Adjuvant Systemic Treatment Personalized Management of Breast Cancer

Adjuvant Systemic Treatment: Personalized Management of Breast Cancer Dr. Law Siu King Department of Surgery United Christian Hospital

Why do the patients with operable breast cancer receive adjuvant systemic therapy?

Role of adjuvant therapy for operable breast cancer Systemic adjuvant treatment is used to tackles the micrometastasis and that accounts for most of the improved survival for operable breast cancer patient seen in the last few decades.

What are the current evidences?
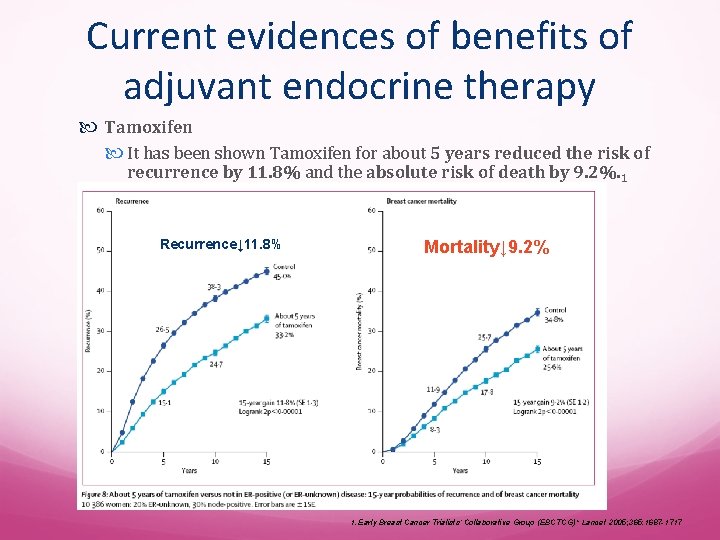
Current evidences of benefits of adjuvant endocrine therapy Tamoxifen It has been shown Tamoxifen for about 5 years reduced the risk of recurrence by 11. 8% and the absolute risk of death by 9. 2%. 1 Recurrence↓ 11. 8% Mortality↓ 9. 2% 1. Early Breast Cancer Trialists’ Collaborative Group (EBCTCG)* Lancet 2005; 365: 1687 -1717

Current evidences of benefits of adjuvant endocrine therapy Aromatase inhibitors AIs (letrozole and anastrozole) have each been shown to improve DFS compared with tamoxifen when given as first line adjuvant therapy for a planned 5 years in postmenopausal women with hormonal receptor-positive early breast cancer 1, 2. 1. BIG 1 -98 Coatest et al J CLin Onco 2007 2. ATAC trial Forbes JF et al Lancet Onco 2008
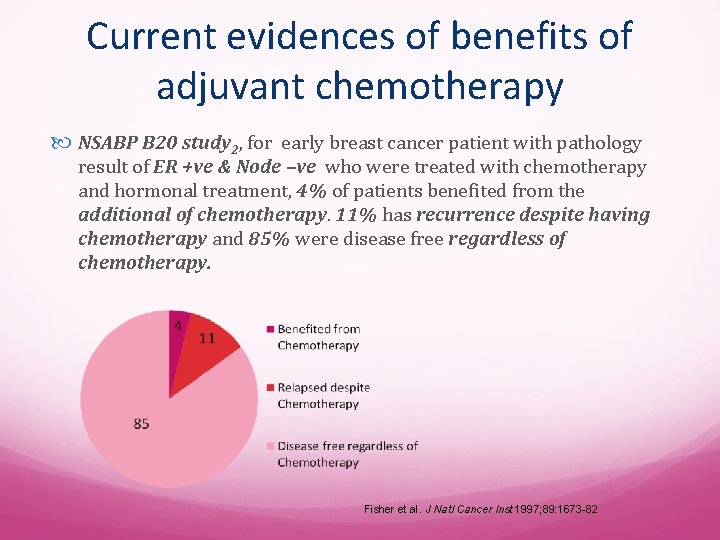
Current evidences of benefits of adjuvant chemotherapy NSABP B 20 study 2, for early breast cancer patient with pathology result of ER +ve & Node –ve who were treated with chemotherapy and hormonal treatment, 4% of patients benefited from the additional of chemotherapy. 11% has recurrence despite having chemotherapy and 85% were disease free regardless of chemotherapy. Fisher et al. J Natl Cancer Inst 1997; 89: 1673 -82
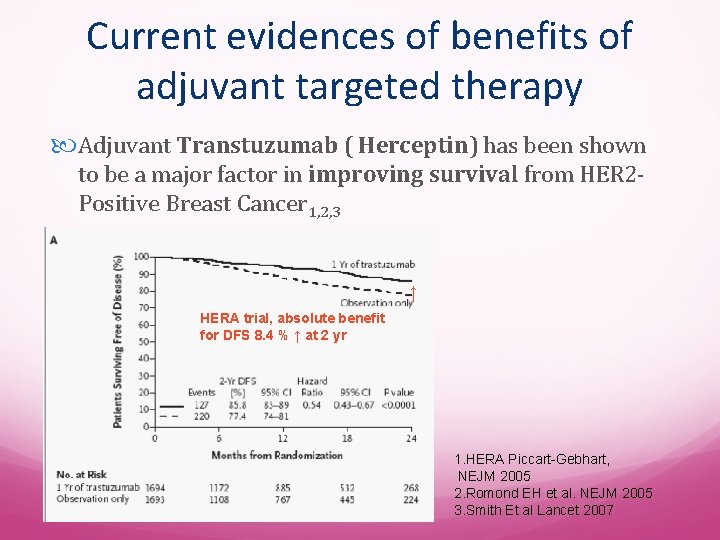
Current evidences of benefits of adjuvant targeted therapy Adjuvant Transtuzumab ( Herceptin) has been shown to be a major factor in improving survival from HER 2 Positive Breast Cancer 1, 2, 3 ↑ HERA trial, absolute benefit for DFS 8. 4 % ↑ at 2 yr 1. HERA Piccart-Gebhart, NEJM 2005 2. Romond EH et al. NEJM 2005 3. Smith Et al Lancet 2007

