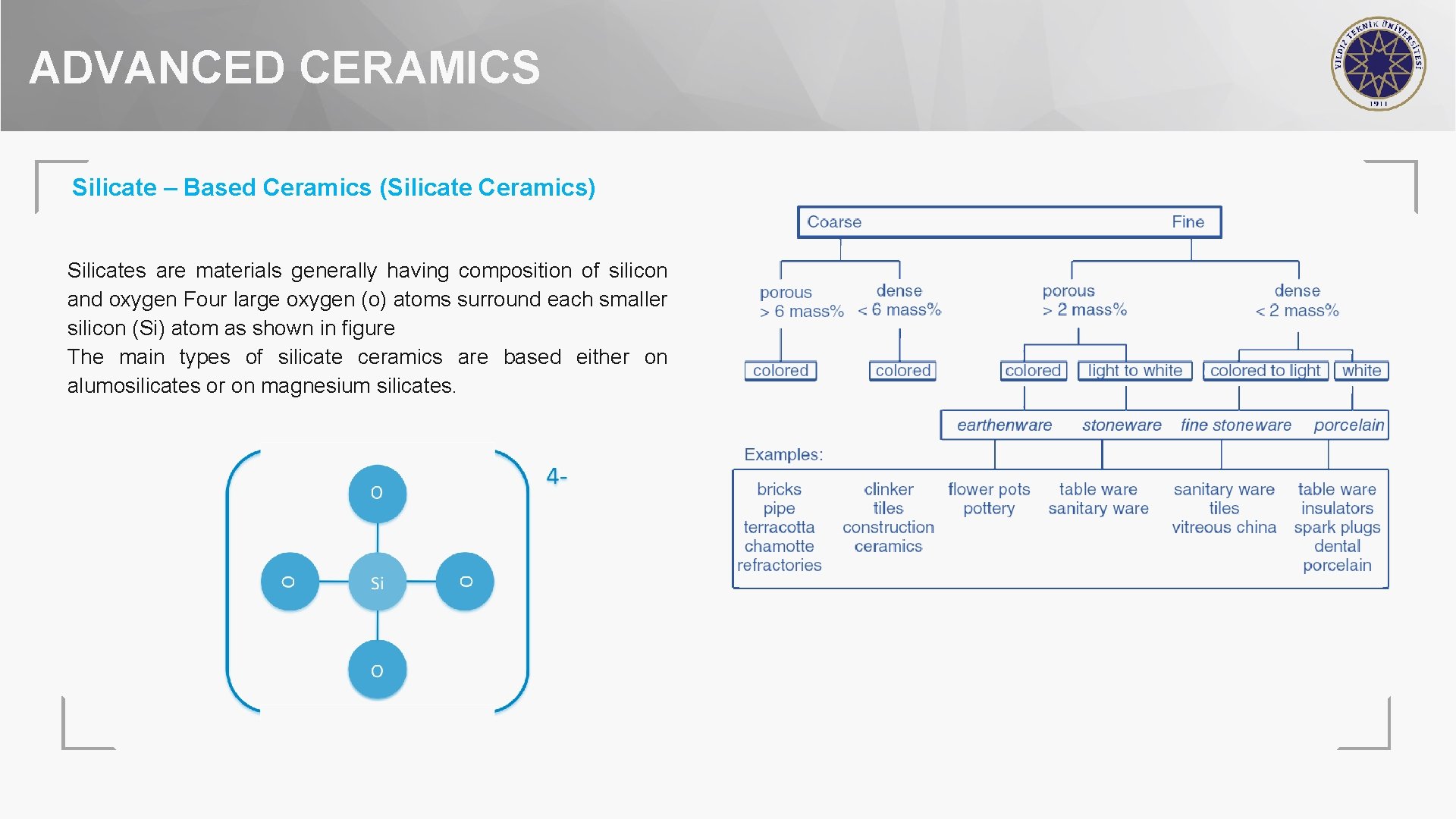
ADVANCED CERAMICS Silicate – Based Ceramics (Silicate Ceramics) Silicates are materials generally having composition of silicon and oxygen Four large oxygen (o) atoms surround each smaller silicon (Si) atom as shown in figure The main types of silicate ceramics are based either on alumosilicates or on magnesium silicates.
