Adsorption and interfacial phenomena Absorption vs Adsorption Absorption

Adsorption and interfacial phenomena

Absorption vs. Adsorption • Absorption is the process in which a fluid is dissolved by a liquid or a solid (absorbent). • Adsorption is the process in which atoms, ions or molecules from a substance (it could be gas, liquid or dissolved solid) adhere to a surface of the adsorbent. • Adsorption is a surface-based process where a film of adsorbate is created on the surface while absorption involves the entire volume of the absorbing substance.

Adsorption phenomena… • Adsorbent – an adsorptive material, such as activated charcoal • The solid substance on the surface of which adsorption occurs is known as adsorbent. • Adsorbate – an adsorbed substance • The substances that get adsorbed on the solid surface due to intermolecular attraction are called adsorbate.

Interfaces • When phases exist together, the boundary between two of them is known as an interface. • The properties of the molecules forming the interface are often sufficiently different from those in the bulk of each phase. • The term surface is used when referring to either a gas–solid or a gas– liquid interface.
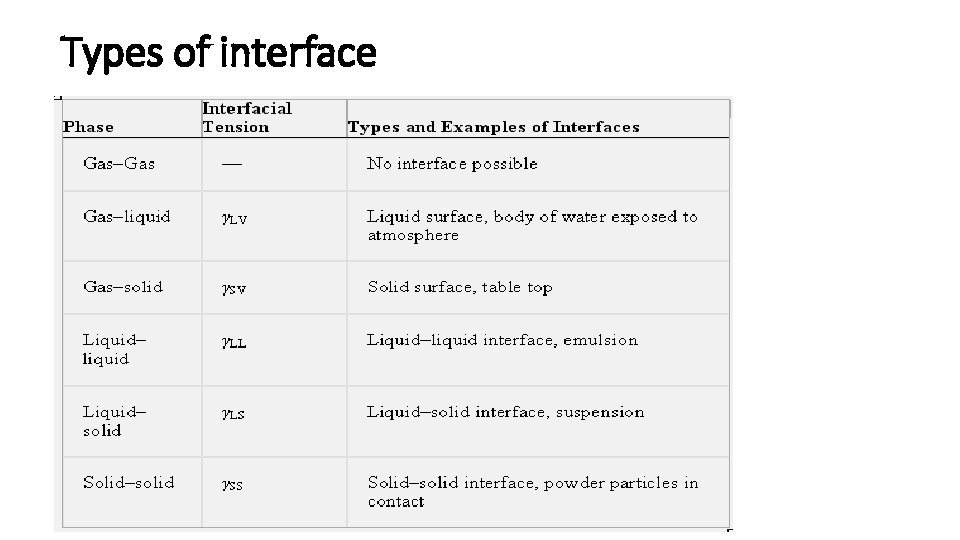
Types of interface

Adhesive and Cohesive Forces • Cohesive forces are the intermolecular forces which cause a tendency in liquids to resist separation. These attractive forces exist between molecules of the same substance. For instance, rain falls in droplets, rather than a fine mist, because water has strong cohesion which pulls its molecules tightly together, forming droplets. • Adhesive forces are the attractive forces between unlike molecules. They are caused by forces acting between two substances, such as mechanical forces (sticking together) and electrostatic forces (attraction due to opposing charges). In the case of a liquid wetting agent, adhesion causes the liquid to cling to the surface on which it rests. When water is poured on clean glass, it tends to spread, forming a thin, uniform film over the glasses surface. This is because the adhesive forces between water and glass are strong.