Adjuvant Systemic Treatment for Breast cancer General principle adjuvant treatment decisions are based on the balance among risk of relapse, proven survival benefits of treatment, and the costs and toxicities of treatment, as well as on patient preference and co-morbidities.

How can we determinate the prognosis ?

Determinants of prognosis For many years , the primary determinants of prognosis included ER, PR and HER 2 status, histology, grade, tumor size and axillary LN status.

Prognostic tools Nottingham prognostic Index( NPI) has been used for many years. It is based 3 factors using the following formula: NPI=p tumor size (cm)x 0. 2+LN stage +histological grade LN stage( score 1, 2 or 3 with 1 =node negative , 2= 1 -3 involved nodes and 3 =more then 3 nodes), histological grade ( score 1, 2, or 3) + histological grade Arbitrary cut-off points of 3. 4 and 5. 4 are used to divide patients into 6 prognostic groups: excellent, good, moderate I and II, Poor and very Poor

Prognostic tools Other pathology based prognostic tool used in breast cancer included Adjuvant!Online Adjuvant! Online can be accessed at www. adjuvantonline. com. It is a decision tool for assessing the risks of an individual patient developing recurrent disease and/or dying within 10 years, when receiving specific treatment on the basis of well validated factors Age, comorbidities, LN status, Tumor size, tumor grade and hormonal receptor status St Gallen Criteria

St. Gallen Consenus St. Gallen derived algorithm for selection of adjuvant systemic therapy for early breast cancer patients includes Tumor size and grade Nodal status, Menopausal status, Peri-tumoral vessels invasion Risk Categories Description Low Risk Node negative AND all of the following features • p. T≤ 2 cm • Grade 1 • Absence of peri-tumoral vascular invasion • HER 2 non-overexpression/ amplification • Age ≥ 35 Intermediate Risk A. Node negative AND at least one of the following features • p. T >2 cm • Grade 2 -3 • Presence of peri-tumoral vascular invasion • HER 2 overexpression/ amplification • Age <35 Endocrine status and HER 2 status. B. Node positive (1 -3 involved nodes) AND HER 2 non-overexpressed/ amplified High Risks A. Node positive (1 -3 involved nodes) AND HER 2 overexpressed/ amplified B. Node positive (4 or more involved nodes) St. Gallen Expert Consensus Meeting 2007

Case discussion

Case Study Madam Choi, 59/F, Excellent Good Past Health Has newly Dx CA Left breast , presented with left breast mass , Confirmed by imaging (MMG/ USG) and histology (Core Biopsy) WLE + Sentinel lymph node biopsy Pathology: Primary tumor - 2. 2 cm , Grade 2, invasive ductal carcinoma, clear resection margins ER 300(+++), PR 300(+++), cerb 2 –ve. No lymphovascular invasion Sentinel lymph nodes – 0/2 Stage IIA (p. T 2 N 0 M 0) What adjuvant systemic therapy should the patient receive? Should the patient receive chemotherapy?

