ADHERENCE GUIDELINES FOR HIV TB AND NCDs REVISED

ADHERENCE GUIDELINES FOR HIV, TB AND NCDs REVISED AGL – SOPs: Minimum Package of Interventions to Support Linkage to Care, Adherence and Retention in Care Updated March 2020 NATIONAL DEPARTMENT OF HEALTH DIRECTORATE: CARE AND TREATMENT 2020

Presentation Outline The orientation session aimed at: • Strengthening interventions to deliver differentiated care to patients along cascade: RSA Constitution and Human Rights COVID-19 Impact and Response DMo. C Strengthening Linkage, Adherence and Retention in Care Strategies for Facility Decongestion Clinical Governance and Treatment Regimens TPT and Decanting Strategies to Manage Advanced HIV disease

Human Rights: TB and HIV, NCD and Mental Health Patients in the Context of COVID-19

Introduction q As country responds to the Coronavirus (COVID-19) pandemic, necessary steps must be taken to ensure that its impact does not lead to secondary tragedy for vulnerable population groups: § These include but not limited to pregnant woman, young children, people with TB, HIV, NCDs and mental illness. q There is an urgent need to relook at strategies to mitigate the impact of disruptions and ensure accessibility of services for the vulnerable population groups

Constitution of South Africa, Act 108 of 1996 and the Mandate to Deliver Health Care Services Section 27 of the Constitution Enshrines q The constitution guarantees all South African Citizens the Right to health services q to ACCESS health care services. Human Rights q Human Rights Law guarantees everyone the right to the highest attainable standard of health and obligates governments to take q to REPRODUCTIVE HEALTH services. q to EMERGENCY MEDICAL TREATMENT steps to prevent threats to public health q All these rights must not be violated

Focus on COVID-19 q This year, 2020, focus has shifted to the q One of the least noticed effects of the COVID- newly emerged pandemic: COVID-19. 19 pandemic is not the disease itself but its q Within a short space of time, COVID-19 has supplanted everything. q Many health services that were previously key significant are no longer receiving much attention. q Significant resources are now diverted to COVID-19 response and management. devastating effect on other medical problems. q The introduction of stay home restrictions put some people at risk of relapse. q There are difficulties to access support groups, treatment and regular check-ups.

Concerns on Neglecting HIV and TB Services q Whilst COVID-19 takes the integral focus, conditions such as mental health, NCDs , HIV and TB are not prioritized in our health facilities. q It is evident that mental health, NCDs , HIV and TB remains part of our livelihoods. q TB is ranked as the top infectious disease that kills more people in the world than any other infectious pathogen. q Therefore, intensifying on-going interventions to address HIV and TB as global health problem remains of paramount important.

Impact of COVID 19 on HIV and TB Services q We cannot afford to lose sight of the business of mental health, NCDs, HIV and TB control because its likely to jeopardize important milestones and gains achieved over many years. q We cannot let the COVID-19 pandemic undo the hard-won gains in the global response to mental health, NCDs, HIV and TB disease. q COVID-19 is deeply affecting, mental health, NCDs TB services efforts in prevention, control , case detection and management. q It is evident that if we do not pay attention to these medical conditions, we will see increase in TB incidence and mortality, compromising the results achieved especially delaying the 2030: End TB strategy.

Impact of COVID 19 on HIV and TB Services q Mental health, NCDs TB and HIV health care workers are now assigned roles in COVID-19 management. q Experts previously involved in other medical conditions have been deployed to manage COVID-19 q Mental health, NCDs and TB diagnosis, adherence to treatment and outcomes are adversely affected. q People living with HIV – as well as young children are at greater risk of progressing from latent TB to active disease. q The importance of not neglecting TB preventive therapy even during COVID-19 is priceless

Access to Health Services q There are reported low stock of mental health, q We need to ensure that people who need mental NCDs treatment and ARVs or Disruptions in the health, NCDs and HIV treatment continue to medication supply. access it. q This could lead to more related deaths. q While there is no cure for HIV nor mental health and q During the past many years in managing mental health, NCDs, TB and HIV, new cases were NCDs, access to treatment can control morbidity significantly reduced, related morbidity and deaths and mortality. fell. q ARVs can control the spread of the virus and prevent sexual transmission to other people. q Million lives were saved globally using mental health, NCDs and ARV treatment. q However, the COVID-19 pandemic is causing disruptions and worsening the situation.

Access to Health Services q We cannot forget that TB remains a global health emergency and needs our attention. q The covid-19 pandemic has overtaken many health issues throughout the world. q The diversion of focus is likely to lead to a reduction in quality of mental health, NCDs TB and HIV care q Furthermore, mental health, NCDs TB and HIV patients often have underlying co-morbidities and lung damage that may make them prone to more severe COVID-19. q The symptoms of TB and COVID-19 can be similar. Access to HIV and TB services q TB symptoms such cough and fever are like the COVID-19. q This can cause diagnostic confusion. q Could worsen the stigmatization, given the fear of COVID-19. q We can maximize the amazingly noticed well coordinated rapid response to covid-19 and collaborate to improve the existing programmes such as mental health, NCDs HIV and TB.

Ethical and Professional Responsibility q We remind health care workers that Responsibilities of all HCW you have an ethical duty and a q We remind you of your responsibilities. professional responsibility to act in q Avoid exposing others to health and safety the best interest of your patients: § Both as a group and as individuals. § All patients, not only COVID-19 risks. q Self-monitor for signs of illness and selfisolate and report illness to managers, if it occurs.

Revised Adherence Guidelines Standard Operating Procedures (SOPs) to Strengthen Linkage, Adherence and Retention in Care
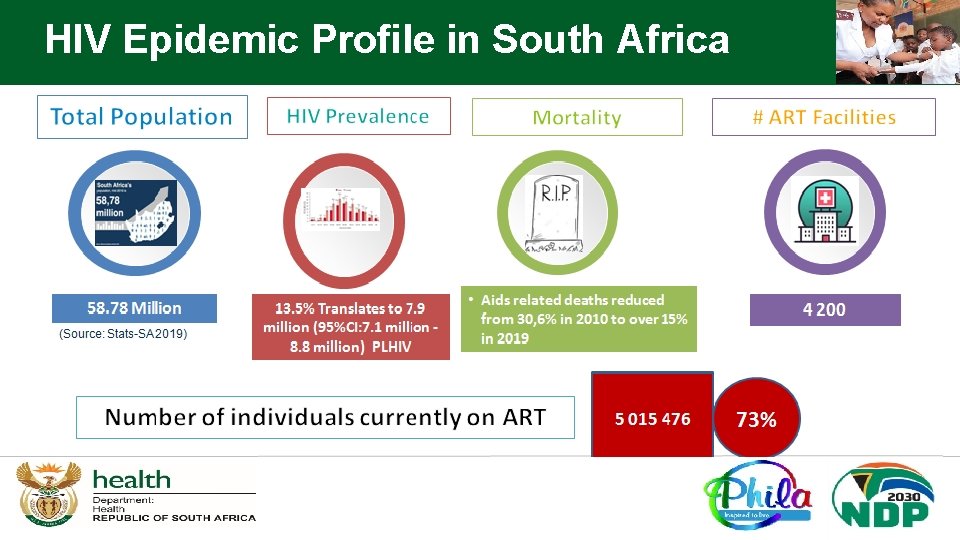
HIV Epidemic Profile in South Africa
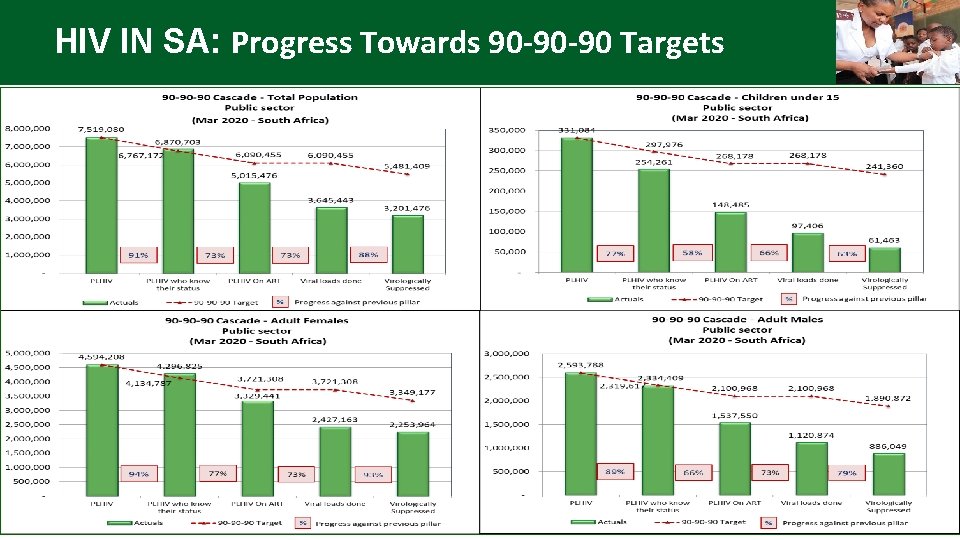
HIV IN SA: Progress Towards 90 -90 -90 Targets

