Additional Aspects of Aqueous Equilibria Chapter 17 Acid

Additional Aspects of Aqueous Equilibria Chapter 17

Acid- Base Rxns – Write Molecular and Net ionic equations Strong Acid and Strong Base HCl(aq) + Na. OH (aq) Strong Acid and Weak Base HCl(aq) + Na. C 2 H 3 O 2(aq) Strong Base and Weak Acid Na. OH(aq) + HC 2 H 3 O 2(aq)

The Common-Ion Effect The extent of ionization of a weak electrolyte is decreased by adding a strong electrolyte that has an ion in common with the weak electrolyte

The Common-Ion Effect If you have a weak acid and a common ion, the [H+] will decrease, causing the p. H to increase

The Common-Ion Effect If you have a weak base and a common ion, the [OH-] will decrease, causing the p. H to decrease

Sample Exercise 17. 1 Calculating the p. H When a Common Ion is Involved What is the p. H of a solution made by adding 0. 30 mol of acetic acid and 0. 30 mol of sodium acetate to enough water to make 1. 0 L of solution? Ka = 1. 8 x 10 -5

Composition and Action of Buffered Solutions Buffers are solns which contain a weak conj. acid-base pair and can resist drastic changes in p. H upon the addition of small amounts of strong acid or strong base

Composition and Action of Buffered Solutions A buffer resists changes in p. H because it contains both an acidic species to neutralize OH- and a basic one to neutralize H+

Composition and Action of Buffered Solutions Consider a buffer composed of weak acid HX and one of its salts MX (X-), where M is Na+, K+, or another cation.

Composition and Action of Buffered Solutions If OH- ions are added, they react with the acid component of the buffer to produce water and base X-. OH- + HX H 2 O + XAs long as the ratio of [HX]/[X-] doesn’t change too much, the change of p. H is small.

Composition and Action of Buffered Solutions If H+ ions are added, they react with the base component of the buffer H+ + X- HX As long as the ratio of [HX]/[X-] doesn’t change too much, the change in p. H is small.


Composition and Action of Buffered Solutions Buffers n n n Made of conjugate acid-base pairs most effectively resist change in p. H when the concentration of weak acid and conjugate base are the same Chosen so that the acid in the buffer has a p. Ka close to the desired p. H

Sample Exercise 17. 3 Calculating the p. H of a Buffer What is the p. H of a buffer that is 0. 12 M in lactic acid [HC 3 H 5 O 3] (Ka = 1. 4 × 10 -4) and 0. 10 M in sodium lactate [Na. C 3 H 5 O 3] ?

Sample Exercise 17. 4 Preparing a Buffer How many moles of NH 4 Cl must be added to 2. 0 L of 0. 10 M NH 3 to form a buffer whose p. H is 9. 00? (Assume that the addition of NH 4 Cl does not change the volume of the solution. )

Addition of Strong Acids or Bases to Buffers To calculate p. H upon addition of a SA or SB 1. Consider the acid-base neutralization rxn and determine its effect on [HX] and [X-] (LR and stoichiometry) 2. Use Ka and the new concentrations of [HX] and [X-] to calculate[H+] (ICE or H -H)

Sample Exercise 17. 5 Calculating p. H Changes in Buffers A buffer is made by adding 0. 300 mol CH 3 COOH and 0. 300 mol CH 3 COONa to enough water to make 1. 00 L of solution. The p. H of the buffer is 4. 74 (Sample Exercise 17. 1). (Ka = 1. 8 x 10 -5) (a) Calculate the p. H of this solution after 0. 020 mol of Na. OH is added. (b) calculate the p. H of the soln after the addition of 0. 020 mol HCl

Acid-Base Titrations Warm up: How many m. L of 0. 105 M HCl are needed to titrate 23. 5 m. L of 0. 117 M KOH to the equivalence point? n Hint: Write a reaction and use stoichiometry