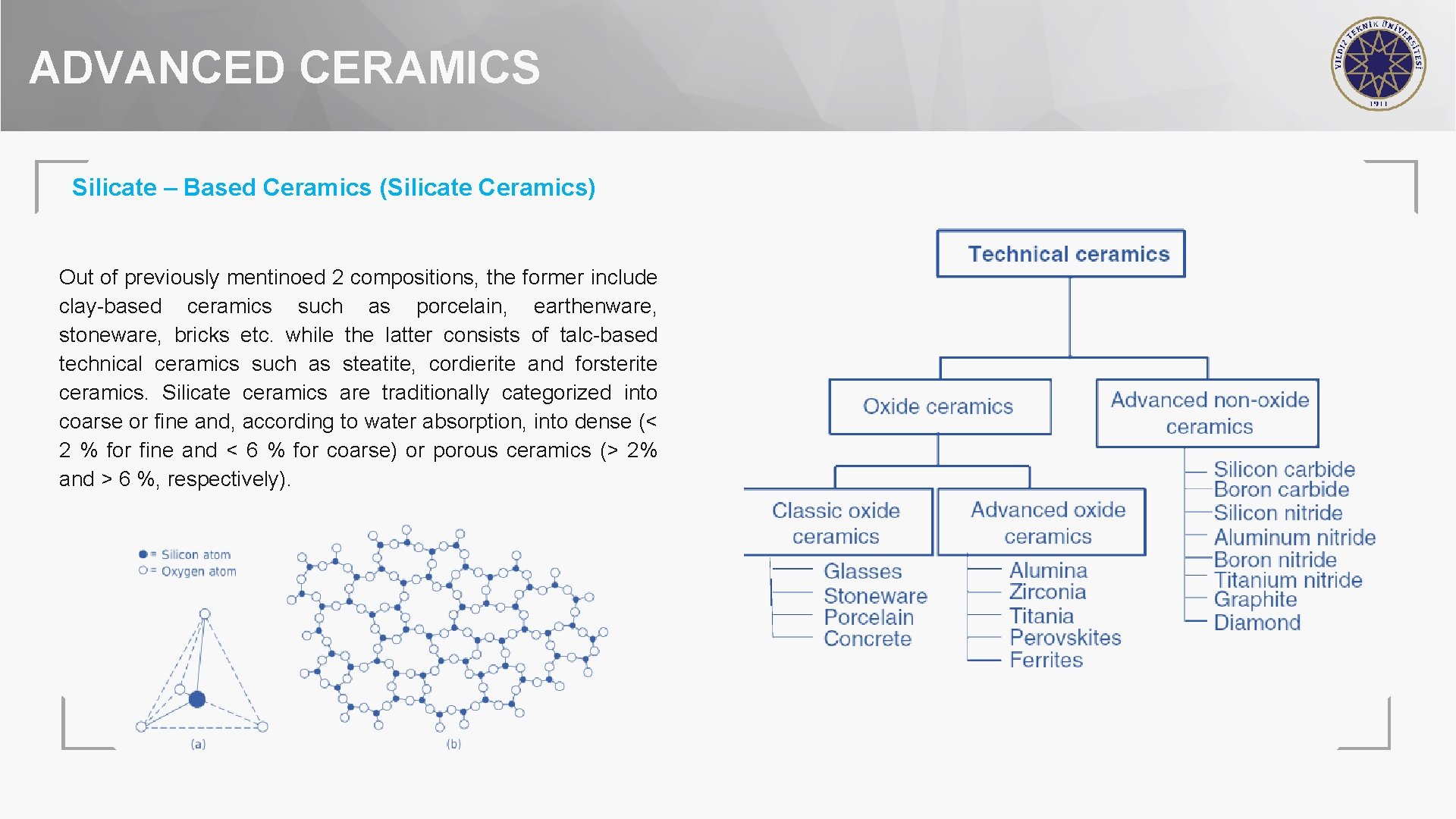
ADVANCED CERAMICS Silicate – Based Ceramics (Silicate Ceramics) Out of previously mentinoed 2 compositions, the former include clay-based ceramics such as porcelain, earthenware, stoneware, bricks etc. while the latter consists of talc-based technical ceramics such as steatite, cordierite and forsterite ceramics. Silicate ceramics are traditionally categorized into coarse or fine and, according to water absorption, into dense (< 2 % for fine and < 6 % for coarse) or porous ceramics (> 2% and > 6 %, respectively).

ADVANCED CERAMICS Oxide Ceramics • Oxide ceramics include alumina, zirconia, silica, aluminium silicate, magnesia and other metal oxide based materials. • These are non-metallic and inorganic compounds by natüre that include oxygen, carbon, or nitrogen. Oxide ceramics possess the following properties: (a) High melting points (b) Low wear resistance (c) An extensive collection of electrical properties These types of ceramics are available with a variety of special features. İ. e; glazes and protective coatings seal porosity, improved water or chemical resistance, and enhanced joining to metals or other materials.

ADVANCED CERAMICS Oxide Ceramics • Oxide ceramics are used in a wide range of applications, which include materials and chemical processing, radio frequency and microwave applications, electrical and high voltage power applications and foundry and metal processing. Aluminium oxide (Al 2 O 3) is the most important technical oxide ceramic material. This synthetically manufactured material consists of aluminium oxide ranging from 80 % to more than 99 %.