Key Identified Challenges Impending Realisation of 90 -90 -90 Targets Case finding Poor linkage to care Gaps in treatment initiation and retention in care High loss to follow-up Gaps in leadership, management and programme integration
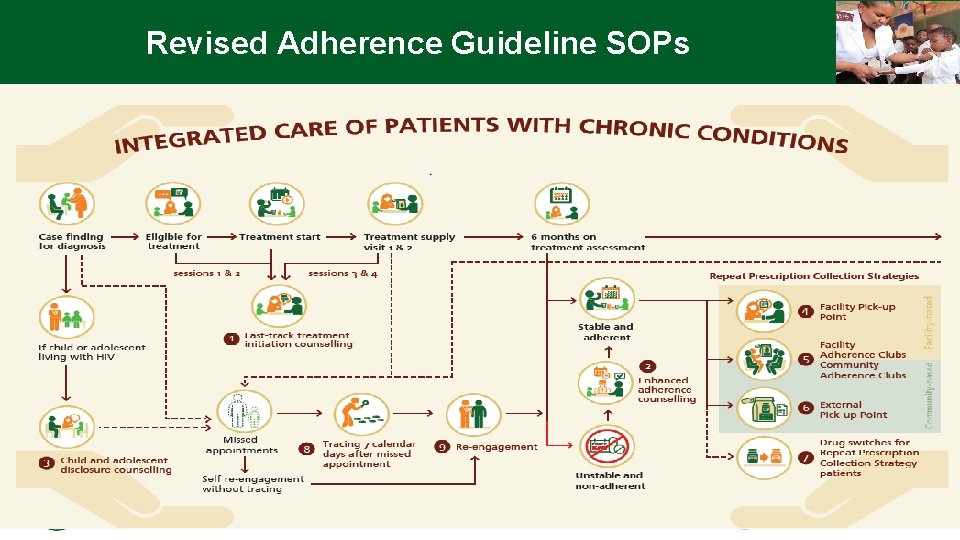
Revised Adherence Guideline SOPs

Overview of Key Revisions q Decanting of stable patients no longer at 12 months but at 6 months q Decanting Criteria: VL < 50 copies/ml and no longer 400 copies/ml; Hb. A 1 c<7% for Diabetes q RPCs modalities: § Facility Pick – up point (Fac – PUP) and no longer SFLA § CCMDD no longer a modality, but a medicine distribution system § Criteria for return to regular care added in all the RPCs q Criteria for children and adolescent included q New SOPs added: § Drug switching § Re-engagement q Terminology: referring to a patient as a defaulter has been discouraged; instead, this is a appointment patient who missed an
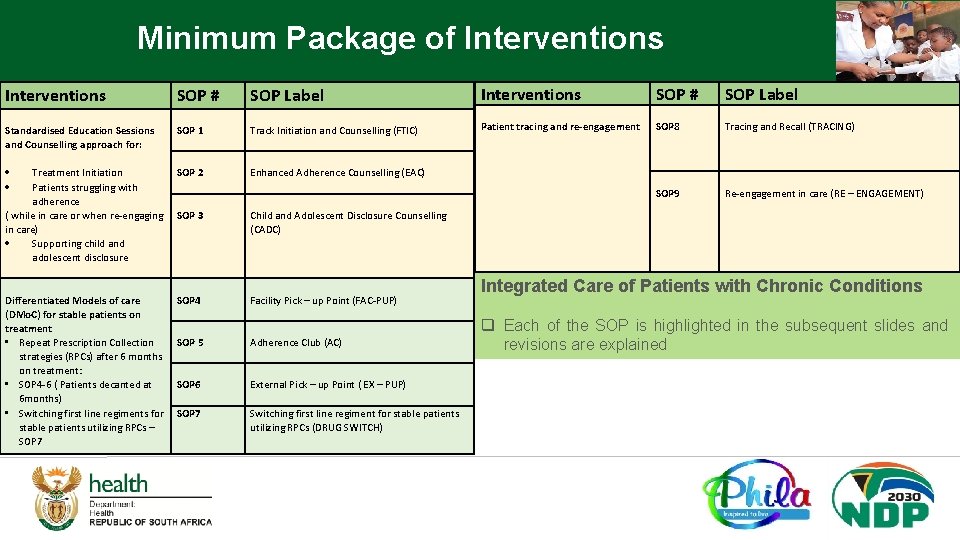
Minimum Package of Interventions SOP # SOP Label Standardised Education Sessions and Counselling approach for: SOP 1 Track Initiation and Counselling (FTIC) Patient tracing and re-engagement SOP 8 Tracing and Recall (TRACING) Treatment Initiation Patients struggling with adherence ( while in care or when re-engaging in care) Supporting child and adolescent disclosure SOP 2 Enhanced Adherence Counselling (EAC) SOP 9 Re-engagement in care (RE – ENGAGEMENT) SOP 3 Child and Adolescent Disclosure Counselling (CADC) Differentiated Models of care (DMo. C) for stable patients on treatment • Repeat Prescription Collection strategies (RPCs) after 6 months on treatment: • SOP 4 -6 ( Patients decanted at 6 months) • Switching first line regiments for stable patients utilizing RPCs – SOP 7 SOP 4 Facility Pick – up Point (FAC-PUP) SOP 5 Adherence Club (AC) SOP 6 External Pick – up Point ( EX – PUP) SOP 7 Switching first line regiment for stable patients utilizing RPCs (DRUG SWITCH) Integrated Care of Patients with Chronic Conditions q Each of the SOP is highlighted in the subsequent slides and revisions are explained

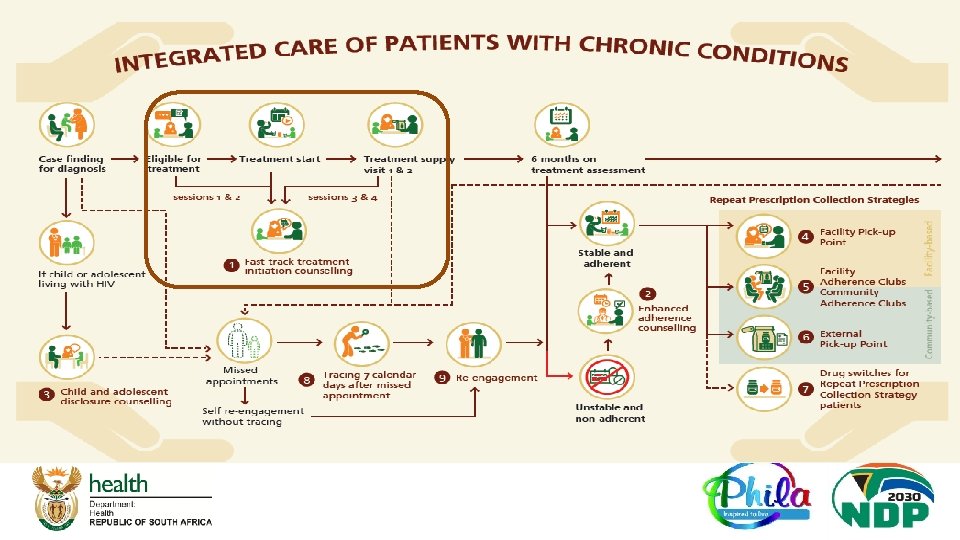
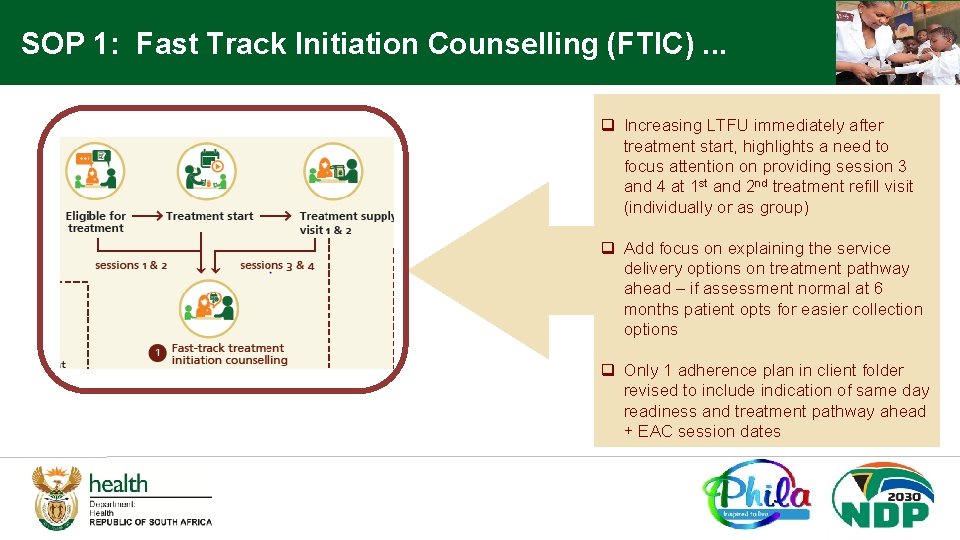
SOP 1: Fast Track Initiation Counselling (FTIC). . . q Increasing LTFU immediately after treatment start, highlights a need to focus attention on providing session 3 and 4 at 1 st and 2 nd treatment refill visit (individually or as group) q Add focus on explaining the service delivery options on treatment pathway ahead – if assessment normal at 6 months patient opts for easier collection options q Only 1 adherence plan in client folder revised to include indication of same day readiness and treatment pathway ahead + EAC session dates

Overview of Fast Track Initiation Counselling Sessions There are four sessions: Session 1: Day of linkage into care - provide education on the health condition and start an adherence plan (It stays in the patient folder follow-up and further completion) Session 2: Day of initiation - continue the adherence plan Session 1 and 2 must be combined if same day initiation Session 3 (New): First treatment refill (1 month from treatment start) - finalize the last steps of the adherence plan and outline the treatment pathway ahead if adherent Session 4 (New): Second treatment refill (2 months from treatment) – educate on assessment, restate goals and treatment pathway ahead if assessment result normal

