Acute postoperative pain management Phakapan Buppha Department of

Acute postoperative pain management Phakapan Buppha Department of Anesthesiology, SWU

Reference Pamela E Macintyre, Stephan A. Schug. Acute pain management : A practical guide. 4 th edition.

Definition of pain � “An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage” Merskey & Bogduk, 1994
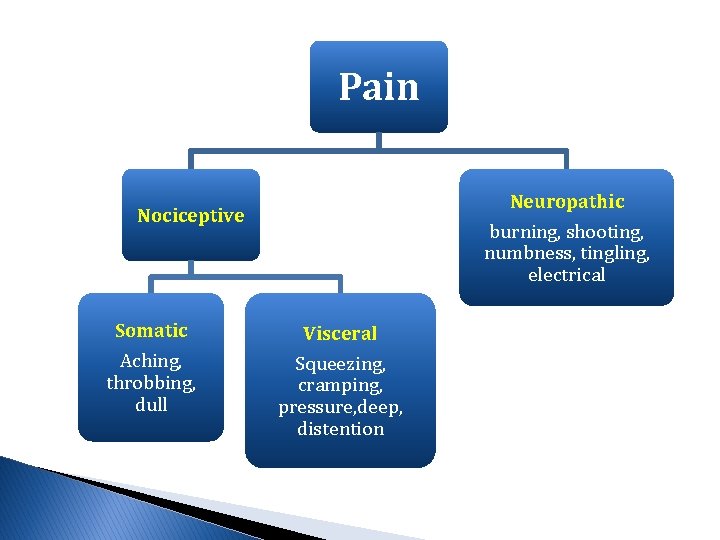
Pain Neuropathic Nociceptive Somatic Aching, throbbing, dull burning, shooting, numbness, tingling, electrical Visceral Squeezing, cramping, pressure, deep, distention
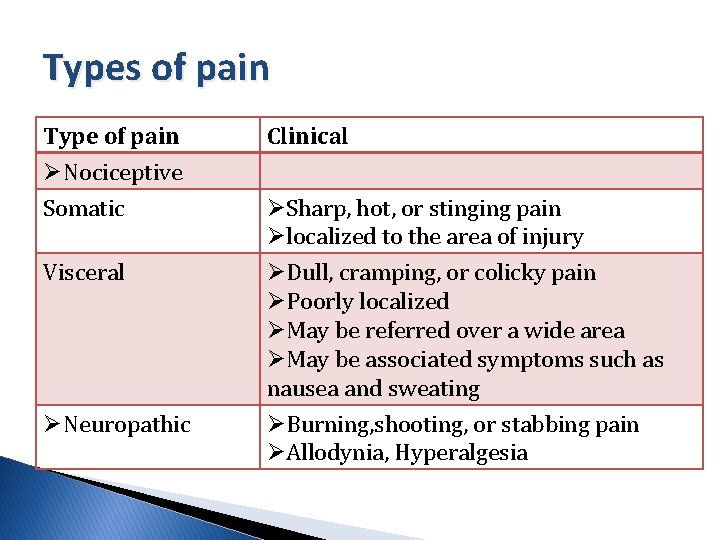
Types of pain Type of pain ØNociceptive Somatic Visceral ØNeuropathic Clinical ØSharp, hot, or stinging pain Ølocalized to the area of injury ØDull, cramping, or colicky pain ØPoorly localized ØMay be referred over a wide area ØMay be associated symptoms such as nausea and sweating ØBurning, shooting, or stabbing pain ØAllodynia, Hyperalgesia
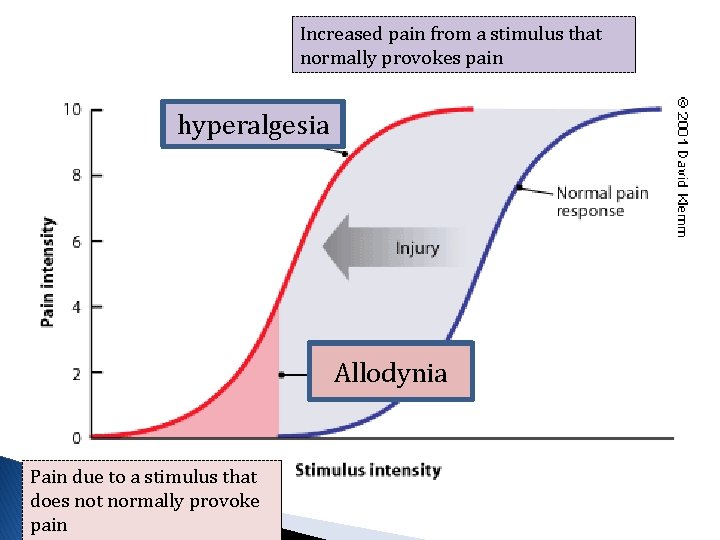
Increased pain from a stimulus that normally provokes pain hyperalgesia Allodynia Pain due to a stimulus that does not normally provoke pain
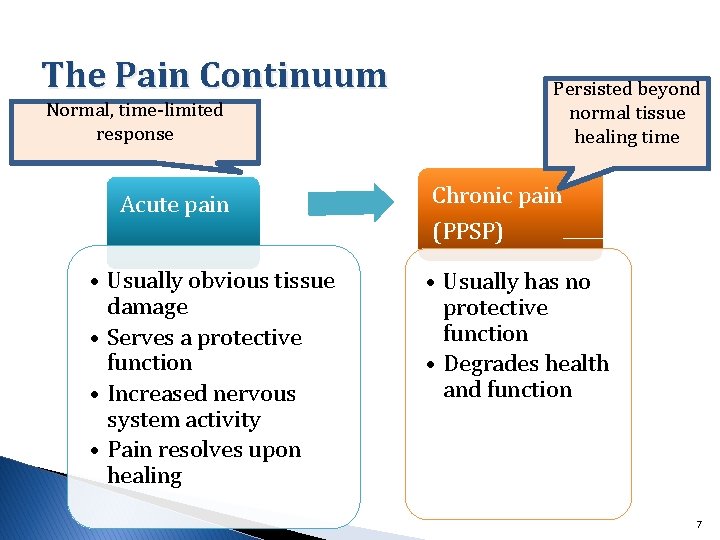
The Pain Continuum Normal, time-limited response Acute pain • Usually obvious tissue damage • Serves a protective function • Increased nervous system activity • Pain resolves upon healing Persisted beyond normal tissue healing time Chronic pain (PPSP) Chronic pain • Usually has no protective function • Degrades health and function 7
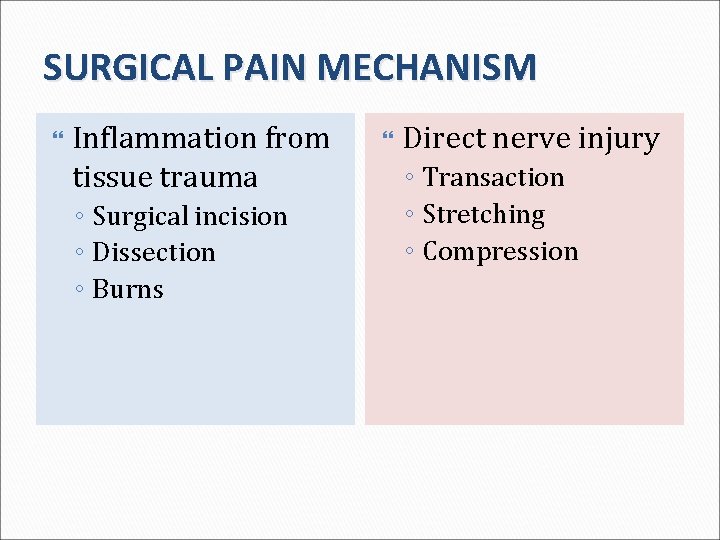
SURGICAL PAIN MECHANISM Inflammation from tissue trauma ◦ Surgical incision ◦ Dissection ◦ Burns Direct nerve injury ◦ Transaction ◦ Stretching ◦ Compression
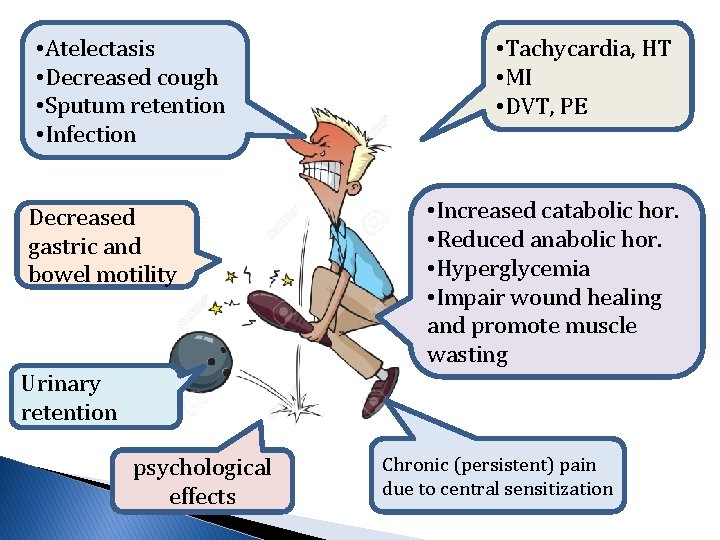
• Atelectasis • Decreased cough • Sputum retention • Infection Decreased gastric and bowel motility • Tachycardia, HT • MI • DVT, PE • Increased catabolic hor. • Reduced anabolic hor. • Hyperglycemia • Impair wound healing and promote muscle wasting Urinary retention psychological effects Chronic (persistent) pain due to central sensitization
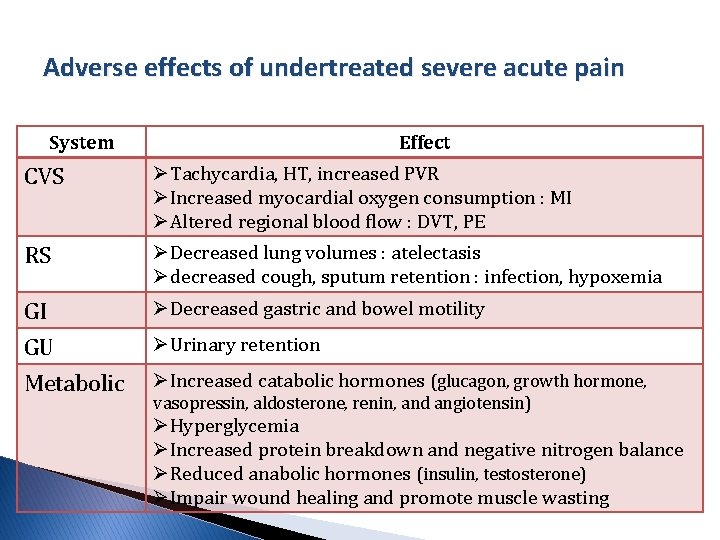
Adverse effects of undertreated severe acute pain System Effect CVS ØTachycardia, HT, increased PVR ØIncreased myocardial oxygen consumption : MI ØAltered regional blood flow : DVT, PE RS ØDecreased lung volumes : atelectasis Ødecreased cough, sputum retention : infection, hypoxemia GI GU Metabolic ØDecreased gastric and bowel motility ØUrinary retention ØIncreased catabolic hormones (glucagon, growth hormone, vasopressin, aldosterone, renin, and angiotensin) ØHyperglycemia ØIncreased protein breakdown and negative nitrogen balance ØReduced anabolic hormones (insulin, testosterone) ØImpair wound healing and promote muscle wasting