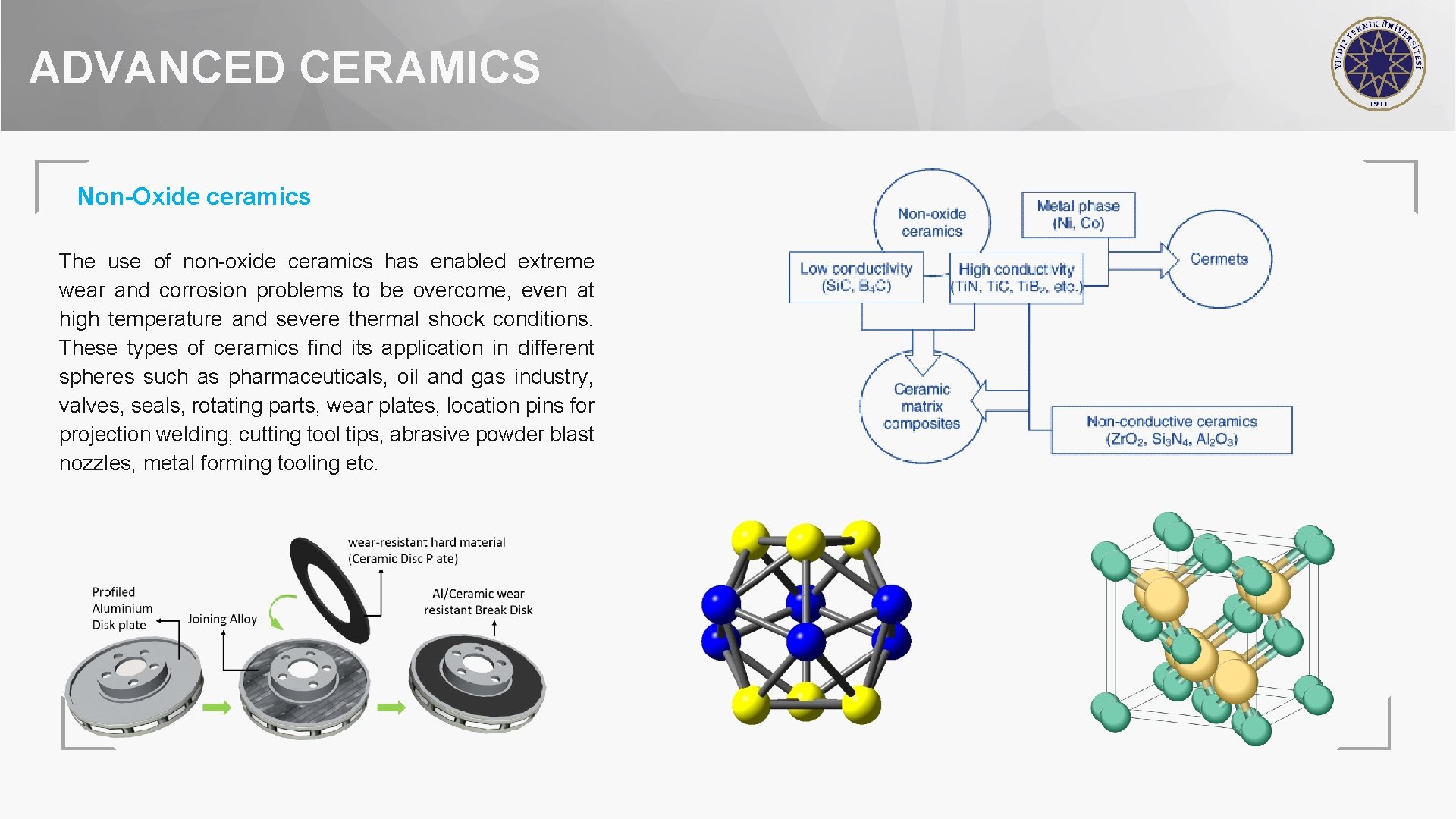
ADVANCED CERAMICS Non-Oxide ceramics The use of non-oxide ceramics has enabled extreme wear and corrosion problems to be overcome, even at high temperature and severe thermal shock conditions. These types of ceramics find its application in different spheres such as pharmaceuticals, oil and gas industry, valves, seals, rotating parts, wear plates, location pins for projection welding, cutting tool tips, abrasive powder blast nozzles, metal forming tooling etc.
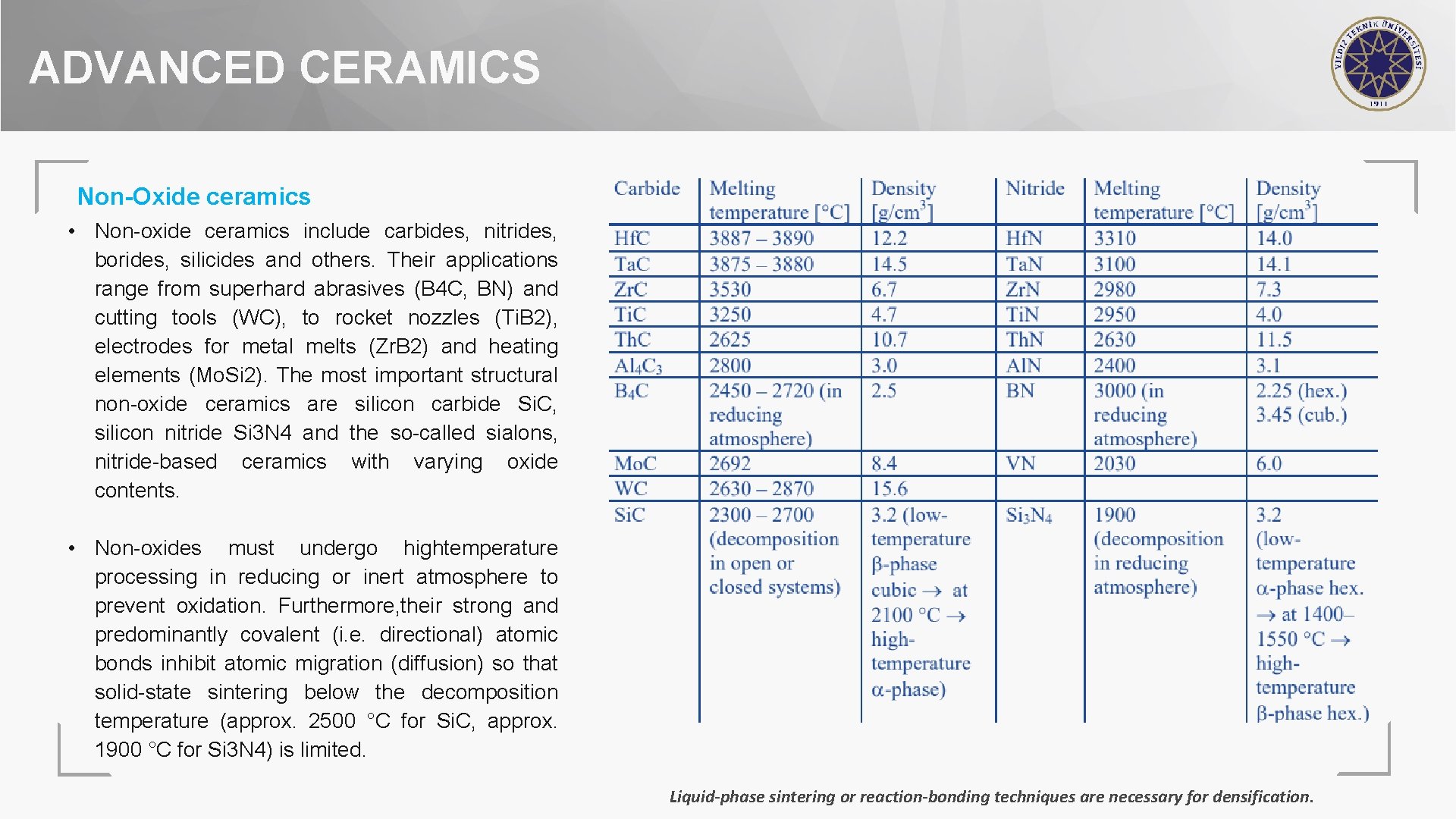
ADVANCED CERAMICS Non-Oxide ceramics • Non-oxide ceramics include carbides, nitrides, borides, silicides and others. Their applications range from superhard abrasives (B 4 C, BN) and cutting tools (WC), to rocket nozzles (Ti. B 2), electrodes for metal melts (Zr. B 2) and heating elements (Mo. Si 2). The most important structural non-oxide ceramics are silicon carbide Si. C, silicon nitride Si 3 N 4 and the so-called sialons, nitride-based ceramics with varying oxide contents. • Non-oxides must undergo hightemperature processing in reducing or inert atmosphere to prevent oxidation. Furthermore, their strong and predominantly covalent (i. e. directional) atomic bonds inhibit atomic migration (diffusion) so that solid-state sintering below the decomposition temperature (approx. 2500 °C for Si. C, approx. 1900 °C for Si 3 N 4) is limited. Liquid-phase sintering or reaction-bonding techniques are necessary for densification.

ADVANCED CERAMICS Ex: Titanium ceramics The ceramic materials based on compounds of titanium, primarily titanium di oxide (Ti. O 2) and barium titanate (Ba. Ti. O 3), that possess the properties of ferr oelectrics. The Ti. O 2 group is characterized by high dielectric constants (ε = 2 0170) and low dielectric losses. This group is widely used in the manufacture of a type of capacitor. Titanium ceramics based on Ba. Ti. O 3 are used in the manufacture of piezoelectric elements. Titanium diboride (Ti. B 2) is a low-density refractory material belonging to the family of ultra-high temperature ceramics (UHTCs).