Acid-Base Titrations p. H titration curve 1. graph of the p. H as a function of the volume of added titrant 2. Shape helps determine equivalence pt, choose indicator, and determine Ka or Kb 3. The equivalence point is the point at which H+ moles = OH- moles 4. Any indicator beginning and ending its color change on the rapid-rise portion of the curve can be used
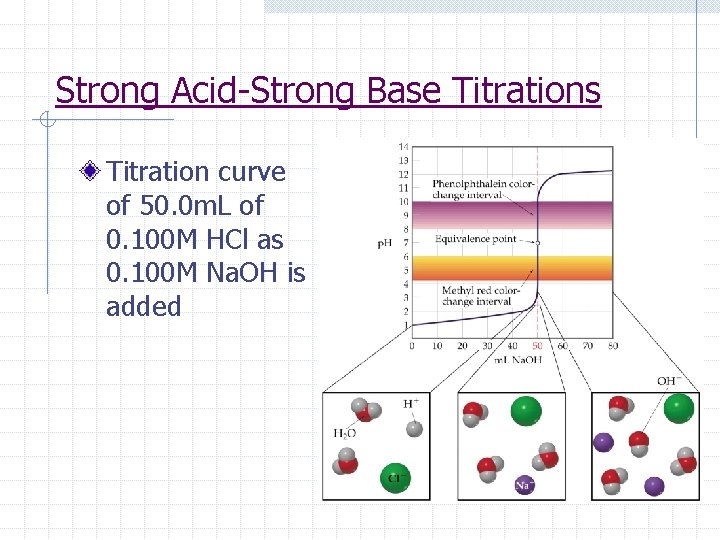
Strong Acid-Strong Base Titrations Titration curve of 50. 0 m. L of 0. 100 M HCl as 0. 100 M Na. OH is added

Strong Acid-Strong Base Titrations (1) The initial p. H before any base is added can be determined from the initial concentration of the strong acid

Strong Acid-Strong Base Titrations (2) Between the initial p. H and the equivalence pt, the p. H is determined by the concentration of the acid that has not yet been neutralized

Strong Acid-Strong Base Titrations (3) At the equivalence point, the moles of H+ = moles of OH- and all that remains in solution is a salt. The p. H is 7. 00.

Strong Acid-Strong Base Titrations (4) After the equivalence point, the p. H can be determined by finding the excess[OH-], the p. OH, and then p. H.

Sample Exercise 17. 6 Calculating p. H for a Strong Acid-Strong Base Titration Calculate the p. H when the following quantities of 0. 100 M Na. OH solution have been added to 50. 0 m. L of 0. 100 M HCl solution: (a) 49. 0 m. L, (b) 51. 0 m. L.
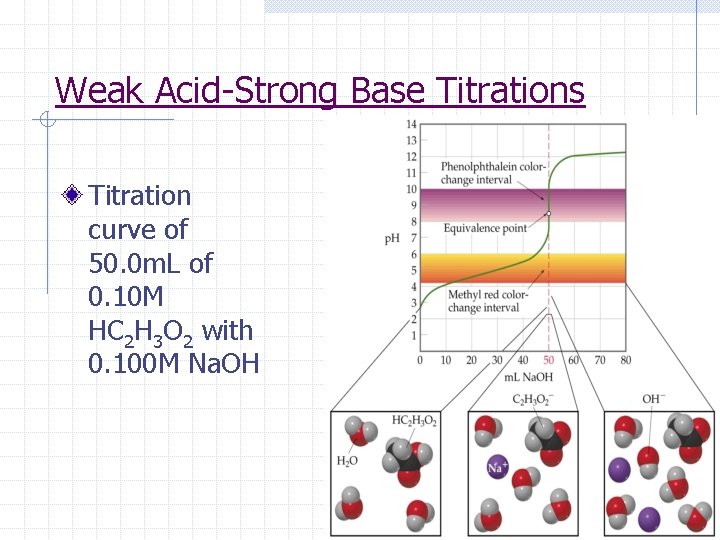
Weak Acid-Strong Base Titrations Titration curve of 50. 0 m. L of 0. 10 M HC 2 H 3 O 2 with 0. 100 M Na. OH

Weak Acid-Strong Base Titrations (1) The initial p. H can be determined by calculating the p. H of weak acid

Weak Acid-Strong Base Titrations (2) Between the initial p. H and the equivalence pt, the p. H can be determined using stoichiometry and the Ka (Note: p. H=p. Ka when [A-] = [HA] or [acid]=[base] which occurs halfway to equivalence)

