Adolescent Immunization Recommendations Immunization opportunities Practical help Contributors

Adolescent Immunization Recommendations Immunization opportunities Practical help Contributors: Jerold Aronson, MD, MPH, FAAP; William Atkinson, MD, MPH; Sharon Cowden, MD, FAAP; Cynthia De. Muth, MD, FAAP; Michael Harkness, MD, FAAP; Kathleen Marker, RN; 1 Jonathan Pletcher, MD; Sharon Shepherd, BS; Amy Wishner, MSN, RN 8/19/2010

Practical tools Handouts include: Immunization schedules “Summary of Recommendations” School requirements Resource list Sample bookmarks and posters – free on request (English and Spanish) 2
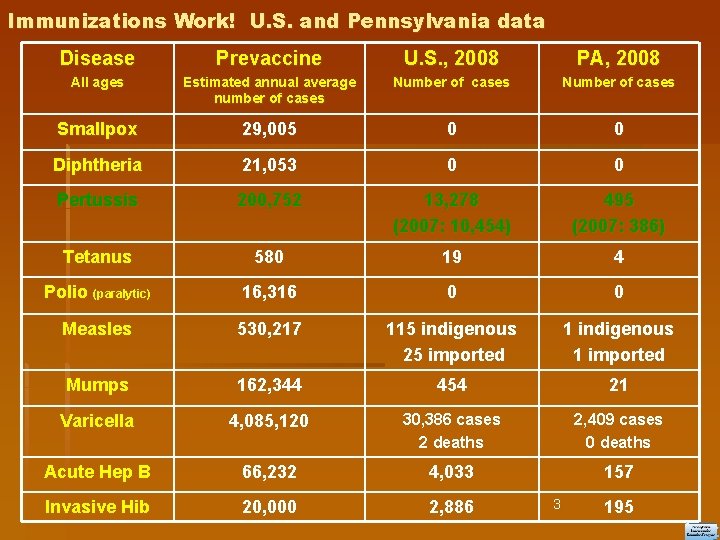
Immunizations Work! U. S. and Pennsylvania data Disease Prevaccine U. S. , 2008 PA, 2008 All ages Estimated annual average number of cases Number of cases Smallpox 29, 005 0 0 Diphtheria 21, 053 0 0 Pertussis 200, 752 13, 278 (2007: 10, 454) 495 (2007: 386) Tetanus 580 19 4 Polio (paralytic) 16, 316 0 0 Measles 530, 217 115 indigenous 25 imported 1 indigenous 1 imported Mumps 162, 344 454 21 Varicella 4, 085, 120 30, 386 cases 2 deaths 2, 409 cases 0 deaths Acute Hep B 66, 232 4, 033 157 Invasive Hib 20, 000 2, 886 3 195
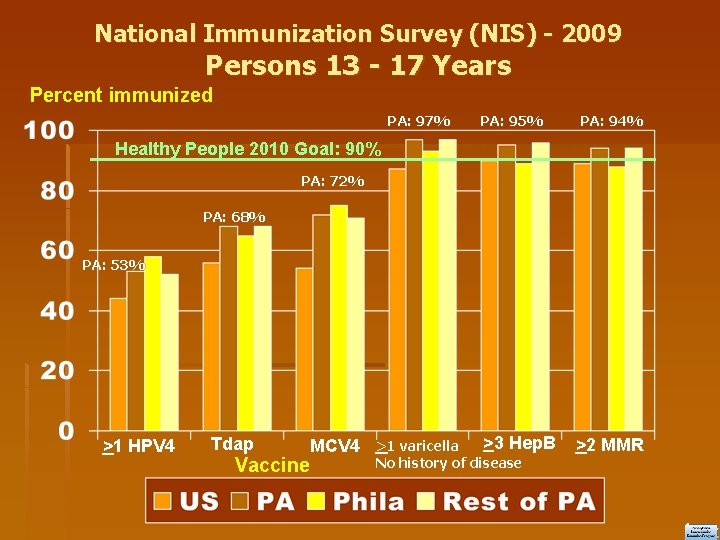
National Immunization Survey (NIS) - 2009 Persons 13 - 17 Years Percent immunized PA: 97% PA: 95% PA: 94% Healthy People 2010 Goal: 90% PA: 72% PA: 68% PA: 53% >1 HPV 4 Tdap MCV 4 Vaccine >3 Hep. B >1 varicella No history of disease 4 >2 MMR