ADVANCED CERAMICS Glass - ceramics • Glass- Ceramics are polycrystalline material manufactured through the controlled crystallization of base glass. • Glass-ceramic materials share many common characteristics with both glasses and ceramics. • Glass-ceramics possess an amorphous phase and more than one crystalline phases. • These are produced by a controlled crystallization procedure. Glassceramics holds the processing advantage of glass and has special characteristics of ceramics.
ADVANCED CERAMICS Glass - ceramics • Glass-ceramics yield an array of materials with interesting properties like zero porosity, fluorescence, high strength, toughness, low or even negative thermal expansion, opacity, pigmentation, high temperature stability, low dielectric constant, machinability, high chemical durability, biocompatibility, superconductivity, isolation capabilities and high resistivity. • These properties can be altered by controlling composition and by controlled heat treatment of the base glass.

ADVANCED CERAMICS • Mineralogy, Structure, and Green Processing of Clay Minerals The natural raw materials utilized to produce silicate - based ceramics can be divided into: • highly plastic materials , such as clays comprising the minerals kaolinite, illite, or montmorillonite; • sparingly plastic minerals for special electro)ceramic applications, such as pyrophyllite and talc; nonplasticmaterials , such as tempering additives (quartz, chamotte) • fluxes (feldspar, apatite, nepheline, calcite, dolomite, etc. ) that are added to clays to alter the chemistry, workability and sintering behavior of the ceramic masses. Natural Clay Minerals Clays are weathering products of feldspars, micas and other rock forming minerals, and as such mechanical mixtures of very different components, each with a different and characteristic grain size distribution. This mechanical mixture generally consists of four main components: (i) fine - grained weathering relics such as quartz, feldspar, sericite, and interlayer - defi cient micas (ii) newly formed clay minerals such as kaolinite, halloysite, illite, and montmorillonite; (iii) remainders of organisms consisting of calcite or aragonite (shells), silica (chert) or graphitic carbon; (iv) neoformations occurring after deposition, such as pyrite, dolomite, or glauconite.

