Adsorption of radioactive noble gases in microporous materials

Adsorption of radioactive noble gases in microporous materials Jose Busto CPPM/ Universite d’Aix-Marseille GDR - neutrino Marseille 26 – 27 Novembre
Background from radioactivity is one of the most hazardous problems and the main limitation for many experiments at low energy and low counting rate.

Background from radioactive noble gases Super. NEMO bb experiment Borexino Solar Neutrino experiment 85 Kr < 0. 2 m. Bq/m 3 39 Ar < 0. 5 m. Bq/m 3 222 Rn < 150 m. Bq/m 3 Gerda bb experiment LUX dark matter experiment 85 Kr < 2 ppt 42 Ar < 40 m. Bq/L

Radioactive noble gases origin Cosmogenic Anthropogenic : Fission producs La Hague Nuclear Reprocesing Plant
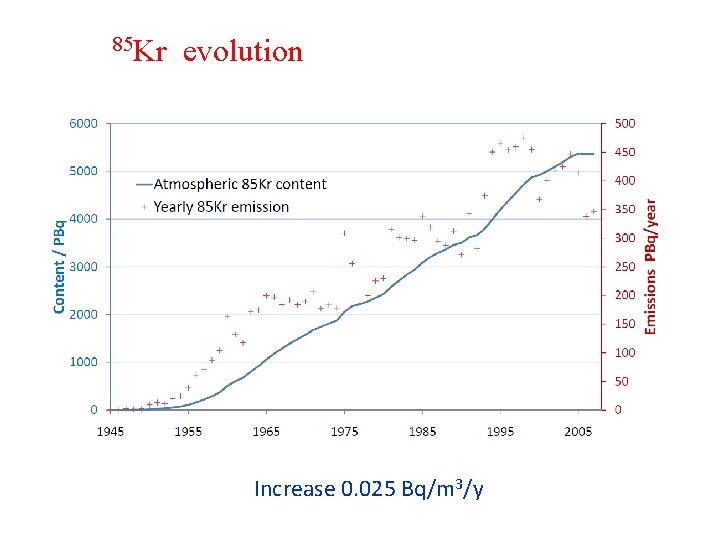
85 Kr evolution Increase 0. 025 Bq/m 3/y

