Acute Respiratory Distress Syndrome DR SABAH HASANI PULMONOGIST

Acute Respiratory Distress Syndrome DR SABAH HASANI PULMONOGIST

• In 2011, the ARDS Definition Task Force of the European Society in Berlin: 1. “acute” respiratory failure must occur within 1 week of predisposing illness 2. ARDS is categorized by severity: mild (200 < Pa. O 2/Fi. O 2 300), moderate (100 < Pa. O 2/Fi. O 2 200), and severe (Pa. O 2/Fi. O 2 ≤ 100). requires a minimum PEEP of 5 cm H 2 O for all severity categories in recognition of the influence of PEEP on the Pa. O 2/Fi. O 2 ratio. 3. use of noninvasive PEEP is allowed but limited to the mild ARDS category
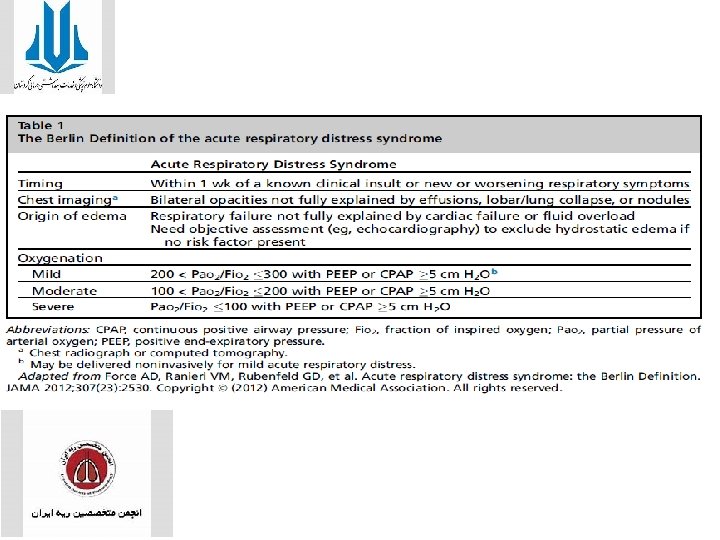

• Ancillary variables : • Radiographic severity, respiratory system compliance, level of PEEP, and exhaled minute ventilation were dropped from the empirical definition because they did not enhance the predictive value of the severe ARDS classification.

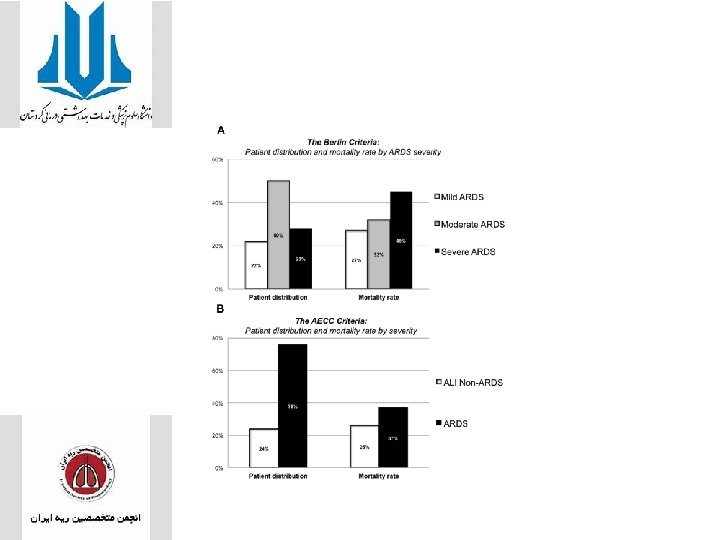
• ARDS severity in the cohort according to the Berlin Definition • was 22% mild, 50% moderate, and 28% severe, with associated mortality rates of 27%, 32%, and 45%, respectivel The Berlin Definition was more predictive of mortality.

EPIDEMIOLOGY • ARDS in patients receiving mechanical ventilation via an endotracheal tube or noninvasive face mask, similarly to the Berlin Definition The incidence of ALI and ARDS was estimated at 78. 9 and 58. 7 cases per 100, 000 patient-years, respectively, • with an age-adjusted extrapolated incidence of 190, 600 cases per year in the United States. Incidence increased with age to a peak of 306 cases per 100, 000 patient-years among 75 - to 84 -year-olds. • Pneumonia (46%) and non pulmonary sepsis (33%) were the most common risk factors. • Overall hospital mortality was 38. 5%.

