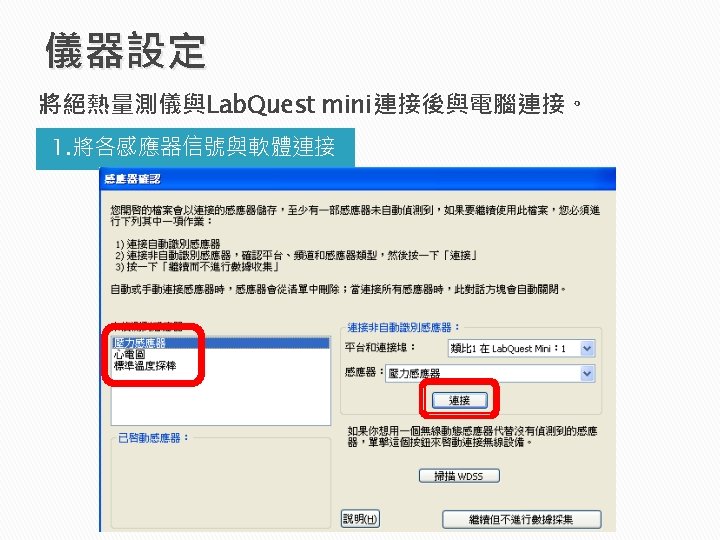
Adiabatic Processes for an Ideal Gas An adiabatic






















Adiabatic Processes for an Ideal Gas An adiabatic process: no energy is transferred by heat between a system and its surroundings. l All three variables in the ideal gas law (P, V, T) can change during an adiabatic process. l Assume an ideal gas is in an equilibrium state PV = n. RT is still valid. l The relation of P and V of an ideal gas at any time during an adiabatic process: l PV = constant. where = CP / CV is assumed to be constant during the process. Section 21. 3 25
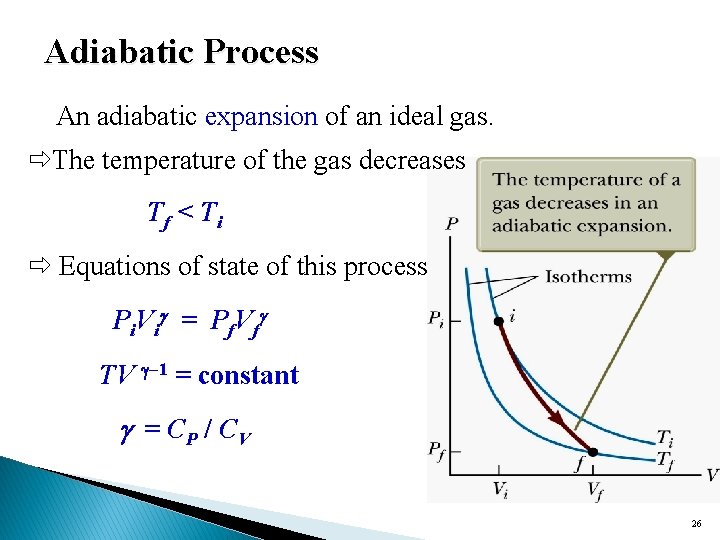
Adiabatic Process An adiabatic expansion of an ideal gas. The temperature of the gas decreases Tf < Ti Equations of state of this process P i V i = P f. V f TV – 1 = constant = CP / CV 26
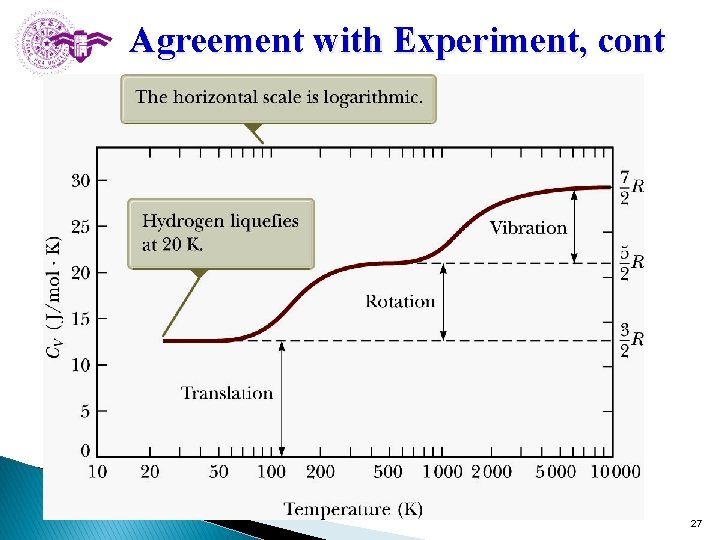
Agreement with Experiment, cont Section 21. 4 27