Case Study Madam Choi, 59/F, Excellent Good Past Health Has newly Dx CA Left breast , presented with left breast mass , Confirmed by imaging (MMG/ USG) and histology (Core Biopsy) WLE + Sentinel lymph node biopsy Pathology: Primary tumor - 2. 2 cm , Grade 2, invasive ductal carcinoma, clear resection margins ER 300(+++), PR 300(+++), cerb 2 –ve. No lymphovascular invasion Sentinel lymph nodes – 0/2 Stage IIA (p. T 2 N 0 M 0) Base on the St. Gallen Criteria , She has intermediate risk (T 2 and Grade 2 tumor)
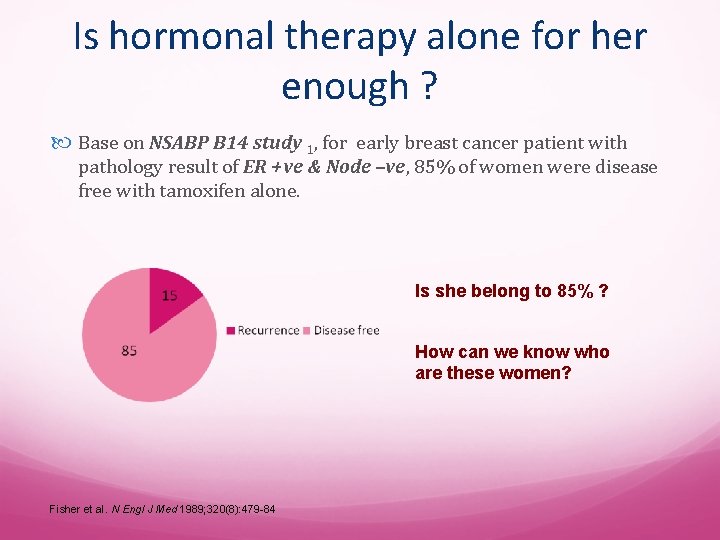
Is hormonal therapy alone for her enough ? Base on NSABP B 14 study 1, for early breast cancer patient with pathology result of ER +ve & Node –ve, 85% of women were disease free with tamoxifen alone. Is she belong to 85% ? How can we know who are these women? Fisher et al. N Engl J Med 1989; 320(8): 479 -84
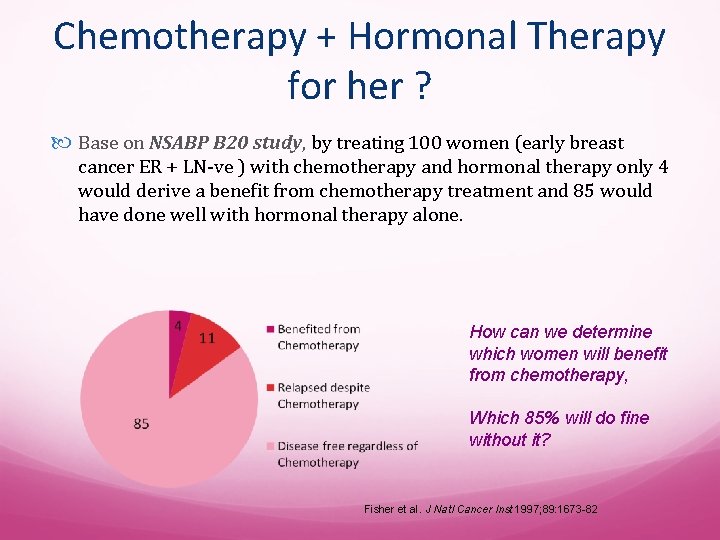
Chemotherapy + Hormonal Therapy for her ? Base on NSABP B 20 study, by treating 100 women (early breast cancer ER + LN-ve ) with chemotherapy and hormonal therapy only 4 would derive a benefit from chemotherapy treatment and 85 would have done well with hormonal therapy alone. How can we determine which women will benefit from chemotherapy, Which 85% will do fine without it? Fisher et al. J Natl Cancer Inst 1997; 89: 1673 -82

Are there any genomic tools that help to determine those patients with highest risks of recurrence and thus benefit most from chemotherapy?

The 21 st Century: genomic era Toward personalized medicine As new technologies emerge in the 21 st century , a number of gene expression signatures have been developed. They can give us information on molecular characteristics of cancer. to assess an individual’s risk of recurrence to provide personalized treatment plan to identify those patients who benefit most from chemotherapy and those who will only benefit from endocrine therapy can be spare the adverse effects of chemotherapy.