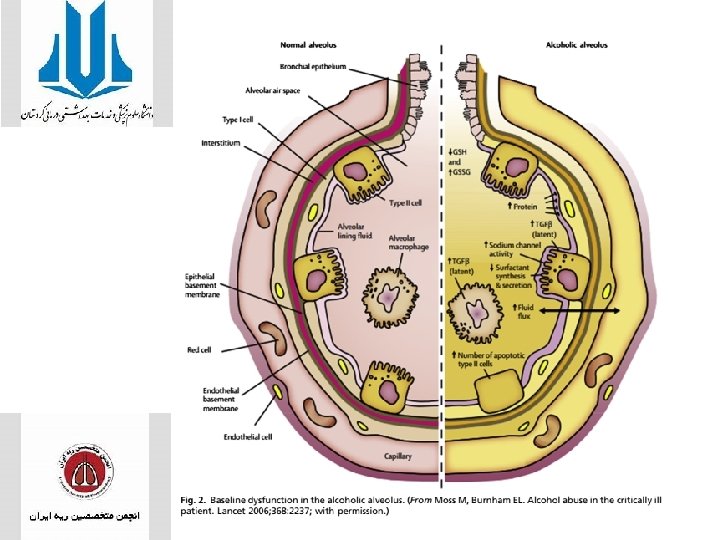
Environmental Risk Factors • ALCOHOL ABUSE • central role for pulmonary immune dysfunction as well as alveolar epithelial dysfunction in the mechanistic link between alcohol and ARDS
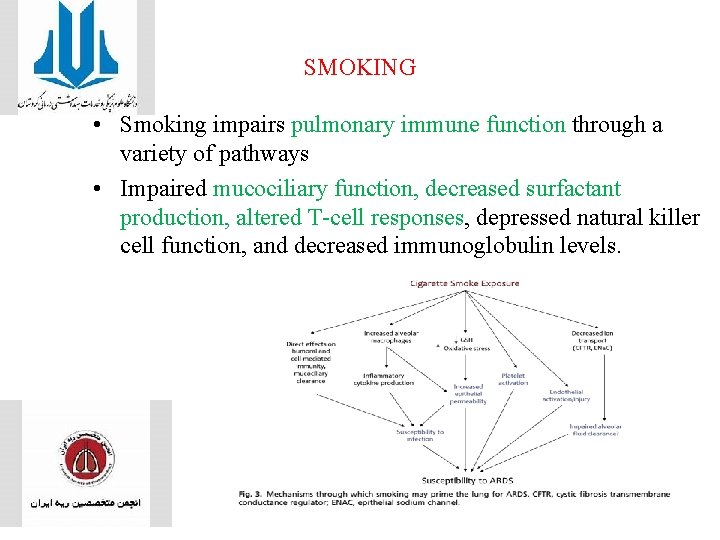
SMOKING • Smoking impairs pulmonary immune function through a variety of pathways • Impaired mucociliary function, decreased surfactant production, altered T-cell responses, depressed natural killer cell function, and decreased immunoglobulin levels.
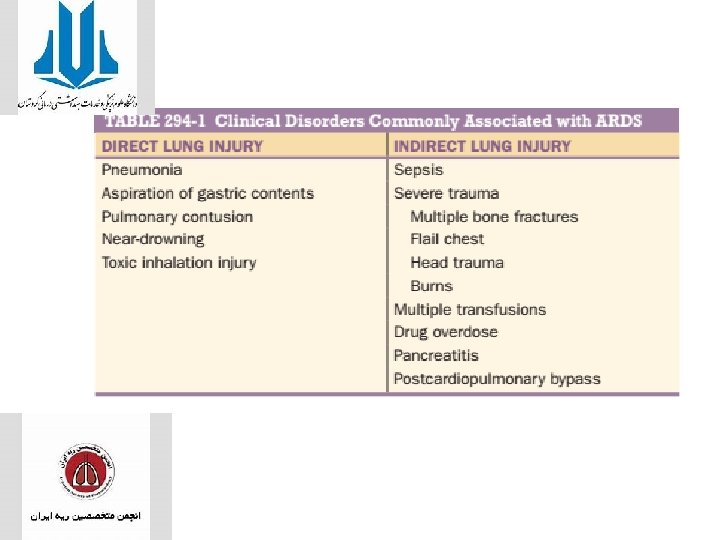

• Predisposing Conditions • established cases of ARDS, with pneumonia (35%– 45%) and non pulmonary sepsis (30%– 35%) being the most common followed by aspiration, trauma, pancreatitis, and multiple transfusions, • Sepsis (33%), pneumonia (27%), and shock (19%) were the most common predisposing • cardiac surgery (15%), traumatic brain injury (12%), aspiration (9%), and acute abdominal surgery, lung contusion, and multiple fractures (7%) were also common. • Chronic alcohol abuse and active and passive tobacco exposure • diabetes mellitus may have a protective effect,

• Diabetes remained protective • race and gender are not clear independent risk factors for developing ARDS. • men had higher rates of developing ALI, but not after controlling for baseline imbalances between sexes • The Berlin Definition did predict mortality better than AECC criteria in the Task Force’s validation cohort;

RISK FACTOR ARDS • Older age, • Neutrophilia • Higher lactate dehydrogenase • D-dimer levels, according to a JAMA Internal Medicine study. methylprednisolone was associated with lower risk for death.

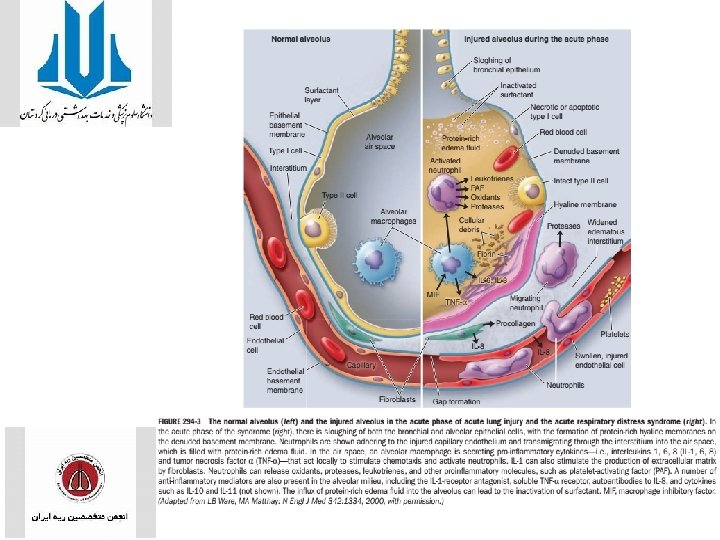
CLINICAL COURSE AND PATHOPHYSIOLOGY • The natural history of ARDS is marked by three phases—exudative, proliferative, and fibrotic. • Exudative Phase • alveolar capillary endothelial cells and type I pneumocytes (alveolar epithelial cells) are injured, with consequent loss of the normally tight alveolar barrier to fluid and macromolecules. Edema fluid that is rich in protein accumulates in the interstitial and alveolar spaces. • Pro-inflammatory cytokines (e. g. , interleukin 1, interleukin 8, and tumor necrosis factor α )and lipid mediators (e. g. , leukotriene B 4) are increased • Leading to the recruitment of leukocytes (especially neutrophils) into the pulmonary interstitium and alveoli