Why emphasize adolescent immunization? Adolescents still need preventive health care! Keep sending the message that immunizations are needed New environments, new risks - travel, summer programs, college Experimentation Sexual activity, smoking, drinking 5

Why emphasize adolescent immunization? Prevent long term sequelae - cervical cancer (HPV), hepatitis A and B, meningitis complications Increase herd immunity (Tdap, varicella, influenza) Stay up-to-date with newer vaccines and recommendations – HPV, Tdap, 2 nd dose varicella, influenza, hepatitis A, meningococcus 6

Adolescent Development Early Adolescence (8 -12 years of age) Rapid physical and pubertal development Testing the boundaries of parental authority Concrete thinkers No clear sexual identity No right or capacity for consent 7

Adolescent Development Middle Adolescence (13 -16 years of age) Slowing physical development, increasing variation Testing, going beyond parental boundaries Beginning abstract thinking, prone to concrete decision-making under duress Sexual identity development through 8 experimentation

Adolescent Development Late Adolescence (17 -19 years of age) Transition to college, work, community Greater independence More able to independently access preventive care Decreased perceived need for preventive health visits 9

Late adolescence (continued) Key time to educate about vaccinepreventable diseases and adult immunizations Take advantage of current insurance coverage, VFC, and college health services Acknowledge risk factors with implications for immunization Chronic illness (sickle cell, cystic fibrosis, asthma, HIV) Sexual behavior 10 Plans for travel and career

Adolescent Consent for Immunization Who can consent? 11 -17 years: need parent/guardian consent, except for hepatitis B vaccine at STD clinics Emancipated Minor can consent Discuss risks and benefits with person providing consent and with adolescent Coming in without a parent Practice should have written policy 11

Compliance complete series Hepatitis B Human Papillomavirus (HPV) Measles Mumps Varicella Hepatitis A 12

Adolescent Vaccines Overview - Routine at 11 -12 years HPV for females, permissive recommendation for males Meningococcal conjugate Tdap (1 dose) Catch-up if not fully immunized previously Hepatitis B Polio MMR Varicella Hepatitis A High-risk Pneumococcal polysaccharide Meningococcal conjugate revaccination Annual influenza vaccine 13

Recommended Immunization Schedules • Are all staff using the same schedule? • Avoid missed opportunities 14

Hepatitis B Disease Transmission: parenteral or mucosal exposure to body fluids containing hepatitis B virus Chronic infection: 25% die prematurely from cirrhosis or liver cancer 1 st anti-cancer vaccine 15

Hepatitis B Vaccine Compliance – need to complete the series No need to restart – immune cell memory Booster doses not routine Adolescent 2 -dose schedule option: Recombivax® adult dose For 11 -15 years of age: time 0 and 4 -6 months PA doing well! In PA, 95% of adolescents 13 -17 years of age have a complete hepatitis B vaccine series. U. S. rate: 90%. 16

Hepatitis B Vaccine If never immunized, hepatitis B immunization is routine care – no need to identify risk factor Household with international adoptee – immunize before child’s arrival Combined hepatitis A-hepatitis B vaccine (Twinrix®) Patients > 18 years of age Two schedules: 0, 1 -, 6 -12 months OR 0, 7, 21 -30 days plus booster at 12 months 17

Diphtheria Can involve almost any mucous membrane Clinical picture may include: Conjunctivitis Pharyngeal membrane “Bullneck” Severe myocarditis Unvaccinated travelers at risk (PA death – 2003) 18 Photos: CDC. (L) 10 year old boy with diphtheria; (R) Thick, grey coating over back of throat

Tetanus 4 Pennsylvania cases in 2008 Rate of at least 1 dose of Tdap among persons ages 13 -17 years National: 56% Pennsylvania: 68% 19 Photo: Sir Charles Bell's portrait of a man with generalized tetanus

Pertussis (whooping cough) 468 confirmed cases in PA in 2009 Incidence peaks: Younger than 6 months (unimmunized) 11 -18 years (waning immunity) 34% of cases are adolescents 11 -18 yrs. May be mild and undiagnosed in older ages (may not seem very ill but can spread disease) Family member is usual source (75%) of infant pertussis Siblings - 20% of family member source cases 20 Bisgard KM et al. Infant pertussis: who was the source? Pediatr Infect Dis J. 2004; 23: 985 -989