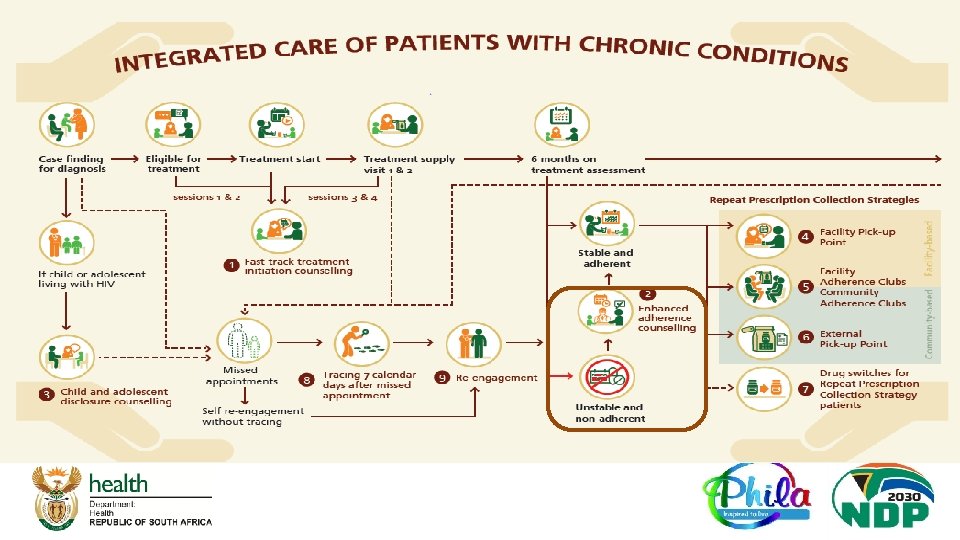

SOP 2: Enhanced Adherence Counselling… q Aligns with new ART clinical guidelines: Clients with VL>50 copies/ml can be referred for EAC by clinician after assessing possible adherence issue after A - E clinician’s assessment (Page 39 ART Consolidated Guidelines) See next slide q All Diabetes Mellitus (DM) patients with Hb. A 1 c > 7% referred for EAC q Also adds explanation to client on easier pick up options if suppressed <50 copies/ml, Hb. A 1 c ≤ 7%

A-E Assessment A thorough assessment is essential for any patient with a viral load measuring ≥ 50 c/ml

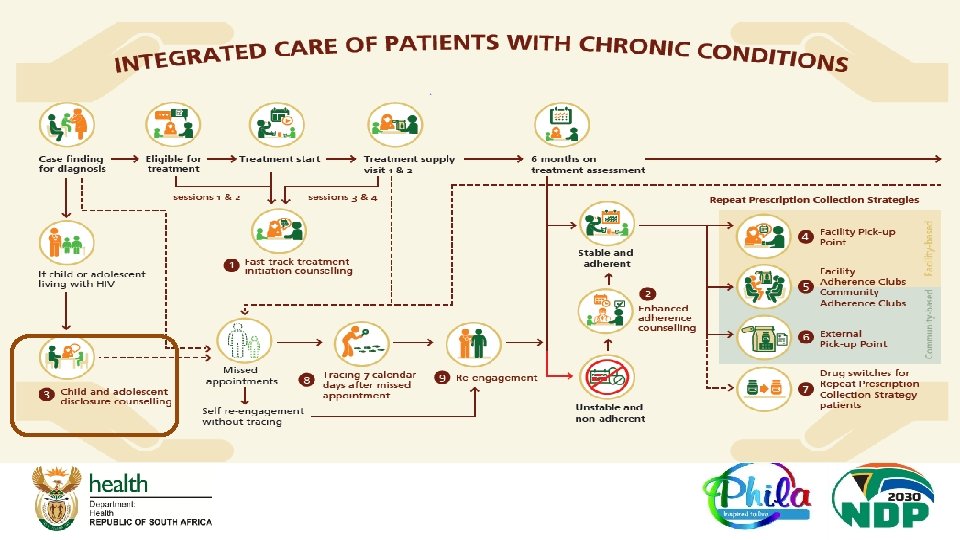

SOP 3: Child and Adolescent Disclosure Counselling q Child and adolescent disclosure support q The counsellors role is to facilitate disclosure not to do the actual disclosure

Repeat Prescription Collection Strategies (RPCs) Facility Pick-up Point: FAC-PUP (SOP 4) Adherence clubs: AC (SOP 5) External Pick-up Point: EX-PUP (SOP 6)
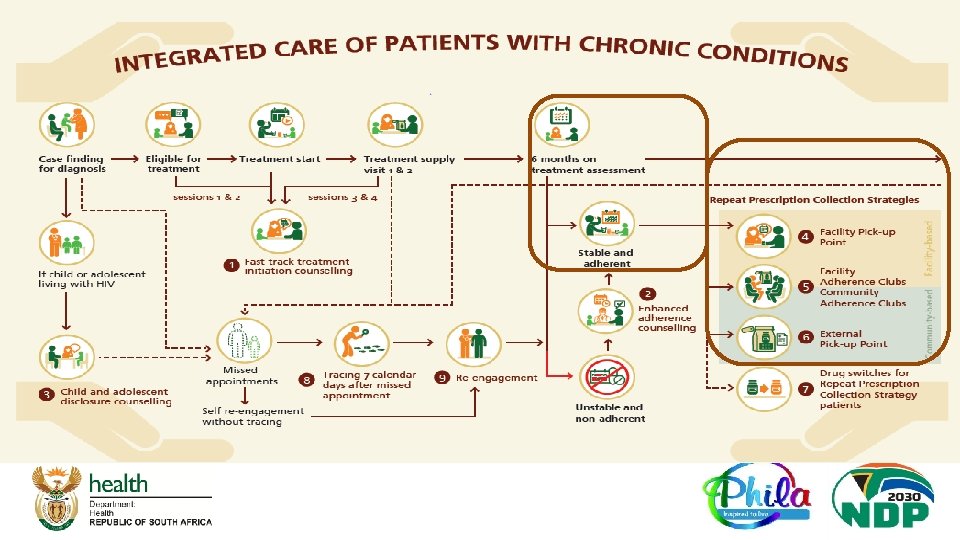
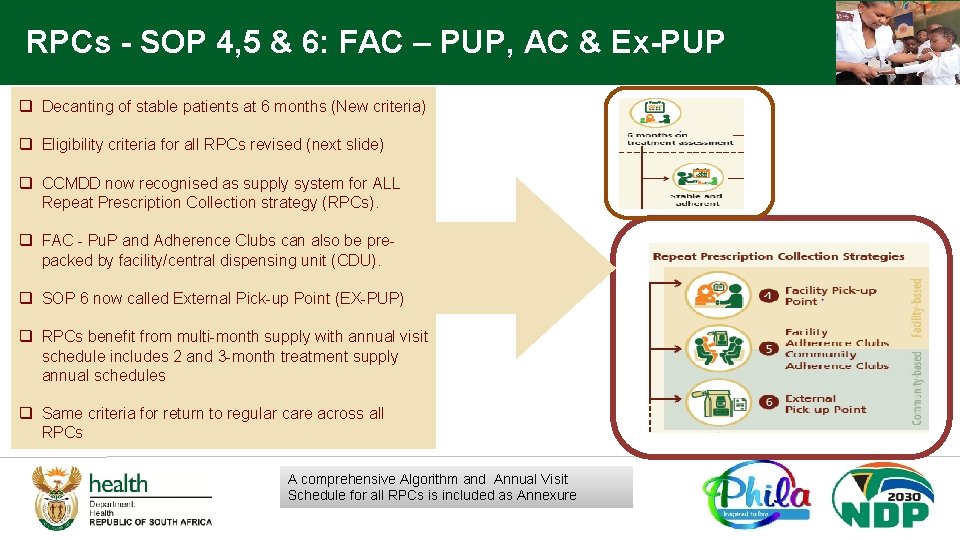
RPCs - SOP 4, 5 & 6: FAC – PUP, AC & Ex-PUP q Decanting of stable patients at 6 months (New criteria) q Eligibility criteria for all RPCs revised (next slide) q CCMDD now recognised as supply system for ALL Repeat Prescription Collection strategy (RPCs). q FAC - Pu. P and Adherence Clubs can also be prepacked by facility/central dispensing unit (CDU). q SOP 6 now called External Pick-up Point (EX-PUP) q RPCs benefit from multi-month supply with annual visit schedule includes 2 and 3 -month treatment supply annual schedules q Same criteria for return to regular care across all RPCs A comprehensive Algorithm and Annual Visit Schedule for all RPCs is included as Annexure

RPCs - SOP 4, 5 & 6: FAC – PUP, AC & Ex-PUP Eligibility Criteria ü No current TB/Medical condition requiring regular clinical consultations ü Clinician confirms eligibility ü Patient voluntarily opts for RPCs option For Adults q Above 18 years For Children and Adolescents q On treatment for at least 6 months q 5 -18 years q Most recent assessment results normal: q On ART for at least 6 months with no regimen or dosage change in § Most recent viral load ( VL) taken in past 6 months < 50 copies/ml for HIV § Most recent Hb. A 1 c taken in past 6 months ≤ 7% for Diabetes § 2 consecutive BP < 140/90 for Hypertension the last 3 months q Most recent VL taken in past 6 months < 50 copies/ml q Care givers counselled on disclosure process q Patient (>12 years/caregiver if patient<12 years) voluntarily opts for the RPCs option

RPCs - SOP 4: Facility Pick Up Point (FAC –Pu. P) - Description q A FAC-Pu. P can take various forms in a facility but q There is only one FAC –Pu. P in each facility, there all forms do not require a patient to attend should not be multiple FAC –Pu. Ps at a facility registry, vital signs or see a clinician. driven by treatment dispensing systems q There is no need to add RPCs patients on facility headcount/utilization rate. q There are no financial implications if these patients do not set their feet in the facility. q The treatment for the FAC-Pu. P can be predispensed by the facility pharmacy or by a Central Dispensing Unit (CDU) or Centralised Chronic Medicines Dispensing and Distribution (CCMDD). ü Implement 2 -month minimum dispensing for all ART and TB patients across all facilities. ü For patients on TLD, 3 month dispensing is encouraged. ü For patients on other chronic medication, 3 months dispensing is encouraged

RPCs - SOP 5: Adherence Clubs (AC) - Description q Adherence clubs can be provided for any group q Adherence clubs can serve as external pick up of people, including from the same geographical point for individual medicine pick up and ( not for area or a specific population of patients groups) q They can take place in or outside of a facility. q The treatment for an adherence club can be pre- q Adherence clubs provide a RPCs for stable dispensed by the facility pharmacy or by a Central patients who value continued psychosocial Dispensing Unit (CDU) or by the Centralised Chronic support and group engagement. Medicines Dispensing and Distribution (CCMDD). ü Implement 2 -month minimum dispensing for all ART and TB patients across all facilities. ü For patients on TLD, 3 month dispensing is encouraged. ü For patients on other chronic medication, 3 months dispensing is encouraged