• condensed plasma proteins aggregate in the air spaces with cellular debris and dysfunctional pulmonary surfactant to form hyaline membrane whorls. • Pulmonary vascular injury • Alveolar edema predominantly involves dependent portions of the lung with diminished aeration. • Collapse of large sections of dependent lung can contribute to decreased lung compliance. • Consequently, intrapulmonary shunting and hypoxemia develop and the work of breathing increases, leading to dyspnea. • The exudative phase encompasses the first 7 days of illness after exposure to a precipitating ARDS risk factor, with the patient experiencing the onset of respiratory symptoms. • Although usually presenting within 12– 36 h after the initial insult, symptoms can be delayed by 5– 7 days

• The chest radiograph • opacities consistent with pulmonary edema and often involves at least three-quarters of the lung fields • While characteristic for ARDS, these radiographic findings are not specific and can be indistinguishable from cardiogenic pulmonary edema. • the chest x-ray in ARDS may not demonstrate cardiomegaly, pleural effusions, or pulmonary vascular redistribution
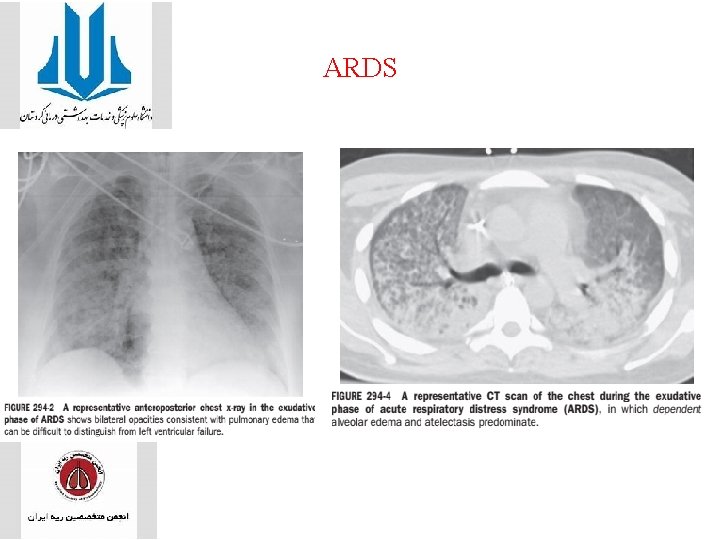
ARDS

• Proliferative Phase • Day 7 to day 21. • Most patients recover rapidly and are liberated from mechanical ventilation during this phase. Despite this improvement, many patients still experience dyspnea, tachypnea, and hypoxemia. • Some patients develop progressive lung injury and early changes of pulmonary fibrosis during the proliferative phase. • Histologically, the first signs of resolution are often evident in this phase, with the initiation of lung repair, the organization of alveolar exudates, and a shift from neutrophil- to lymphocyte-predominant pulmonary infiltrates. • pulmonary fibrosis during the proliferative phase. • Fibrotic Phase • While many patients with ARDS recover lung function 3– 4 weeks after the initial pulmonary injury

• Fibrotic phase • require long-term support on mechanical ventilators and/or supplemental oxygen. • Histologically, the alveolar edema and inflammatory exudates of earlier phases convert to extensive alveolar-duct and interstitial fibrosis. • Marked disruption of acinar architecture leads to emphysema-like changes, with large bullae. • Intimal fibro proliferation in the pulmonary microcirculation causes progressive vascular occlusion and pulmonary hypertension. • The physiologic consequences include an increased risk of pneumothorax, reductions in lung compliance, and increased pulmonary dead space.
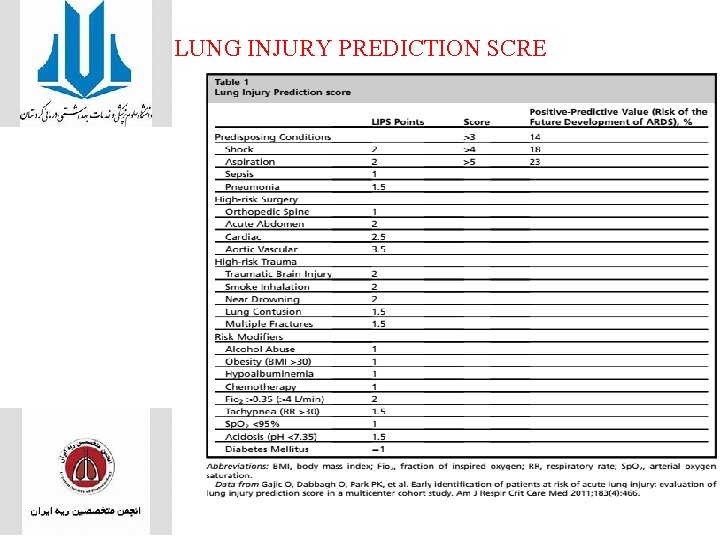
LUNG INJURY PREDICTION SCRE

• In the original study, an LIPS of greater than 4 was found to have good discriminatory power, in that 97% of patients with a score of less than or equal to 4 did not go on to develop ARDS, • Whereas 18% of patients with a score greater than 4 went on to develop ARDS