ADVANCED CERAMICS Most Important Natural Clays are; • Kaolinite group Consists of polymorphs of formula Al 2 Si 2 O 5(OH)4. The repeat unit is a single silicate sheet condensed with alumina octahedra. • Illite-Smectite group Clays have a general formula (K, H)Al 2(Si, Al)4 O 10(OH)2 -x. H 2 O, where x represents a variable amount of water. The structure of this group is similar to the montmorillonite group with a sandwich-type structure. • Montmorillonite-Smectite group Includes talc, vermiculite, montmorillonite and others. The general formula is (Ca, Na, H)(Al, Mg, Fe, Zn)2(Si, Al)4 O 10(OH)2 -x. H 2 O, where x represents a variable amount of water. These minerals all have the sandwich structure with tetrahedral silicate layers strongly bonded to octahedral aluminum or magnesium atoms.
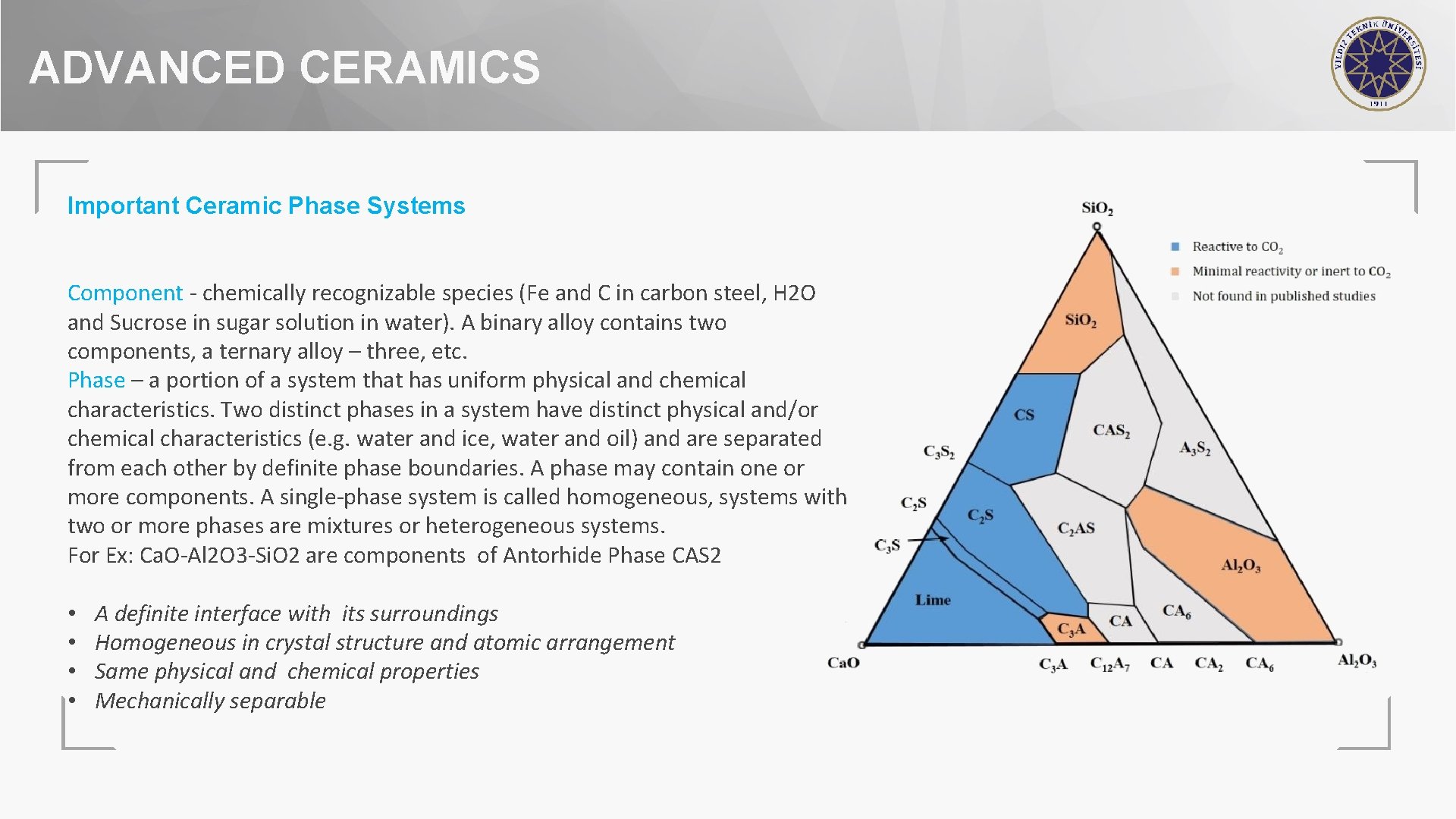
ADVANCED CERAMICS Important Ceramic Phase Systems Component - chemically recognizable species (Fe and C in carbon steel, H 2 O and Sucrose in sugar solution in water). A binary alloy contains two components, a ternary alloy – three, etc. Phase – a portion of a system that has uniform physical and chemical characteristics. Two distinct phases in a system have distinct physical and/or chemical characteristics (e. g. water and ice, water and oil) and are separated from each other by definite phase boundaries. A phase may contain one or more components. A single-phase system is called homogeneous, systems with two or more phases are mixtures or heterogeneous systems. For Ex: Ca. O-Al 2 O 3 -Si. O 2 are components of Antorhide Phase CAS 2 • • A definite interface with its surroundings Homogeneous in crystal structure and atomic arrangement Same physical and chemical properties Mechanically separable

ADVANCED CERAMICS • Phase diagrams are one of the most important sources of information concerning the behavior of elements, compounds and solutions. • They provide us with the knowledge of phase composition and phase stability as a function of temperature (T), pressure (P) and composition (C). • Furthermore, they permit us to study and control important processes such as phase separation, solidification, sintering, purification, growth and doping of single crystals for technological and other applications. • Although phase diagrams provide information about systems at equilibrium, they can also assist in predicting phase relations, compositional changes and structures in systems not at equilibrium. • In general terms, a “phase” is a homogeneous, physically distinct, mechanically separable portion of a material with a given chemical composition. To illustrate this definition, let us look at a few examples of common multi-phase systems. Ice cubes in water constitute a two-phase system (ice and liquid water), unless we include the vapor above the glass in our system, which would make it a three-phase system. A mixture of oil and water would also be a two-phase system. Just as oil and water represent two distinct liquid phases, two regions of a solid with distinctly different composition or structure separated by boundaries represent two solid phases.
