Adjuvant Denosumab In Early Breast Cancer First Results

Adjuvant Denosumab In Early Breast Cancer — First Results From The International Multicenter Randomized Phase III Placebo-controlled D-CARE Study R Coleman, D Finkelstein, C Barrios, M Martin, H Iwata, J Glaspy, Y Zhou, D Jandial, A Chan on behalf of the D-CARE Global Investigators Robert E. Coleman
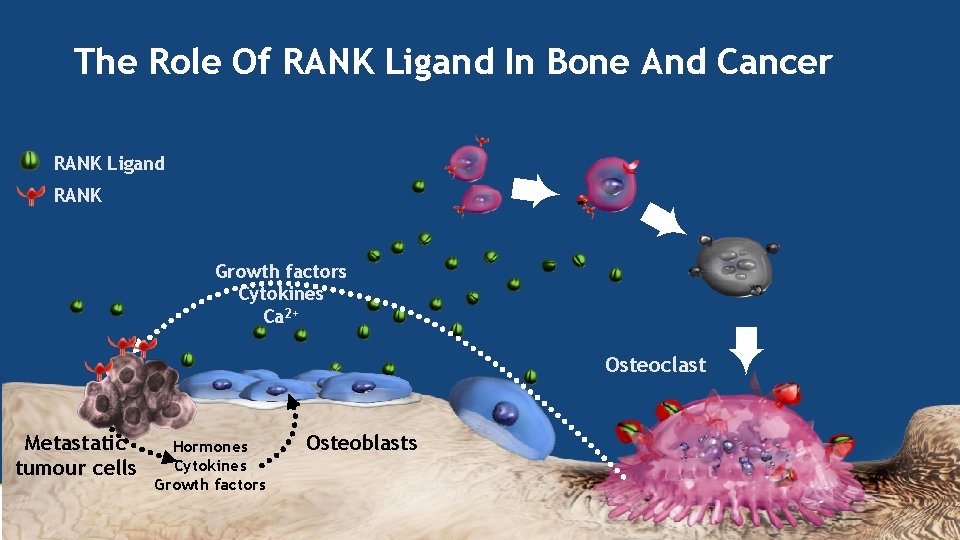
The Role Of RANK Ligand In Bone And Cancer RANK Ligand ➨ RANK ➨ Osteoclast Metastatic tumour cells Hormones Cytokines Growth factors Osteoblasts Robert E. Coleman ➨ Growth factors Cytokines Ca 2+
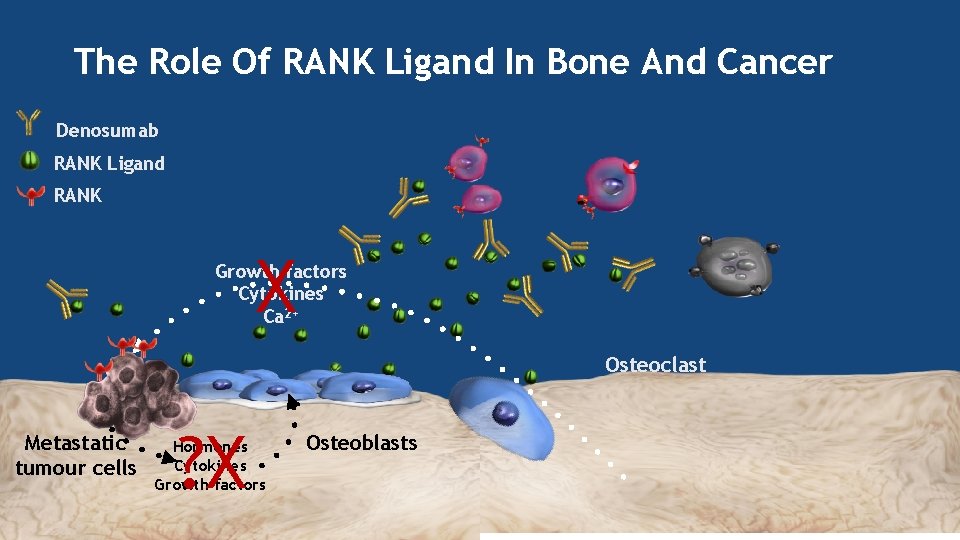
The Role Of RANK Ligand In Bone And Cancer Denosumab RANK Ligand RANK X Growth factors Cytokines Ca 2+ Osteoclast Metastatic tumour cells ? X Hormones Cytokines Growth factors Osteoblasts Robert E. Coleman
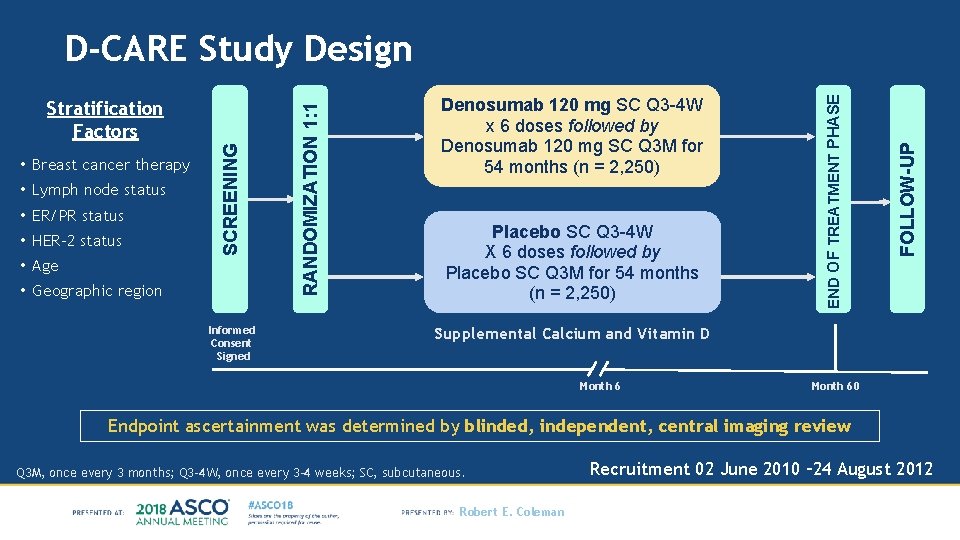
• ER/PR status • HER-2 status • Age • Geographic region Informed Consent Signed Placebo SC Q 3 -4 W X 6 doses followed by Placebo SC Q 3 M for 54 months (n = 2, 250) FOLLOW-UP • Lymph node status Denosumab 120 mg SC Q 3 -4 W x 6 doses followed by Denosumab 120 mg SC Q 3 M for 54 months (n = 2, 250) END OF TREATMENT PHASE • Breast cancer therapy SCREENING Stratification Factors RANDOMIZATION 1: 1 D-CARE Study Design Supplemental Calcium and Vitamin D Month 60 Endpoint ascertainment was determined by blinded, independent, central imaging review Q 3 M, once every 3 months; Q 3 -4 W, once every 3 -4 weeks; SC, subcutaneous. Robert E. Coleman Recruitment 02 June 2010 – 24 August 2012

Key Eligibility Criteria • Histologically-confirmed, AJCC 7. 0 stage II or III breast cancer • No metastases on baseline CT scan of chest, abdomen and pelvis and radionuclide bone scan • High risk • Regional node positive disease • Tumor size > 5 cm (T 3) or locally advanced disease (T 4) • Documented ER, PR and HER-2 status • Scheduled to receive standard of care (neo)adjuvant systemic and loco-regional treatments according to institutional guidelines • ≤ 12 weeks between definitive surgery and randomization • No prior invasive breast cancer or other malignancy unless disease-free > 5 years • No current or prior use of IV bisphonates or denosumab or oral bisphonate within 1 year Robert E. Coleman