Weak Acid-Strong Base Titrations (3) At the equivalence pt, the p. H must be calculated by finding p. H of weak base formed
![Weak Acid-Strong Base Titrations (4) After the equivalence pt, the excess [OH-] from the Weak Acid-Strong Base Titrations (4) After the equivalence pt, the excess [OH-] from the](http://slidetodoc.com/presentation_image_h/0a77137412578b0161013ed2b49938db/image-30.jpg)
Weak Acid-Strong Base Titrations (4) After the equivalence pt, the excess [OH-] from the addition of the strong base will account for the p. H

Sample Exercise 17. 8 Calculating p. H for a Weak Acid-Strong Base Titration Calculate the p. H of the solution formed when 45. 0 m. L of 0. 100 M Na. OH is added to 50. 0 m. L of 0. 100 M CH 3 COOH (Ka = 1. 8 × 10 -5).

Sample Exercise 17. 9 Calculating the p. H at the Equivalence Point Calculate the p. H at the equivalence point in the titration of 50. 0 m. L of 0. 100 M CH 3 COOH with 0. 100 M Na. OH.
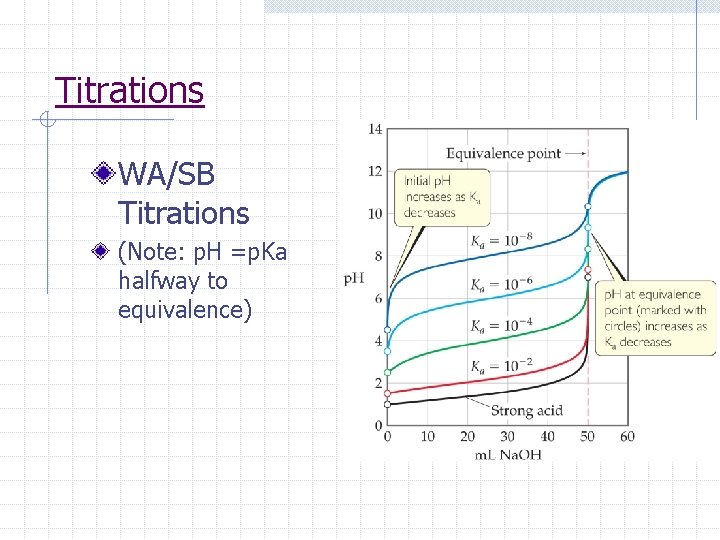
Titrations WA/SB Titrations (Note: p. H =p. Ka halfway to equivalence)
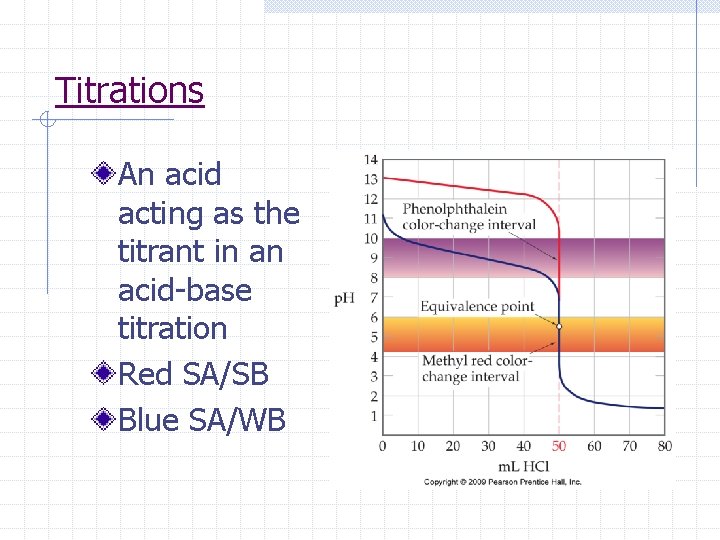
Titrations An acid acting as the titrant in an acid-base titration Red SA/SB Blue SA/WB
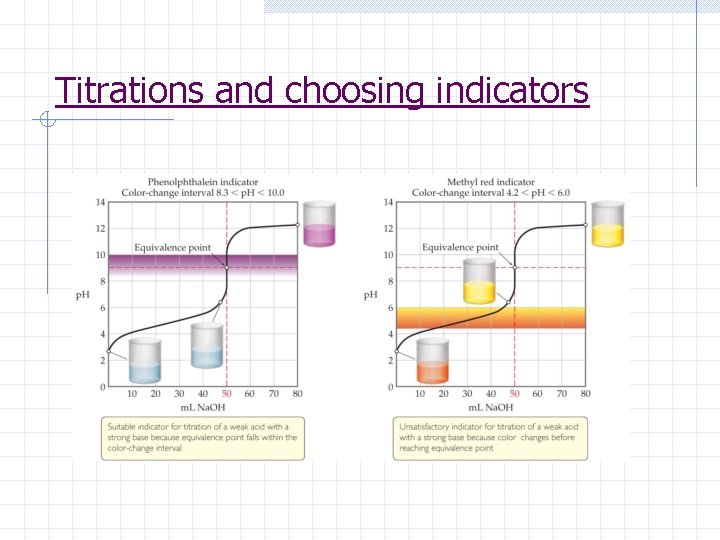
Titrations and choosing indicators
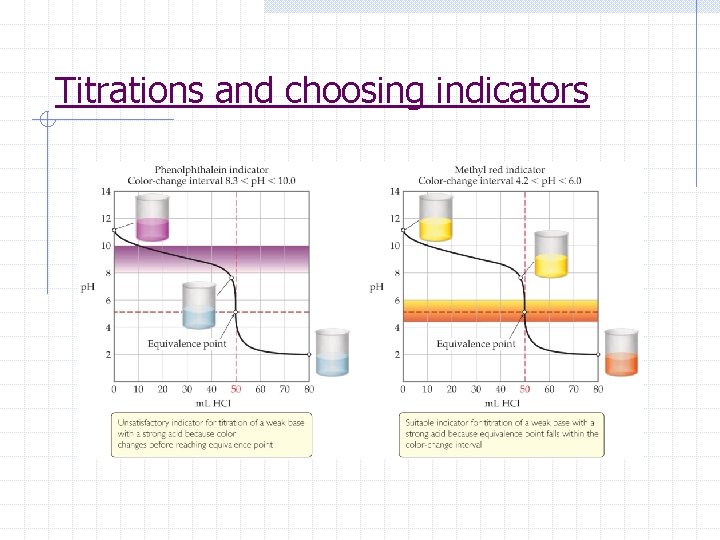
Titrations and choosing indicators
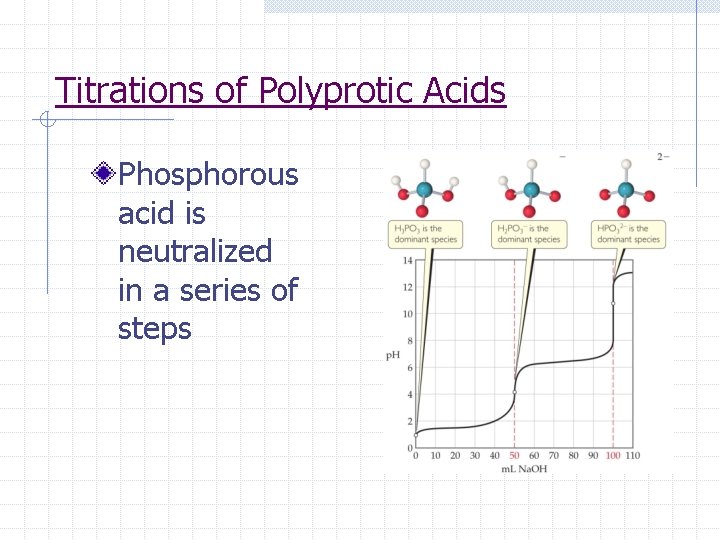
Titrations of Polyprotic Acids Phosphorous acid is neutralized in a series of steps