Multi-gene assays for Breast Cancer Two commercially available assays that use in the clinical setting have been developed. The 21 -gene recurrence score ( Oncotype DX®; Genomic Health, Red-wood City, Calif. ) and The Amsterdam 70 -gene signature ( Mammaprint®; Agendia, Amsterdam, The Netherlands ) Gene-Expression Signatures in Breast Cancer Molecular Origins of Cancer Christos Sotiriou et al. The New England Journal of Medicine. Boston: Feb 19, 2009. Vol. 360, Iss. 8; pg. 790

What is 21 -gene recurrence score assay ( Oncotype DX®)?
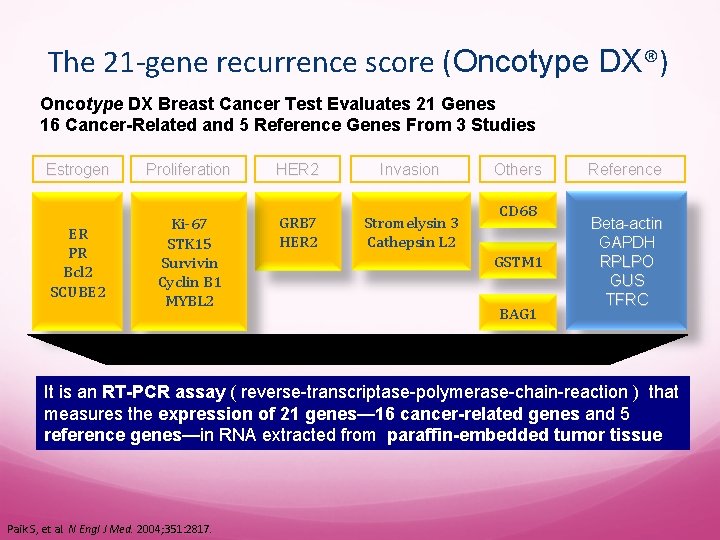
The 21 -gene recurrence score (Oncotype DX®) Oncotype DX Breast Cancer Test Evaluates 21 Genes 16 Cancer-Related and 5 Reference Genes From 3 Studies Estrogen Proliferation HER 2 Invasion ER PR Bcl 2 SCUBE 2 Ki-67 STK 15 Survivin Cyclin B 1 MYBL 2 GRB 7 HER 2 Stromelysin 3 Cathepsin L 2 Others CD 68 GSTM 1 BAG 1 Reference Beta-actin GAPDH RPLPO GUS TFRC It is an RT-PCR assay ( reverse-transcriptase-polymerase-chain-reaction ) that measures the expression of 21 genes— 16 cancer-related genes and 5 reference genes—in RNA extracted from paraffin-embedded tumor tissue Paik S, et al. N Engl J Med. 2004; 351: 2817.
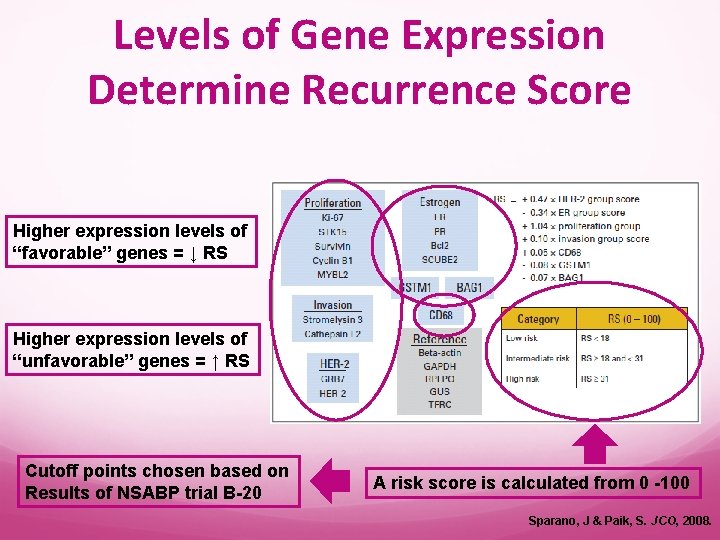
Levels of Gene Expression Determine Recurrence Score Higher expression levels of “favorable” genes = ↓ RS Higher expression levels of “unfavorable” genes = ↑ RS Cutoff points chosen based on Results of NSABP trial B-20 A risk score is calculated from 0 -100 Sparano, J & Paik, S. JCO, 2008.
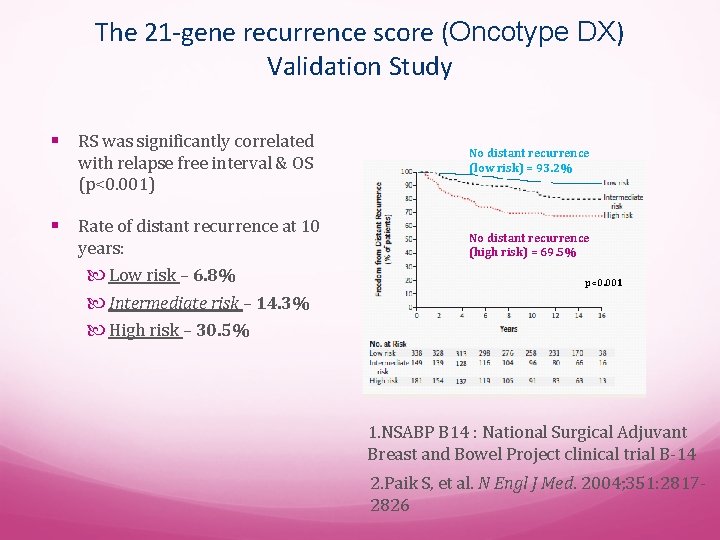
The 21 -gene recurrence score (Oncotype DX) Validation Study § RS was significantly correlated with relapse free interval & OS (p<0. 001) § Rate of distant recurrence at 10 years: Low risk – 6. 8% Intermediate risk – 14. 3% High risk – 30. 5% No distant recurrence (low risk) = 93. 2% No distant recurrence (high risk) = 69. 5% p<0. 001 1. NSABP B 14 : National Surgical Adjuvant Breast and Bowel Project clinical trial B-14 2. Paik S, et al. N Engl J Med. 2004; 351: 28172826
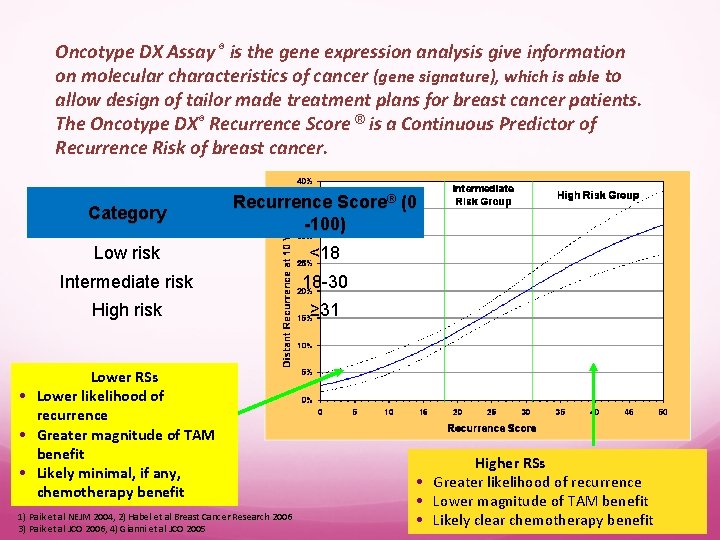
Oncotype DX Assay ® is the gene expression analysis give information on molecular characteristics of cancer (gene signature), which is able to allow design of tailor made treatment plans for breast cancer patients. The Oncotype DX® Recurrence Score ® is a Continuous Predictor of Recurrence Risk of breast cancer. Category Recurrence Score® (0 -100) Low risk <18 Intermediate risk 18 -30 High risk ≥ 31 Lower RSs • Lower likelihood of recurrence • Greater magnitude of TAM benefit • Likely minimal, if any, chemotherapy benefit 1) Paik et al NEJM 2004, 2) Habel et al Breast Cancer Research 2006 3) Paik et al JCO 2006, 4) Gianni et al JCO 2005 Higher RSs • Greater likelihood of recurrence • Lower magnitude of TAM benefit • Likely clear chemotherapy benefit