Example 21. 3 Q: Air at 20. 0 o. C in the cylinder of a diesel engine is compressed from an initial pressure of 1. 00 atm and volume of 800. 0 cm 3 to a volume of 60. 0 cm 3. Assume air behaves as an ideal gas with g = 1. 40 and the compression is adiabatic. Find the final pressure and temperature of the air. l. Solution: 28
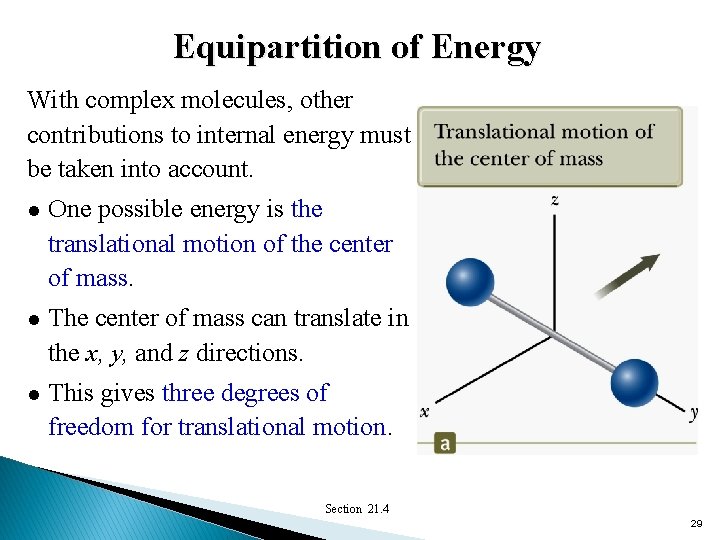
Equipartition of Energy With complex molecules, other contributions to internal energy must be taken into account. l One possible energy is the translational motion of the center of mass. l The center of mass can translate in the x, y, and z directions. l This gives three degrees of freedom for translational motion. Section 21. 4 29
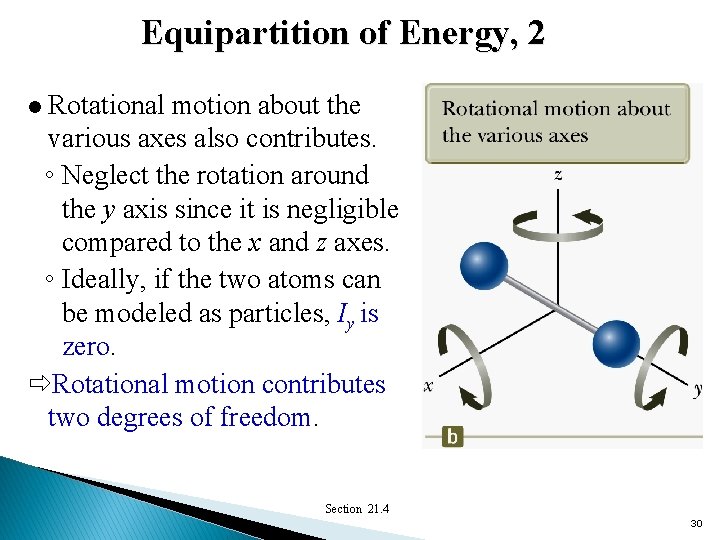
Equipartition of Energy, 2 Rotational motion about the various axes also contributes. ◦ Neglect the rotation around the y axis since it is negligible compared to the x and z axes. ◦ Ideally, if the two atoms can be modeled as particles, Iy is zero. Rotational motion contributes two degrees of freedom. l Section 21. 4 30

Equipartition of Energy, 3 l The molecule can also vibrate. There is kinetic energy and potential energy associated with the vibrations. The vibrational mode adds two more degrees of freedom. Section 21. 4 31

Equipartition of Energy, 4 l Taking into account the degrees of freedom from just the translation and rotation contributions. Eint = 5/2 n. RT CV = 5/2 R CP = 7/2 R Be in good agreement with data for diatomic molecules = 7/5 = 1. 40 l. However, the vibrational motion adds two more degrees of freedom. Eint = 7/2 n. RT & CV = 7/2 R = 9/7 This is inconsistent with experimental results. Section 21. 4 32

Molar Specific Heat: Agreement with Experiment Molar specific heat is a function of temperature CV(T). (1) At low temperatures: a diatomic gas acts like a monatomic gas. CV = 3/2 R l (2) At about room temperature: the value increases to CV = 5/2 R. - Be consistent with adding rotational energy but not vibrational energy. (3) At high temperatures: the value increases to CV = 7/2 R. l. This includes vibrational energy as well as rotational and translational. 33
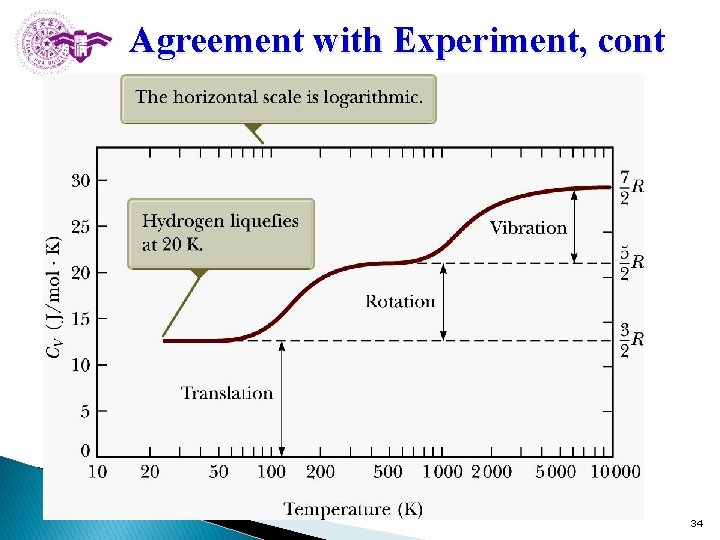
Agreement with Experiment, cont Section 21. 4 34

Complex Molecules For molecules with more than two atoms, the vibrations are more complex. 2. The number of degrees of freedom is larger. 3. The more degrees of freedom available to a molecule, the more “ways” there are to store energy. This results in a higher molar specific heat. 1. Section 21. 4 35



- Slides: 45