Pertussis Clinical picture in adolescents: 72%-100% - paroxysmal cough, difficulty breathing and sleeping 30%-65% - whoop 50%-70% - post-tussive vomiting 1%-2% - hospitalized, pneumonia, or rib fracture 0. 2%-1% - seizures or loss of consciousness Lee GM, Lett. S, Schauer S, et al. Societal costs and morbidity of pertussis in adolescents and adults. Clin Infect Dis. 2004: 39: 1572 -1580. 21

Tdap Vaccine – one dose (for now) Boostrix (GSK): for 10 -64 years Adacel (sanofipasteur): for 11 -64 years ® ® Recommended: 7 to 9 years: Td for wound care or catch-up (ACIP recommendations for pertussis protection for this age group coming) 10 years: Boostrix for wound care or catch-up 11 years: one dose of either Tdap 12 to 64 years: one dose of Tdap for wound care or 22 catch up, then use Td ®

Tdap Vaccine Minimum interval between Td and Tdap - ACIP and AAP did NOT set a minimum interval Canadian data on safety of 2 year interval between Td and Tdap – minimal side effects Consider family, community pertussis – use clinical judgment (may use shorter interval than 2 years) 23

Tdap Contraindications Severe allergic reaction to vaccine component or following a prior dose (true for all vaccines) Encephalopathy not due to another identifiable cause occurring within 7 days after vaccination with a pertussiscontaining vaccine 24

Tdap Precautions History of Arthus-type reaction following previous tetanus- or diphtheria-containing vaccine Progressive neurologic disorder, uncontrolled epilepsy, progressive encephalopathy Severe (anaphylactic) latex allergy History of Guillain Barré Syndrome within 6 weeks of previous dose of tetanus-containing vaccine Moderate or severe illness 25

Tdap Precautions – different than for DTa. P Tdap is given to older children and adults (more mature immune system) NOT precautions to Tdap: Post-DTa. P/DT history of: Temperature > 105° Collapse or shock-like state Persistent crying lasting 3 hours or more Convulsions with or without fever Limb swelling Stable neurologic disorder Pregnancy Breastfeeding Immunosuppression including HIV infection Minor illness 26 Antibiotic use
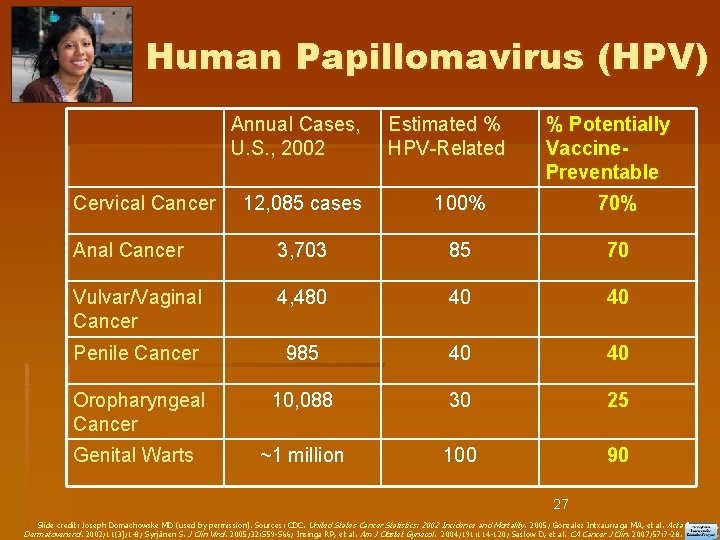
Human Papillomavirus (HPV) Annual Cases, U. S. , 2002 Cervical Cancer Estimated % HPV-Related % Potentially Vaccine. Preventable 12, 085 cases 100% 70% Anal Cancer 3, 703 85 70 Vulvar/Vaginal Cancer 4, 480 40 40 Penile Cancer 985 40 40 10, 088 30 25 ~1 million 100 90 Oropharyngeal Cancer Genital Warts 27 Slide credit: Joseph Domachowske MD (used by permission). Sources: CDC. United States Cancer Statistics: 2002 Incidence and Mortality. 2005; Gonzalez Intxaurraga MA, et al. Acta Dermatovenerol. 2002; 11(3); 1 -8; Syrjänen S. J Clin Virol. 2005; 32: S 59 -S 66; Insinga RP, et al. Am J Obstet Gynecol. 2004; 191: 114 -120; Saslow D, et al. CA Cancer J Clin. 2007; 57: 7 -28.