What is 70 -gene profile (Mammaprint )? Web Source: Agendia
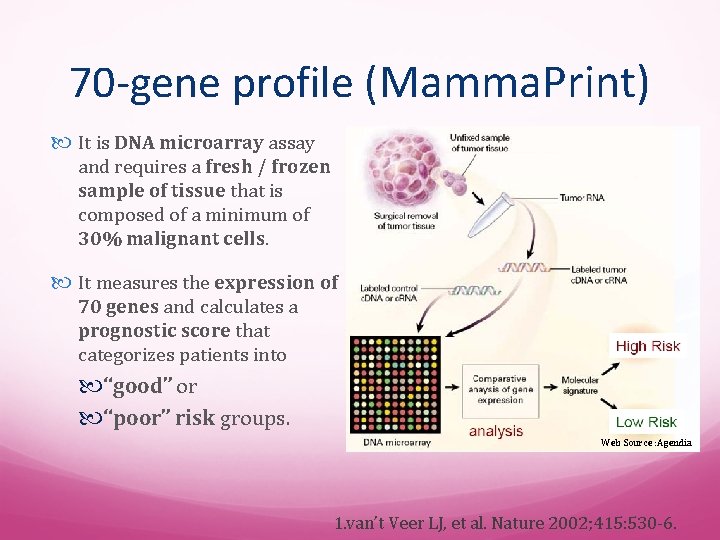
70 -gene profile (Mamma. Print) It is DNA microarray assay and requires a fresh / frozen sample of tissue that is composed of a minimum of 30% malignant cells. It measures the expression of 70 genes and calculates a prognostic score that categorizes patients into “good” or “poor” risk groups. Web Source : Agendia 1. van’t Veer LJ, et al. Nature 2002; 415: 530 -6.
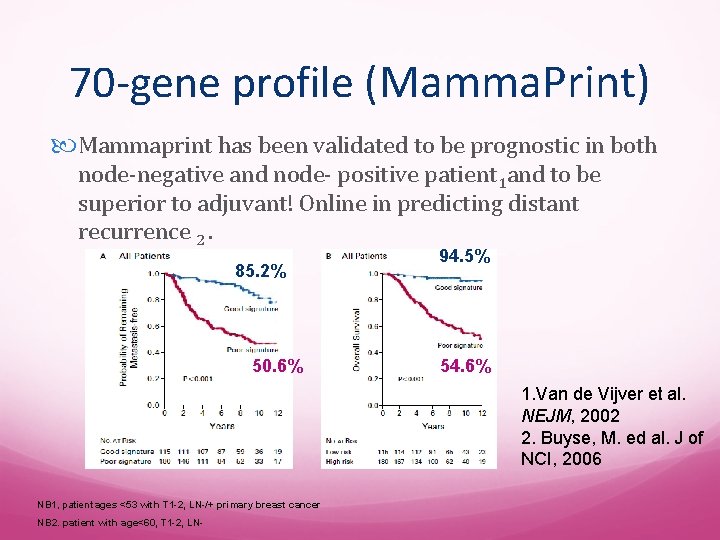
70 -gene profile (Mamma. Print) Mammaprint has been validated to be prognostic in both node-negative and node- positive patient 1 and to be superior to adjuvant! Online in predicting distant recurrence 2. 85. 2% 50. 6% 94. 5% 54. 6% 1. Van de Vijver et al. NEJM, 2002 2. Buyse, M. ed al. J of NCI, 2006 NB 1, patient ages <53 with T 1 -2, LN-/+ primary breast cancer NB 2. patient with age<60, T 1 -2, LN-