Acute pain management and patient outcomes System Effect Musculoskeletal ØMuscle spasm, immobility (risk of DVT) ØMuscle wasting (prolonged recovery of function) Central nervous ØChronic (persistent) pain due to central system sensitization Psychological ØAnxiety, fear ØSleep deprivation (increased pain and potential long-term psychological effects)
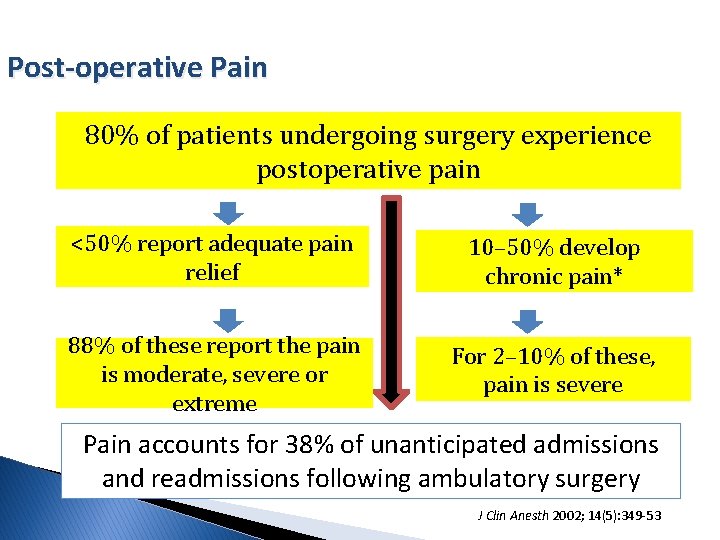
Post-operative Pain 80% of patients undergoing surgery experience postoperative pain <50% report adequate pain relief 10– 50% develop chronic pain* 88% of these report the pain is moderate, severe or extreme For 2– 10% of these, pain is severe Pain accounts for 38% of unanticipated admissions and readmissions following ambulatory surgery J Clin Anesth 2002; 14(5): 349 -53

Aim of treatment Humanitarian (Relieves suffering, Increased patient satisfaction) Better outcome ◦ Short term Decrease risk of postop. pulmonary and cardiac complication Early return of bowel function “early recovery after surgery” (ERAS) protocols ◦ Long term Persistent postsurgical pain