Liquid Interfaces Surface and Interfacial Tensions • molecules in the bulk liquid are surrounded in all directions by other molecules for which they have an equal attraction. • molecules at the surface (i. e. , at the liquid–air interface) can only develop attractive cohesive forces with other liquid molecules that are situated below and adjacent to them. They can develop adhesive forces of attraction with the molecules constituting the other phase involved in the interface, although, in the case of the liquid–gas interface, this adhesive force of attraction is small.
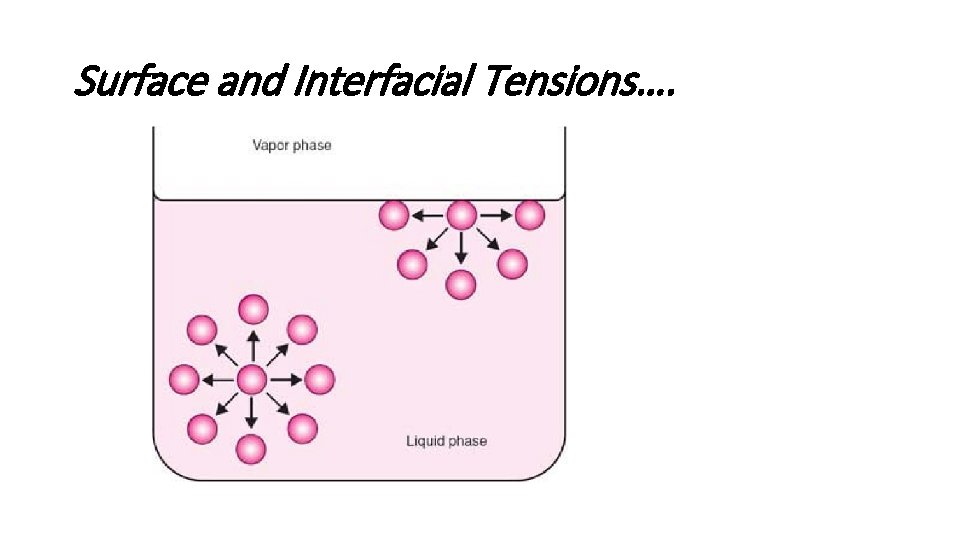
Surface and Interfacial Tensions….

• The net effect is that the molecules at the surface of the liquid experience an inward force toward the bulk. • Such a force pulls the molecules of the interface together and, as a result, contracts the surface, resulting in a surface tension. • This “tension” in the surface is the force per unit length that must be applied parallel to the surface so as to counterbalance the net inward pull. • Interfacial tension is the force per unit length existing at the interface between two immiscible liquid phases and. • the surface and interfacial tensions, have the units of dynes/cm or N/m.

Surface Free Energy and Surface Tension • The surface layer of a liquid possesses additional energy as compared to the bulk liquid. • This energy increases when the surface of the same mass of liquid increases and is therefore called surface free energy. • To move a molecule from the inner layers to the surface, work needs to be done against the force of surface tension.

The work W required to create a unit area of surface is known as SURFACE FREE ENERGY/UNIT AREA (ergs/cm 2) erg = dyne. cm W=γ∆A Its equivalent to the surface tension γ Thus the greater the area A of interfacial contact between the phases, the greater the free energy. For equilibrium, the surface free energy of a system must be at a minimum. Thus Liquid droplets tend to assume a spherical shape since a sphere has the smallest surface area per unit volume.

SURFACE ACTIVE AGENTS / SURFACTANT • A surface active agent (surfactant) is a substance which lowers the surface tension of the medium in which it is dissolved. • Ex- Soap, Detergent, Emulsifier, Foaming agent etc. • Surfactants in solution are often association with colloids, that is, they tend to form micelles.

Emulsifier • An emulsifier (also known as an "emulgent") is a substance that stabilizes an emulsion by increasing its kinetic stability. Emulsifiers are compounds that typically have a polar or hydrophilic (i. e. watersoluble) part and a non-polar (i. e. hydrophobic or lipophilic) part.

HLB Value • HLB (Hydrophile-Lipophile Balance) is an empirical expression for the relationship of the hydrophilic ("water-loving") and hydrophobic ("water-hating") groups of a surfactant. Surfactants are typically ampiphilic molecules that contain both hydrophilic and lipophilic groups. • The hydrophile-lipophile balance (HLB) number is used as a measure of the ratio of these groups. It is a value between 0 -60 defining the affinity of a surfactant for water or oil. Mainly their range assume 020. • The higher the HLB value, the more water-soluble the surfactant.
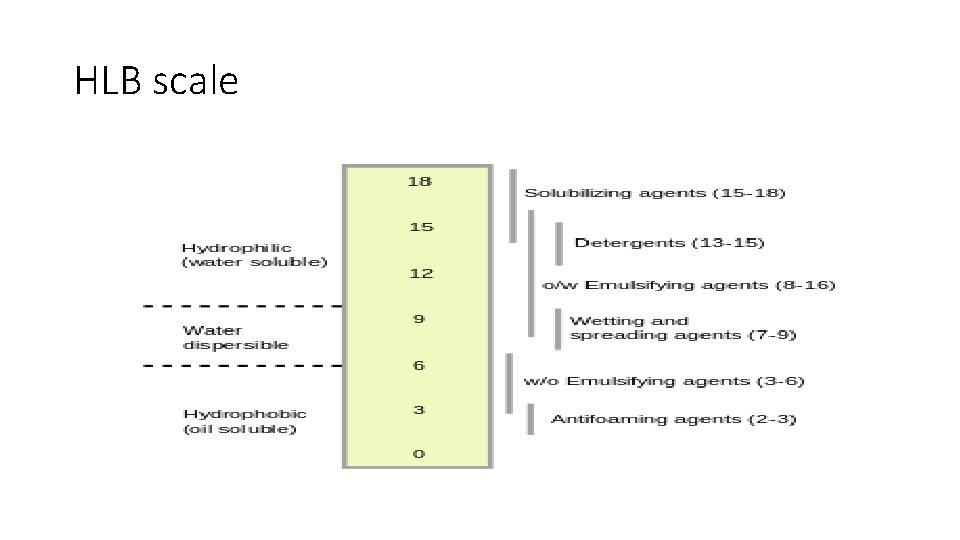
HLB scale