Prospective Validation of mammaprint : The MINDACT Trial There is an ongoing prospective randomized European Study, the Microarray in Node- Negative Disease may Avoid Chemotherapy ( MINDACT ) to study the ability of the 70 gene profile to predict clinical benefit from chemotherapy

Readiness of multi-gene assays for clinical use? Oncotype DX® has been endorsed by ASCO 1 and NCCN 2 guidelines to assist clinicians in making decisions about treatment. Mammaprint® has been cleared by the FDA for clinical use. In St. Gallen Consensus 2009 , the panel agreed that validated multi-gene tests, if readily available, could assist in deciding whether to add chemotherapy in cases where its use was uncertain after conventional markers. 1. Harris L, et al. J Clin Oncol. 2007; 25(33): 5287 -312. 2. NCCN Clinical Practice Guidelines in Oncology™ Breast Cancer, (Version 1. 2011)

Case Study Revisited Madam Choi, 59/F, Excellent Good Past Health Has newly Dx CA Left breast , presented with left breast mass , Confirmed by imaging (MMG/ USG) and histology (Core Biopsy) WLE + Sentinel lymph node biopsy Pathology: Primary tumor - 2. 2 cm , Grade 2, invasive ductal carcinoma, clear resection margins ER 300(+++), PR 300(+++), cerb 2 –ve. No lymphovascular invasion Sentinel lymph nodes – 0/2 Stage IIA (p. T 2 N 0 M 0) Base on the St. Gallen Criteria, she was classified to have intermediate risks. So she decided for further gene expression signatures test , Oncotype Dx
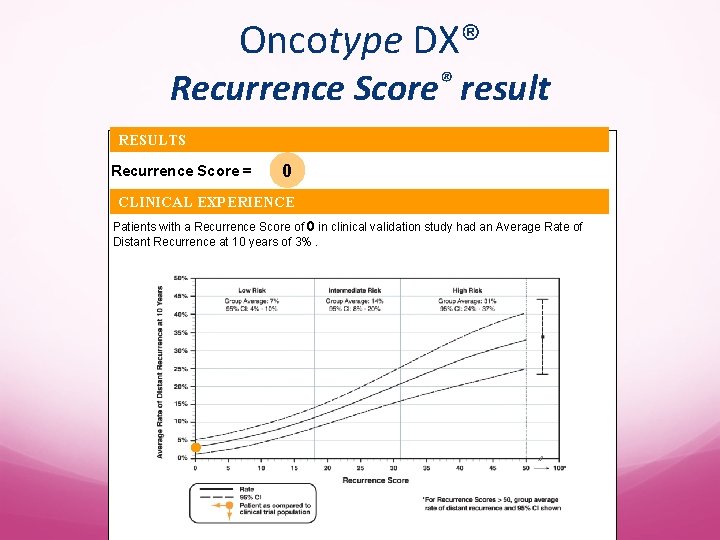
Oncotype DX® Recurrence Score® result RESULTS Recurrence Score = 0 CLINICAL EXPERIENCE Patients with a Recurrence Score of 0 in clinical validation study had an Average Rate of Distant Recurrence at 10 years of 3%.