Pain pathway

Pain pathway

Pain pathway
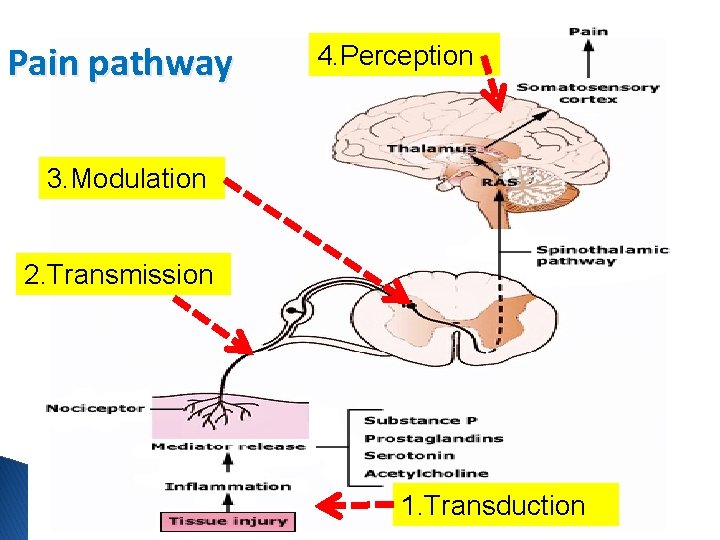
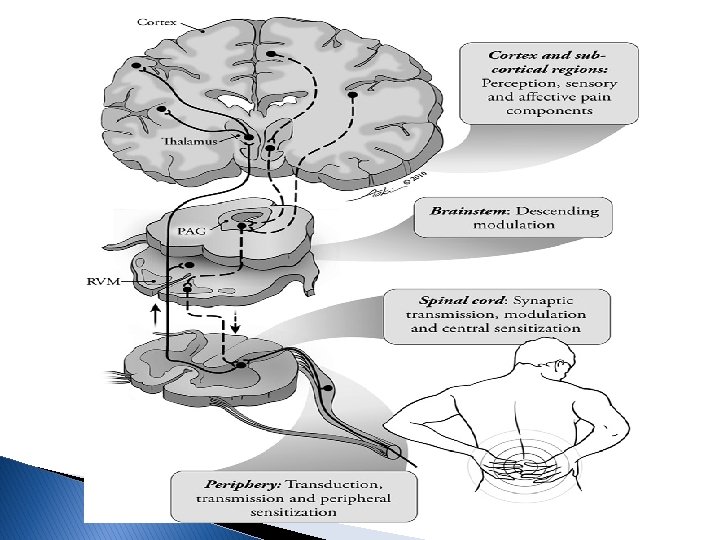
Pain pathway 4. Perception 3. Modulation 2. Transmission 1. Transduction
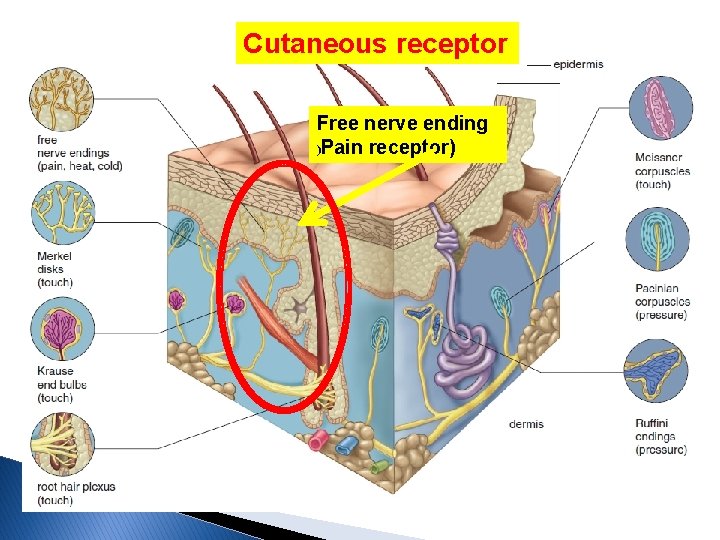
Cutaneous receptor Free nerve ending )Pain receptor)
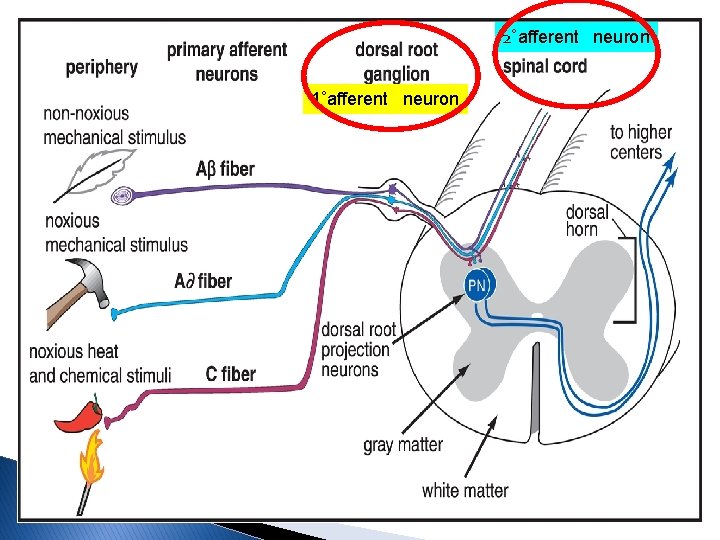
2˚afferent neuron 1˚afferent neuron
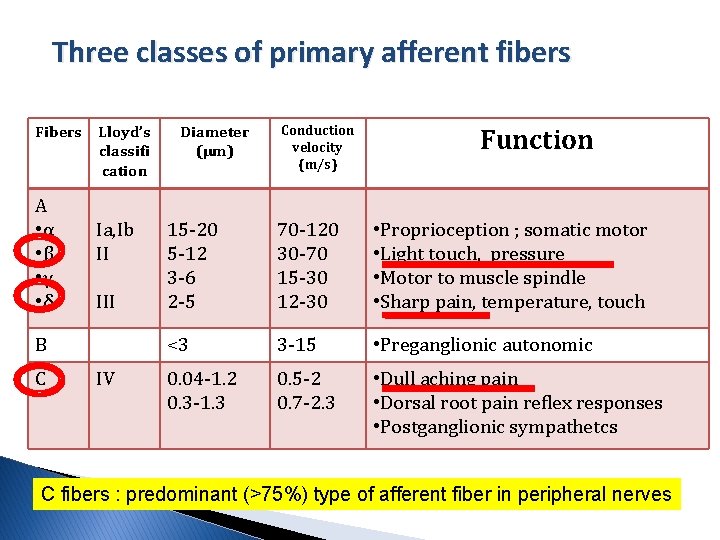
Three classes of primary afferent fibers Fibers A • α • β • γ • δ Lloyd’s classifi cation Ia, Ib II III B C IV Diameter (µm) Conduction velocity (m/s) Function 15 -20 5 -12 3 -6 2 -5 70 -120 30 -70 15 -30 12 -30 • Proprioception ; somatic motor • Light touch, pressure • Motor to muscle spindle • Sharp pain, temperature, touch <3 3 -15 • Preganglionic autonomic 0. 04 -1. 2 0. 3 -1. 3 0. 5 -2 0. 7 -2. 3 • Dull aching pain • Dorsal root pain reflex responses • Postganglionic sympathetcs C fibers : predominant (>75%) type of afferent fiber in peripheral nerves
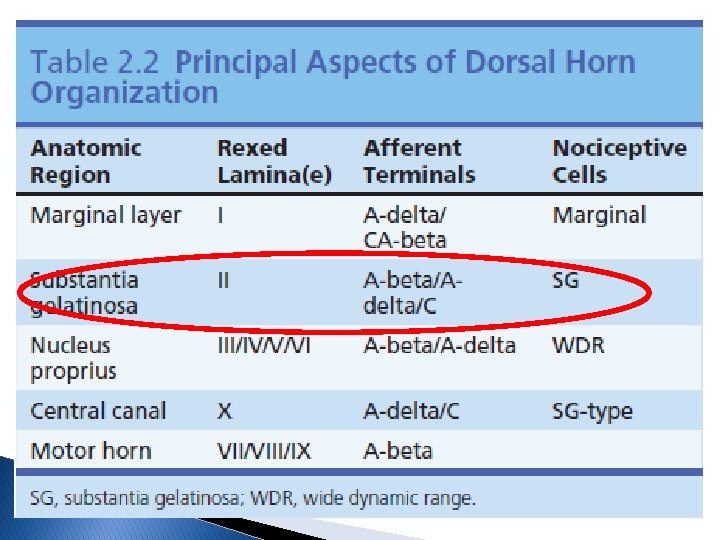
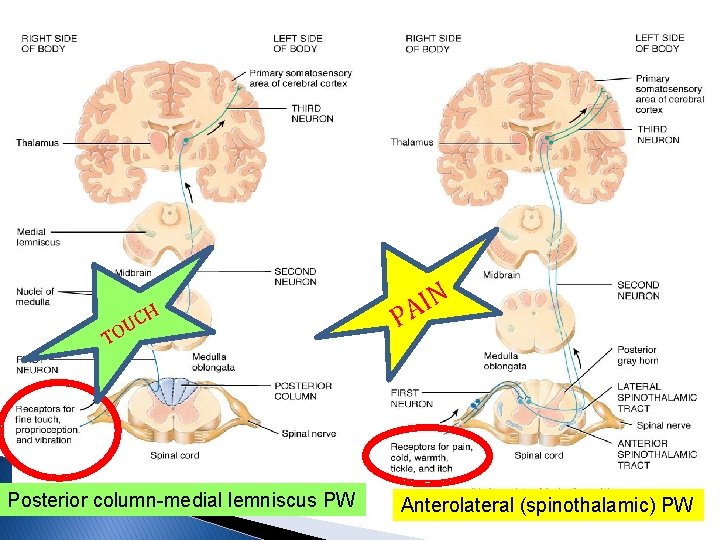
H UC O T Posterior column-medial lemniscus PW IN A P Anterolateral (spinothalamic) PW
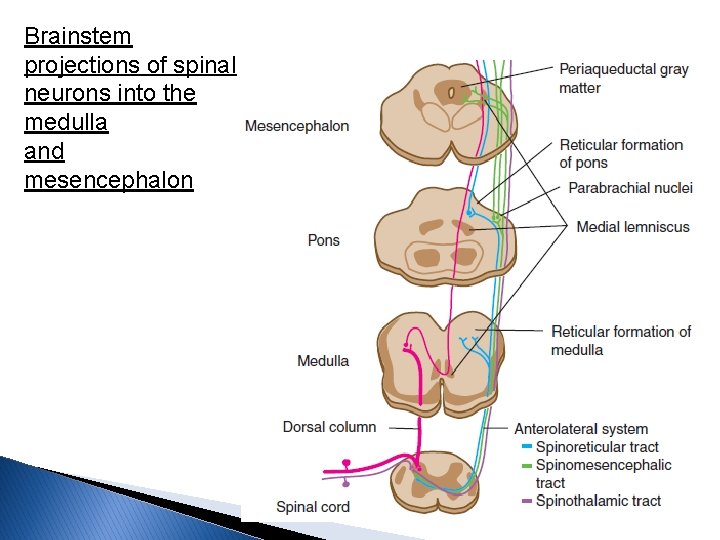
Brainstem projections of spinal neurons into the medulla and mesencephalon
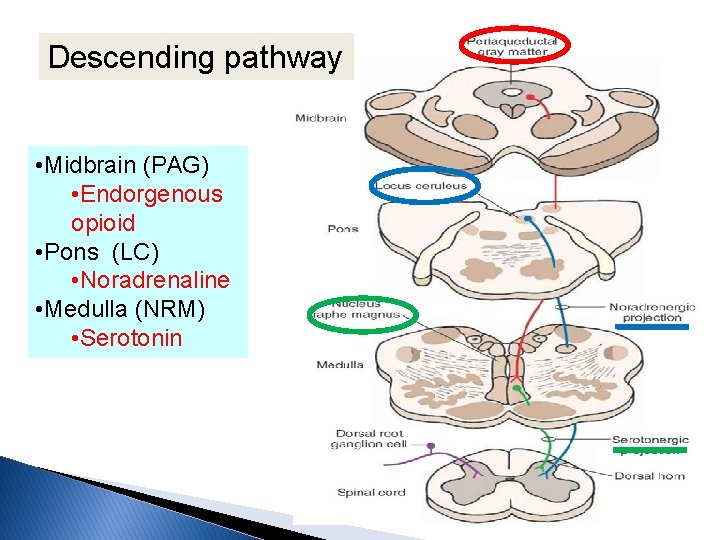
Descending pathway • Midbrain (PAG) • Endorgenous opioid • Pons (LC) • Noradrenaline • Medulla (NRM) • Serotonin
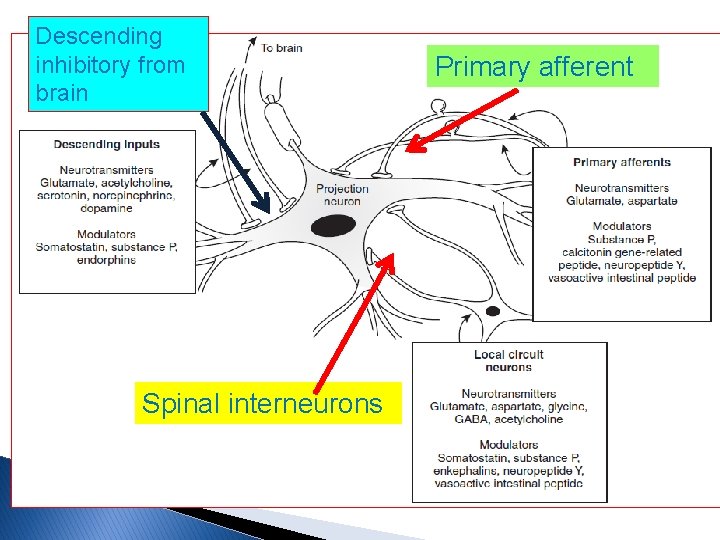
Descending inhibitory from brain Primary afferent . Spinal interneurons
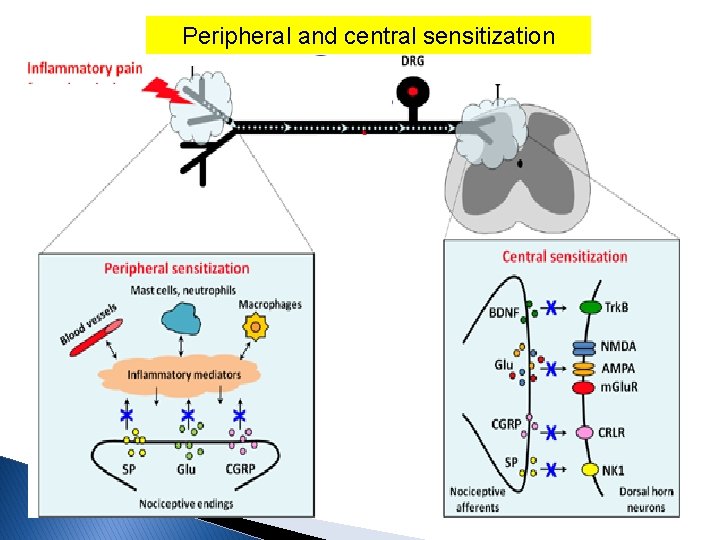
Peripheral and central sensitization
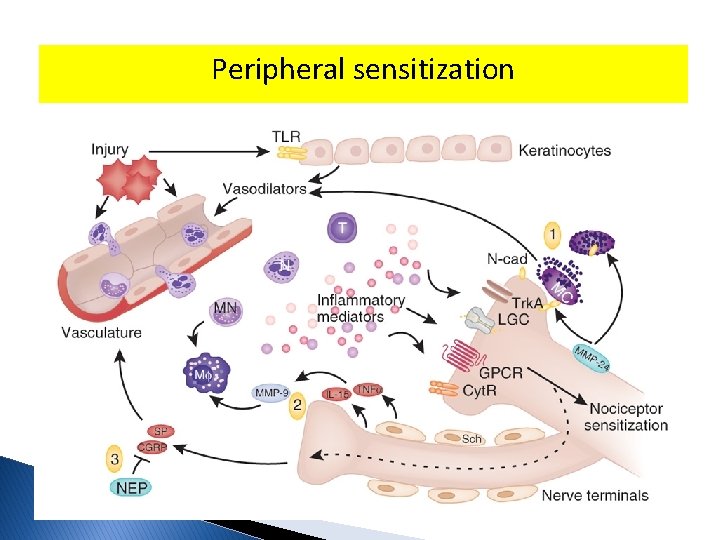
Peripheral sensitization
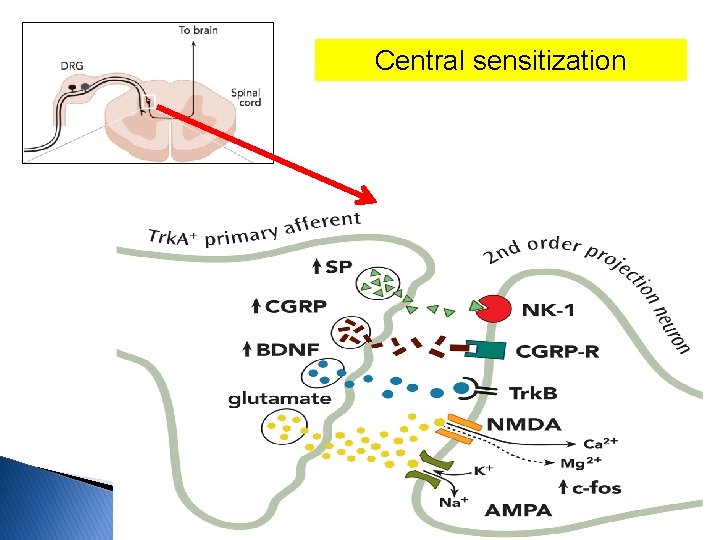
Central sensitization