Plasma Biomarkers for the Risk Prediction • Adding Angiopoietin-2 levels to the LIPS further improved discriminatory power. • other plasma biomarkers such as club cell protein 16, interleukin 8, and tissue factor have also shown promise in identifying at-risk patients
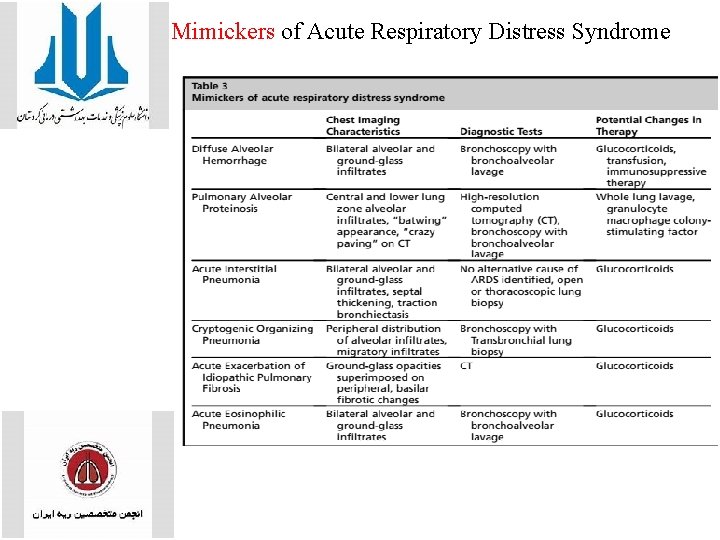
Mimickers of Acute Respiratory Distress Syndrome
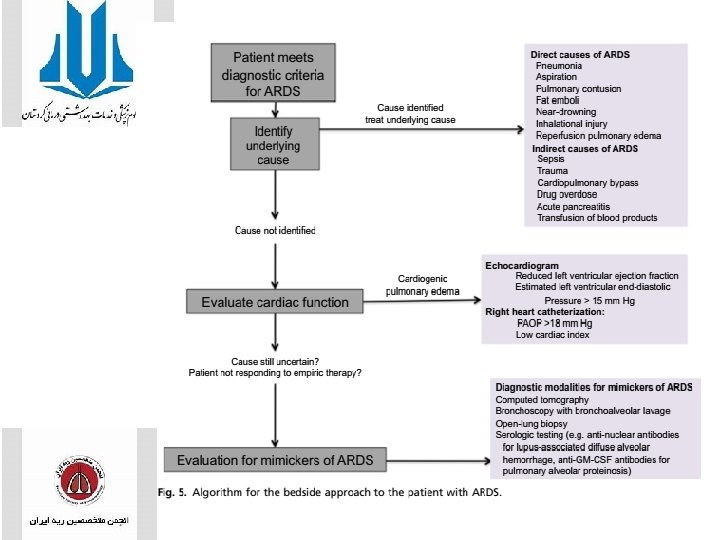

Invasive Evaluation of Acute Respiratory Distress Syndrome • Flexible bronchoscopy with bronchoalveolar lavage may play a role in determining the cause of ARDS and evaluating for mimickers of ARDS. sensitivity as high as 60% • a differential cell count of 48% eosinophils on bronchoalveolar lavage and was diagnosed with acute eosinophilic pneumonia. • ventilator-associated pneumonia diagnosed by bronchoalveolar lavage occurs in approximately 36% of patients with ARDS. • Open lung biopsy may also play a similar role in patients with undifferentiated ARDS

TREATMENT • VENTILATOR-INDUCED LUNG INJURY • Classically, the components of VILI include: barotrauma, volutrauma, and atelectrauma. • Alveolar over distention coupled with cyclical recruitment and atelectasis are the primary causes of VIL. • Barotrauma is generally recognized as pneumothoraces, pneumomediastinum, and subcutaneous emphysema. • TARGETING VOLUTRAUMA • A meta-analysis including 6 RCTs (1297 patients) comparing ventilation strategies targeting a Tidal volume of 7 m. L/kg or less and plateau pressures of 30 cm. H 2 O or less

LUNG Protective Strategy

Principle 1. Minimise atelectrauma • open up alveoli with recruitment manoeuvre • keep alveoli open (prevent de-recruitment) by applying optimal PEEP 2. Minimise volutrauma (over-distension injury) • keep plateau pressure < 30 cm H 2 O • use low tidal volume ventilation 6 ml/kg IBW 3. Minimise O 2 toxicity • maintain Fi. O 2 below 0. 6 4. Accept physiologic target outside normal range • permissive hypercapnia • permissive hypoxemia

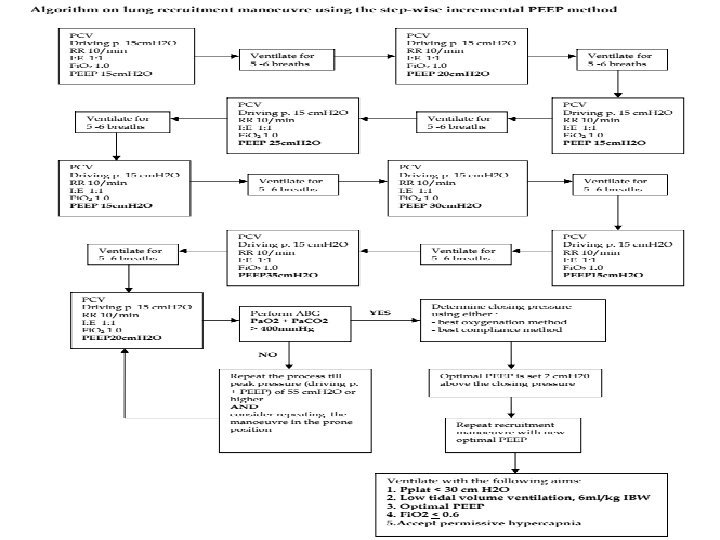
Recruitment manoeuvre • The patient must have relatively stable haemodynamics • Choose one of the 2 options: A. Step-wise incremental PEEP B. PCV with PEEP method