D-CARE Study Endpoints Primary Endpoint Exploratory Endpoints • Bone metastasis-free survival (BMFS) Time to event: • Time to first bone metastasis (excluding death) • Time to bone metastasis as site of first recurrence • Time to first on-study symptomatic bone metastasis • Time to disease recurrence • Time to distant recurrence Fracture related: • Time to first on-study fracture (prior to bone metastasis) • Time to first on-study SRE (following bone metastasis) • Time to first on-study SRE or hypercalcemia (following bone metastasis) Breast effects: • Breast density • Response to neoadjuvant therapy Pharmacokinetics, Pharmacodynamics Biomarker Development Secondary Endpoints • • Disease-free survival (DFS) in the full study population DFS in the postmenopausal subset Overall survival (OS) Distant recurrence-free survival (DRFS) Safety Endpoints • Safety and tolerability Patient-Reported Outcome Exploratory Endpoints • Analgesic use • Pain (Brief Pain Inventory Short Form) • Health Index Scores (Euro. Qo. L-5 Dimensions) Robert E. Coleman

D-CARE Statistical Design Primary Analysis: Amended due to low event rate Event-driven Time-driven Interim Analysis 1 Interim Analysis 2 Primary Analysis (PA) • 367 patients BMFS • 555 patients DFS • 551 patients BMFS • 833 patients DFS • 735 patients BMFS (Assumed HR=0. 8, power=85%) • 1110 patients DFS (Assumed HR=0. 83, power=80%) • 512 patients DFS in the postmenopausal subset (Assumed HR=0. 76, power=80%) Interim Analysis Primary Analysis (PA) Robert E. Coleman • 367 patients BMFS • 555 patients DFS (Data cut-off 12 JAN 15) After all enrolled patients had the opportunity to complete 5 years of treatment (Data cut-off 31 AUG 17) Actually observed # of events at PA • BMFS: 597 • DFS: 875 • DFS in postmenopausal subset: 417
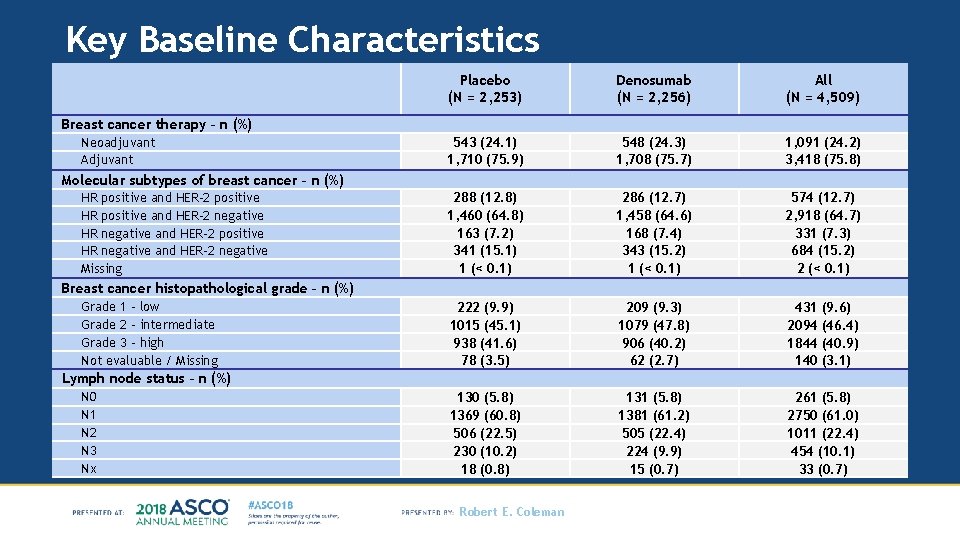