AMPA and NMDA receptor

Assessment and monitoring
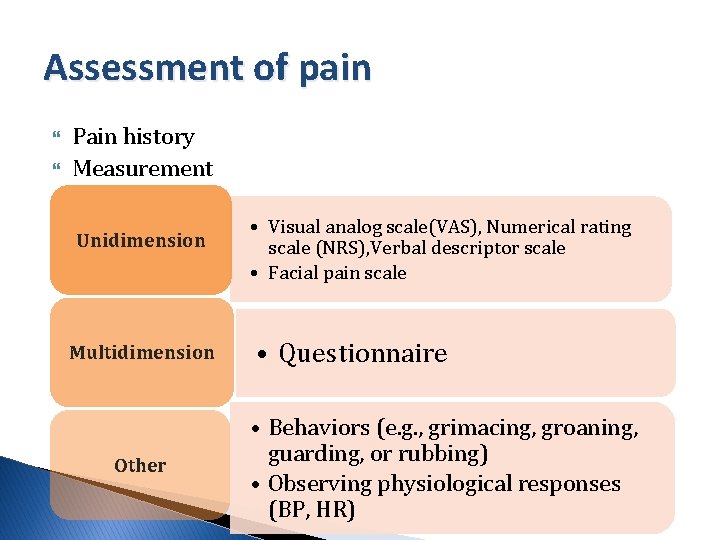
Assessment of pain Pain history Measurement Unidimension Multidimension Other • Visual analog scale(VAS), Numerical rating scale (NRS), Verbal descriptor scale • Facial pain scale • Questionnaire • Behaviors (e. g. , grimacing, groaning, guarding, or rubbing) • Observing physiological responses (BP, HR)
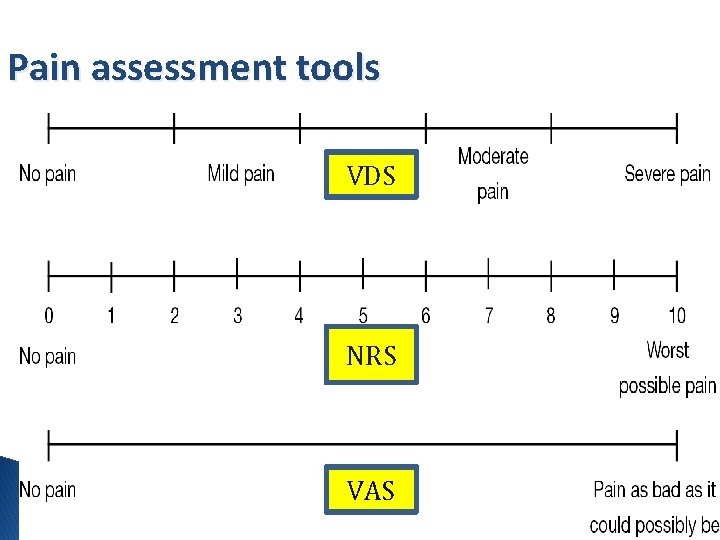
Pain assessment tools VDS NRS VAS
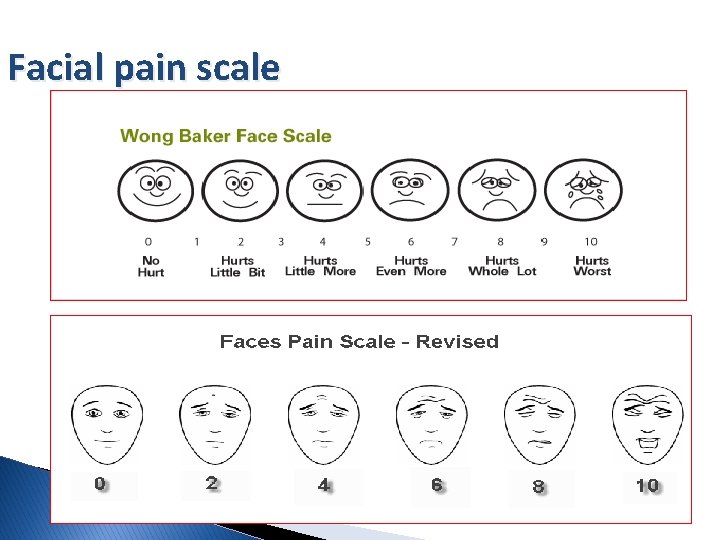
Facial pain scale

Oucher scale

Children’s Revised Impact of Event Scale (CRIES)

Face Legs Activity Cry and Consolability (FLACC) Young children

Treatment

Treatment Opioids Non-opioids ◦ Acetaminophen ◦ NSAIDs Adjuvant ◦ Gabapentin and pregabalin ◦ Ketamine ◦ Alpha 2 agonists (clonidine, dexmedetomidine) Neuraxial block ◦ Spinal block ◦ Epidural block Peripheral nerve blocks local anesthetic wound infiltration Continuous wound infusion techniques
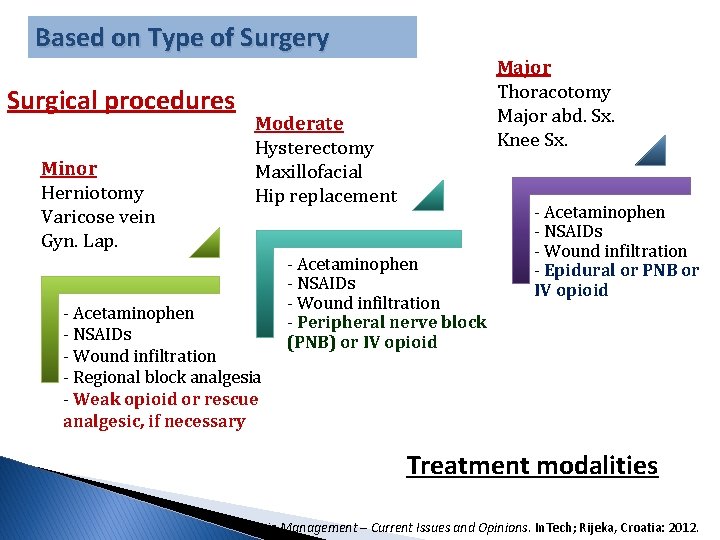
Based on Type of Surgery Surgical procedures Minor Herniotomy Varicose vein Gyn. Lap. Major Thoracotomy Major abd. Sx. Knee Sx. Moderate Hysterectomy Maxillofacial Hip replacement - Acetaminophen - NSAIDs - Wound infiltration - Regional block analgesia - Weak opioid or rescue analgesic, if necessary - Acetaminophen - NSAIDs - Wound infiltration - Peripheral nerve block (PNB) or IV opioid - Acetaminophen - NSAIDs - Wound infiltration - Epidural or PNB or IV opioid Treatment modalities Pain Management – Current Issues and Opinions. In. Tech; Rijeka, Croatia: 2012.
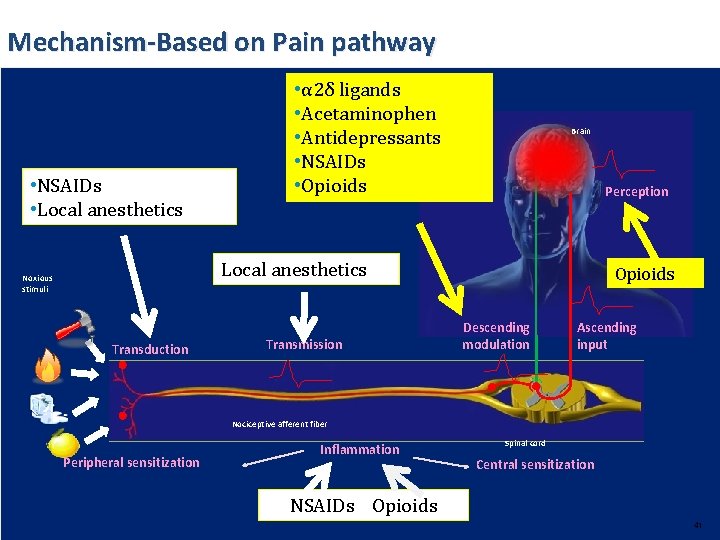
Mechanism-Based on Pain pathway • NSAIDs • Local anesthetics • α 2δ ligands • Acetaminophen • Antidepressants • NSAIDs • Opioids Brain Perception Local anesthetics Noxious stimuli Transduction Transmission Opioids Descending modulation Ascending input Nociceptive afferent fiber Peripheral sensitization Inflammation Spinal cord Central sensitization NSAIDs Opioids 41