Uranium and Thorium Radioactive Chain
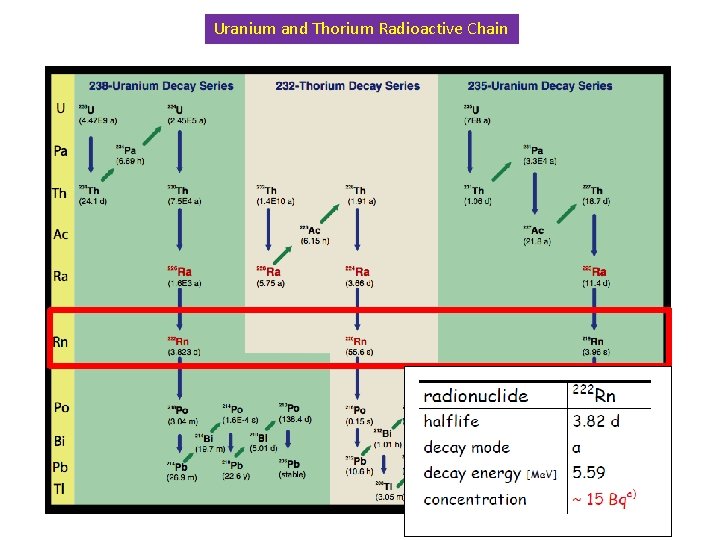
Uranium and Thorium Radioactive Chain
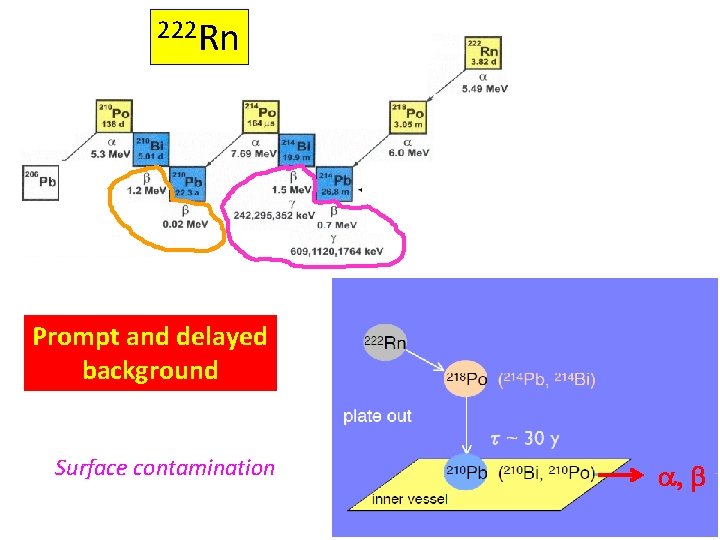
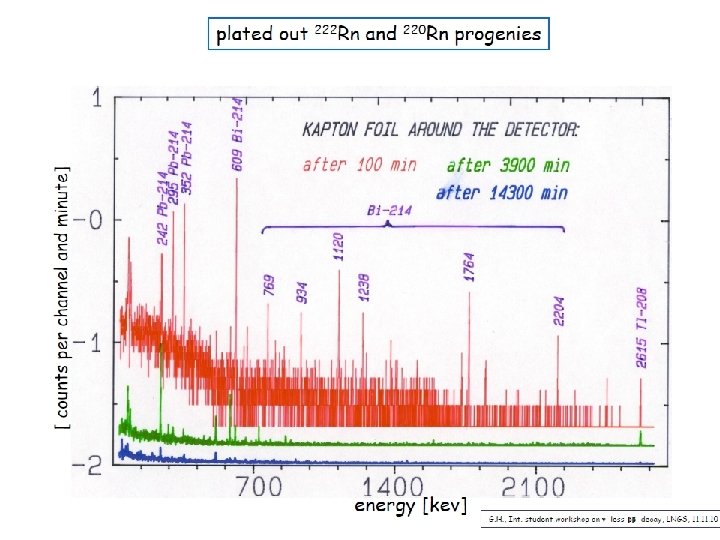
222 Rn Prompt and delayed background Surface contamination a, b

Capture of noble gases Noble gases Full outer shell ( 8 e-) => no valence electrons => very small reactivity Nevertheless Possible compounds for heavier noble elements Kr, Xe Rn must interact with matter better than Xe However radon is a pure radioactive short period gas = > poor knowledge of chemical properties Chemistry of noble gases is possible but very poor Very difficult capture by chemisorption
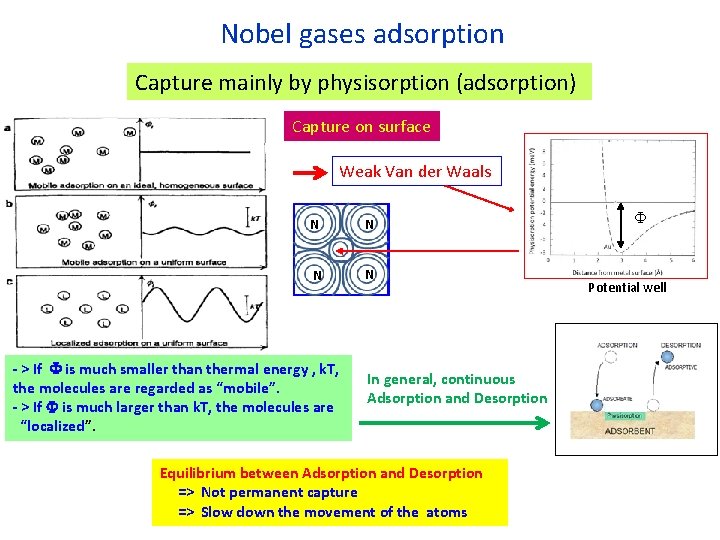
Nobel gases adsorption Capture mainly by physisorption (adsorption) Capture on surface Weak Van der Waals N N - > If F is much smaller than thermal energy , k. T, the molecules are regarded as “mobile”. - > If F is much larger than k. T, the molecules are “localized”. In general, continuous Adsorption and Desorption Equilibrium between Adsorption and Desorption => Not permanent capture => Slow down the movement of the atoms F Potential well