Key Baseline Characteristics Placebo (N = 2, 253) Denosumab (N = 2, 256) All (N = 4, 509) 543 (24. 1) 1, 710 (75. 9) 548 (24. 3) 1, 708 (75. 7) 1, 091 (24. 2) 3, 418 (75. 8) 288 (12. 8) 1, 460 (64. 8) 163 (7. 2) 341 (15. 1) 1 (< 0. 1) 286 (12. 7) 1, 458 (64. 6) 168 (7. 4) 343 (15. 2) 1 (< 0. 1) 574 (12. 7) 2, 918 (64. 7) 331 (7. 3) 684 (15. 2) 2 (< 0. 1) 222 (9. 9) 1015 (45. 1) 938 (41. 6) 78 (3. 5) 209 (9. 3) 1079 (47. 8) 906 (40. 2) 62 (2. 7) 431 (9. 6) 2094 (46. 4) 1844 (40. 9) 140 (3. 1) 130 (5. 8) 1369 (60. 8) 506 (22. 5) 230 (10. 2) 18 (0. 8) 131 (5. 8) 1381 (61. 2) 505 (22. 4) 224 (9. 9) 15 (0. 7) 261 (5. 8) 2750 (61. 0) 1011 (22. 4) 454 (10. 1) 33 (0. 7) Breast cancer therapy – n (%) Neoadjuvant Adjuvant Molecular subtypes of breast cancer – n (%) HR positive and HER-2 positive HR positive and HER-2 negative HR negative and HER-2 positive HR negative and HER-2 negative Missing Breast cancer histopathological grade – n (%) Grade 1 - low Grade 2 - intermediate Grade 3 - high Not evaluable / Missing Lymph node status – n (%) N 0 N 1 N 2 N 3 Nx Robert E. Coleman
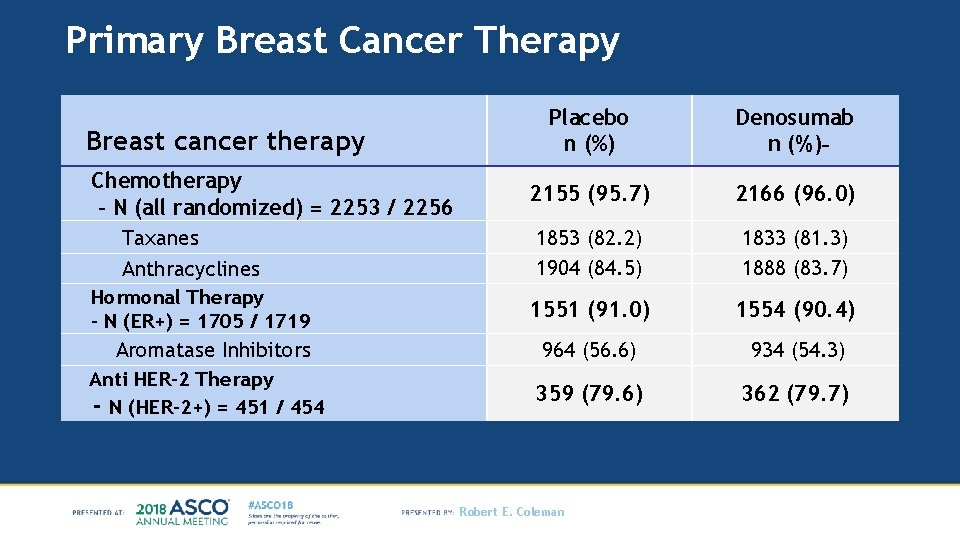
Primary Breast Cancer Therapy Breast cancer therapy Chemotherapy - N (all randomized) = 2253 / 2256 Taxanes Anthracyclines Hormonal Therapy - N (ER+) = 1705 / 1719 Aromatase Inhibitors Anti HER-2 Therapy - N (HER-2+) = 451 / 454 Placebo n (%) Denosumab n (%) 2155 (95. 7) 2166 (96. 0) 1853 (82. 2) 1904 (84. 5) 1833 (81. 3) 1888 (83. 7) 1551 (91. 0) 1554 (90. 4) 964 (56. 6) 934 (54. 3) 359 (79. 6) 362 (79. 7) Robert E. Coleman
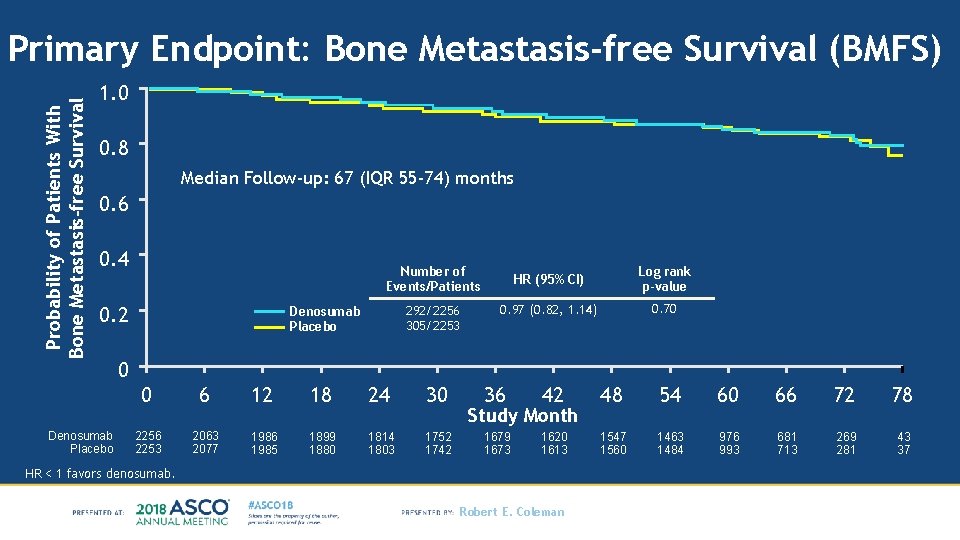
Probability of Patients With Bone Metastasis-free Survival Primary Endpoint: Bone Metastasis-free Survival (BMFS) 1. 0 0. 8 Median Follow-up: 67 (IQR 55 -74) months 0. 6 0. 4 Number of Events/Patients 0. 2 Denosumab Placebo 292/2256 305/2253 Denosumab Placebo HR (95% CI) Log rank p-value 0. 97 (0. 82, 1. 14) 0. 70 0 0 6 12 18 24 30 2256 2253 2063 2077 1986 1985 1899 1880 1814 1803 1752 1742 36 42 48 54 60 66 72 78 1679 1673 1620 1613 1547 1560 1463 1484 976 993 681 713 269 281 43 37 Study Month HR < 1 favors denosumab. Robert E. Coleman
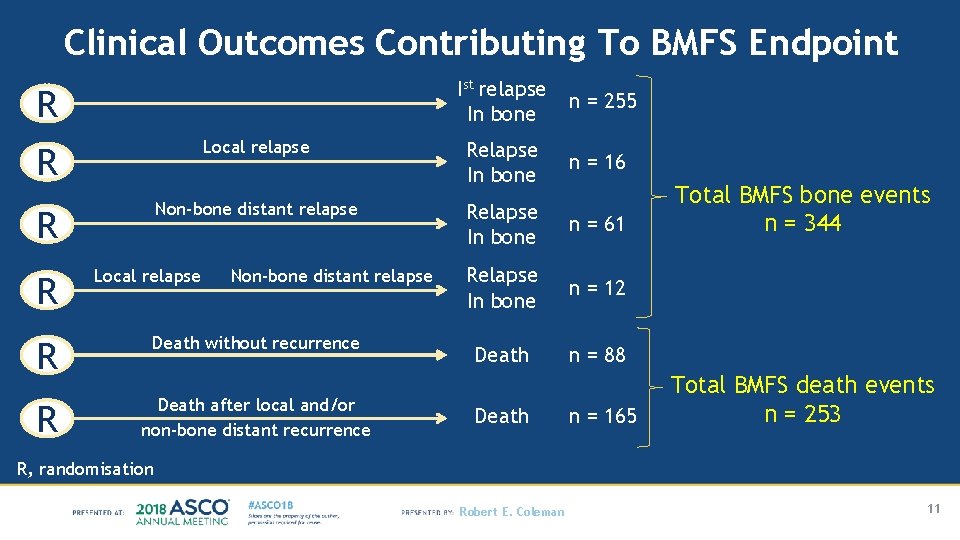