A. Step-wise incremental PEEP Patient may need to be paralysed during the manoeuvre • Mode of ventilation: Pressure-controlled ventilation • Driving pressure (inspiratory pressure) : 15 cm H 2 O (up to 20 cm H 2 O) • Respiratory rate 10 – 12/min; I: E ratio 1: 1; Fi. O 2 1. 0 • Begin with PEEP 15 cm H 2 O. Ventilate for 5 - 6 breaths. Increase PEEP by 5 cm H 2 O to 20 cm H 2 O. Ventilate for 5 - 6 breaths. Reduce PEEP back to 15 cm H 2 O ventilate for 5 - 6 breaths. increasing PEEP of 5 cm H 2 O (PEEP 25 cm H 2 O, then 30 cm H 2 O and 35 cm H 2 O) each time till a peak pressure of 50 cm H 2 O is achieved.

• ABG • Ventilatory settings • PCV driving pressure 15 cm H 2 O, RR 10/min, I: E 1: 2, PEEP 20 cm H 2 O and Fi. O 2 1. 0. • GOAL • Pa. O 2 + Pa. CO 2 > 400 mm. Hg suggests that there is less than 5% of the alveoli which are still collapsed.

Don’t achieve the goal • repeat the recruitment manoeuvre to a higher peak pressure (driving pressure + PEEP) e. g. 55 cm H 2 O or 60 cm H 2 O or even higher while maintaining the driving pressure at 15 cm H 20. • If still unsuccessful, consider repeating the recruitment • manoeuvre in the prone position.

Terminate manoeuvre o Sp. O 2 <85% o MAP < 60 o HR < 60 or > increase more than 20% from baseline o New arrhythmia

PCV with PEEP method Sedate and paralyse the patient • Pressure control ventilation • Fi. O 2 1. 0 • I: E 1: 1 • Rate 8 -10/min • PEEP 20 cm H 2 O The peak inspiratory pressure is slowly increased to 50 - 55 cm H 2 O. • Ventilation at this peak pressure for about 2 minutes, after which the peak inspiratory pressure is lowered to 30 -35 cm H 2 O.

Optimal PEEP • Search for the optimal PEEP follows a recruitment manoeuvre using the decremental PEEP technique. • Set PEEP at 20 cm. H 2 O and reduce the PEEP gradually (1 cm H 2 O every 5 min). • A new recruitment manoeuvre is performed at the optimal PEEP level. • In extra-pulmonary ARDS, the optimal PEEP level is usually 16 - 18 cm. H 2 O in the initial phase and 8 -12 cm H 2 O is enough after 1 -2 days.

Determine closing pressure A. Best oxygenation method • Perform ABG at each PEEP level. • The PEEP level at which there is a reduction in Pa. O 2 of more than 10% from the previous indicates the collapse pressure. • The optimal PEEP is set at 2 cm H 2 O above the collapse pressure. B. Best compliance method • Determine the compliance at each PEEP level. • The PEEP level at which there is a marked decrease in compliance from the previous indicates the collapse pressure. • The optimal PEEP is set at 2 cm H 2 O above the collapse pressure.

Low tidal volume ventilation • Calculate Ideal body weight(IBW ) Male = 50 + 0. 91 [height (cm) - 152. 4] Female = 45. 5 + 0. 91 [height (cm) - 152. 4] • Mode: Pressure-controlled ventilation (preferred) or volume controlled ventilation. • Aim for tidal volume of 6 ml/kg IBW while not exceeding plateau pressure of 30 cm H 2 O. • In PCV, plateau airway pressure is equivalent to peak airway pressure

• volume-controlled ventilation • the plateau pressure needs to be measured regularly e. g. 2 – 4 hourly. • If Pplat > 30 cm H 2 O, decrease tidal volume by 1 ml/kg steps to 5 ml/kg or if necessary to 4 ml/kg IBW. • breath stacking or severe dyspnoea occurs, • Tidal volume may be increased (not required) to 7 or 8 ml/kg IBW if Pplat remains ≤ 30 cm H 2 O. Oxygenation: Pa. O 2( 55 – 80) mm. Hg or Sp. O 2 88 -95% • Arterial p. H: aim > 7. 1 The respiratory rate may be increased to a maximum of 35 /min

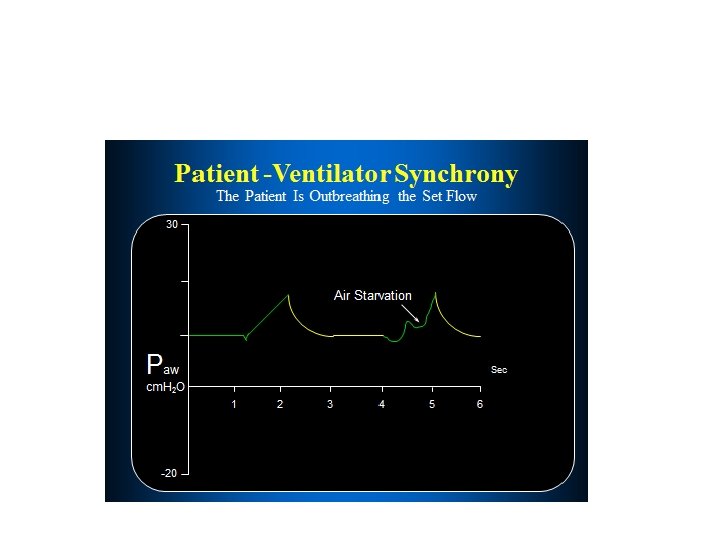
• Spontaneous Breathing • spontaneous breathing in patients with severe ARDS may induce patient–ventilator asynchrony and rapid–shallow breathing, further potentiating atelectrauma.