Detergents • A detergent is a surfactant or a mixture of surfactants with "cleaning properties in dilute solutions. " • These substances are usually alkylbenzene sulfonates, a family of compounds that are similar to soap but are more soluble in hard water.

Mechanism of Detergents • This is the structure of a detergent molecule • One end (the head) of the molecule is hydrophilic, this part has a strong affinity for water molecule. The other end (the tail) of the molecule is hydrophobic, this part tries to stay away from water molecule. However it does love dirt and grime.

• Thus the hydrophobic tail attaches itself to dirt and grime while the hydrophilic head gets affixed to water molecule. • Therefore when the dirty cloths are swished vigorously, dirt is pulled away from the cloth and washed away in the water leaving the cloth sparkling clean.

Chemical classification of surfactants/detergents • Detergents are classified into three broad groupings, depending on the electrical charge of the surfactants. • Anionic detergents: • Cationic detergents • Non-ionic and zwitter ionic detergents

• Anionic surfactants contain anionic functional groups at their head, such as sulfate, sulfonate, phosphate, and carboxylates. Prominent alkyl sulfates include ammonium lauryl sulfate, sodium lauryl sulfate (sodium dodecyl sulfate, SLS, or SDS), and the related alkylether sulfates sodium laureth sulfate (sodium lauryl ether sulfate or SLES), and sodium myreth sulfate. • Cationic surfactants contain cationic functional groups at their head, such as Cetrimonium bromide (CTAB), Cetylpyridinium chloride (CPC), Benzalkonium chloride (BAC), Benzethonium chloride (BZT) • Zwitterionic (amphoteric) surfactants have both cationic and anionic centers attached to the same molecule. The cationic part is based on primary, secondary, or tertiary amines or quaternary ammonium cations. The anionic part can be more variable and include sulfonates.

Defoamer • A defoamer or an anti-foaming agent is a chemical additive that reduces and hinders the formation of foam in industrial process liquids. • In industrial processes, foams pose serious problems. They cause defects on surface coatings. They prevent the efficient filling of containers. A variety of chemical formulas are available to prevent formation of foams.

• Exogenous pulmonary surfactant as a drug delivering agent & influence of antibiotics on surfactant activity • It has been proposed to use exogenous pulmonary surfactant as a drug delivery system for antibiotics to the alveolar compartment of the lung. Little, however, is known about interactions between pulmonary surfactant and antimicrobial agents. • This study investigated the activity of a bovine pulmonary surfactant after mixture with amphotericin B, amoxicillin, ceftazidime, pentamidine or tobramycin. • Therefore, before using surfactant-antibiotic mixtures in clinical trials, interactions between the two agents should be carefully evaluated.

Pharmaceutical applications of foam products • To extinguish gasoline and oil fires the fire department will spread this over a fire and it remains on top of the substance robbing it of oxygen thus putting out the flames. • Airplanes that have mechanical problems with their landing gear often use a foam spread on the runway to prevent ignition of the jet fuel • This surface tension is used to make whipping cream in cooking. • Insulating foams also can be shot into confined spaces and then expand filling the cavity with foam.