Clinical Outcomes Contributing To BMFS Endpoint R R Local relapse R Non-bone distant relapse R Local relapse Non-bone distant relapse R Death without recurrence R Death after local and/or non-bone distant recurrence Ist relapse In bone n = 255 Relapse In bone n = 16 Relapse In bone n = 61 Relapse In bone n = 12 Death n = 88 Death n = 165 Total BMFS bone events n = 344 Total BMFS death events n = 253 R, randomisation Robert E. Coleman 11
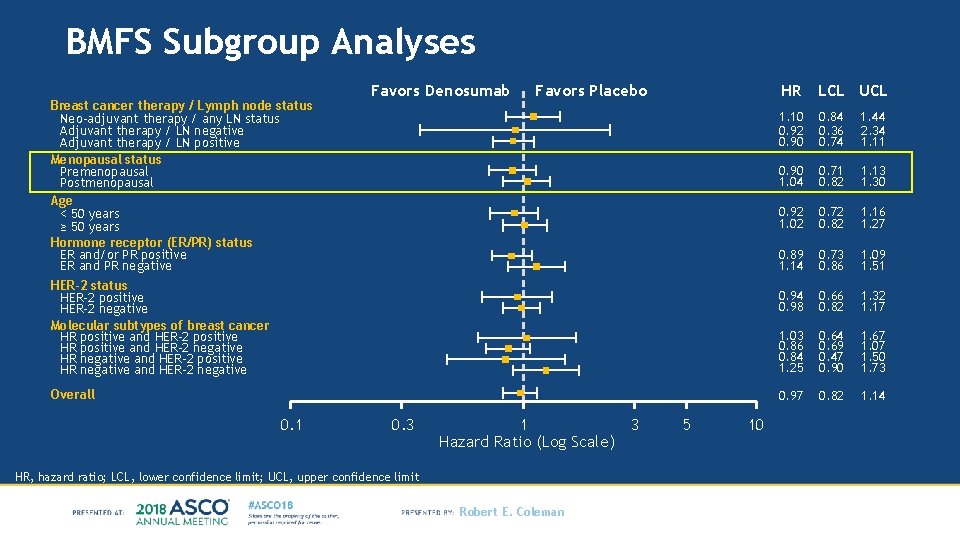
BMFS Subgroup Analyses Breast cancer therapy / Lymph node status Neo-adjuvant therapy / any LN status Adjuvant therapy / LN negative Adjuvant therapy / LN positive Menopausal status Premenopausal Postmenopausal Age < 50 years ≥ 50 years Hormone receptor (ER/PR) status ER and/or PR positive ER and PR negative Favors Denosumab Favors Placebo HER-2 status HER-2 positive HER-2 negative Molecular subtypes of breast cancer HR positive and HER-2 positive HR positive and HER-2 negative HR negative and HER-2 positive HR negative and HER-2 negative Overall 0. 1 0. 3 1 Hazard Ratio (Log Scale) HR, hazard ratio; LCL, lower confidence limit; UCL, upper confidence limit Robert E. Coleman 3 5 10 HR LCL UCL 1. 10 0. 92 0. 90 0. 84 0. 36 0. 74 1. 44 2. 34 1. 11 0. 90 1. 04 0. 71 0. 82 1. 13 1. 30 0. 92 1. 02 0. 72 0. 82 1. 16 1. 27 0. 89 1. 14 0. 73 0. 86 1. 09 1. 51 0. 94 0. 98 0. 66 0. 82 1. 32 1. 17 1. 03 0. 86 0. 84 1. 25 0. 64 0. 69 0. 47 0. 90 1. 67 1. 07 1. 50 1. 73 0. 97 0. 82 1. 14
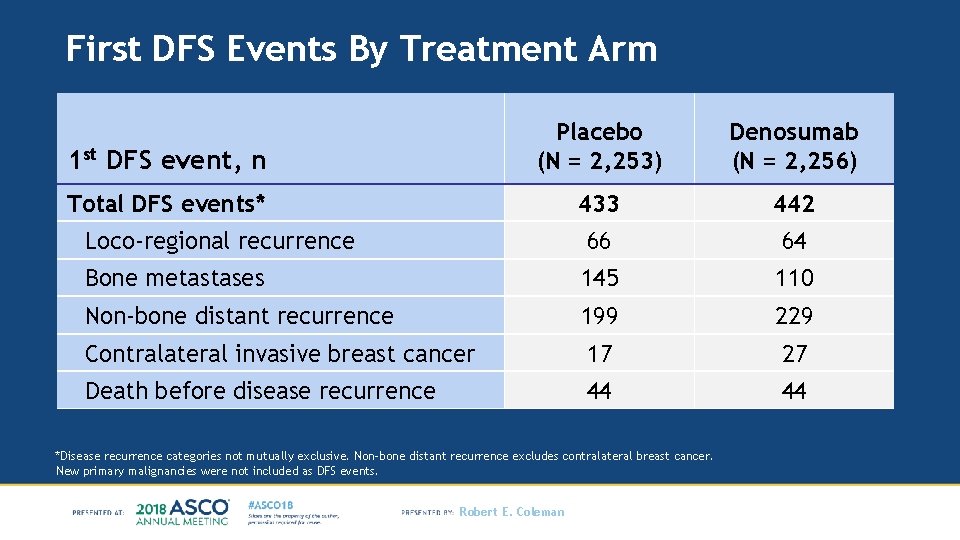
First DFS Events By Treatment Arm 1 st DFS event, n Placebo (N = 2, 253) Denosumab (N = 2, 256) Total DFS events* 433 442 Loco-regional recurrence 66 64 Bone metastases 145 110 Non-bone distant recurrence 199 229 Contralateral invasive breast cancer 17 27 Death before disease recurrence 44 44 *Disease recurrence categories not mutually exclusive. Non-bone distant recurrence excludes contralateral breast cancer. New primary malignancies were not included as DFS events. Robert E. Coleman
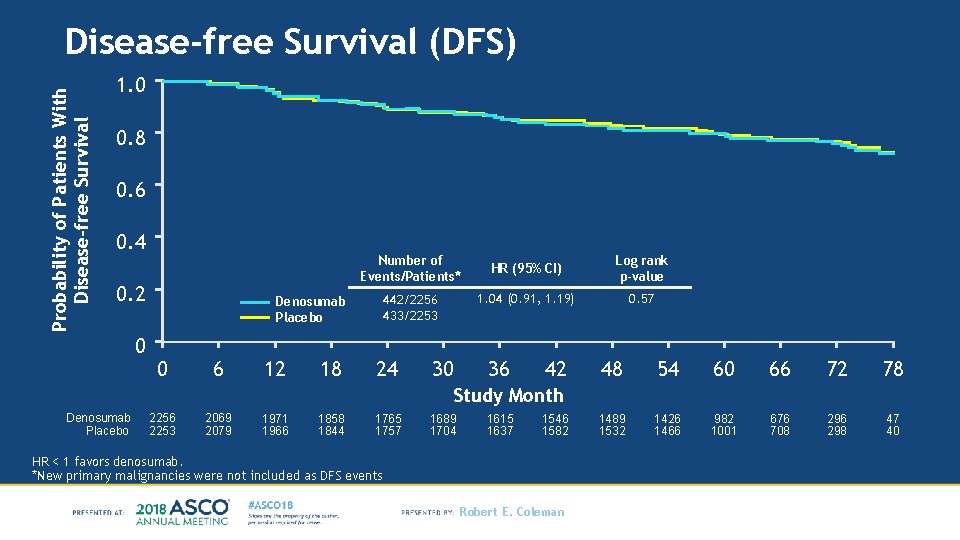
Probability of Patients With Disease-free Survival (DFS) 1. 0 0. 8 0. 6 0. 4 Number of Events/Patients* 0. 2 0 442/2256 433/2253 Denosumab Placebo 0 6 12 18 24 30 HR (95% CI) Log rank p-value 1. 04 (0. 91, 1. 19) 0. 57 36 42 48 54 60 66 72 78 1489 1532 1426 1466 982 1001 676 708 296 298 47 40 Study Month Denosumab Placebo 2256 2253 2069 2079 1971 1966 1858 1844 1765 1757 1689 1704 1615 1637 1546 1582 HR < 1 favors denosumab. *New primary malignancies were not included as DFS events Robert E. Coleman Full Analysis Set