Human Papillomavirus (HPV) Type 16 and 18 6 and 11 Women 70% of cervical cancer v 70% of anal/genital cancer v Transmission v 70% of anal and penile cancer v Transmission v 90% of genital warts v 90% of Recurrent Respiratory Papillomatosis v 90% of RRP (RRP) lesions v Transmission v Cervarix (GSK): 16 and 18 only Photos: (L) Georgia AAP; (R) PA AAP Men Gardasil (Merck): 6, 28 11, 16 and 18

HPV Vaccines Gardasil (Merck) – licensed by FDA for: Females ages 9 through 26 years of age Males ages 9 through 26 years of age Cervarix (GSK) – licensed by FDA for females 10 through 25 years of age ACIP harmonized HPV vaccine recommendations for females: 3 -dose series recommended at 11 -12 years old Schedule: 0, 1 -2 months, 6 months after first dose O. K. as young as 9 years of age Different VISs for Gardasil and Cervarix 29

HPV Vaccines No evidence of effectiveness against pre-existing infection or disease Even if prior HPV infection, immunize May not be infected with all vaccine strains Not for older females in U. S. (yet) – pending FDA decision Gardasil studies show effectiveness through age 45 years 30

HPV Vaccine Pregnancy: Ask but don’t test Defer start or continuation of HPV vaccine if pregnant If HPV vaccine given during pregnancy Gardasil: www. gardasil. com or 1 -800 -986 -8999 Cervarix: 1 -888 -452 -9622 No change in cervical cancer screening 31

HPV Vaccine for Males FDA licensed Gardasil (not Cervarix) for males 9 -26 years for prevention of genital warts caused by HPV types 6 and 11 ACIP: Eligible males 9 through 26 years of age “may be vaccinated” (rather than stronger “recommended”) VFC covers – Check with private insurers Same schedule as females 32

HPV Vaccine Safety Study of Vaccine Adverse Event Reporting System (VAERS) 6/2006 – 12/2009 Pain, swelling at injection site, headache, nausea, fever Syncope – have adolescent patients wait 15 minutes after any immunization Guillain-Barré Syndrome (GBS) or blood clots – no increase above background rate Deaths – 49 U. S. deaths reported following Gardasil: 28 confirmed, others missing data. No pattern suggesting deaths caused by vaccine. 33

Myths About HPV Vaccine HPV vaccine encourages my daughter/son to have sex HPV is sexually transmitted and my daughter/son is not sexually active - no need for HPV vaccine Free poster (English or Spanish) from iep@paaap. org 34

Meningococcal Disease At least 13 serogroups Most invasive disease: A, B, C, Y, W-135 Relative importance of strains depends on geography and age Transmission: respiratory droplet spread or direct contact Abrupt onset, rapid course of disease Case fatality rate: 9%-12% (up to 40% with meningococcemia) 1%-19% of survivors suffer serious sequelae including deafness, neurologic deficit, or limb loss 35

Meningococcal Disease 15 year old boy Waterhouse-Friderichsen Syndrome – Neisseria meningitidis Risk factors for invasive disease include: Host: asplenia, smoking, upper respiratory infection Crowding College freshmen living in dorm – College students not living in dorm are not at higher risk than others of similar age 36 Photo ©Massachusetts Medical Society. All rights reserved.

Meningococcal Conjugate Vaccines (MCV 4) Menactra (ages 2 -55 yrs. ) Menveo (ages 11 -55 yrs. ) Reduce asymptomatic carriage of N. meningitidis ACIP recommendations – 1 dose of MCV 4 All persons 11 -18 years Person 2 -55 years at increased risk (includes college freshmen in dorms, travel to or residence in endemic countries) 15 -minute wait recommended after Menveo 37 (syncope risk) Photo: Meningococcemia / AAP

Meningococcal Revaccination of high-risk children/teens through age 18 years Received 1 dose MCV 4 or MPSV 4 at ages 2 -6 years, give MCV 4 three years after previous dose Received 1 dose MCV 4 or MPSV 4 after age 6 years, give MCV 4 five years after previous dose Revaccination at 5 year intervals as long as in increased risk group - with MCV 4 if < 55 years and with MPSV 4 if > 56 years 38

Meningococcal Vaccination College Freshmen ACIP does not recommend that college freshmen living in dormitories who were previously vaccinated with MCV 4 be revaccinated College freshmen living in dormitories who were vaccinated with MPSV 4 ≥ 5 years previously are recommended to be vaccinated with MCV 4 MMWR September 25, 2009 39