RPCs - SOP 6: External Pick - Up Point (EX -PUP) - Description q EX-Pu. P can take various forms but all involve the q EX-Pu. P treatment is pre-dispensed to the EX-Pu. P patient collecting their treatment supply individually from service provider by the Centralised Chronic pick-up point outside of the facility or from an automated Medicines Dispensing and Distribution (CCMDD) system. q Examples of EX-Pu. Ps: § Treatment supply pick-up from a private pharmacy § Treatment supply pick-up from a designated community venue ( which can also be AC) § Treatment supply pick-up from a post box/ATM or similar automated system located inside or outside of a facility ü Implement 2 -month minimum dispensing for all ART and TB patients across all facilities. ü For patients on TLD, 3 month dispensing is encouraged. ü For patients on other chronic medication, 3 months dispensing is encouraged

Criteria for Return fo Regular Care for RPCs Criteria for Return to Regular Care q FAC – Pu. P, AC or EX-Pu. P did not return their RPCs collection point within 7 calendar days of their missed scheduled collection date q RPCs patient screens positive for TB q Other safety lab test results are abnormal: q Other indications assessed on individual clinical consultation q RPCs patient becomes pregnant and should be referred to integrated Maternal, Neonatal, Child and Women Health services (MNCWH) q All patients must be advised that they are being § For HIV: VL > 1000 copies/ml ( where VL is 50 -1000 returned to regular care to ensure more frequent copies/ml: the patient can remain in the RPCs but clinical care until they are stable again. Patients can must see a clinician 3 months after the date of return to RPCs after a single normal result and elevated VL for further VL assessment meeting other RPCs criteria in the future ( see Re- § For diabetes: Hb. A 1 c >7% § For Hypertension: BP > 140/90 engagement SOP 9)

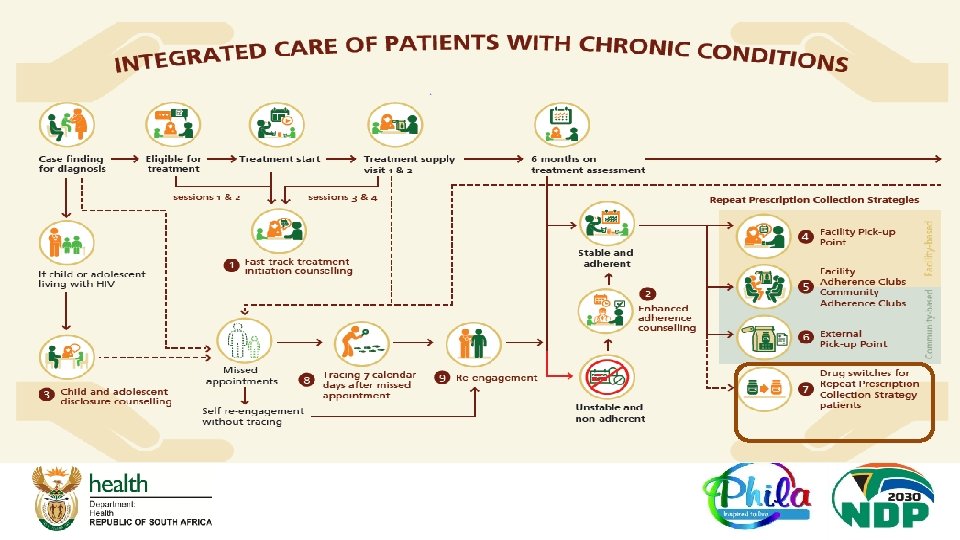

NEW SOP 7: RPCs Drug Switching SOP q NEW SOP – supporting access for stable clients in RPCs to new regimens while remaining in their RPCs q SOP refrains from referring to specific regimen so that it can be used for future regimens beyond DTG. q Overview of the Drug Switching procedure ( See next slide): § Review the patient’s recent viral load result (not older than 6 months) § Assess the stability of the patient: o Either VL<50 copies/ml o OR second VL assessment between 501000 copies/ml § Remember the A - E Assessment
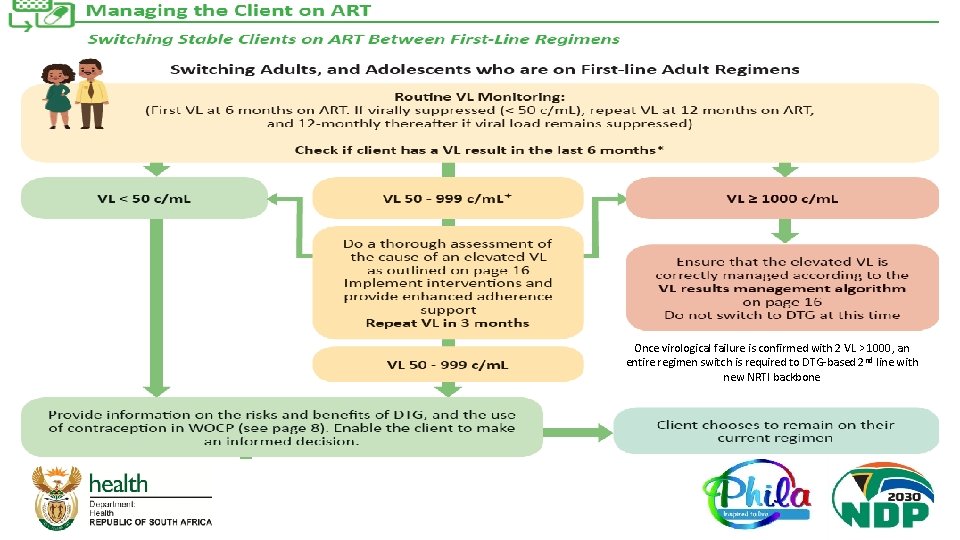
Once virological failure is confirmed with 2 VL > 1000, an entire regimen switch is required to DTG-based 2 nd line with new NRTI backbone

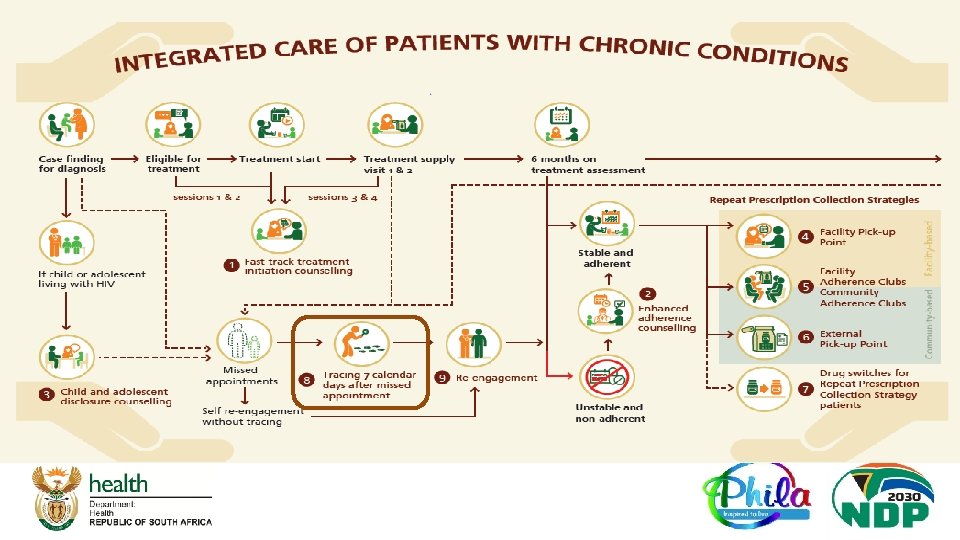

SOP 8: Tracing and Recall q Stops using term ‘defaulter’ , instead refers to a client who missed an appointment