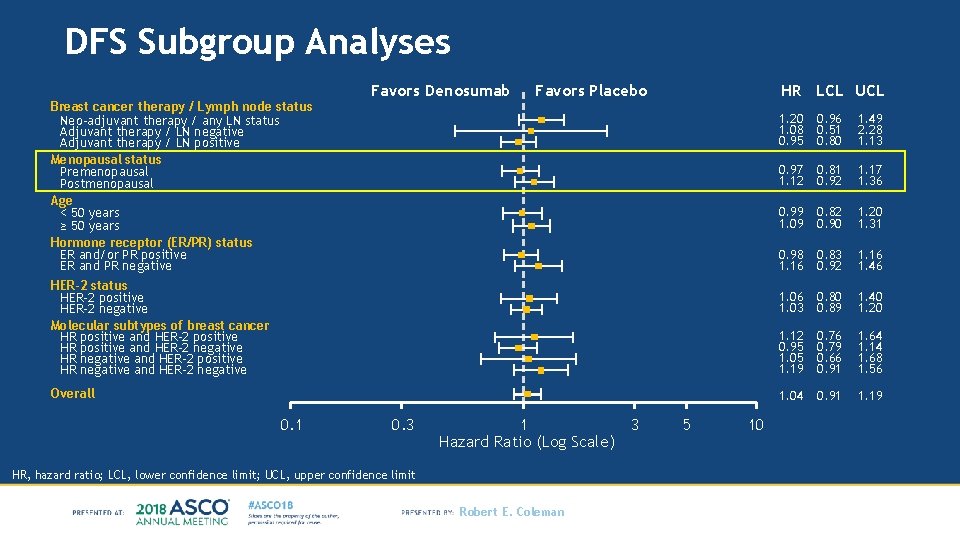
DFS Subgroup Analyses Breast cancer therapy / Lymph node status Neo-adjuvant therapy / any LN status Adjuvant therapy / LN negative Adjuvant therapy / LN positive Menopausal status Premenopausal Postmenopausal Age < 50 years ≥ 50 years Hormone receptor (ER/PR) status ER and/or PR positive ER and PR negative Favors Denosumab Favors Placebo HR LCL UCL HER-2 status HER-2 positive HER-2 negative Molecular subtypes of breast cancer HR positive and HER-2 positive HR positive and HER-2 negative HR negative and HER-2 positive HR negative and HER-2 negative Overall 0. 1 0. 3 1 Hazard Ratio (Log Scale) HR, hazard ratio; LCL, lower confidence limit; UCL, upper confidence limit Robert E. Coleman 3 5 10 1. 20 1. 08 0. 95 0. 96 0. 51 0. 80 1. 49 2. 28 1. 13 0. 97 1. 12 0. 81 0. 92 1. 17 1. 36 0. 99 1. 09 0. 82 0. 90 1. 20 1. 31 0. 98 1. 16 0. 83 0. 92 1. 16 1. 46 1. 03 0. 80 0. 89 1. 40 1. 20 1. 12 0. 95 1. 05 1. 19 0. 76 0. 79 0. 66 0. 91 1. 64 1. 14 1. 68 1. 56 1. 04 0. 91 1. 19
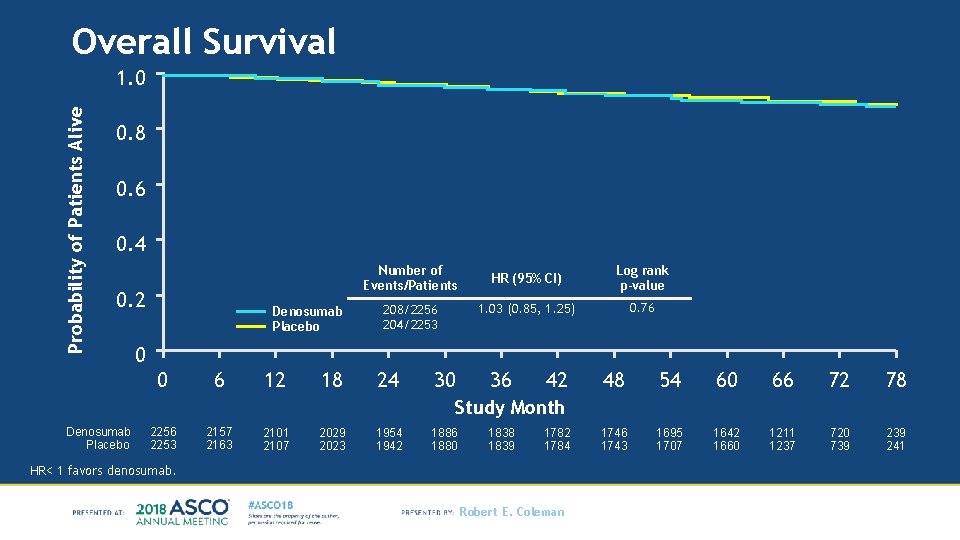
Overall Survival Probability of Patients Alive 1. 0 0. 8 0. 6 0. 4 Number of Events/Patients 0. 2 Denosumab Placebo 208/2256 204/2253 HR (95% CI) Log rank p-value 1. 03 (0. 85, 1. 25) 0. 76 0 0 6 12 18 24 30 36 42 48 54 60 66 72 78 1746 1743 1695 1707 1642 1660 1211 1237 720 739 241 Study Month Denosumab Placebo 2256 2253 2157 2163 2101 2107 2029 2023 1954 1942 1886 1880 1838 1839 1782 1784 HR< 1 favors denosumab. Robert E. Coleman
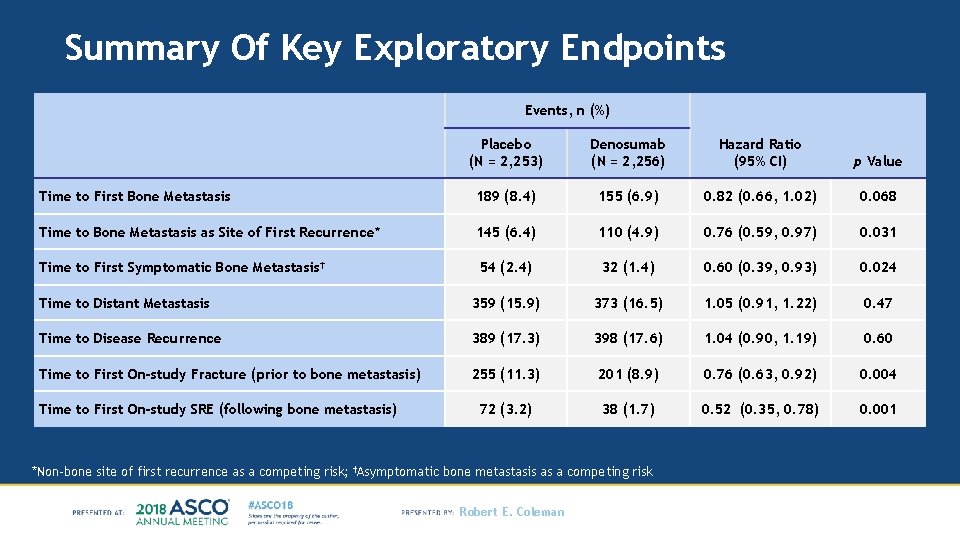
Summary Of Key Exploratory Endpoints Events, n (%) Placebo (N = 2, 253) Denosumab (N = 2, 256) Hazard Ratio (95% CI) p Value Time to First Bone Metastasis 189 (8. 4) 155 (6. 9) 0. 82 (0. 66, 1. 02) 0. 068 Time to Bone Metastasis as Site of First Recurrence* 145 (6. 4) 110 (4. 9) 0. 76 (0. 59, 0. 97) 0. 031 Time to First Symptomatic Bone Metastasis† 54 (2. 4) 32 (1. 4) 0. 60 (0. 39, 0. 93) 0. 