Pneumococcal Disease Transmission: person-to-person via respiratory droplets Outbreaks – uncommon, occur in crowded environments Complications: pneumonia, bacteremia, meningitis Risk factors: asplenia, sickle cell disease, HIV infection, cochlear implant, smoking or exposure to passive smoking Some populations (African American, Alaskan Native, some Native American) – reasons for 40 higher risk unclear

PCV 13 Recommendations see tables in MMWR Children 6 -18 years at increased risk for invasive pneumococcal disease (IPD) because of sickle cell disease, HIV infection or other immunocompromising condition, cochlear implant, or cerebrospinal fluid leaks, regardless of previous PCV 7 or PPSV 23 - single dose of PCV 13 may be administered 41

Pneumococcal Polysaccharide Vaccine PPV 23 (Pneumovax 23® and Pnu-Immune®) For high-risk adolescents Chronic heart, lung, liver, kidney disease (e. g. , C. O. P. D or emphysema, but not asthma) Asplenic Sickle cell disease Cochlear implant Immunocompromised HIV infection Living in institutional environment 42

Pneumococcal Polysaccharide Vaccine (PPV 23) (Pneumovax 23® and Pnu-Immune®) Revaccinate once (not every 5 years) Revaccinate after 5 years if still high risk Why only 1 revaccination? Trials show same result with single and multiple boosters 43

Influenza in Adolescents Illness, hospitalization Miss school, sports, work (Parents miss work) VECTORS – spread to younger siblings, older adults, schools Contacts of infants too young to be immunized Vaccines do not work as well in seniors; may not be well-protected even if immunized Spread influenza to teachers, classmates Death 44

Influenza Vaccine Pandemic 2009 H 1 N 1 first detected in U. S. in April 2009 Identified after manufacturers started making seasonal influenza vaccine (so two influenza vaccines) 2010 -2011 influenza season: Universal annual influenza immunization – all people 6 months of age and older (unless medical contraindication) One vaccine for 2010 -2011! Includes pandemic 2009 H 1 N 1 vaccine strain 45

Influenza Vaccine Egg allergy – currently contraindicated but check with allergist Start giving vaccine as soon as it arrives Immunize throughout influenza season Recommended for pregnant women since 2004 (CDC and ACOG) 46

Influenza – Immunize all Eligible Practice Staff Nationally 34. 7% of HCP received both influenza vaccines 2009 -2010 Don’t get the flu - your coworkers need you! Don’t spread the flu - you can spread influenza even before symptoms appear The first doses of your first shipment of flu vaccine are for your practice 47

Hepatitis A Disease Lost school and activity time, hospitalization, death Fecal-oral transmission Food-borne outbreaks – PA, 2003 (Chi -Chi’s, green onions) 601 cases, 124 hospitalizations, 3 deaths Risk factors include child or employee in day care (baby sitting), foreign travel, household contact of adoptee from 48 hepatitis A-endemic country