To increase the capture capacity we need : - large surface area => high porosity - reduced the temperature Typical porosity of active charcoals : Enormous surface area of active charcoal The effective surface area is distributed between different porous size

The size and the shape of the pores are also important parameters Potential energy Enhancement of the adsorption energy in a slit shaped pore of various widths Da Dp Narrow pore Da Dp Large pore
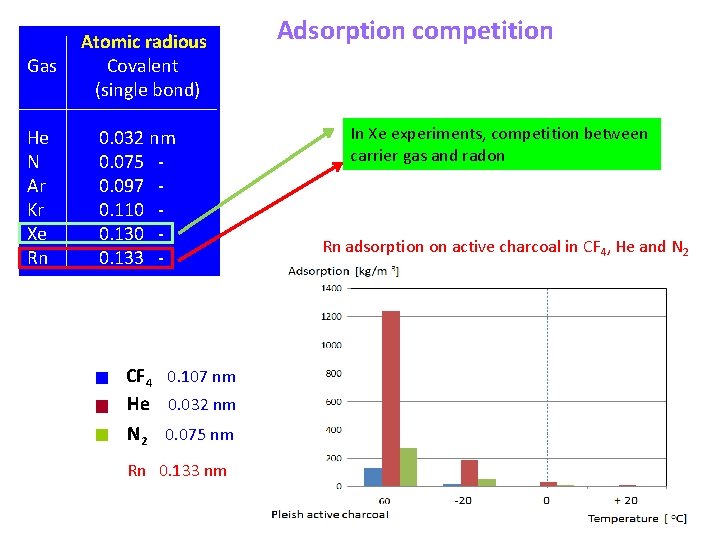
Atomic radious Gas Covalent (single bond) He N Ar Kr Xe Rn 0. 032 nm 0. 075 0. 097 0. 110 0. 133 - CF 4 0. 107 nm He 0. 032 nm N 2 0. 075 nm Rn 0. 133 nm Adsorption competition In Xe experiments, competition between carrier gas and radon Rn adsorption on active charcoal in CF 4, He and N 2

Radon capture measurements at CPPM Characterization of radon adsorption on microporous materials

Radon capture measurement by “dynamic” technique Input Rn Rn + gaz carrier Cg Adsorbant Trap Output Rn Rn detector : Rn concentration in gas Trap in equilibrium Equilibrium Adsorption – desorption t time

Radon capture measurement by “dynamic” technique Radon in filter material by g-Ge spectrometry Cm : Rn concentration in the trap Radon concentration

Residence time v. s. adsorption capacity Residence time : t From gas chromatography Filter Column ( mass : M) Flow : F Mass chr Flow If K is big enough => t >> T 1/2(Rn) therefore, Rn concentration after the radon trap became negligible

Experimental setup Dynamic Rn adsorption Rn Buffer, 10 L Flowmeter 10 L / H Trap Pressure Temperature Rn concentration (diffusion Rn detector) Carrier gas : Nitrogen, Helium Flow : 10 L / H Rn concentration : ~ 900 Bq / m 3 Trap temperature : + 20 o. C to – 50 o. C Pressure : 1 bar RAD 7 N 2 T = 12 o C Particle filter Continuous Rn detector To exhaust

Materials tested T = 20 o. C, -30 o. C, -50 o. C, P = 1 bar Gas = Nitrogen, He • Classical activated carbon : very wide pore size distribution • Synthetic carbon - Carboact : synthetic very low activity charcoal • Commercial Carbon molecular sieve : well defined pore size • Metal Organic Framework : very large surface area