024 Time to Distant Metastasis 359 (15. 9) 373 (16. 5) 1. 05 (0. 91, 1. 22) 0. 47 Time to Disease Recurrence 389 (17. 3) 398 (17. 6) 1. 04 (0. 90, 1. 19) 0. 60 Time to First On-study Fracture (prior to bone metastasis) 255 (11. 3) 201 (8. 9) 0. 76 (0. 63, 0. 92) 0. 004 72 (3. 2) 38 (1. 7) 0. 52 (0. 35, 0. 78) 0. 001 Time to First On-study SRE (following bone metastasis) *Non-bone site of first recurrence as a competing risk; †Asymptomatic bone metastasis as a competing risk Robert E. Coleman
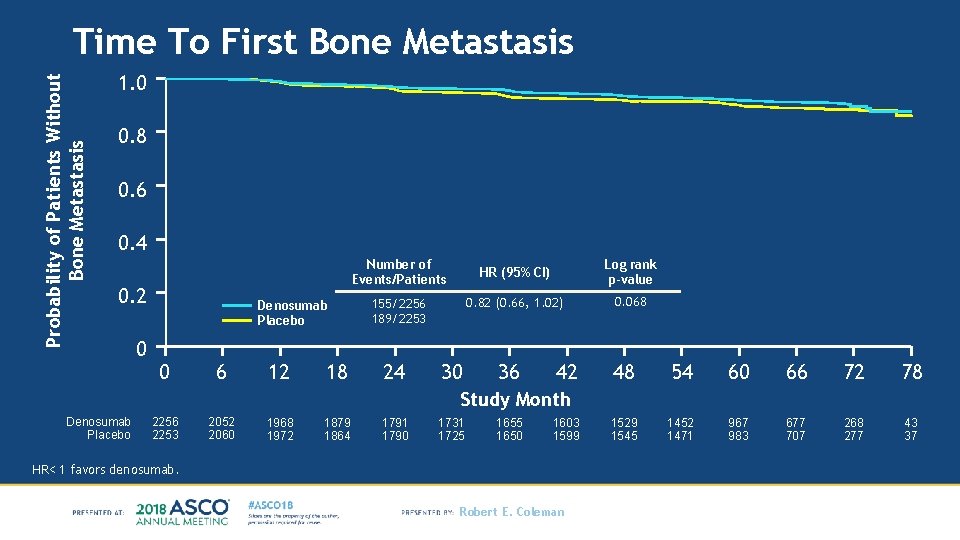
Probability of Patients Without Bone Metastasis Time To First Bone Metastasis 1. 0 0. 8 0. 6 0. 4 Number of Events/Patients 0. 2 0 Denosumab Placebo 0 6 12 18 155/2256 189/2253 24 30 HR (95% CI) Log rank p-value 0. 82 (0. 66, 1. 02) 0. 068 36 42 48 54 60 66 72 78 1529 1545 1452 1471 967 983 677 707 268 277 43 37 Study Month Denosumab Placebo 2256 2253 2052 2060 1968 1972 1879 1864 1791 1790 1731 1725 1650 1603 1599 HR< 1 favors denosumab. Robert E. Coleman
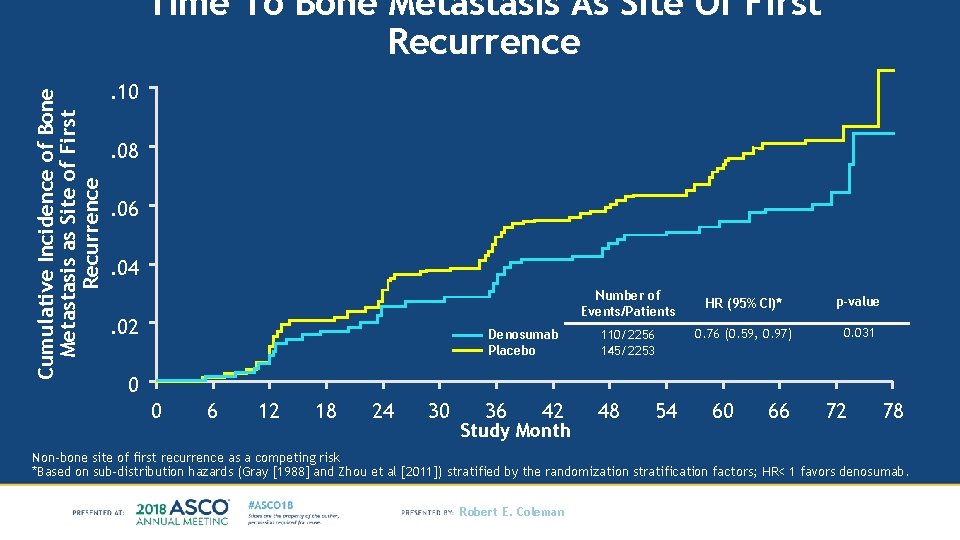
Cumulative Incidence of Bone Metastasis as Site of First Recurrence Time To Bone Metastasis As Site Of First Recurrence. 10. 08. 06. 04 Number of Events/Patients . 02 Denosumab Placebo 110/2256 145/2253 36 48 HR (95% CI)* p-value 0. 76 (0. 59, 0. 97) 0. 031 0 0 6 12 18 24 30 42 Study Month 54 60 66 72 78 Non-bone site of first recurrence as a competing risk *Based on sub-distribution hazards (Gray [1988] and Zhou et al [2011]) stratified by the randomization stratification factors; HR< 1 favors denosumab. Robert E. Coleman
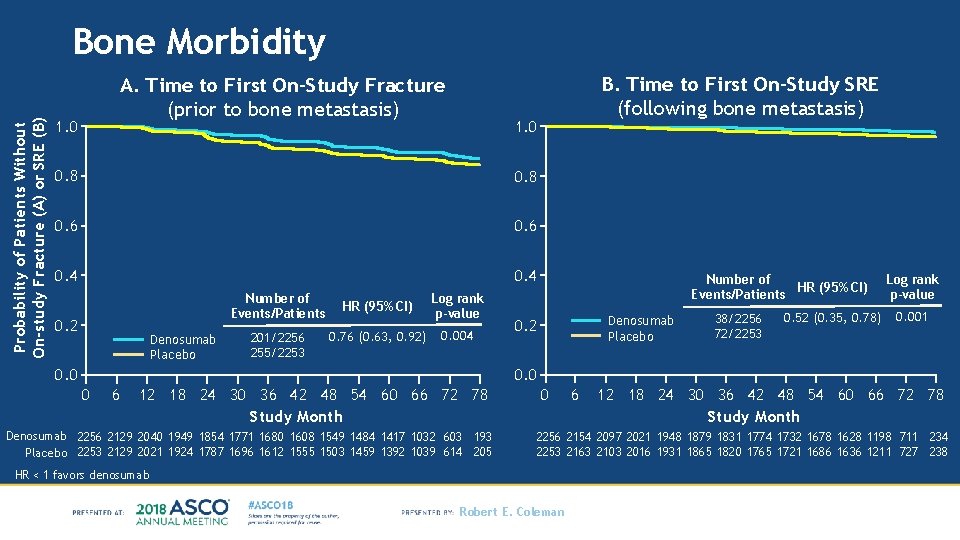
Probability of Patients Without On-study Fracture (A) or SRE (B) Bone Morbidity A. Time to First On-Study Fracture (prior to bone metastasis) 1. 0 0. 8 0. 6 0. 4 Number of Events/Patients 0. 2 Denosumab Placebo 201/2256 255/2253 HR (95% CI) 0. 76 (0. 63, 0. 92) B. Time to First On-Study SRE (following bone metastasis) Log rank p-value 0. 004 0. 0 Number of HR (95% CI) Events/Patients Denosumab Placebo 0. 2 38/2256 72/2253 0. 52 (0. 35, 0. 78) Log rank p-value 0. 001 0. 