ADSORPTION ISOTHERM • A mathematical equation, which describes the relationship between pressure (p) of the gaseous adsorbate and the extent of adsorption at any fixed temperature, is called adsorption isotherm. • The extent of adsorption is expressed as mass of the adsorbate adsorbed on one unit mass of the adsorbent. • Thus, if x g of an adsorbate is adsorbed on m g of the adsorbent, then • Extent of adsorption = x/m

Freundlich equation • The Freundlich equation or Freundlich adsorption isotherm, an adsorption isotherm, is a curve relating the concentration of a solute on the surface of an adsorbent to the concentration of the solute in the liquid with which it is in contact. • The Freundlich Adsorption Isotherm is mathematically expressed as • It is also written as • • Where, x = mass of adsorbate m = mass of adsorbent p = Equilibrium pressure of adsorbate; K and n are constants

• The equation above equation is comparable with the equation of straight line, y = m x + c where, m represents slope of the line and c represents intercept on y axis. • Plotting a graph between log(x/m) and log p, we will get a straight line with value of slope equal to 1/n and log k as y-axis intercept. . • log(x/m) vs. log p graph

• Limitation of Freundlich Adsorption Isotherm • Experimentally it was determined that extent of adsorption varies directly with pressure till saturation pressure Ps is reached. Beyond that point rate of adsorption saturates even after applying higher pressure. Thus Freundlich Adsorption Isotherm failed at higher pressure.

Langmuir equation • The Langmuir equation (also known as the Langmuir isotherm) relates the adsorption of molecules on a solid surface to gas pressure or concentration of a medium above the solid surface at a fixed temperature. The equation is stated as: • θ is the fractional coverage of the surface, P is the gas pressure or concentration, α is a constant. • The constant α is the Langmuir adsorption constant and increases with an increase in the binding energy of adsorption and with a decrease in temperature.

• Equation Derivation: • The Langmuir equation is derived starting from the equilibrium between empty surface sites (S*), particles (P) and filled particle sites (SP) • The equilibrium constant K is thus given by the equation: • Due to the number of filled surface sites (SP) is proportional to θ, the number of unfilled sites (S*) is proportional to 1 -θ, and the number of particles is proportional to the gas pressure or concentration (p), the equation can be rewritten as:

• Where α is a constant. Rearranging this as follows: • Thus Langmuir equation:

BET theory • Brunauer–Emmett–Teller (BET) theory aims to explain the physical adsorption of gas molecules on a solid surface and serves as important analysis technique for the measurement of the specific surface area of a material. • The resulting BET equation is • where ρ and ρo are the equilibrium and the saturation pressure of adsorbates at the temperature of adsorption, v is the adsorbed gas quantity, and vm is the monolayer adsorbed gas quantity.

• c is the BET constant, • Where E 1 is the heat of adsorption for the first layer, and ELis that for the second and higher layers and T is equal to the heat of liquefaction.

Electrical double layer, EDL • Electrical double layer (EDL) is a structure that appears on the surface of an object when it is exposed to a fluid. The object might be a solid particle, a gas bubble, a liquid droplet or a porous body. • The EDL refers to two parallel layers of charge surrounding the object. • The first layer, the surface charge (either positive or negative), comprises ions adsorbed onto the object due to chemical interactions. • The second layer is composed of ions attracted to the surface charge via the coulomb force, electrically screening the first layer. This second layer is loosely associated with the object • The EDL is closely related to electro kinetic phenomena and electro acoustic phenomena.

Zeta potential • Zeta potential is a scientific term for electrokinetic potential in colloidal systems. It is usually denoted using the Greek letter zeta (ζ). • Zeta potential is the potential difference between the dispersion medium and the stationary layer of fluid attached to the dispersed particle. • Zeta potential [m. V] illustrates the stability behavior of the colloids. • The significance of zeta potential is that its value can be related to the stability of colloidal dispersions.

Gibbs Free Energy • Four quantities called "thermodynamic potentials" are useful in the chemical thermodynamics of reactions and non-cyclic processes. They are internal energy, the enthalpy, the Helmholtz free energy and the Gibbs free energy. The Gibbs free energy G is defined by

• The change in Gibbs free energy, ΔG, in a reaction is a very useful parameter. It can be thought of as the maximum amount of work obtainable from a reaction. • For example, in the oxidation of glucose, the change in Gibbs free energy is ΔG = 686 kcal = 2870 k. J. This reaction is the main energy reaction in living cells.
- Slides: 36