ADVANCED CERAMICS Phase Diagrams For obvious reasons it is desirable to know for any given material the conditions (P, T) under which the solid state, the liquid state and the gaseous state are stable, as well as the conditions under which the solid and liquid phases may coexist. These conditions are graphically presented in equilibrium phase diagrams, which can be experimentally determined.
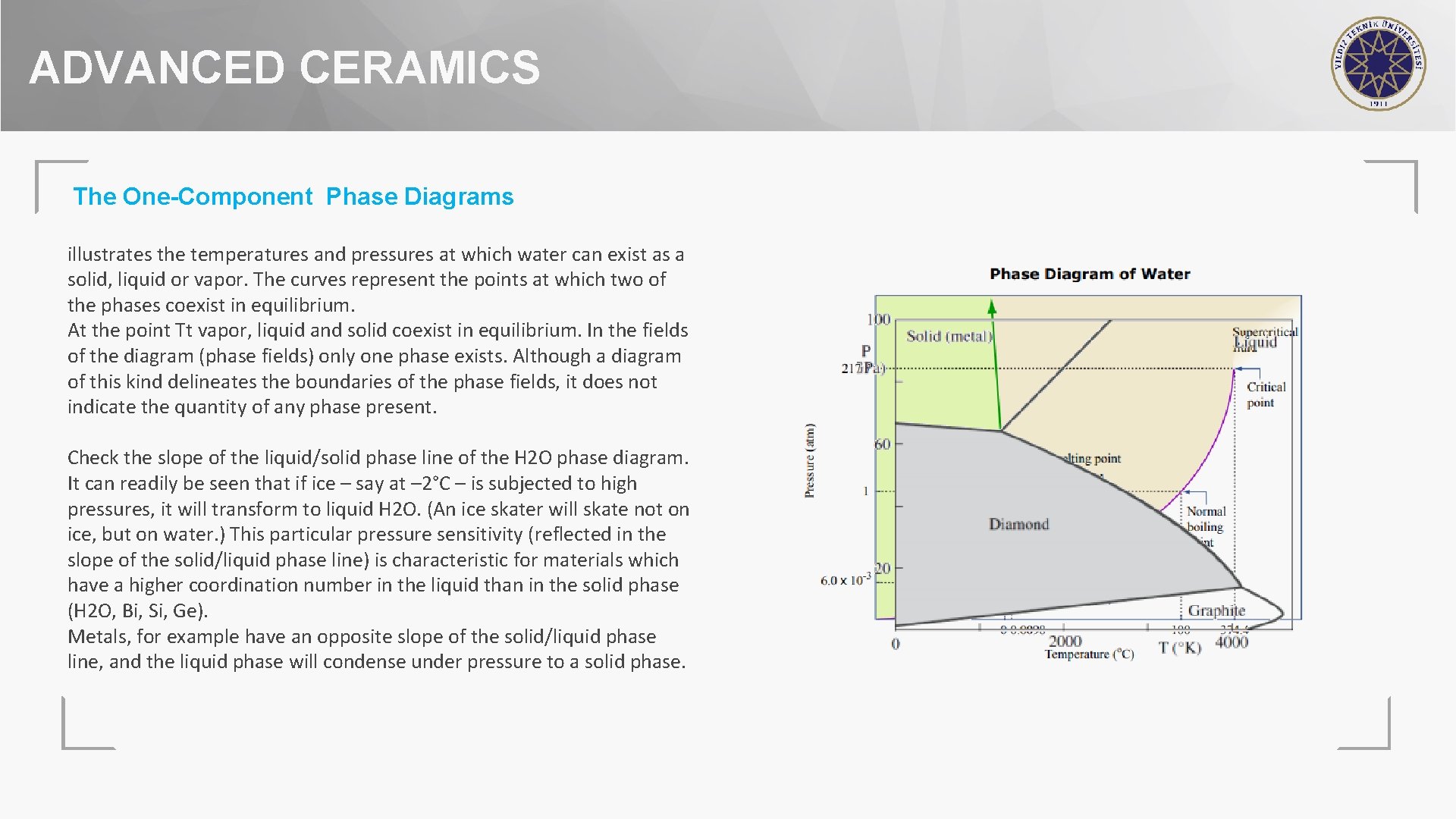
ADVANCED CERAMICS The One-Component Phase Diagrams illustrates the temperatures and pressures at which water can exist as a solid, liquid or vapor. The curves represent the points at which two of the phases coexist in equilibrium. At the point Tt vapor, liquid and solid coexist in equilibrium. In the fields of the diagram (phase fields) only one phase exists. Although a diagram of this kind delineates the boundaries of the phase fields, it does not indicate the quantity of any phase present. Check the slope of the liquid/solid phase line of the H 2 O phase diagram. It can readily be seen that if ice – say at – 2°C – is subjected to high pressures, it will transform to liquid H 2 O. (An ice skater will skate not on ice, but on water. ) This particular pressure sensitivity (reflected in the slope of the solid/liquid phase line) is characteristic for materials which have a higher coordination number in the liquid than in the solid phase (H 2 O, Bi, Si, Ge). Metals, for example have an opposite slope of the solid/liquid phase line, and the liquid phase will condense under pressure to a solid phase.