0 0 6 12 18 24 30 36 42 48 54 60 66 72 78 Study Month Denosumab 2256 2129 2040 1949 1854 1771 1680 1608 1549 1484 1417 1032 603 193 Placebo 2253 2129 2021 1924 1787 1696 1612 1555 1503 1459 1392 1039 614 205 0 6 12 18 24 30 36 42 48 54 60 66 72 78 Study Month 2256 2154 2097 2021 1948 1879 1831 1774 1732 1678 1628 1198 711 234 2253 2163 2103 2016 1931 1865 1820 1765 1721 1686 1636 1211 727 238 HR < 1 favors denosumab Robert E. Coleman

Treatment-emergent Adverse Events Placebo (N = 2, 218) Denosumab (N = 2, 241) All treatment-emergent adverse events, n (%)* Serious adverse events Leading to discontinuation of investigational product Leading to discontinuation of study Fatal adverse events CTCAE Grade 3, 4 , or 5 Treatment-related adverse events, † n (%) Serious adverse events Leading to discontinuation of investigational product Leading to discontinuation of study Fatal adverse events CTCAE Grade 3, 4, or 5 2, 166 (97. 7) 675 (30. 4) 59 (2. 7) 3 (0. 1) 39 (1. 8) 1, 162 (52. 4) 703 (31. 7) 30 (1. 4) 24 (1. 1) 0 (0. 0) 2 (< 0. 1) 78 (3. 5) 2, 195 (97. 9) 701 (31. 3) 79 (3. 5) 6 (0. 3) 25 (1. 1) 1, 228 (54. 8) 856 (38. 2) 70 (3. 1) 52 (2. 3) 3 (0. 1) 0 (0. 0) 106 (4. 7) N = number of patients who received ≥ 1 dose of investigational product. *Treatment-emergent adverse events starting after the first dose of IP and up to 30 days after the last dose of IP †Includes only treatment-emergent adverse events deemed related by the investigator CTCAE version v 3. 0. Robert E. Coleman
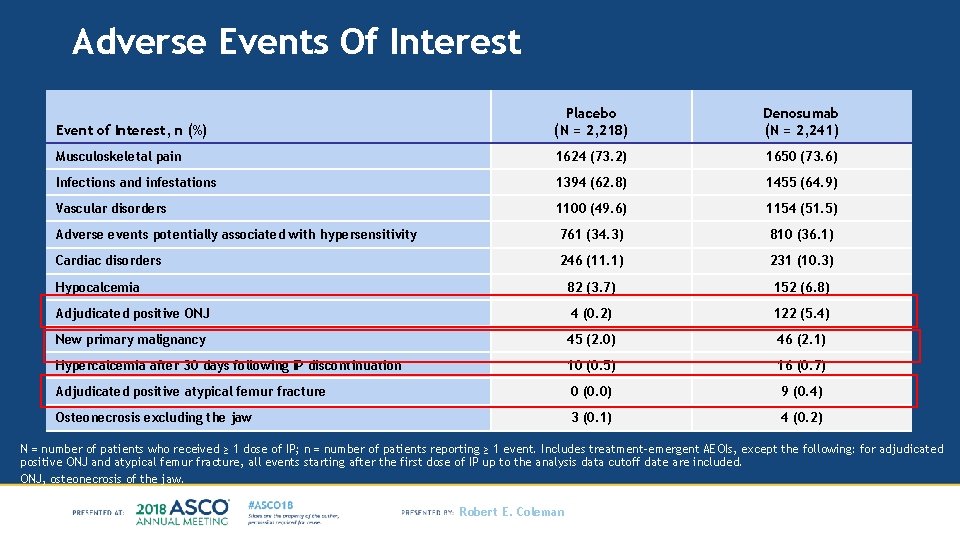
Adverse Events Of Interest Event of Interest, n (%) Placebo (N = 2, 218) Denosumab (N = 2, 241) Musculoskeletal pain 1624 (73. 2) 1650 (73. 6) Infections and infestations 1394 (62. 8) 1455 (64. 9) Vascular disorders 1100 (49. 6) 1154 (51. 5) Adverse events potentially associated with hypersensitivity 761 (34. 3) 810 (36. 1) Cardiac disorders 246 (11. 1) 231 (10. 3) Hypocalcemia 82 (3. 7) 152 (6. 8) Adjudicated positive ONJ 4 (0. 2) 122 (5. 4) New primary malignancy 45 (2. 0) 46 (2. 1) Hypercalcemia after 30 days following IP discontinuation 10 (0. 5) 16 (0. 7) Adjudicated positive atypical femur fracture 0 (0. 0) 9 (0. 4) Osteonecrosis excluding the jaw 3 (0. 1) 4 (0. 