• Molecular Cages : well defined structue Curcubiturils • Carbon Aerogel : very low density large surface areas • Sucrose carbon micro spheres : very high microporosity Porous organic cages CC 3 (Liverpool Univ. )

K_factor for several materials in N 2 20 o C m 3/kg 25 20 15 10 5 Standard active charcoal 69 5 Ca rb ox en 1 00 0 III ie rb ox Ca rb os Ca en ve ct o. A rb Ca 10 K 6 Ca ar Nu cle K 4 8 S rb 0 Carbon Molecular Sieve

K_factor for several materials in N 2 20 o C m 3/kg -30 o C m 3/kg Standard active charcoal Carbon Molecular Sieve Standard active charcoal 9 56 n Ca rb ox e 1 00 0 III ie rb ox Ca rb os Ca en ve ct o. A rb Ca 10 K 6 Ca ar 69 5 Ca rb ox en 1 00 0 III ie rb ox Ca rb os Ca en ve ct o. A rb Ca 10 K 6 Ca ar Nu cle K 4 8 S rb 0 S 5 8 10 cle 15 K 4 20 rb 180 160 140 120 100 80 60 40 20 0 Nu 25 Carbon Molecular Sieve

rb os ie ve Ca II rb I ox en 1 00 Ca 0 rb ox en 5 69 Ca ct m 3/kg rb o. A -50 o C 800 700 600 500 400 300 200 100 0 Ca 9 0 56 n 00 III ve 1 en ie ox e rb rb ox Ca rb os ct o. A rb Ca 10 K 6 rb 0 Ca 5 ar 10 S 15 8 20 cle 69 0 25 K 4 5 00 m 3/kg Ca 1 en rb ox III ve 20 o C Nu Ca ie en rb ox Ca rb os Ca ct o. A 10 K 6 rb Ca ar S 8 K 4 rb Ca cle Nu m 3/kg Ca 0 K 6 1 rb S Ca ar cle Nu K 4 8 K_factor for several materials in N 2 -30 o C 180 160 140 120 100 80 60 40 20 0

ct r Ae oge ro l J ge l J 18 D ge l ro -30 o C Ae Ae m 3/kg Ca Ca 9 0 56 n 00 III ve 1 en ie ox e rb rb ox Ca rb os Ca ct o. A rb Ca 10 K 6 rb Ca m 3/kg rb os rb ieve ox II Ca en 1 I rb 0 ox 00 en 5 69 Ca 800 700 600 500 400 300 200 100 0 rb o. A -50 o C ar 0 S 5 8 10 cle 69 0 15 K 4 5 00 20 Ca 1 en rb ox III ve 25 Nu Ca ie en rb ox Ca rb os Ca ct o. A rb Ca 10 K 6 rb Ca ar S 8 K 4 20 o C Nu K 48 cle S ar Ca rb K 6 10 rb os ie ve Ca II rb I ox en 1 00 Ca 0 rb ox en 5 69 Ca ct m 3/kg rb o. A cle Nu m 3/kg Ca 0 K 6 1 rb S Ca ar cle Nu K 4 8 K_factor for several materials in N 2 -30 o C 180 160 140 120 100 80 60 40 20 0 Aerogel 400 350 300 250 200 150 100 50 0

Ca Ca 15 10 5 0 m 3/kg 800 700 600 500 400 300 200 100 0 10 20 K 6 rb Ca ar S 8 K 4 25 0 cle Nu m 3/kg K 6 1 rb S Ca ar cle Nu K 4 8 20 o C 180 160 140 120 100 80 60 40 20 0 -50 o C 400 350 300 250 200 150 100 50 0 ct ct r Ae oge ro l J ge l J 18 D ge l ro -30 o C Ae Ae m 3/kg Ca Ca 9 0 56 n 00 III ve 1 en ie ox e rb rb ox Ca ct o. A 10 K 6 S rb Ca ar 0 69 8 5 00 K 4 rb Ca cle rb os Ca 1 III ve en rb ox Nu Ca ie en rb ox Ca rb os ct o. A rb m 3/kg rb os rb ieve ox II Ca en 1 I rb 0 ox 00 en 5 69 Ca rb o. A Ca Nu K 48 cle S ar Ca rb K 6 10 rb os ie ve Ca II rb I ox en 1 00 Ca 0 rb ox en 5 69 Ca rb o. A s t l n a e r o b r c e r t o t a s e h d a hb ec w uc tiv e N m ac e d r r a da n a t s n a th Ca K_factor for several materials in N 2 -30 o C Aerogel