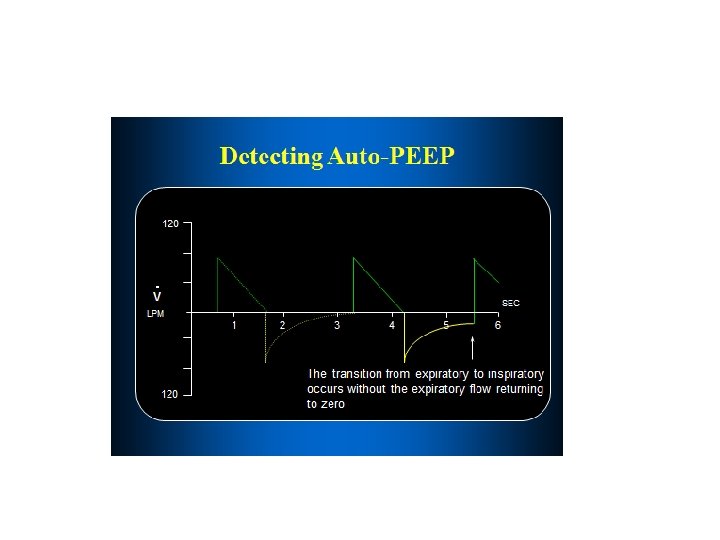
TARGETING ATELECTRAUMA AND LUNG RECRUITMENT • PEEP • has been used to prevent end-expiratory collapse of unstable lung units, resulting in cyclic (tidal) recruitment– derecruitment • overall findings were relatively consistent, with non significant reductions in short-term mortality with higher PEEP. • HIGH-FREQUENCY OSCILLATORY VENTILATION • Tidal over distension is avoided through the delivery of very small tidal volumes (1– 4 m. L/kg PBW), • A systematic review and meta-analysis including 6 trials (365 patients) revealed a significant reduction in shortterm mortality in favor of HFOV. • improved oxygenation (Pa. O 2/Fi. O 2 ratio)

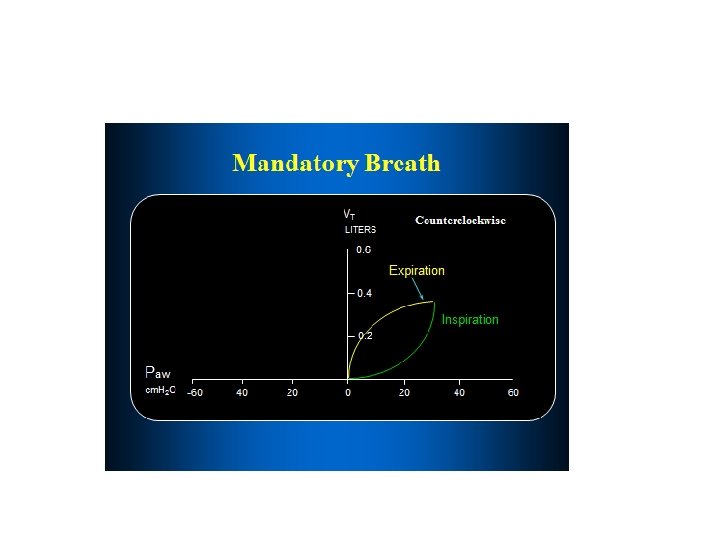
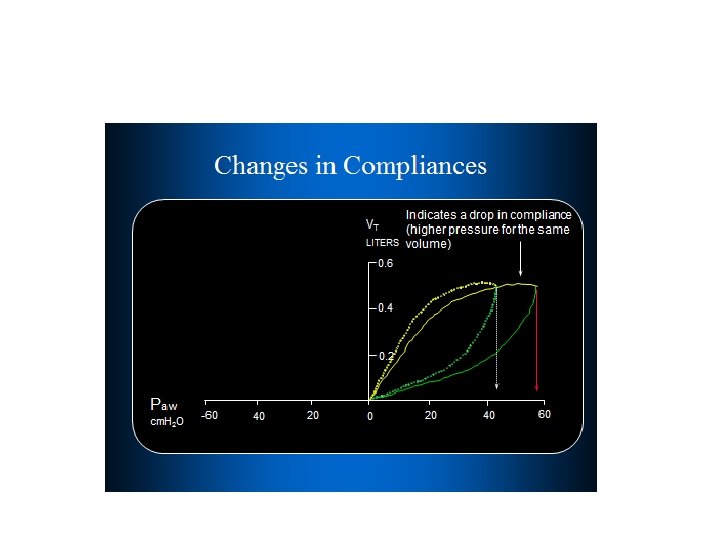
MONITORING AT THE BEDSIDE • Lung Mechanics • the pressure–volume (PV) curve, may help guide PEEP titration. • In ARDS , The PV curve exhibits sigmoid inspiratory and expiratory limbs separated by hysteresis. • On the inspiratory limb, the lower inflection point is felt to represent opening pressure. • The upper inflection point is felt to represent airway pressures at which over distension occurs. • Logically, it follows that first strategy would be to set PEEP above the lower inflection point • deep sedation and often paralysis

• Esophageal Pressure Monitoring and Trans pulmonary Pressure • Trans pulmonary pressures (PL). • the pressure at the airway opening (Paw) minus the pleural pressure (Ppl), esophageal pressure (Pes) monitoring has been used as a surrogate for the more difficult to measure pleural pressure • PEEP was set to maintain a positive trans pulmonary pressure of 0 to 10 cm H 2 O at end expiration. Although tidal volumes were limited to keep PL at less than 25 H 2 O at end inspiration, the initial setting of 6 m. L/kg PBW never had to be reduced for this rule.