Titration Simulation http: //group. chem. iastate. edu/Greenbo we/sections/projectfolder/flashfiles/stoic hiometry/acid_base. html http: //chemilp. net/lab. Techniques/Acid. Base. Idicator. S imulation. htm

Solubility and Salts http: //phet. colorado. edu/en/si mulation/soluble-salts

Solubility Equilibria Solubility the amount of a substance that dissolves in a given amount of solvent at a certain T to form a saturated soln

Solubility Equilibria Unsaturated Solutions – more solute can be added to solvent. It will continue to dissolve solute into solvent. Saturated solutions – no more solute can be added to the solvent. It can no longer dissolve any more solute.

Solubility Equilibria A saturated solution of a slightly soluble salt is one in which the solution is in equilibrium with undissolved solute The solubility product constant Ksp tells us how soluble a solid is in water

Solubility Equilibria Predict which solution would have a greater solubility Ag. Cl Ksp = 1. 8 x 10 -10 n Ag. Br Ksp = 5. 0 x 10 -13 n Ag. I Ksp = 8. 3 x 10 -17 n

Sample Exercise 17. 9 Writing Solubility-Product (Ksp) Expressions Write the expression for the solubility product constant for Ca. F 2, and look up the corresponding Ksp value in Appendix D.

Sample Exercise 17. 10 Calculating Ksp from Solubility Solid silver chromate is added to pure water at 25 ºC. Some of the solid remains undissolved at the bottom of the flask. The mixture is stirred for several days to ensure that equilibrium is achieved between the undissolved Ag 2 Cr. O 4(s) and the solution. Analysis of the equilibrated solution shows that its silver ion concentration is 1. 3 × 10 -4 M. Calculate Ksp for this compound.

Sample Exercise 17. 11 Calculating Solubility from Ksp The Ksp for Ca. F 2 is 3. 9 × 10 -11 at 25 ºC. Calculate the solubility of Ca. F 2 in grams per liter.

Factors that affect solubility Solubility of a substance is affected by T

Factors that affect solubility Solubility of a slightly soluble salt is decreased by the presence of a second solute that has a common ion

Sample Exercise 17. 12 Calculating the Effect of a Common Ion on Solubility Calculate the molar solubility of Ca. F 2 at 25 °C in a solution that is (a) 0. 010 M in Ca(NO 3)2, (b) 0. 010 M in Na. F.

Factors that affect solubility The solubility of any substance whose anion is basic will be affected to some extent by the p. H of the soln

Factors that affect solubility A saturated solution of Mg(OH)2 has a p. H of 10. 52 and contains 1. 7 x 10 -4 M Mg 2+. The p. H is then changed to 9. 00, what is [Mg 2+]?

Precipitation and Separation of ions If Q > Ksp, precipitation occurs until Q=Ksp If Q = Ksp, equilibrium exists (saturated solution) If Q < Ksp, solid dissolves until Q=Ksp (unsaturated)

Sample Exercise 17. 15 Predicting Whether a Precipitate Will Form Will a precipitate form when 0. 10 L of 8. 0 x 10 -3 M Pb(NO 3)2 is added to 0. 40 L of 5. 0 x 10 -3 M Na 2 SO 4?

Precipitation and Separation of ions Selective precipitation - Ions can be separated from each other based upon the solubilities of their salts

Sample Exercise 17. 16 Calculating Ion Concentrations for Precipitation A solution contains 1. 0 × 10 -2 M Ag+ and 2. 0 × 10 -2 M Pb 2+. When Cl– is added to the solution, both Ag. Cl (Ksp = 1. 8× 10 -10) and Pb. Cl 2 (Ksp = 1. 7× 10 -5) precipitate from the solution. What concentration of Cl– is necessary to begin the precipitation of each salt? Which salt precipitates first?

Practice Exercise 17. 16 Calculating Ion Concentrations for Precipitation A solution consists of 0. 050 M Mg 2+ and 0. 020 M Cu 2+. Which ion will precipitate first as OH– is added to the solution? What concentration of OH– is necessary to begin the precipitation of each cation? [Ksp = 1. 8 × 10 -11 for Mg(OH)2, and Ksp = 4. 8 × 10 -20 for Cu(OH)2. ]

Practice Exercise 17. 7 (a) Calculate the p. H in the solution formed by adding 10. 0 m. L of 0. 050 M Na. OH to 40. 0 m. L of 0. 0250 M benzoic acid (C 6 H 5 COOH, Ka = 6. 3 × 10 -5).

Practice Exercise 17. 7 (b) Calculate the p. H in the solution formed by adding 10. 0 m. L of 0. 100 M HCl to 20. 0 m. L of 0. 100 M NH 3. (Kb=1. 8 x 10 -5)

Practice Exercise 17. 8 Calculate the p. H at the equivalence point when (a) 40. 0 m. L of 0. 025 M benzoic acid (C 6 H 5 COOH, Ka = 6. 3 × 10 -5 ) is titrated with 0. 050 M Na. OH
- Slides: 59