2) N = number of patients who received ≥ 1 dose of IP; n = number of patients reporting ≥ 1 event. Includes treatment-emergent AEOIs, except the following: for adjudicated positive ONJ and atypical femur fracture, all events starting after the first dose of IP up to the analysis data cutoff date are included. ONJ, osteonecrosis of the jaw. Robert E. Coleman
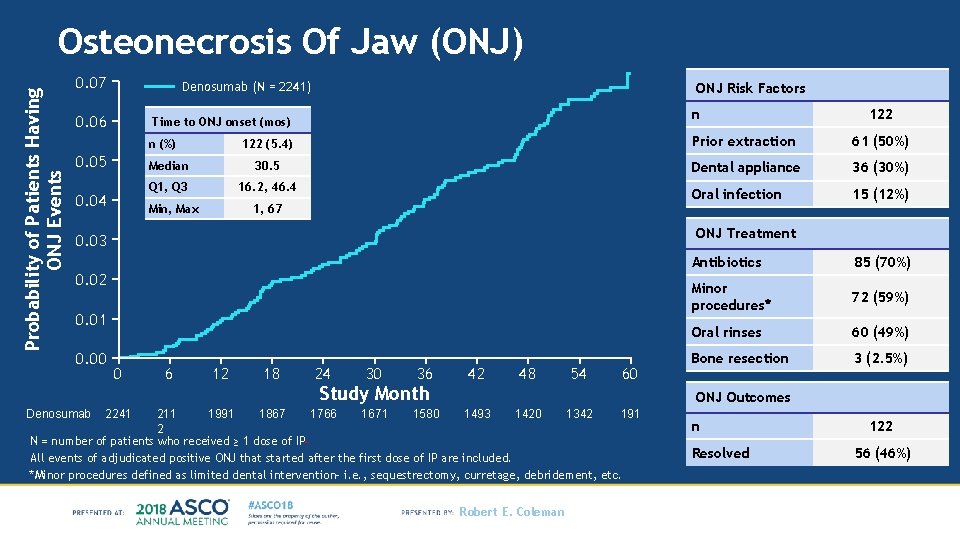
Probability of Patients Having ONJ Events Osteonecrosis Of Jaw (ONJ) 0. 07 Denosumab (N = 2241) 0. 06 ONJ Risk Factors n Time to ONJ onset (mos) 122 (5. 4) Prior extraction 61 (50%) Median 30. 5 Dental appliance 36 (30%) Q 1, Q 3 16. 2, 46. 4 Oral infection 15 (12%) n (%) 0. 05 0. 04 Min, Max 1, 67 ONJ Treatment 0. 03 0. 02 0. 01 0. 00 Denosumab 122 0 6 12 18 24 30 36 Study Month 42 48 54 60 Antibiotics 85 (70%) Minor procedures* 72 (59%) Oral rinses 60 (49%) Bone resection 3 (2. 5%) ONJ Outcomes 2241 211 1991 1867 1766 1671 1580 1493 1420 1342 191 2 N = number of patients who received ≥ 1 dose of IP All events of adjudicated positive ONJ that started after the first dose of IP are included. *Minor procedures defined as limited dental intervention– i. e. , sequestrectomy, curretage, debridement, etc. Robert E. Coleman n Resolved 122 56 (46%)

Conclusions • Adjuvant denosumab at this intensive dosing schedule: • • Did not improve BMFS in women with high-risk breast cancer No subgroup identified that obtained benefit in BMFS or DFS Reduced first recurrence in bone Reduced fracture incidence • Denosumab was generally well tolerated and events were consistent with the known safety profile: • Was associated with a 5% frequency of ONJ • BMFS is not an appropriate endpoint for trials of adjuvant therapy for early breast cancer Robert E. Coleman

Acknowledgements • We would like to thank: • the 4509 patients and their families who took part in the study, • the physicians and study teams at all 407 participating centres • the independent trial steering and data monitoring committees for study oversight. • We thank Richard Davis for medical writing support and Tian Dai for biostatistics support. • This study was funded by Amgen Inc. Robert E. Coleman
- Slides: 25