Imaging—Computed Tomography and Electrical Impedance Tomography • CT has greatly improved the understanding of the morphologic features and distribution of parenchymal changes in ARDS. • comparison of tissue densities representing derecruited dense lung zones, recruited aerated lung zones, and hypodense areas of hyperinflation • under different airway pressures, CT has now shown promise in determining the potential for lung recruitment and titration of PEEP

NEUROMUSCULAR BLOCKING AGENTS • ACQUIRED WEAKNESS • The incidence of ICU-acquired weakness (ICUAW) was 34% to 60% in patients with ARDS. • ICUAW is responsible for severe and durable morbidity and, since it was first described in the early 1980 s, has become a major concern. • Independent risk factors such as female sex, multiple organ dysfunctions, duration of MV, and administration of corticosteroids , Duration of vasopressor support , duration of ICU stay, hyperglycemia, low serum albumin, and neurologic failure
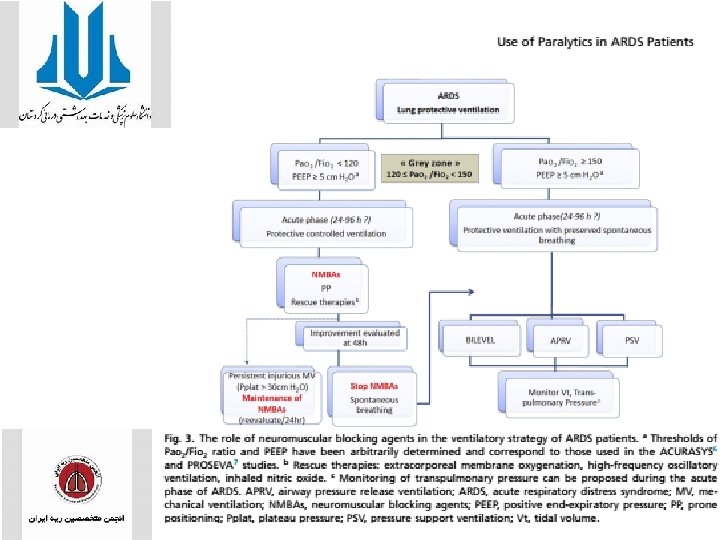


Steroids • Glucocorticoids are potent antiinflammatories that act primarily by binding to cytoplasmic glucocorticoid receptors. • Once bound, the glucocorticoid-receptor complexes regulate the transcription of glucocorticoid-response elements such as nuclear factor receptor-kb (NF-kb). • The transcription of many proinflammatory cytokines (including interleukins 1 a, 1 b, 2, 3, 5, 6, 8, and 12; tumor necrosis factor alpha; and interferon gamma) is modulated by NF-kb. • A large multicenter study was conducted simultaneously by Bone and colleagues testing the efficacy of methylprednisolone on reducing mortality in patients with septic shock. • This study found no benefit of high-dose steroids on mortality, and a subgroup analysis found an association between randomization to steroids and increased mortality

Prone Positioning • Computed tomography (CT) imaging and autopsy studies indicate that atelectasis preferentially develops and is more extensive in the dorsal caudal portions of the lung when normal individuals and patients with ALI/ARDS are supine. To the extent that these regions receive perfusion (Q) low ventilation-to-perfusion areas or shunt result • Most human clinical trials have reported that prone ventilation improves oxygenation in most (but not all) patients with ARDS (ie, 60%– 70%). • These benefits can, on occasion, be maintained after returning the patient to the supine position after a few hours but are more frequently lost on turning supine, requiring that prone ventilation be continued longer. The first 4 randomized trials found that, despite improvements in oxygenation, prone ventilation had no effect on mortality

obesity
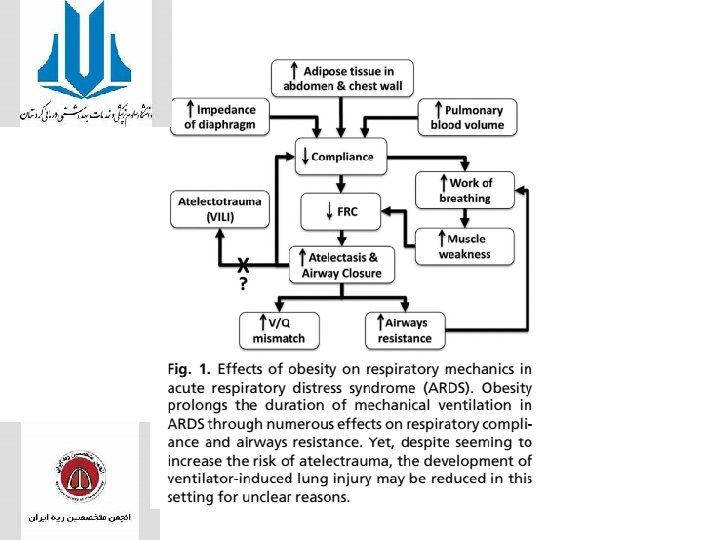
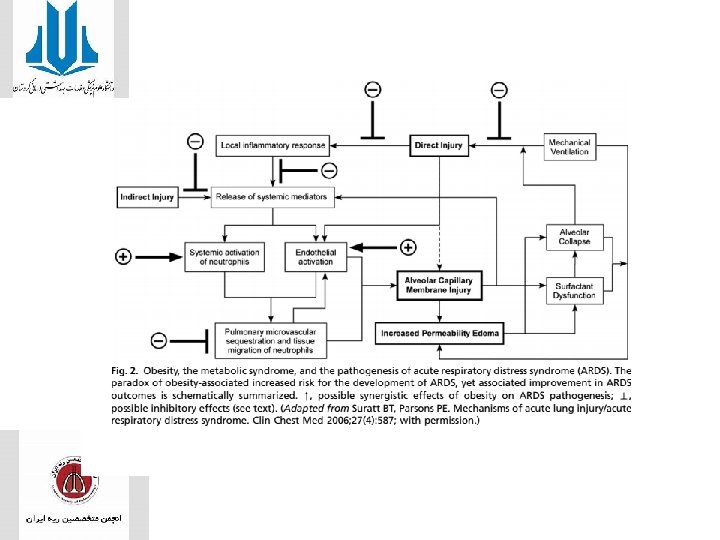