The WHO Ladder : 5 key principles By Mouth By the Clock By the Ladder For the Individual Attention to detail
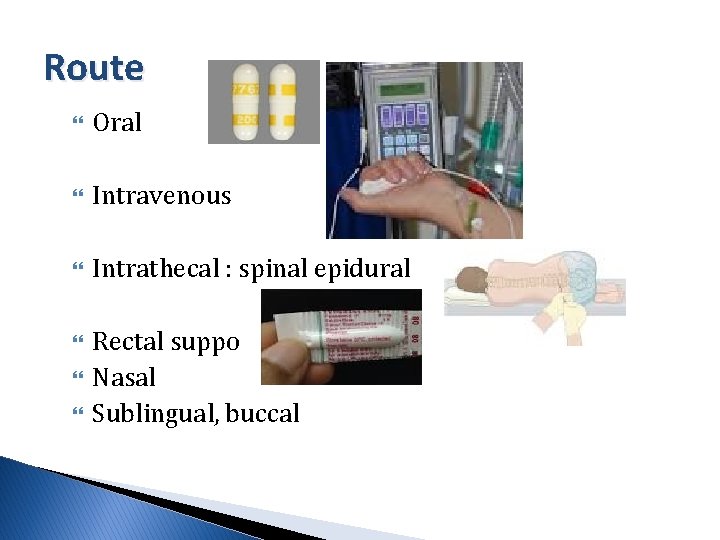
Route Oral Intravenous Intrathecal : spinal epidural Rectal suppo Nasal Sublingual, buccal
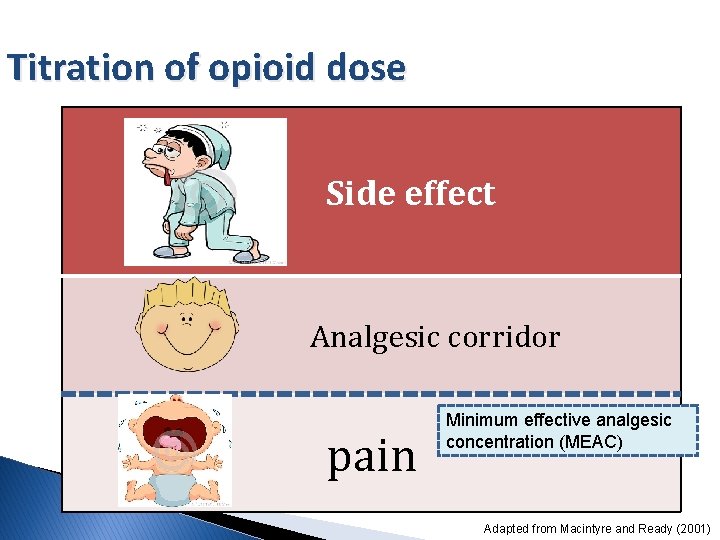
Titration of opioid dose Side effect Analgesic corridor pain Minimum effective analgesic concentration (MEAC) Adapted from Macintyre and Ready (2001)
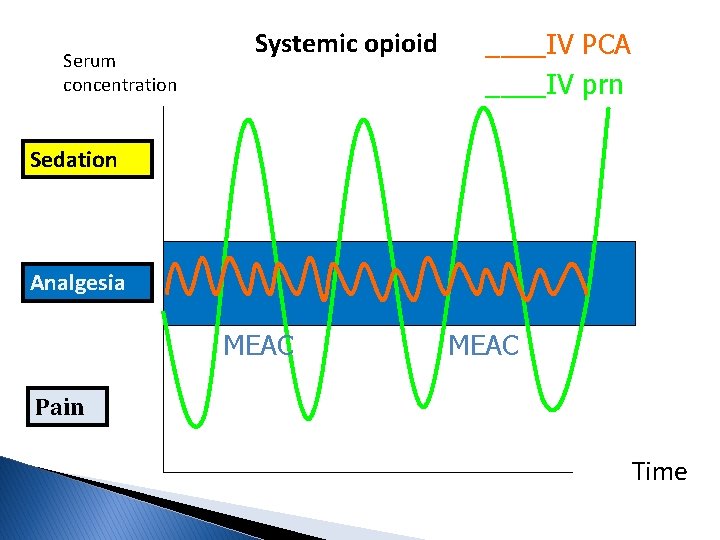
Serum concentration Systemic opioid ____IV PCA ____IV prn Sedation Analgesia MEAC Pain Time
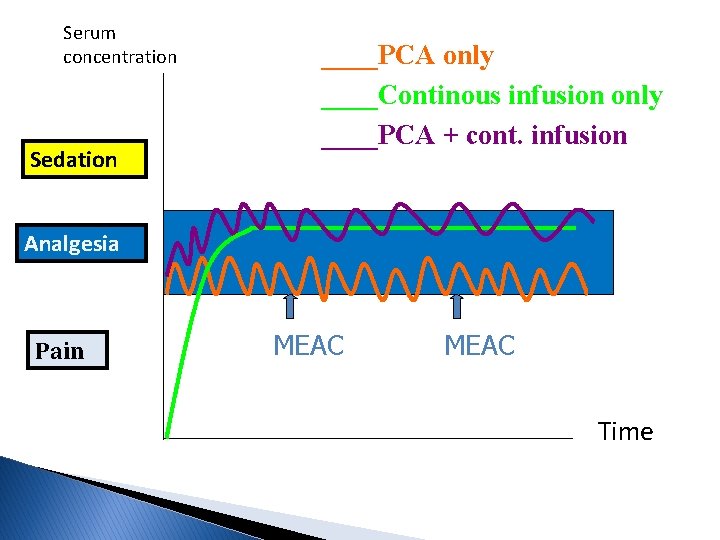
Serum concentration Sedation ____PCA only ____Continous infusion only ____PCA + cont. infusion Analgesia Pain MEAC Time
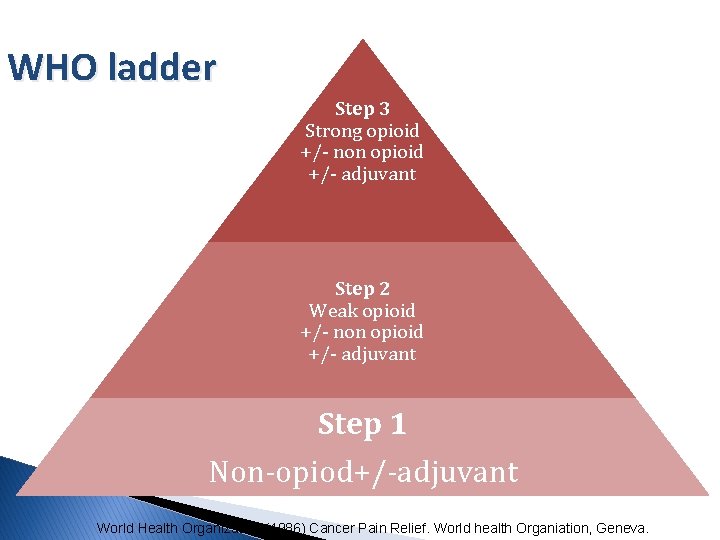
WHO ladder Step 3 Strong opioid +/- non opioid +/- adjuvant Step 2 Weak opioid +/- non opioid +/- adjuvant Step 1 Non-opiod+/-adjuvant World Health Organization (1986) Cancer Pain Relief. World health Organiation, Geneva.