Oncotype DX® Recurrence Score® result RESULTS Recurrence Score = 0 CLINICAL EXPERIENCE Patients with a Recurrence Score of 0 in clinical validation study had an Average Rate of Distant Recurrence at 10 years of 3%. Her Recurrence score is 0 and her average rate of distant recurrence at 10 years is 3%. Lower likelihood of recurrence Greater magnitude of tamoxifen benefit Likely minimal, if any, chemotherapy benefit

Case study revisited She was recommended RT + Tamoxifen/ Aromatase Inhibitor after joint discussion with oncologist and surgeon. She finally decided for RT + Tamoxifen alone as postoperative systemic adjuvant treatment. She can avoid the S/E of chemotherapy.

Conclusion Systemic adjuvant treatment improves survival for operable breast cancer patient by tackling the micro-metastasis. Decision for adjuvant treatment are made after balancing the risks and benefits. Gene expression signatures (Oncotype. Dx and Mammaprint) allow us to determine individual’s risk of recurrence and to aid selection of optimal therapy for individual patients. There are ongoing prospective randomized trial for the gene expression signatures ( TAILORx and MINDACT).

Thank You!

Q&A


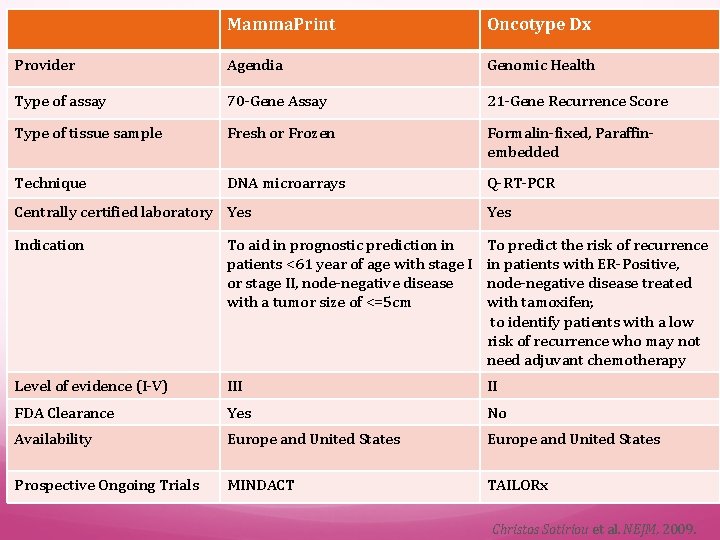
Mamma. Print Oncotype Dx Provider Agendia Genomic Health Type of assay 70 -Gene Assay 21 -Gene Recurrence Score Type of tissue sample Fresh or Frozen Formalin-fixed, Paraffinembedded Technique DNA microarrays Q-RT-PCR Centrally certified laboratory Yes Indication To aid in prognostic prediction in patients <61 year of age with stage I or stage II, node-negative disease with a tumor size of <=5 cm To predict the risk of recurrence in patients with ER-Positive, node-negative disease treated with tamoxifen; to identify patients with a low risk of recurrence who may not need adjuvant chemotherapy Level of evidence (I-V) III II FDA Clearance Yes No Availability Europe and United States Prospective Ongoing Trials MINDACT TAILORx Christos Sotiriou et al. NEJM, 2009.
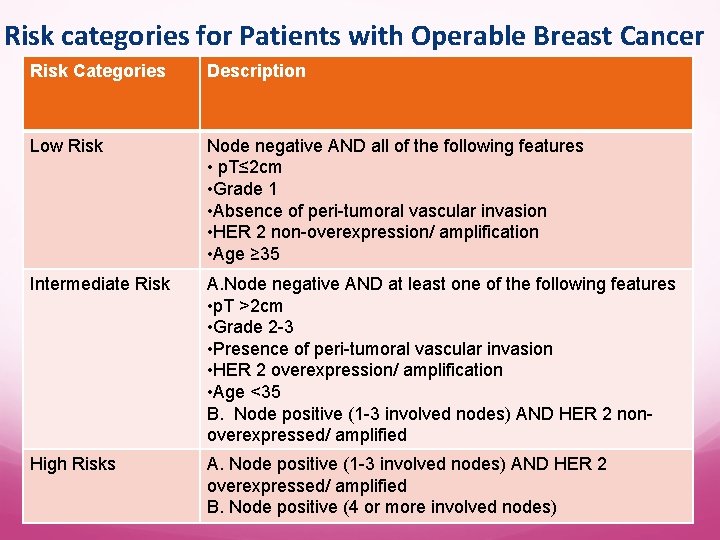
Risk categories for Patients with Operable Breast Cancer Risk Categories Description Low Risk Node negative AND all of the following features • p. T≤ 2 cm • Grade 1 • Absence of peri-tumoral vascular invasion • HER 2 non-overexpression/ amplification • Age ≥ 35 Intermediate Risk A. Node negative AND at least one of the following features • p. T >2 cm • Grade 2 -3 • Presence of peri-tumoral vascular invasion • HER 2 overexpression/ amplification • Age <35 B. Node positive (1 -3 involved nodes) AND HER 2 nonoverexpressed/ amplified High Risks A. Node positive (1 -3 involved nodes) AND HER 2 overexpressed/ amplified B. Node positive (4 or more involved nodes)