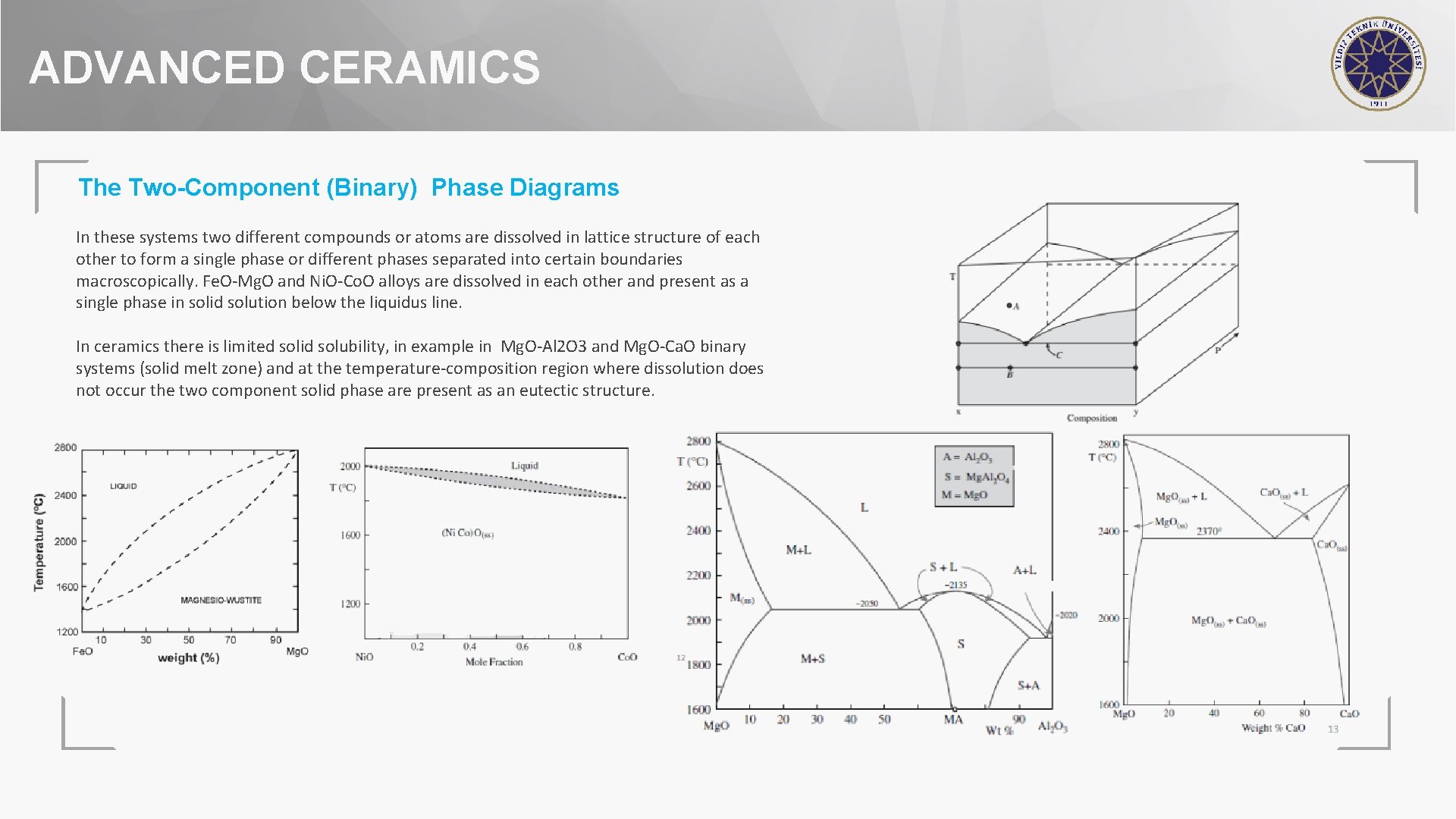
ADVANCED CERAMICS The Two-Component (Binary) Phase Diagrams In these systems two different compounds or atoms are dissolved in lattice structure of each other to form a single phase or different phases separated into certain boundaries macroscopically. Fe. O-Mg. O and Ni. O-Co. O alloys are dissolved in each other and present as a single phase in solid solution below the liquidus line. In ceramics there is limited solid solubility, in example in Mg. O-Al 2 O 3 and Mg. O-Ca. O binary systems (solid melt zone) and at the temperature-composition region where dissolution does not occur the two component solid phase are present as an eutectic structure.