Opioids

Opioids All endogenous and exogenous substances with morphine- like properties All compounds that work at the opioid receptors
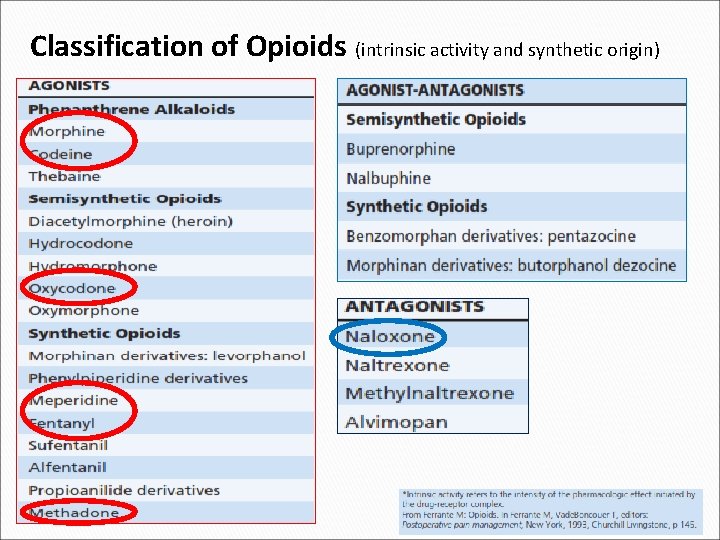
Classification of Opioids (intrinsic activity and synthetic origin)
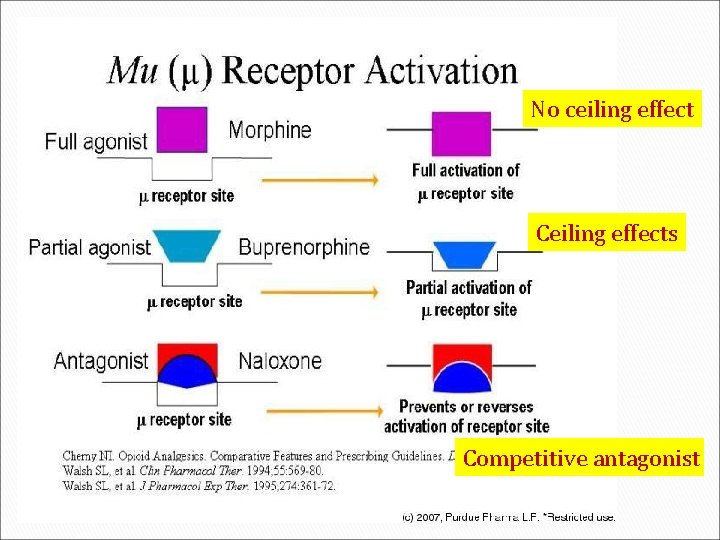
No ceiling effect Ceiling effects Competitive antagonist

World health organization classification of opioids Weak opioids Strong opioids ØCodeine ØMorphine ØTramadol ØFentanyl ØPethidine ØOxycodone ØHydromophone ØMethadone
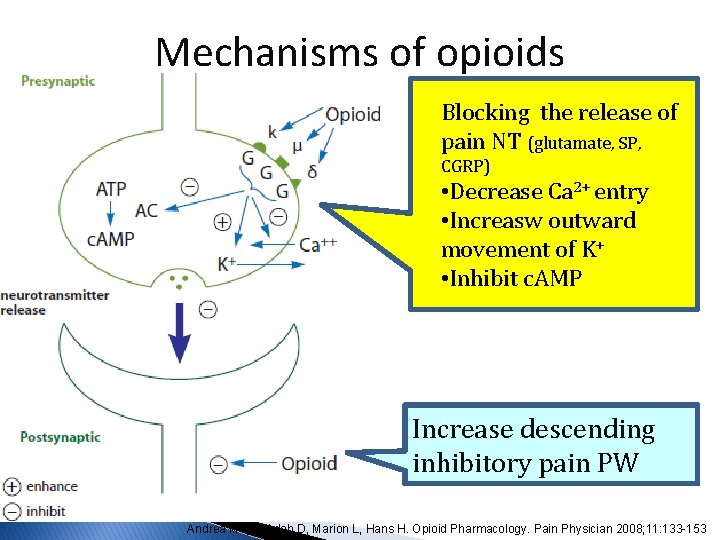
Mechanisms of opioids Blocking the release of pain NT (glutamate, SP, CGRP) • Decrease Ca 2+ entry • Increasw outward movement of K+ • Inhibit c. AMP Increase descending inhibitory pain PW Andrea MT, Sukdeb D, Marion L, Hans H. Opioid Pharmacology. Pain Physician 2008; 11: 133 -153
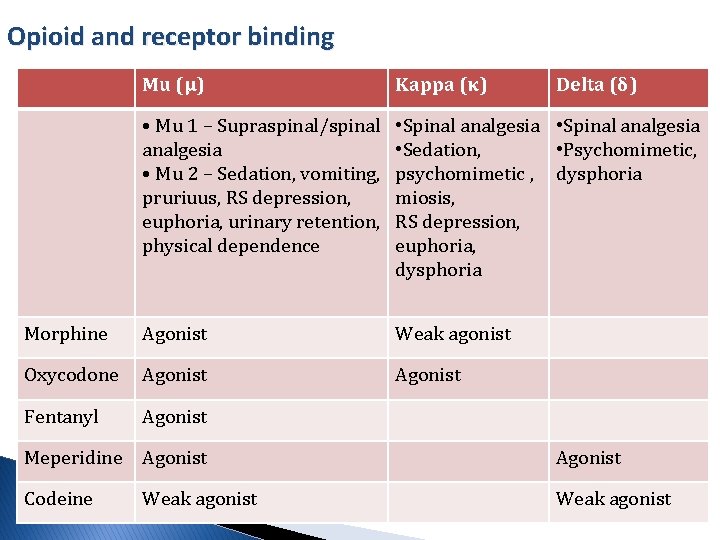
Opioid and receptor binding Mu (μ) Kappa (κ) Delta (δ) • Mu 1 – Supraspinal/spinal analgesia • Mu 2 – Sedation, vomiting, pruriuus, RS depression, euphoria, urinary retention, physical dependence • Spinal analgesia • Sedation, • Psychomimetic, psychomimetic , dysphoria miosis, RS depression, euphoria, dysphoria Morphine Agonist Weak agonist Oxycodone Agonist Fentanyl Agonist Meperidine Agonist Codeine Weak agonist

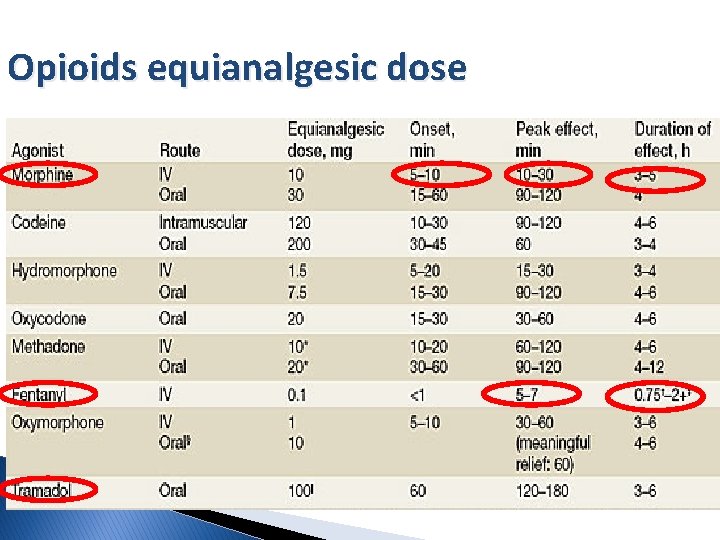
Opioids equianalgesic dose
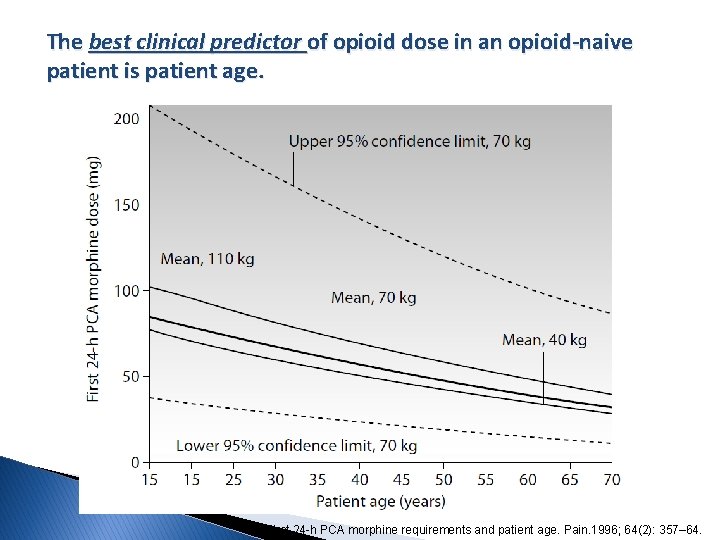
The best clinical predictor of opioid dose in an opioid-naive patient is patient age. First 24 -h PCA morphine requirements and patient age. Pain. 1996; 64(2): 357– 64.

Morphine ØMetabolism : liver ØM-3 -G : no analgesic property ØM-6 -G : more potent than morphine ØHistamine release

Fentanyl ØRapid onset & short duration ØInactive metabolite ØNo histamine release

Meperidine ØAtropine like effect ØTachycardia , dry mouth ØMetabolism liver ØNormeperidine CNS excitation ØShivering treatment ØInteraction with MAOI ØHyperpyrexia, convulsion , hypertension , coma

Tramadol Ø Multiple mechanism ØWeak µ-receptor agonist ØInhibit serotonin & NE reuptake Ø Dose : 50 -100 mg PO q 4 -6 hr. Ø Max. 400 mg/d (oral)
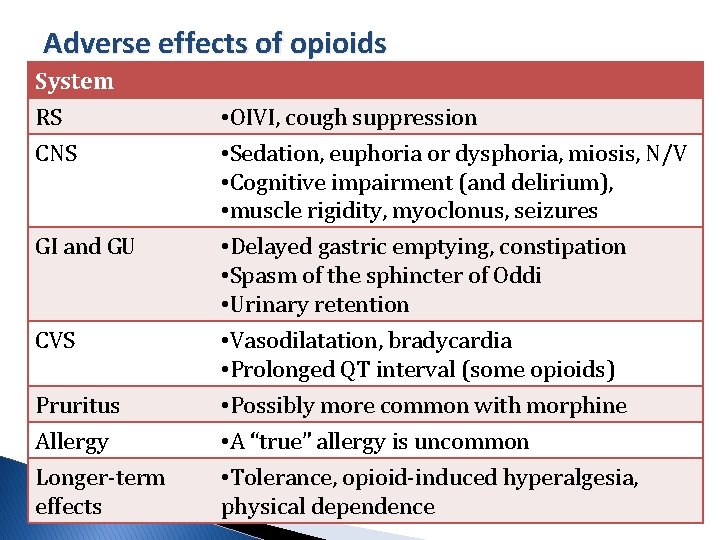
Adverse effects of opioids System RS CNS GI and GU CVS Pruritus Allergy Longer-term effects • OIVI, cough suppression • Sedation, euphoria or dysphoria, miosis, N/V • Cognitive impairment (and delirium), • muscle rigidity, myoclonus, seizures • Delayed gastric emptying, constipation • Spasm of the sphincter of Oddi • Urinary retention • Vasodilatation, bradycardia • Prolonged QT interval (some opioids) • Possibly more common with morphine • A “true” allergy is uncommon • Tolerance, opioid-induced hyperalgesia, physical dependence