K_factor for several materials in He / N 2 He -50 o C 12000 10000 8000 N 2 6000 4000 2000 11 I K ge l ro IE OS RB Ae VE II CT CA RB OA l CA sp ec ia K 4 8 8 J 1 ge l ro IE OS RB CA Ae VE II CT RB OA l CA sp ec ia K 4 8 I 0 Very low competition between Rn and He

Randon capture v. s. porosity Volume microporeux total Volume ultramicro % ultra Volume supermicro Aext ABET Volume total t-plot as-plot DR as-plot m 2. g-1 cm 3. g-1 K 610 940 0, 381 0, 370 0, 410 0, 348 0, 174 42 0, 236 40 Carboxen 569 290 0, 484 0, 080 0, 064 0, 117 0, 064 100 0, 000 164 K 48 special 1268 0, 512 0, 505 0, 507 0, 520 0, 274 54 0, 233 5 Carboact 1182 0, 534 0, 470 0, 462 0, 476 0, 290 63 0, 172 26 Carboxen 1000 931 0, 842 0, 357 0, 359 0, 366 0, 253 70 0, 106 20 Carbosieve SIII 1362 0, 565 0, 510 0, 535 0, 355 70 0, 155 8 Echantillons Ultramicropore : diamètre<1, 062 nm

Adsorption v. s. microscopic properties Adsorption ( m 3 /kg) v. s volume of ultramicropores ( < 1. 62 nm) m 3 /kg (@ - 50 o. C) 800. 0 Carbosieve III 700. 0 Carboxene 1000 600. 0 500. 0 400. 0 300. 0 200. 0 Carboact K 48 S K 610 carboxen 569 100. 000 0. 050 0. 100 0. 150 0. 200 0. 250 0. 300 0. 350 0. 400 Very nice correlation Þ Modelization of radon adsorption ? ? ? cm 3/g

Not only, surface area, and porous distributions are important Adsorption ( m 3 /kg) v. s volume of ultramicropores ( < 1. 62 nm) m 3 /kg (@ - 50 o. C) 800. 0 Carbosieve III 700. 0 Carboxene 1000 600. 0 500. 0 400. 0 300. 0 200. 0 Carboact K 48 S K 610 carboxen 569 100. 000 0. 050 0. 100 0. 150 0. 200 0. 250 0. 300 0. 350 0. 400 cm 3/g Carboxen 1000 : pore size ~ 10 – 12 Å = > high adsorption Carbosieve III S : pore size ~ 4 – 10 Å => high adsorption Zeolite 13 X : pore size ~ 10 Å = > NO adsorption Carbon Molecular Sieve Aluminium Molecular Sieve Need further measurements and a better knowledge of the structure

Last but not least : Emanation Radioactivity of some active charcoals Radon emanation from the trap it self § Very pure adsorbent ( synthetic materials ) § Mass of adsorbent as low as possible § Self adsorption can help

Conclusion Ø Radioactivity from noble gases is a very important background source in many low energy and low count rate experiments. Ø Increase Rn adsorption in new microporous materials is possible. We need nevertheless a better understanding of the porosity and structure of the adsorbent. Ø High radio-purity is mandatory ( synthetic materials) Ø Optimization of production process (price of adsorbent) is also required.


Modelization is possible in “symmetric” carbon cages CC 3 cage – University of Liverpool Predicted adsorption isotherme for Kr, Xe and Rn
- Slides: 35