St. Gallen Consensus 2009 Treatment Modality Indication Comment Endocrine therapy Any ER staining ER Negative and PR Positive probably artefactual Anti. HER 2 therapy ASCO/CAP HER 2 positive(>30% intense and complete staining (IHC) or FISH>2. 2+) May use clinical trial definitions In HER 2 -postive disease (with anti-HER 2 therapy) Trial evidence for transtuzumab is limited to use with or following chemotherapy Combined endocrine therapy + Anti-HER 2 therapy without chemotherapy in strongly ER-positive, HER 2 -positive is logical but unproven In triple-negative disease Most Patients No proven alternatives; most at elevate risk In ER-positive, HER 2 negative disease (with Variable according to risk See table 3 Chemotherapy
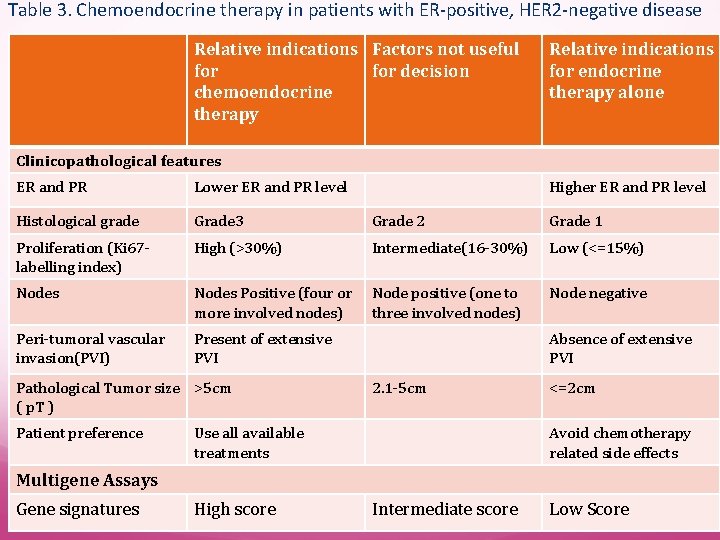
Table 3. Chemoendocrine therapy in patients with ER-positive, HER 2 -negative disease Relative indications Factors not useful for decision chemoendocrine therapy Relative indications for endocrine therapy alone Clinicopathological features ER and PR Lower ER and PR level Histological grade Grade 3 Grade 2 Grade 1 Proliferation (Ki 67 labelling index) High (>30%) Intermediate(16 -30%) Low (<=15%) Nodes Positive (four or more involved nodes) Node positive (one to three involved nodes) Node negative Peri-tumoral vascular invasion(PVI) Present of extensive PVI Pathological Tumor size >5 cm ( p. T ) Patient preference Higher ER and PR level Absence of extensive PVI 2. 1 -5 cm Use all available treatments <=2 cm Avoid chemotherapy related side effects Multigene Assays Gene signatures High score Intermediate score Low Score

Trastuzumab ( Herceptin) Trastuzumab, a monoclonal antibody was successfully developed to attack the cell Surface receptors of Her 2/Neu oncogene found in breast cancer cells

Prognostic significance – HER 2/neu 20% of breast cancer patients have HER 2/neu gene amplification Gene amplification ® cytoplasm glucoprotein overexpression HER 2 amplification or overexpression has been associated with higher tumour grade, negative ER status, higher tumour proliferation (e. g. Ki-67); and therefore poor prognosis IHC (expression) vs. FISH (gene amplification)
- Slides: 47