Assessment of adverse effects Opioid-induced ventilatory impairment (OIVI) ◦ Sedation score and respiratory rate ◦ oxygen and carbon dioxide levels
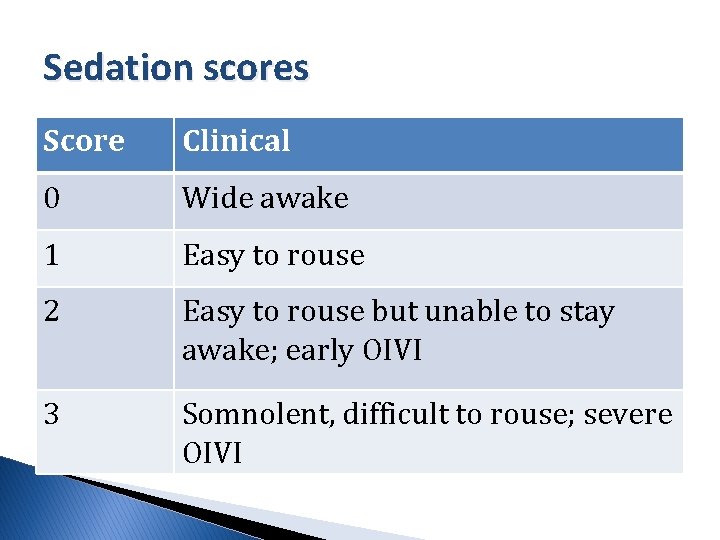
Sedation scores Score Clinical 0 Wide awake 1 Easy to rouse 2 Easy to rouse but unable to stay awake; early OIVI 3 Somnolent, difficult to rouse; severe OIVI
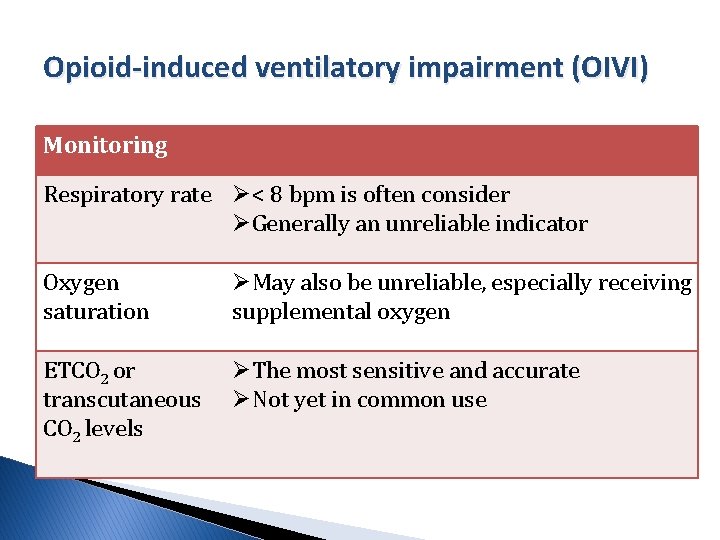
Opioid-induced ventilatory impairment (OIVI) Monitoring Respiratory rate Ø< 8 bpm is often consider ØGenerally an unreliable indicator Oxygen saturation ØMay also be unreliable, especially receiving supplemental oxygen ETCO 2 or transcutaneous CO 2 levels ØThe most sensitive and accurate ØNot yet in common use

Non opioids

Acetaminophen

Acetaminophen �Effective analgesic �Action ◦ Analgesic ◦ Antipyretic ◦ Anti-inflammatory agent �Relative safety �Effective for the musculoskeletal aches, joint stiffness

Acetaminophen �Disadvantage ◦ Dose-dependent hepatotoxicity, GI upset ◦ Agranulocytosis �Dosage ◦ 650 -1000 mg PO q 4 hr. ◦ Max. 4 g/d �Reduce dose 50 -70% in patient with significant hepatic impairment

Non steroidal antiinflammatory drugs (NSAID)
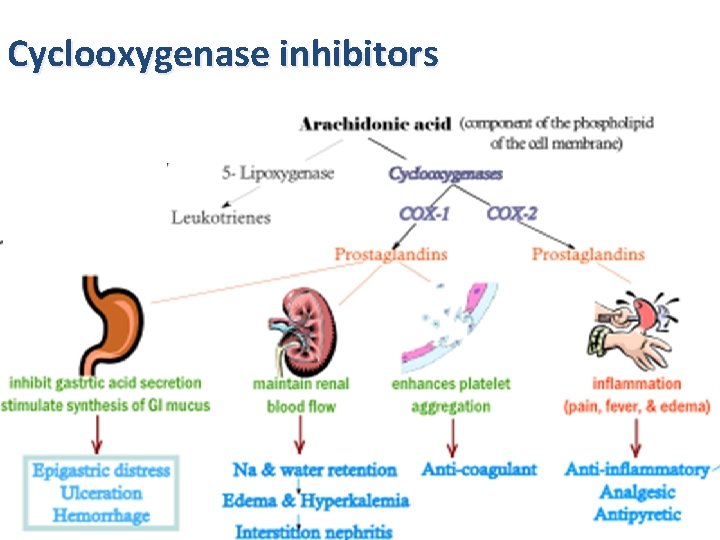
Cyclooxygenase inhibitors
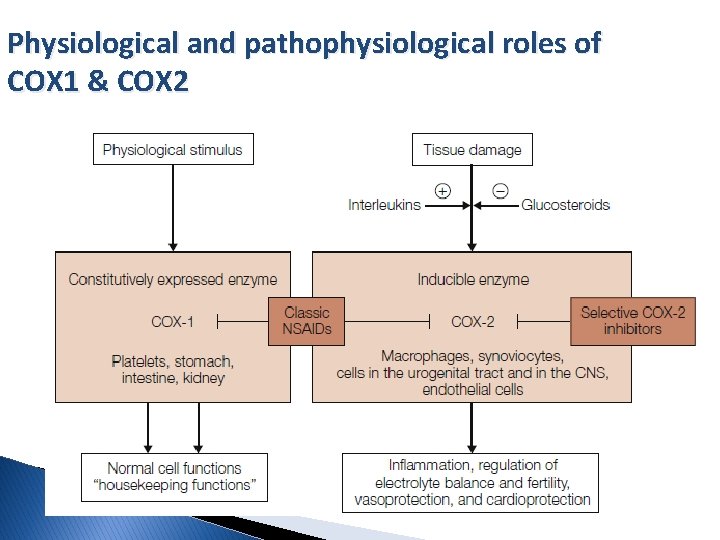
Physiological and pathophysiological roles of COX 1 & COX 2
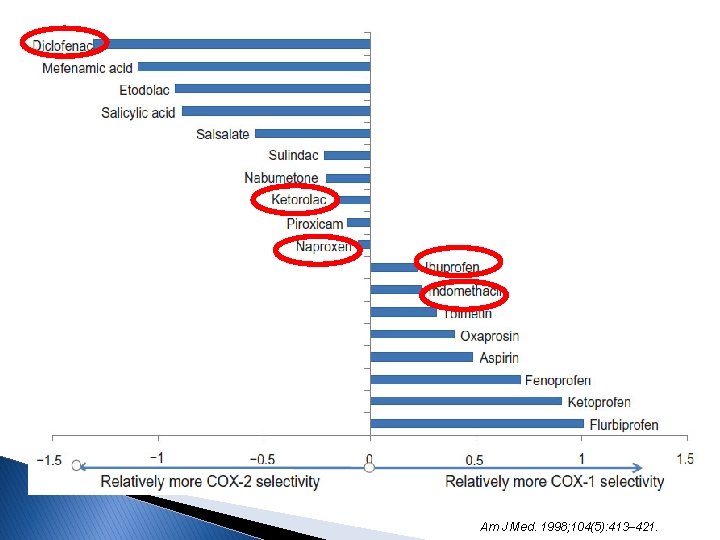
Am J Med. 1998; 104(5): 413– 421.
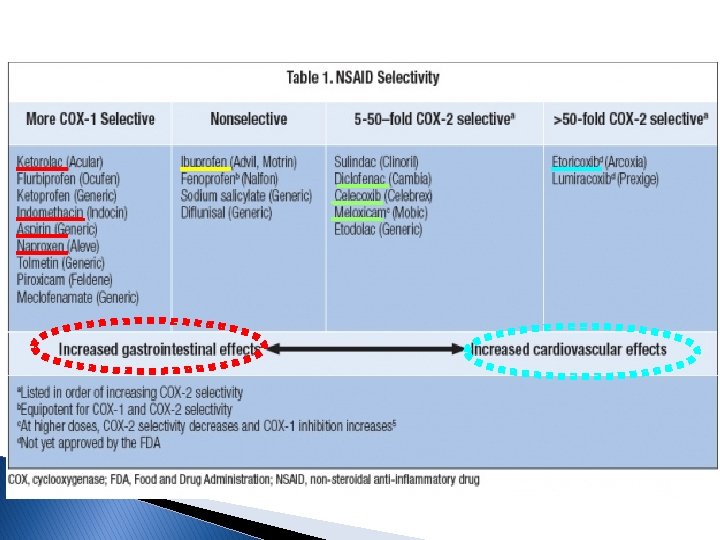
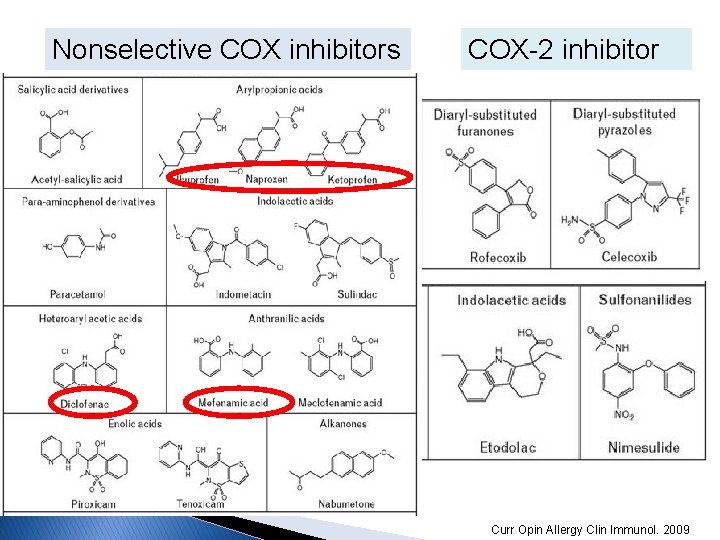
Nonselective COX inhibitors COX-2 inhibitor Curr Opin Allergy Clin Immunol. 2009