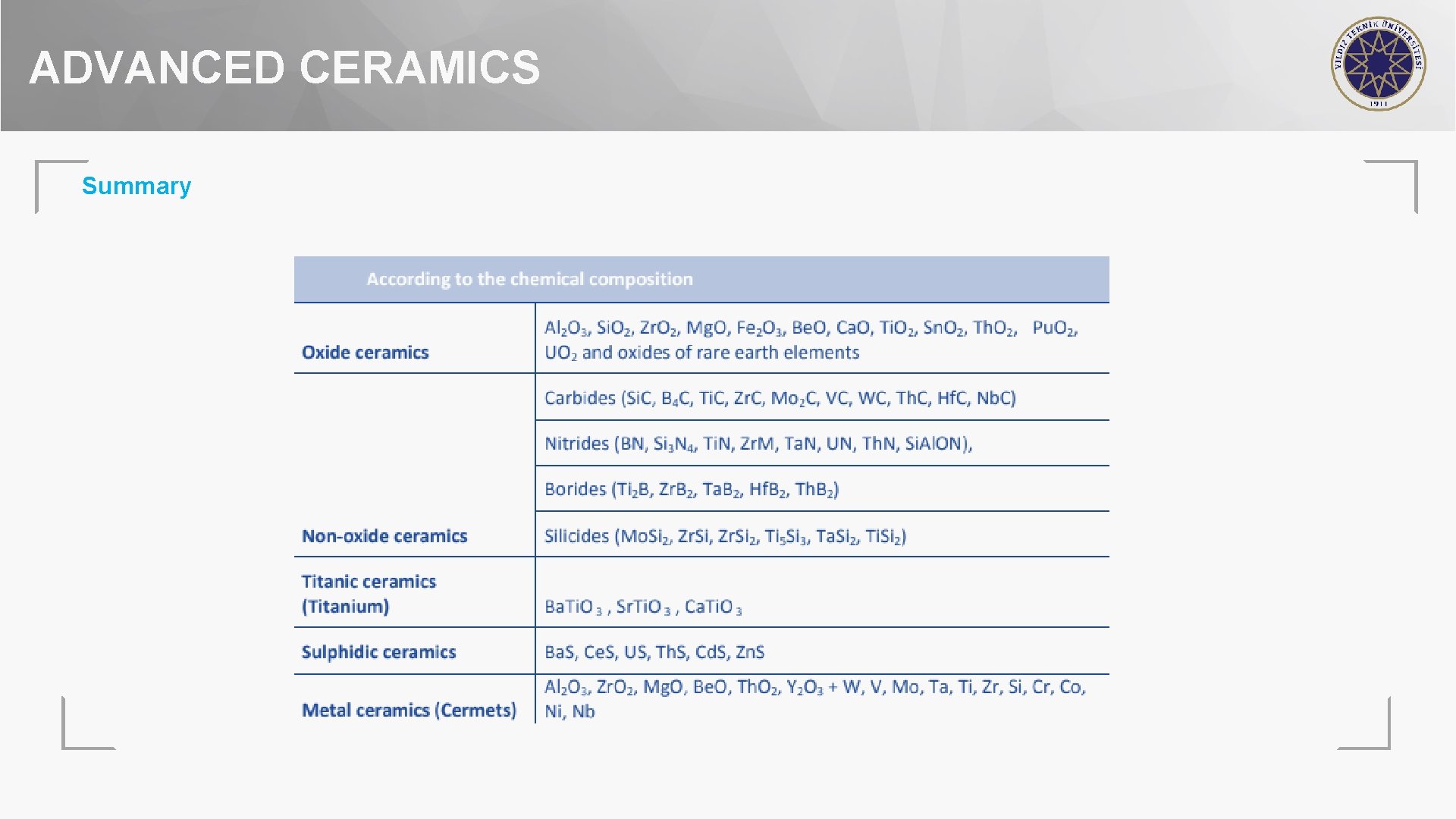
ADVANCED CERAMICS Summary

ADVANCED CERAMICS • Mineralogy, Structure, and Green Processing of Clay Minerals • Formation of Clay Minerals • Structure of Important Clay Minerals • Kaonlinite, Illıte, Montmorlionit • Synthetic Raw materials • Dry Wet forming methods of ceramics • Viscosity • Thixotropy • Zeta potential
l 1 on ne uti Ch an Dis o uti % 10 10 % n l 6 ne an trib Dis Ch 30 an ne l 4 on uti ne on uti l 3 trib an Ch Dis % 10 % 20% Ch Channel 5 trib Distribution Channels 20% Distribution Channel 2
- Slides: 28