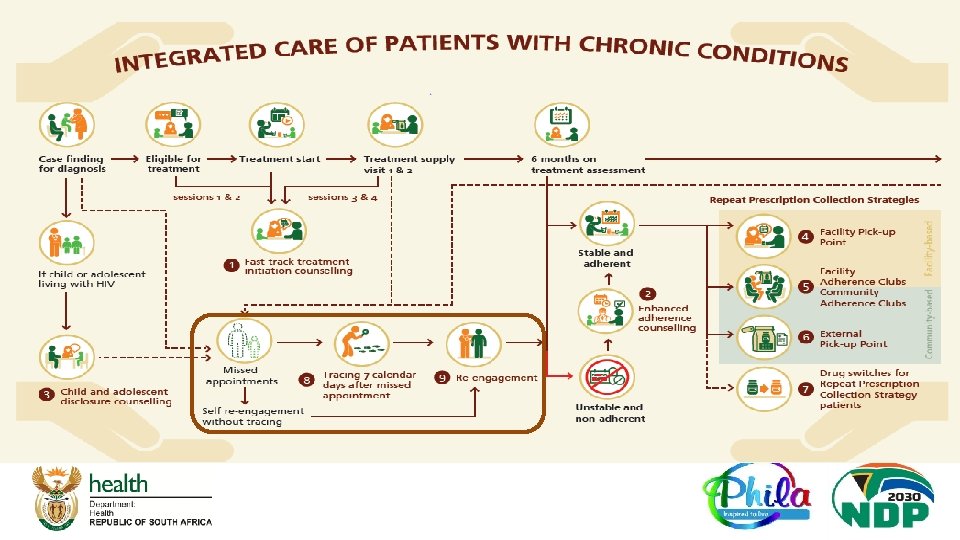
NEW SOP 9: Re - engagement SOP q NEW SOP – supports re-engagement after tracing or self re-engagement. q Acknowledging need to differentiate adherence support interventions for clients re-engaging. q Involves assessing treatment interruption and adherence challenges, including reviewing documented suppressed viral load. q Patient can be referred for EAC or preferred RPCs q Introduces algorithm to assess appropriate adherence support intervention to offer and aligns with procedure in new ART clinical guideline
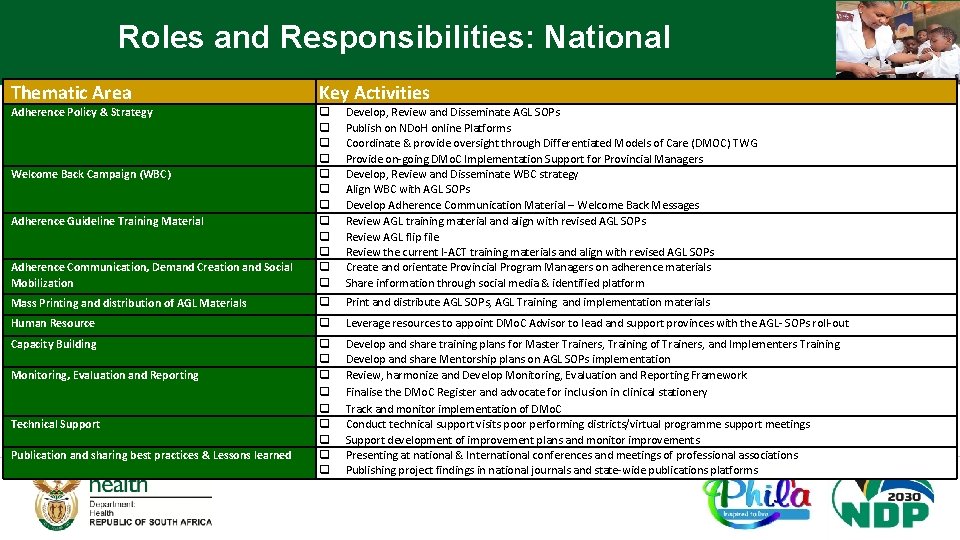
Roles and Responsibilities: National Thematic Area Key Activities Adherence Policy & Strategy Adherence Communication, Demand Creation and Social Mobilization q q q Develop, Review and Disseminate AGL SOPs Publish on NDo. H online Platforms Coordinate & provide oversight through Differentiated Models of Care (DMOC) TWG Provide on-going DMo. C Implementation Support for Provincial Managers Develop, Review and Disseminate WBC strategy Align WBC with AGL SOPs Develop Adherence Communication Material – Welcome Back Messages Review AGL training material and align with revised AGL SOPs Review AGL flip file Review the current I-ACT training materials and align with revised AGL SOPs Create and orientate Provincial Program Managers on adherence materials Share information through social media & identified platform Mass Printing and distribution of AGL Materials q Print and distribute AGL SOPs, AGL Training and implementation materials Human Resource q Leverage resources to appoint DMo. C Advisor to lead and support provinces with the AGL- SOPs roll-out Capacity Building q q q q q Develop and share training plans for Master Trainers, Training of Trainers, and Implementers Training Develop and share Mentorship plans on AGL SOPs implementation Review, harmonize and Develop Monitoring, Evaluation and Reporting Framework Finalise the DMo. C Register and advocate for inclusion in clinical stationery Track and monitor implementation of DMo. C Conduct technical support visits poor performing districts/virtual programme support meetings Support development of improvement plans and monitor improvements Presenting at national & International conferences and meetings of professional associations Publishing project findings in national journals and state-wide publications platforms Welcome Back Campaign (WBC) Adherence Guideline Training Material Monitoring, Evaluation and Reporting Technical Support Publication and sharing best practices & Lessons learned

Roles and Responsibilities: Provinces Thematic Area Key Activities Adherence Policy & Strategy q q q q Welcome Back Campaign (WBC) Adherence Guideline Training Material Adherence Communication, Demand Creation and Social Mobilization Mass Printing and distribution of AGL Materials Human Resource Capacity Building Monitoring, Evaluation and Reporting Technical Support Publication and sharing best practices & Lessons learned q q q q q Publish AGL SOPs on PDo. H online Platforms Coordination & provide oversight through Differentiated Models of Care (DMOC) Provincial TWGs Provide on-going DMo. C implementation Support for District Program Managers Align the provincial Welcome Back Campaign Strategy with the National Strategy Develop operational plans for WBC roll-out Adopt NDo. H adherence training materials Support implementation of AGL flip file and I-ACT in the districts Orientate District Program Managers on adherence materials Share DMo. C information through Provincial identified platforms (e. g. Radio Stations and social media) Participate in provincial IMBIZO and ensure linkage, adherence and retention in care is part of the agenda Coordinate the distribution of printed AGL SOPs, AGL Training and implementation materials Monitor and support the optimal implementation of the AGL materials in the districts Designate HAST Managers in Provinces to lead the DMo. C roll-out and support Districts & Sub-Districts HAST Managers Operationalize training plans for Master Trainers, Training of Trainers, and Implementers Training Where possible, integrate AGL training with other trainings e. g. HTS for non-clinicians Adopt and own the monitoring, Evaluation and Reporting Framework Capacitate M&E officials on the DMo. C M&E framework Encourage reporting at all levels using the provided programme reporting tools Conduct programme performance reviews Conduct technical support visits poor performing districts/ facilities Conduct monthly Virtual programme support meetings Support development of improvement plans and monitor improvements Conduct provincial review and learning platforms workshops/ seminars (Virtual or face to face)
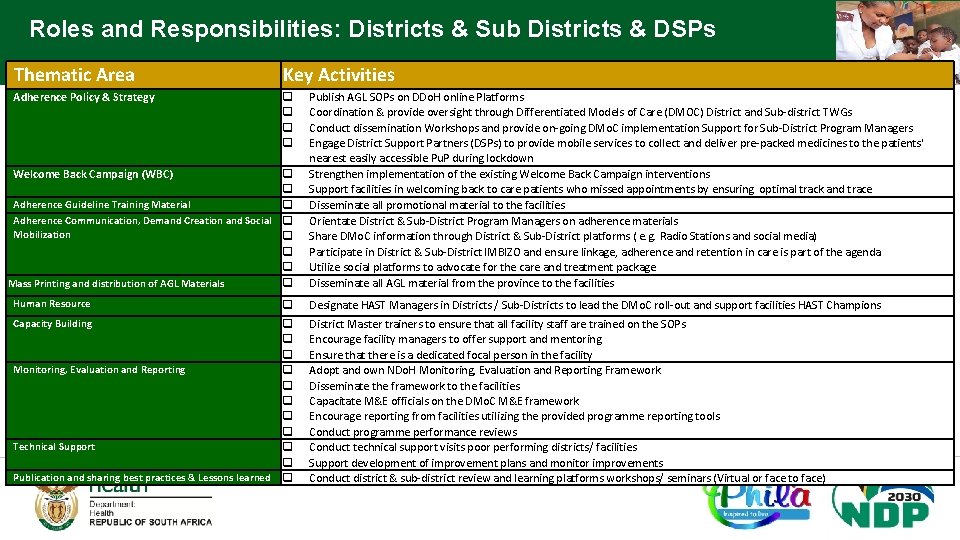
Roles and Responsibilities: Districts & Sub Districts & DSPs Thematic Area Key Activities Adherence Policy & Strategy q q Welcome Back Campaign (WBC) q q Adherence Guideline Training Material q Adherence Communication, Demand Creation and Social q Mobilization q q q Mass Printing and distribution of AGL Materials q Human Resource Capacity Building q q Monitoring, Evaluation and Reporting q q q Technical Support q q Publication and sharing best practices & Lessons learned q Publish AGL SOPs on DDo. H online Platforms Coordination & provide oversight through Differentiated Models of Care (DMOC) District and Sub-district TWGs Conduct dissemination Workshops and provide on-going DMo. C implementation Support for Sub-District Program Managers Engage District Support Partners (DSPs) to provide mobile services to collect and deliver pre-packed medicines to the patients' nearest easily accessible Pu. P during lockdown Strengthen implementation of the existing Welcome Back Campaign interventions Support facilities in welcoming back to care patients who missed appointments by ensuring optimal track and trace Disseminate all promotional material to the facilities Orientate District & Sub-District Program Managers on adherence materials Share DMo. C information through District & Sub-District platforms ( e. g. Radio Stations and social media) Participate in District & Sub-District IMBIZO and ensure linkage, adherence and retention in care is part of the agenda Utilize social platforms to advocate for the care and treatment package Disseminate all AGL material from the province to the facilities Designate HAST Managers in Districts / Sub-Districts to lead the DMo. C roll-out and support facilities HAST Champions District Master trainers to ensure that all facility staff are trained on the SOPs Encourage facility managers to offer support and mentoring Ensure that there is a dedicated focal person in the facility Adopt and own NDo. H Monitoring, Evaluation and Reporting Framework Disseminate the framework to the facilities Capacitate M&E officials on the DMo. C M&E framework Encourage reporting from facilities utilizing the provided programme reporting tools Conduct programme performance reviews Conduct technical support visits poor performing districts/ facilities Support development of improvement plans and monitor improvements Conduct district & sub-district review and learning platforms workshops/ seminars (Virtual or face to face)
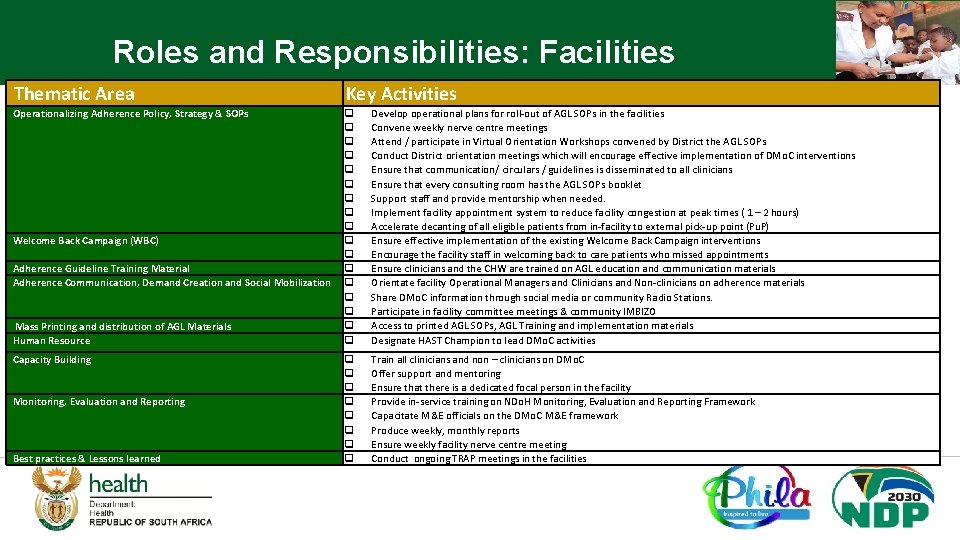
Roles and Responsibilities: Facilities Thematic Area Key Activities Operationalizing Adherence Policy, Strategy & SOPs q q q q q Develop operational plans for roll-out of AGL SOPs in the facilities Convene weekly nerve centre meetings Attend / participate in Virtual Orientation Workshops convened by District the AGL SOPs Conduct District orientation meetings which will encourage effective implementation of DMo. C interventions Ensure that communication/ circulars / guidelines is disseminated to all clinicians Ensure that every consulting room has the AGL SOPs booklet Support staff and provide mentorship when needed. Implement facility appointment system to reduce facility congestion at peak times ( 1 – 2 hours) Accelerate decanting of all eligible patients from in-facility to external pick-up point (Pu. P) Ensure effective implementation of the existing Welcome Back Campaign interventions Encourage the facility staff in welcoming back to care patients who missed appointments Ensure clinicians and the CHW are trained on AGL education and communication materials Orientate facility Operational Managers and Clinicians and Non-clinicians on adherence materials Share DMo. C information through social media or community Radio Stations. Participate in facility committee meetings & community IMBIZO Access to printed AGL SOPs, AGL Training and implementation materials Designate HAST Champion to lead DMo. C activities q q q q Train all clinicians and non – clinicians on DMo. C Offer support and mentoring Ensure that there is a dedicated focal person in the facility Provide in-service training on NDo. H Monitoring, Evaluation and Reporting Framework Capacitate M&E officials on the DMo. C M&E framework Produce weekly, monthly reports Ensure weekly facility nerve centre meeting Conduct ongoing TRAP meetings in the facilities Welcome Back Campaign (WBC) Adherence Guideline Training Material Adherence Communication, Demand Creation and Social Mobilization Mass Printing and distribution of AGL Materials Human Resource Capacity Building Monitoring, Evaluation and Reporting Best practices & Lessons learned