Hepatitis A Vaccine 2 doses at least 6 months apart Post-exposure – vaccine preferred to IG for healthy persons 12 months through 40 years Easier to administer, long-term protection, equivalent efficacy Children/adolescents 2 -18 years – VFC will cover Consider adolescent risk factors Travel, work, sexual activity Consider Twinrix® (Hep A/Hep B) for > 18 years 49
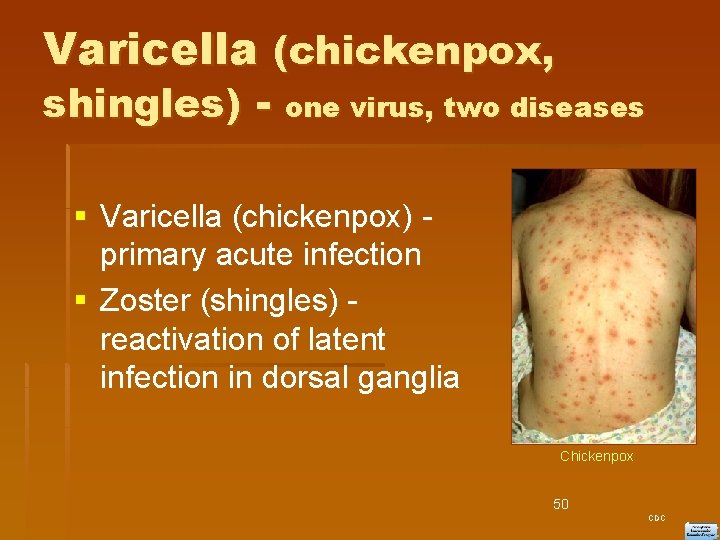
Varicella (chickenpox, shingles) - one virus, two diseases Varicella (chickenpox) - primary acute infection Zoster (shingles) - reactivation of latent infection in dorsal ganglia Chickenpox 50 CDC
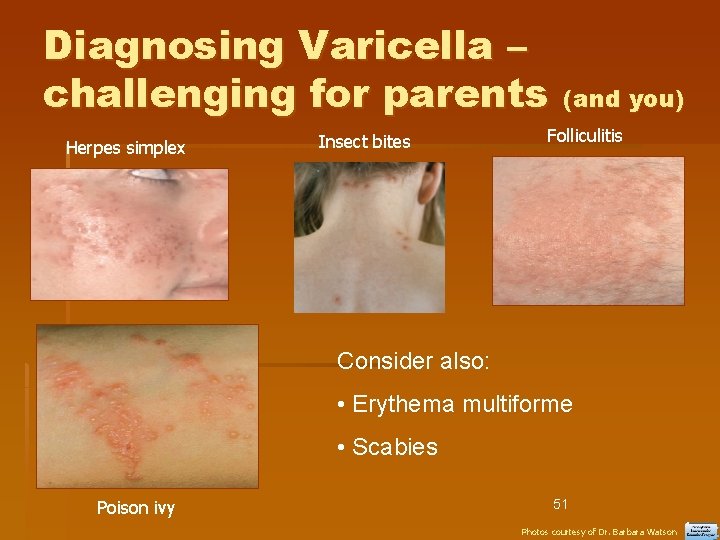
Diagnosing Varicella – challenging for parents Herpes simplex Insect bites (and you) Folliculitis Consider also: • Erythema multiforme • Scabies Poison ivy 51 Photos courtesy of Dr. Barbara Watson

Varicella - do not assume immunity Parent history alone - no longer evidence of immunity (except for school requirements) Immunity to varicella: • Documentation of age-appropriate vaccination • Lab evidence of immunity or disease • Born in the U. S. prior to 1980 - except for healthcare workers and pregnant women • Health care provider diagnosis • Verified history of herpes zoster 52

Varicella Vaccine No evidence of immunity? Immunization: 2 doses of vaccine Minimum interval between doses 12 months – 12 years of age: 3 months 13 years and older: 4 weeks BUT – do not repeat dose if interval was at least 28 days 53

Poliovirus Fecal-oral transmission Up to 95% of infections are asymptomatic Single lifetime booster for travel to polioendemic countries (Afghanistan, Nigeria, India, Pakistan) VAPP in unvaccinated U. S. traveler, 2005 Infections in Amish, 2005 54

Measles and Mumps College-age outbreaks Un- or under-immunized cohort plus travel and geographic mixing 2010 measles outbreak in Vancouver, Canada 2 importations (Japan/China and India); 2 cases from Canada imported into U. S. Mumps Outbreaks – U. K. , U. S. , Canada NY and NJ, 2009 -2010: 3, 100+ cases tied to 11 yr. old who travelled to U. K. (U. K. : 7, 400+ cases in 2009), spread to California in May, 2010 3 rd dose MMR used in Orange County, NY – results pending Review all immunizations for travel Assess staff immunity 55

Rubella immunity – 1 dose of vaccine or serology Important for adolescent females as they approach childbearing age – prevent congenital rubella syndrome 56

MMR Vaccine Two doses – complete the series Egg allergy not a contraindication Separate measles, mumps, rubella antigens not available (No scientific evidence of any advantage) 57

Summary Tdap: Single dose to replace Td booster, wound care, consider community pertussis Human papillomavirus (HPV): 3 doses, females 9 -26 years, recommended 11 -12 years; permissive recommendation for males Pneumococcal polysaccharide: high-risk Influenza: high-risk and household contacts, all who wish 58 Photos: PA AAP

Summary Hepatitis A: can complete series Hepatitis B: complete series Polio: catch up unvaccinated, consider booster for travel Measles-Mumps-Rubella (MMR): 2 doses routine, beware unvaccinated travelers Varicella: catch-up 2 nd dose Meningococcal: 1 dose MCV 4 for 11 -18 years Revaccinate if high-risk, as discussed 59 Photos: PA AAP