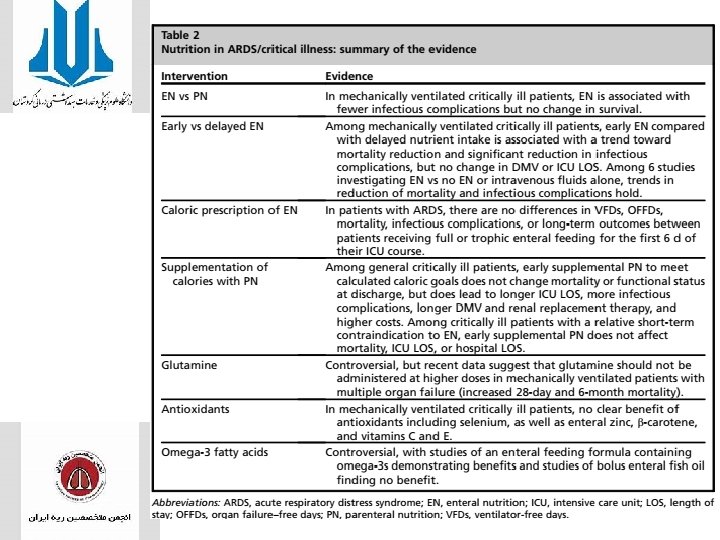
• Malnutrition is coupled with impairment of immune function and increased morbidity and mortality in critically ill patients. • Roughly 25 kcal/kg IBW is frequently the standard practice, and other equations such as Harris-Benedict, Ireton-Jones, and Weir are commonly used • Aggregated data suggest that patients with ARDS should preferentially receive EN, and this should be started early (within 48 hours of ICU admission) to preserve GI lumen integrity and prevent infectious complications.

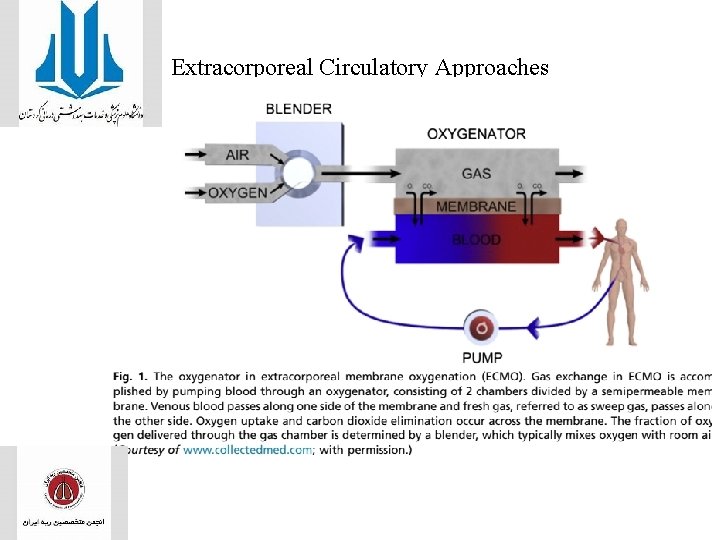
Extracorporeal Circulatory Approaches
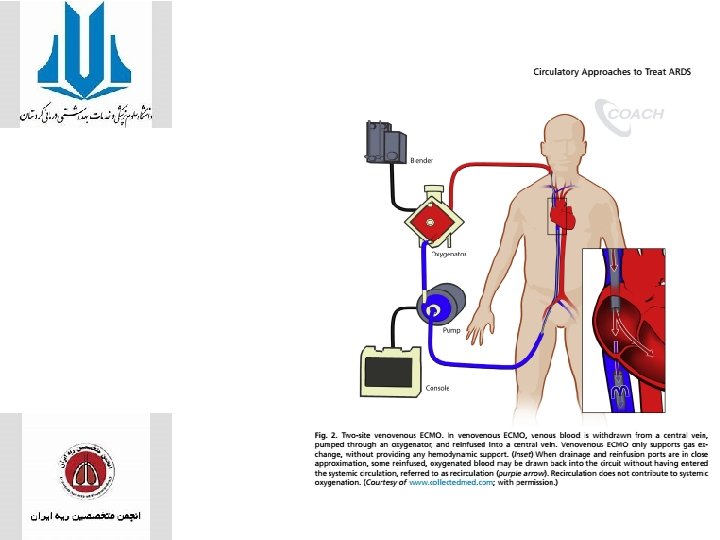



- Slides: 67