CLINICAL GOVERNANCE AND TREATMENT REGIMENS
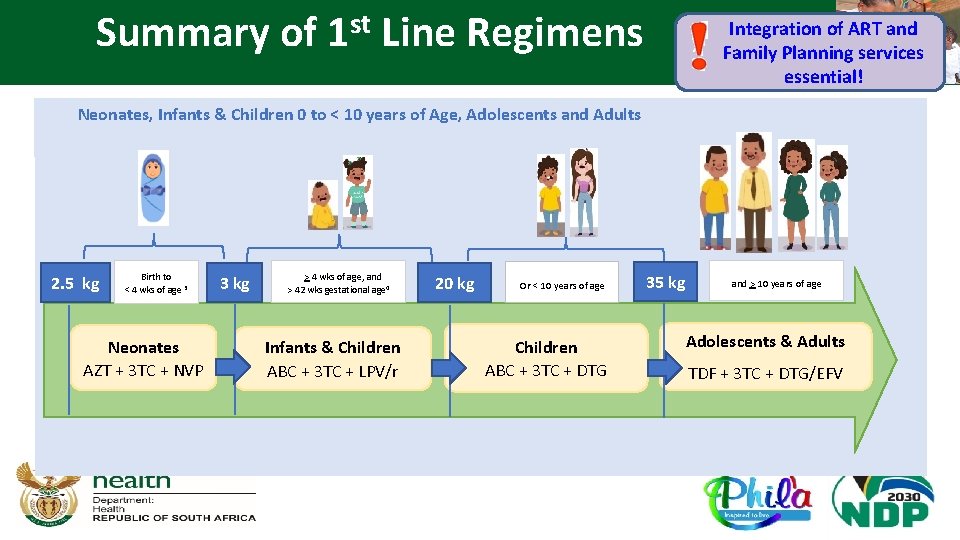
Summary of 1 st Line Regimens Integration of ART and Family Planning services essential! Neonates, Infants & Children 0 to < 10 years of Age, Adolescents and Adults 2. 5 kg Birth to < 4 wks of age 3 Neonates AZT + 3 TC + NVP 3 kg > 4 wks of age, and > 42 wks gestational age 4 Infants & Children ABC + 3 TC + LPV/r 20 kg Or < 10 years of age Children ABC + 3 TC + DTG 35 kg and > 10 years of age Adolescents & Adults TDF + 3 TC + DTG/EFV
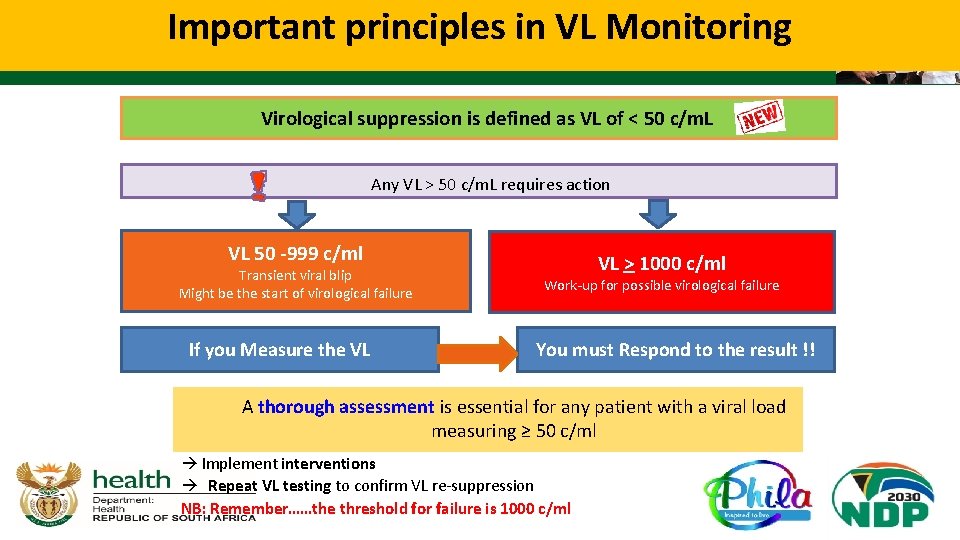
Important principles in VL Monitoring Virological suppression is defined as VL of < 50 c/m. L Any VL > 50 c/m. L requires action VL 50 -999 c/ml Transient viral blip Might be the start of virological failure If you Measure the VL VL > 1000 c/ml Work-up for possible virological failure You must Respond to the result !! A thorough assessment is essential for any patient with a viral load measuring ≥ 50 c/ml Implement interventions Repeat VL testing to confirm VL re-suppression NB: Remember……the threshold for failure is 1000 c/ml
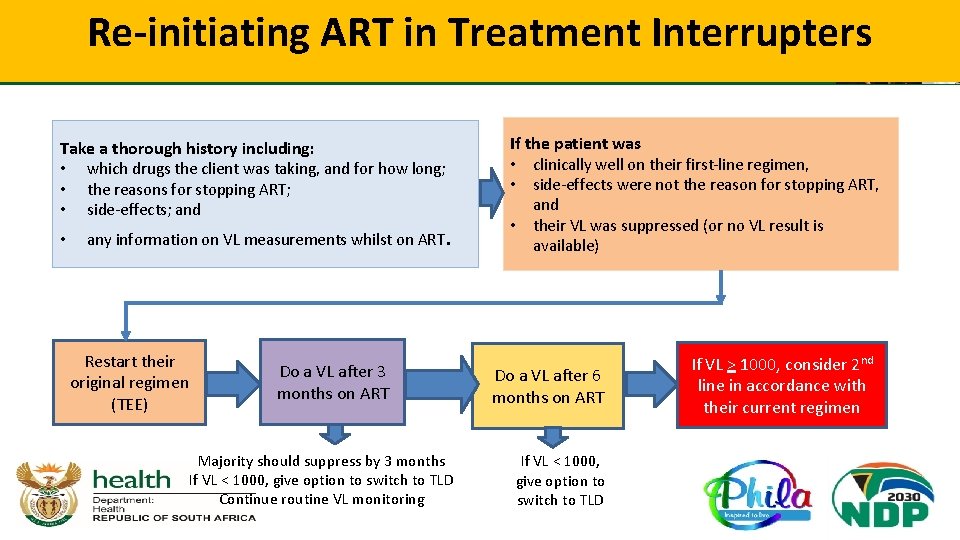
Re-initiating ART in Treatment Interrupters Take a thorough history including: • which drugs the client was taking, and for how long; • the reasons for stopping ART; • side-effects; and • any information on VL measurements whilst on ART. Restart their original regimen (TEE) Do a VL after 3 months on ART Majority should suppress by 3 months If VL < 1000, give option to switch to TLD Continue routine VL monitoring If the patient was • clinically well on their first-line regimen, • side-effects were not the reason for stopping ART, and • their VL was suppressed (or no VL result is available) Do a VL after 6 months on ART If VL < 1000, give option to switch to TLD If VL > 1000, consider 2 nd line in accordance with their current regimen
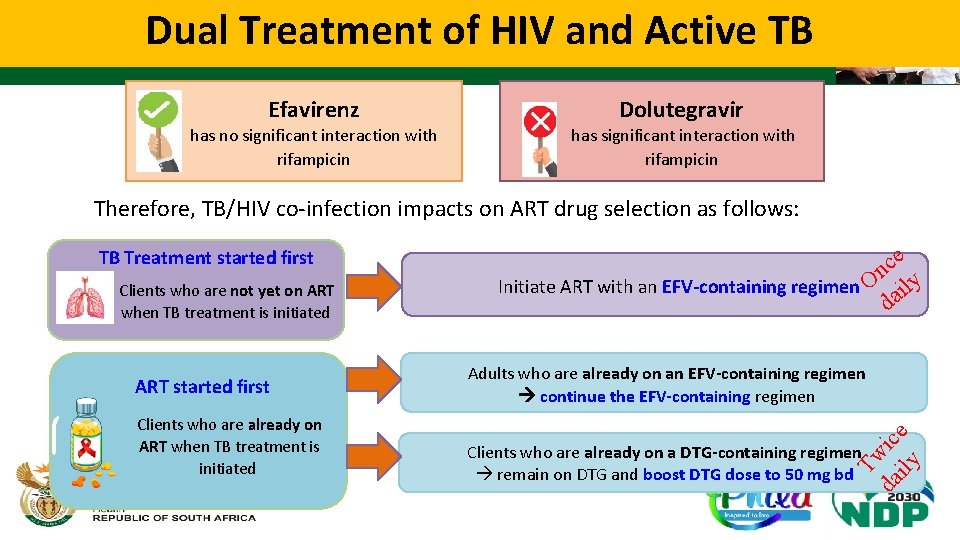
Dual Treatment of HIV and Active TB Efavirenz Dolutegravir has no significant interaction with rifampicin has significant interaction with rifampicin Therefore, TB/HIV co-infection impacts on ART drug selection as follows: Clients who are already on a DTG-containing regimen remain on DTG and boost DTG dose to 50 mg bd ily Clients who are already on ART when TB treatment is initiated ice ART started first Adults who are already on an EFV-containing regimen continue the EFV-containing regimen da Clients who are not yet on ART when TB treatment is initiated ce n Initiate ART with an EFV-containing regimen O ily da Tw TB Treatment started first


