Addressing COVID19 in recently completed clinical trials Bryan

Addressing COVID-19 in recently completed clinical trials Bryan Goldman DSBS webinar 26 -Nov-2020

2 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® Global Phase 3 a Program: a timeline 2020 Jan Feb Mar Apr Trial 1 Trial 2 Trial 3 Trial 4 Last Patient Last Treatment Last Patient Last Visit Lockdown (DK) May Jun

3 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® Initial Considerations How to handle protocol deviations? How to capture reason for discontinuation &/or withdrawal? How to identify impacted visits? Allow any home assessments? Which ones?

4 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® Initial Decisions Structured entry of all COVID-related protocol deviations Visits impacted by COVID identified from protocol deviations Few COVID-related discontinuations & withdrawals entered as free text Captured body weight, pregnancy assessed at home. Excluded from summaries, analyses

5 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® FDA expectations for the Clinical Trial Report Sponsors should describe in appropriate sections of the clinical study report (or in a separate study specific document): 1. Contingency measuresimplemented to manage study conduct during disruption of the study as a result of COVID-19 control measures. 2. A listing of all participants affectedby the COVID-19 related study disruption by unique subject numberidentifier and by investigational site, and a description of how the individual’s participation was altered. 3. Analyses and corresponding discussions that address the impact of implemented contingency measures (e. g. , trial participant discontinuationfrom investigational product and/or study, alternative proceduresused to collect critical safety and/or efficacy data) on the safety and efficacy results reported for the study.

6 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® Clinical Trial Report Example COVID-19 – Summary of impact • The global pandemic occurred when almost all subjects had completed their last visits. Therefore, the impact of COVID-19 was low in this trial. The main trial procedures affected by COVID-19 comprised: • Source data verification was abolished for the last part of the trial. . . All data were still entered into the electronic data capture system and checked for completeness. Furthermore, data cleaning and casebook sign-off was ensured. • Approximately 213 subjects had their follow-up visits… converted to phone visits… AE reporting and safety follow-up information was ensured for these subjects. • . . . No subjects discontinued treatment or withdrew from the trial due to COVID-19.
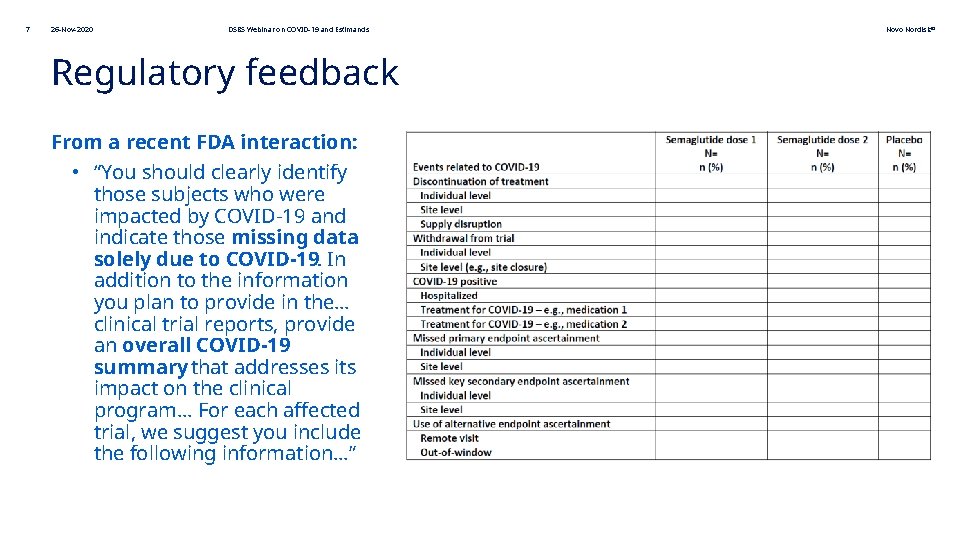
7 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Regulatory feedback From a recent FDA interaction: • “You should clearly identify those subjects who were impacted by COVID-19 and indicate those missing data solely due to COVID-19. In addition to the information you plan to provide in the… clinical trial reports, provide an overall COVID-19 summary that addresses its impact on the clinical program… For each affected trial, we suggest you include the following information…” Novo Nordisk®

8 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® Updated strategy • COVID-related protocol deviations tabulated manually to derive a list of subjects and visits converted from site visit to phone visit due to COVID-19 • Used to identify both subjects with missing assessments due to COVID-19 and those with home assessments performed at a COVID-impacted visit. • This list used to flag: • Home assessments to be excluded from analyses, summaries • Subject visits impacted by COVID-19 • Based on these data enhancements, listings and summary tables of subjects with data missing or excluded due to COVID-19 can now be produced

9 26 -Nov-2020 DSBS Webinar on COVID-19 and Estimands Novo Nordisk® Questions? ?
- Slides: 9