Adolescent Immunization Schedule (ideal) 11 -12 years Primary platform – recommended by ACIP, AAP, Society for Adolescent Medicine 14 -18 years Catch-up and complete series Take advantage of having VFC or other coverage 60

Adolescent Immunization Take advantage of every opportunity to immunize (camp, sports, driver, and pre-college physicals; acute care visits) Provide multiple vaccines in a single visit Address patient/family resistance Recommend vaccines! You have an important impact on immunization rates! 61 Photo: PA AAP

Adolescent Immunization Outreach to high-risk groups “Vaccinate before you graduate” and “Vaccines aren’t just for babies” programs Collaborate with local immunization coalitions Measure how you are doing Assess immunizations at every visit Don’t miss an opportunity to immunize 62 Photo: PA AAP

Reminder/Recall What do you do now? Electronic health record pop-ups or reminders Consider standing orders – see samples: www. immunize. org Immunization registries can help 63

Vaccine Storage & Handling Ø Work with VFC Ø See IEP Resource Packet Ø Refrigerator: 35 - 46°F (2 -8°C) • Freezer: < 5°F (-15°C) Ø Check & record temps twice daily Ø Keep logs for 3 years Ø Use only certified calibrated thermometers Ø Power failure plan Ø Have alternate vaccine storage site Ø Know local source for dry ice (vaccine transport) Ø Inventory & rotate vaccines 64

Out of range temperatures? Take action! Determine cause and correct Contact manufacturers, follow directives Do not use impacted vaccine until manufacturer says o. k. Record length of time temperature was out of range Record all actions taken VFC sites: Use VFC Vaccine Storage and Handling Incident Worksheet Report to VFC 65

Immunization Registries Search patient history, print records, generate reminder/recall, print VAERS forms, find patient contraindications and precautions, maintain inventory, provide VFC reports SIIS – Pennsylvania outside of Philadelphia Web site: www. health. state. pa. us/pasiis KIDS – Philadelphia and providers nearby Mandatory reporting all ages Web site: https: //kids. phila. gov 66

Adverse Events Syncope and teens (all vaccines, especially HPV) Have them sit during & wait 15 minutes after immunization Acute reactions - have emergency plan, practice it, involve new staff Report adverse events to VAERS [www. vaers. hhs. gov] and immunization registry You do not have to decide if the vaccine caused the adverse event 67

Vaccine-hesitant Patient or Family Use Vaccine Education Center materials: www. vaccine. chop. edu Recommended web sites: www. cdc. gov/vaccines (CDC) www. immunize. org (Immunization Action Coalition) www. aap. org/immunization/ (AAP) www. ecbt. org (Every Child by Two) www. getvaxed. org (teen site of Parents of Kids with Infectious Diseases PKIDS) Document patient refusal 68

Vaccine Safety Concerns Nothing is completely “safe” but vaccines are among the most thoroughly tested and safest things we put into our bodies Refusing a vaccine means assuming the known risks of the disease and of spreading the disease to others PA school law mandates keeping an unimmunized adolescent out of school if there is a case of the disease 69

Vaccine Safety Concerns “Natural” immunization (from disease) comes at a high cost – risks of disease, permanent damage, death Clear, consistent, reproducible research: MMR vaccine does not cause autism Thimerosal does not cause autism Multiple vaccines at one time do not cause autism Anti-vaccine groups frequently change targets 70

Vaccine Schedules Other Than CDC/AAP/AAFP Recommendations “Slippery slope” once you stop following the science-based immunization schedule Organization-wide policy may help – see sample at www. immunize. org Uncompensated time with families, uncompensated nurse-only additional vaccine visits See Jan. 2009 Pediatrics (Offit, Moser) article debunking “Dr. Bob’s” schedule 71

Immunization Champion Help lead and coordinate your team Identify materials, web sites, resources Update practice on new recommendations Ensure office policies and procedures are up-to-date 72

Plans to Improve What can you do to improve? List 2 -3 things Who is your Immunization Champion? Can we help? 73

Final Questions? Thank you for your attention Thank you for your hard work an caring every day! Contact us: PA Immunization Education Program iep@paaap. org (800) 375 -5214 www. paiep. org Facebook: PA Immunization Education Program 74
- Slides: 74