TB Preventative Therapy (TPT) and Decanting
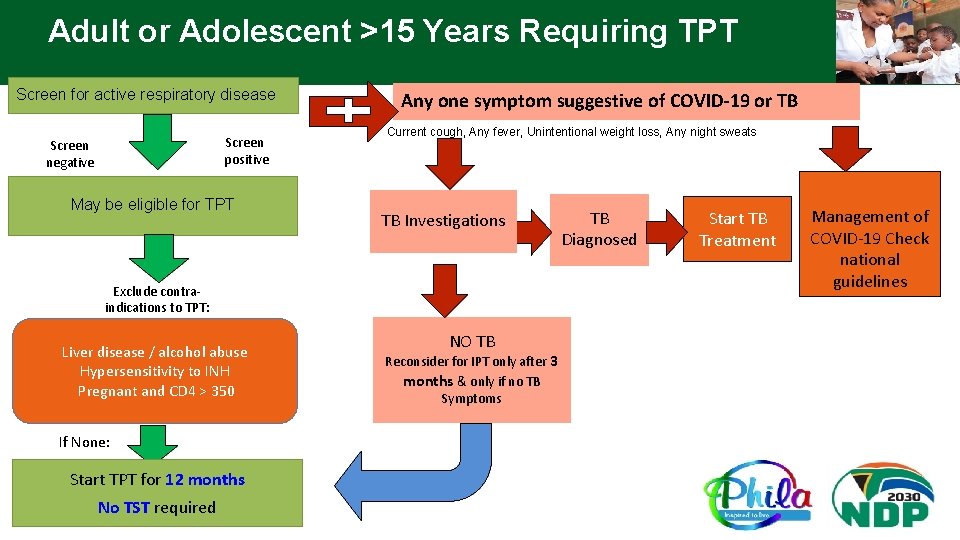
Adult or Adolescent >15 Years Requiring TPT Screen for active respiratory disease Screen positive Screen negative May be eligible for TPT Any one symptom suggestive of COVID-19 or TB Current cough, Any fever, Unintentional weight loss, Any night sweats TB Investigations Exclude contraindications to TPT: Liver disease / alcohol abuse Hypersensitivity to INH Pregnant and CD 4 > 350 If None: Start TPT for 12 months No TST required NO TB Reconsider for IPT only after 3 months & only if no TB Symptoms TB Diagnosed Start TB Treatment Management of COVID-19 Check national guidelines
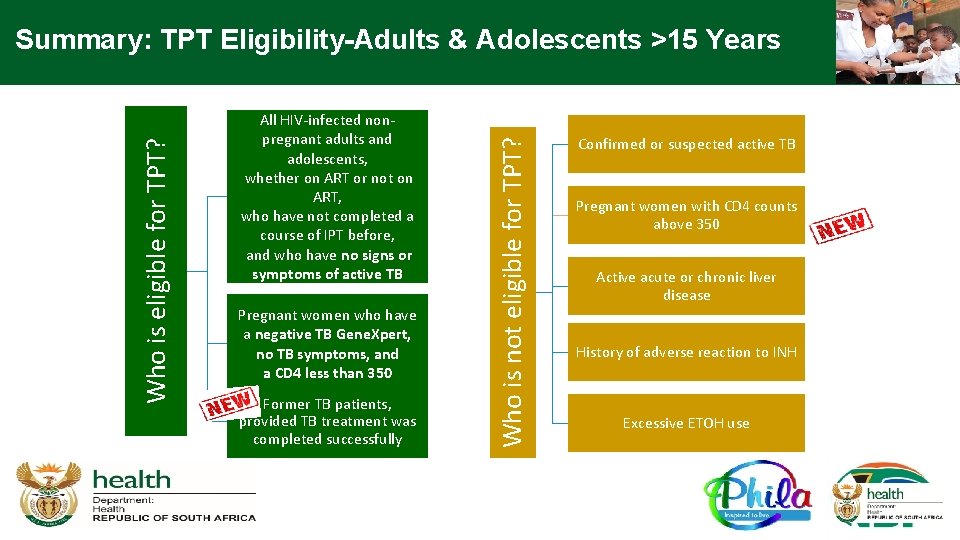
All HIV-infected nonpregnant adults and adolescents, whether on ART or not on ART, who have not completed a course of IPT before, and who have no signs or symptoms of active TB Pregnant women who have a negative TB Gene. Xpert, no TB symptoms, and a CD 4 less than 350 Former TB patients, provided TB treatment was completed successfully Who is not eligible for TPT? Who is eligible for TPT? Summary: TPT Eligibility-Adults & Adolescents >15 Years Confirmed or suspected active TB Pregnant women with CD 4 counts above 350 Active acute or chronic liver disease History of adverse reaction to INH Excessive ETOH use

TPT and Children q TB disease prevalence extremely high, equivalent to 16, 400 per 100, 000 young child contacts (Jaganath D, et al. Clin Infect Dis 2013) q Child contacts have significant increased risk of all-cause mortality compared to children living in non-TB households in same community –If mother had TB, 8 -fold increase (MRR 7. 82 (95% CI 2. 1 -30) AF Gomes et al, Thorax 2011) q Missed opportunities for IPT were common (71%) in at-risk children that later presented with confirmed TB disease – 81% were <3 years of age, 25% had disseminated TB and 5% died –TB source case was the mother or father in 74/156 (47. 4%) children (K Du Preez et al, Ann Trop Paediatric 2011)

TPT and Children q Screening children should include: q Asking about TB contacts § Contact with a TB infected person within the last 12 months q Asking about TB symptoms § Cough / fever / loss of weight / night sweats q If positive TB contact and no active disease offer TPT for 6 months q In all children <5 years q In all HIV-positive children up to 15 years

Summary of the Differences in TPT for Adults and Children Adults Children q Any HIV positive adult may be q A child with a positive TB contact eligible for TPT, regardless of may be eligible for TPT, if less than presence of known TB contact or 5 years old or HIV positive not q INH for 6 months q INH for 12 months q TPT with every new exposure to TB q Usually only given once per lifetime

TPT Regimen and Monitoring in Adults and Adolescents Dosage INH 5 mg/kg/day (max 300 mg/day) For 12 months in adults Children 10 mg/kg of INH with pyridoxine for 6 months Pyridoxine 25 mg/day Comments No TST required to determine TPT duration Prevents peripheral neuropathy Clinical monitoring Adherence: Check adherence to TPT and provide ongoing counselling and patient education Side Effects: Early identification and management of side effects TB symptom screening: If symptoms positive, investigate for TB as per the NTCP guidelines Ensure TPT visits coincide with ART visits

TPT Adherence Guidance q Identify newly diagnosed HIV patient that has completed the 1 st six months of ART, IPT and has a VL<50 copies/ML. q Complete a prescription for the patient containing the ARV medication and TPT items. q TPT multi month script to be aligned to ARV script(2 or 3 months) q Record patient on THIS for TB and Tier. Net q Once TPT is completed (after 12 months from initiation and 6 months on Repeat Prescription Collection Strategies-RPCS), patient should be reviewed q If clinically stable could continue with collection through RPCS for ART.

ADVANCED HIV DISEASE(AHD) / HIV ADVANCED CLINICAL CARE (ACC)

Advance HIV Disease (AHD) /Advance Clinical Care (ACC) WHO definition of AHD q For adults and adolescents, and children ≥ 5 years old, advanced HIV disease is defined as the presence of a CD 4 cell count <200 cells/ mm 3 or a WHO clinical stage 2, 3 or 4 event. q All children < 5 years old with HIV infection are considered as having advanced HIV disease q People with advanced HIV disease are at high risk of death, even after starting ART, with this risk increasing with decreasing CD 4 cell count. The most common causes of death are tuberculosis (TB), severe bacterial infections, and cryptococcal meningitis.

Advance HIV Disease (AHD) /ACC. . AHD: Advance HIV Disease q WHO guidelines recommend that a defined package of care interventions includes: q Screening, q Treatment and prophylaxis for major opportunistic infections, q Rapid initiation of ART and q Intensified treatment adherence support, q Provided to patients presenting with advanced HIV disease to reduce associated morbidity and mortality. q Offered to all people presenting with advanced HIV disease including those who are re-engaging with care after a period of ART interruption.
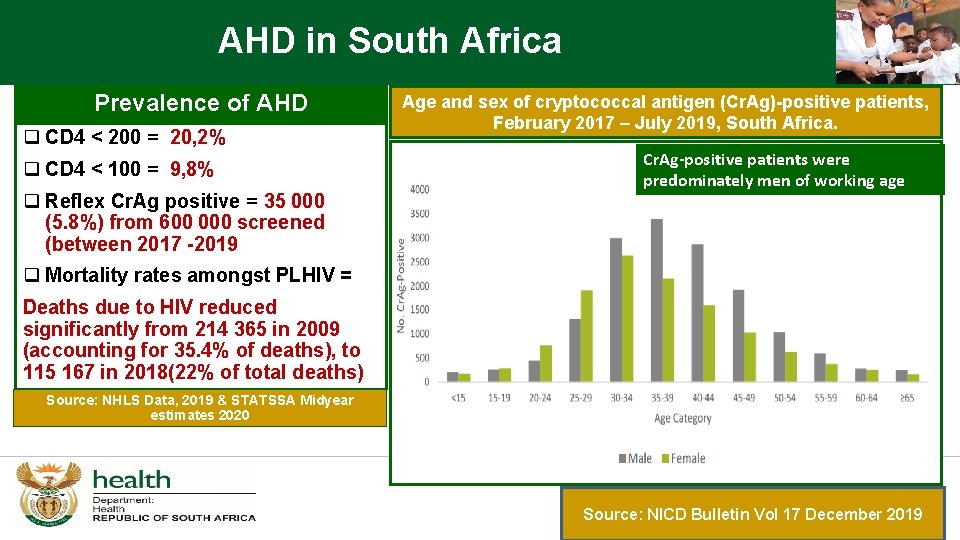
AHD in South Africa Prevalence of AHD q CD 4 < 200 = 20, 2% q CD 4 < 100 = 9, 8% q Reflex Cr. Ag positive = 35 000 (5. 8%) from 600 000 screened (between 2017 -2019 Age and sex of cryptococcal antigen (Cr. Ag)-positive patients, February 2017 – July 2019, South Africa. Cr. Ag-positive patients were predominately men of working age q Mortality rates amongst PLHIV = Deaths due to HIV reduced significantly from 214 365 in 2009 (accounting for 35. 4% of deaths), to 115 167 in 2018(22% of total deaths) Source: NHLS Data, 2019 & STATSSA Midyear estimates 2020 Source: NICD Bulletin Vol 17 December 2019
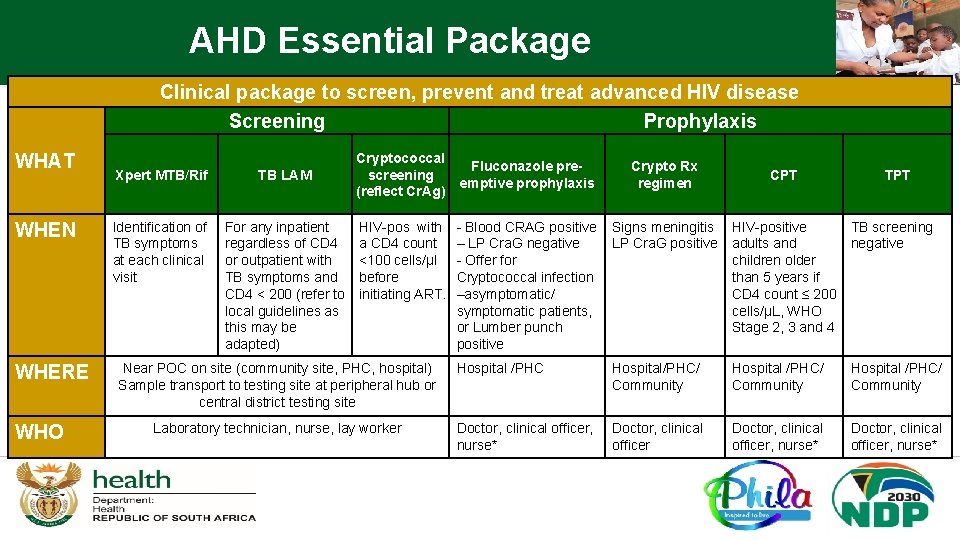
AHD Essential Package Clinical package to screen, prevent and treat advanced HIV disease Screening Prophylaxis WHAT WHEN WHERE WHO Xpert MTB/Rif TB LAM Identification of TB symptoms at each clinical visit For any inpatient regardless of CD 4 or outpatient with TB symptoms and CD 4 < 200 (refer to local guidelines as this may be adapted) Cryptococcal screening (reflect Cr. Ag) HIV-pos with a CD 4 count <100 cells/μl before initiating ART. Near POC on site (community site, PHC, hospital) Sample transport to testing site at peripheral hub or central district testing site Laboratory technician, nurse, lay worker Fluconazole preemptive prophylaxis Crypto Rx regimen - Blood CRAG positive – LP Cra. G negative - Offer for Cryptococcal infection –asymptomatic/ symptomatic patients, or Lumber punch positive Signs meningitis LP Cra. G positive HIV-positive TB screening adults and negative children older than 5 years if CD 4 count ≤ 200 cells/μL, WHO Stage 2, 3 and 4 Hospital /PHC Hospital/PHC/ Community Hospital /PHC/ Community Doctor, clinical officer, nurse* CPT TPT
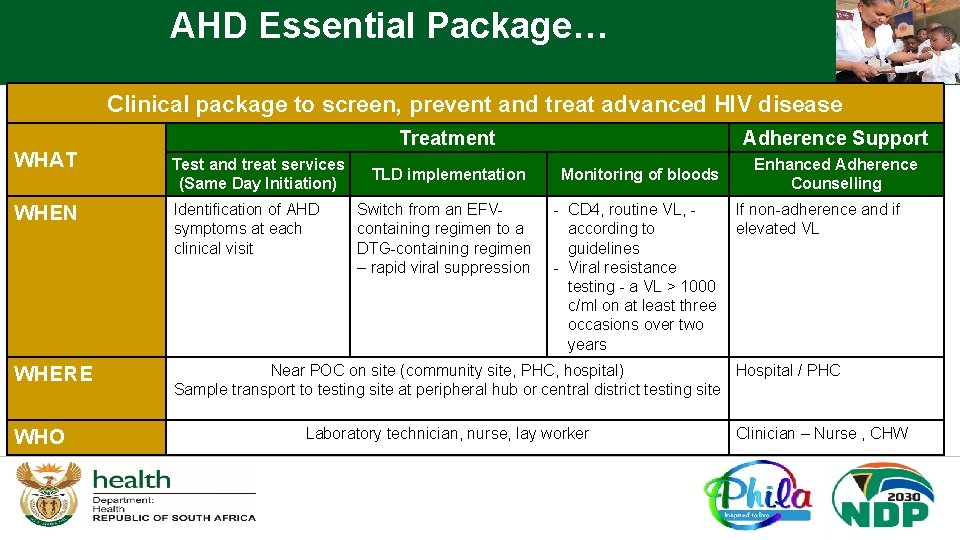
AHD Essential Package… Clinical package to screen, prevent and treat advanced HIV disease Treatment Adherence Support WHAT Test and treat services (Same Day Initiation) WHEN Identification of AHD symptoms at each clinical visit WHERE Near POC on site (community site, PHC, hospital) Hospital / PHC Sample transport to testing site at peripheral hub or central district testing site WHO TLD implementation Monitoring of bloods Switch from an EFVcontaining regimen to a DTG-containing regimen – rapid viral suppression - CD 4, routine VL, according to guidelines - Viral resistance testing - a VL > 1000 c/ml on at least three occasions over two years Laboratory technician, nurse, lay worker Enhanced Adherence Counselling If non-adherence and if elevated VL Clinician – Nurse , CHW

Monitoring & Evaluation q Track LTFU patients as they have interrupted treatment ( refer to SOP 8 – Tracing and Recall) § Return in care with AHD, § provide package of care according to ART guidelines (includes AHD management) q Monitor blood requested , results received and act on them – eg high VL or failure, HIV resistant testing – attend to abnormal results q Tier. net system be captured with clinical information as part of tracking/monitoring and take action on time. q Analyse NHLS/NICD reports on CD 4 count, VL, HIV resistant , Cr. Ag, etc

We need to Maximize Low Hanging Fruits… Ensure TPT and Cotrimoxazole are optimally implemented according to the ART Guidelines

…. But it will take us a heavy push, to avoid major setbacks that will lead to AHD…. Let's intensify CD 4 Testing to clinically manage our patients and avert mortality that could have been prevented…

AHD Policies and Guidelines Overarching – Country HIV/AIDS Response • South African - National Strategic plan on HIV, TB and STIs (2017 -2022) NDOH – HIV/AIDS/TB/ PMTCT & AHD response • National Consolidated HIV treatment guidelines, includes AHD clinical management • AGL-SOP - Minimum package of interventions to support linkage to care, adherence and retention in care Operationalization • Training material developed, complete. Plan to conduct national training in August 2020. Due to current COVID-19 pandemic, a 3 days Virtual training underway (face to face takes 5 days)

Priorities for 2020 – 2021 National Advance Clinical Care (ACC)/AHD training package developed and finalized: q Conduct national virtual ACC/AHD training in August 2020, q Provinces to cascade the ACC trainings to districts and health facilities.

Acknowledgements The final booklet produced q DMo. C TWG q NDOH Clusters: q IAS q CDC q HIV/AIDS & STIs, TB, MNCWH, q NCDs, PHC, CCMDD, q TIER. NET, Affordable Medicine q RTC and Knowledge Hub q USAID q HE 2 RO

Adherence Guideline Web Links Knowledge Hub: https: //www. knowledgehub. org. za/elibrary/adherenceguidelines-hiv-tb-and-ncds-standard-operating-procedures-2020 National Department of Health: http: //www. health. gov. za/index. php/component/phocadow nload/category/672
- Slides: 79