PREVENTION STRATEGIES in patients with GI risk � General Rules ◦ Use ‘‘safer’’ NSAIDs (coxibs, diclofenac, ibuprofen. ) ◦ Use lowest effective dose for shortest period of time. ◦ Less use of NSAIDs with highest GI toxicity (ketorolac, piroxicam, ketoprofen) ◦ Avoid concomitant therapy (anticoagulants, corticosteroids, low -dose aspirin or antiplatelet agents) ◦ Eradicate H. pylori infection in pts with prior ulcer Hx. � Use of Prevention Strategies

Patients at increased risk for NSAID GI toxicity High risk 1. History of a previously complicated ulcer, especially recent 2. Multiple (>2) risk factors Moderate risk (1 – 2 risk factors) 1. Age >65 years 2. High dose NSAID therapy 3. A previous history of uncomplicated ulcer 4. Concurrent use of aspirin (including low dose) corticosteroids or anticoagulants Low risk 1. No risk factors H. pylori is an independent and additive risk factor and needs to be addressed separately Am J Gastroenterol 2009; 104: 728 – 738

Summary of recommendations for prevention of NSAID-related ulcer complications CVS risk No risk (no aspirin) Risk (aspirin) Gastrointestinal risk Low NSAID alone Moderate High • NSAID+PPI/ misoprostol • Alternative therapy if possible or (lowest • COX-2 effective inhibitor+PPI/misopr dose) ostol Naproxen+PPI/ • Avoid NSAIDs or COX PPI/misoprostol -2 inhibitors ostol • Use alternative therapy Am J Gastroenterol 2009; 104: 728 – 738
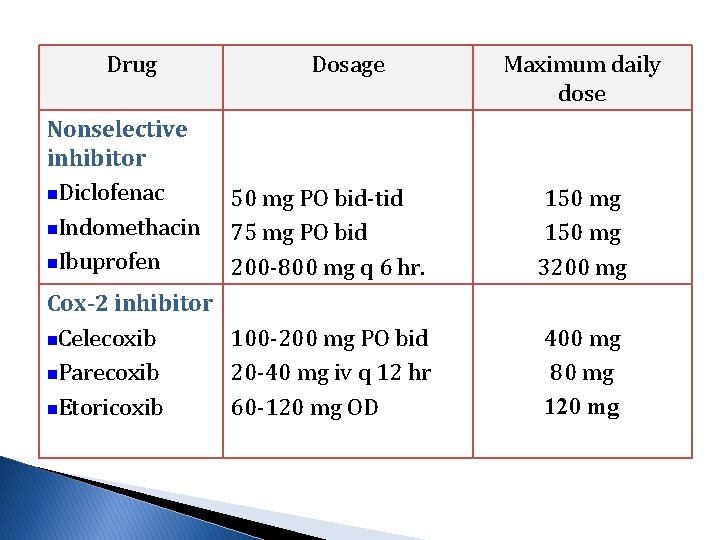
Drug Nonselective inhibitor n. Diclofenac n. Indomethacin n. Ibuprofen Dosage Maximum daily dose 50 mg PO bid-tid 75 mg PO bid 200 -800 mg q 6 hr. 150 mg 3200 mg Cox-2 inhibitor n. Celecoxib 100 -200 mg PO bid n. Parecoxib 20 -40 mg iv q 12 hr n. Etoricoxib 60 -120 mg OD 400 mg 80 mg 120 mg

Local anesthetic drugs
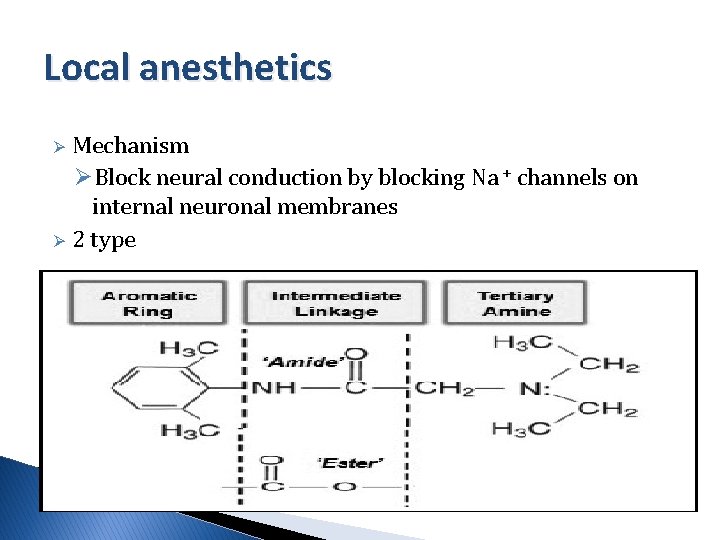
Local anesthetics Mechanism ØBlock neural conduction by blocking Na + channels on internal neuronal membranes Ø 2 type Ø

Local anesthetic (LA) drugs Ester LAs • Cocaine • Procaine • Chloroprocaine Metabolize Plama pseudocholineaterase Allergy Para aminobenzoic acid(PABA) Amide • Lidocaine • Bupivacaine • Ropivacaine • Levobupivacaine • Prilocaine hepatic Methylparaben (preservatives)
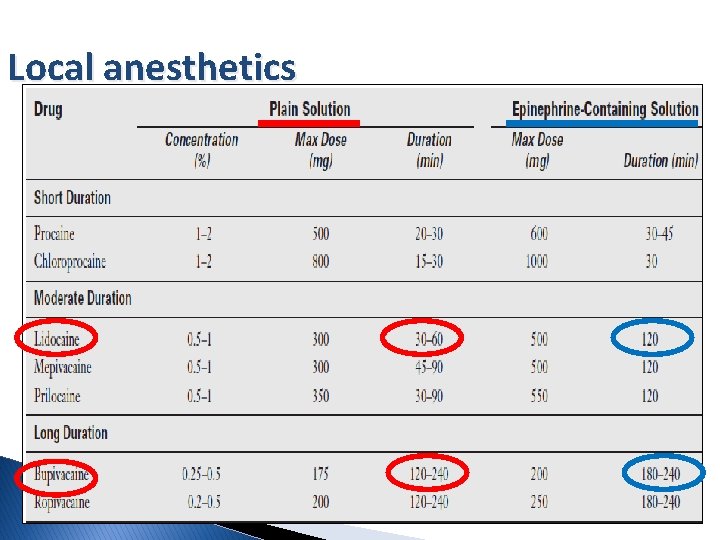
Local anesthetics

Toxicity Ø Local Anesthetic Systemic toxicity (LAST) Ø Local toxicity ØMyotoxicity (few case report) ØEffect on articular cartilage (few case report) ØNeurotoxicity : highly concentrated solutions / intraneural injections
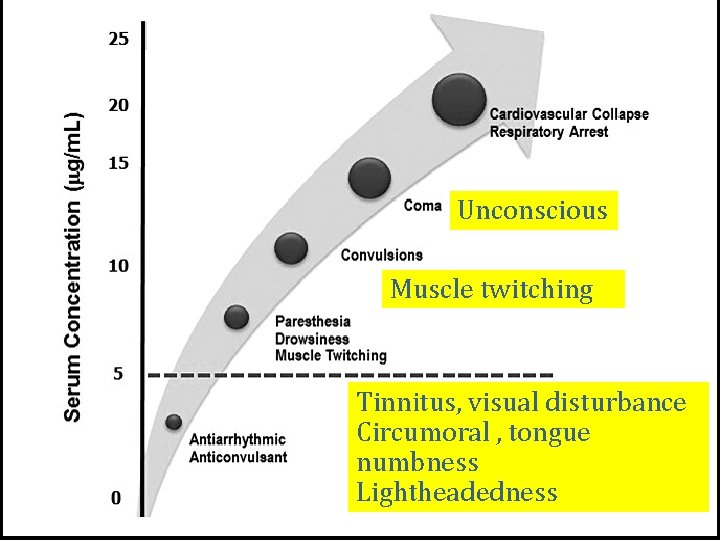
Unconscious Muscle twitching Tinnitus, visual disturbance Circumoral , tongue numbness Lightheadedness

Management of LAST
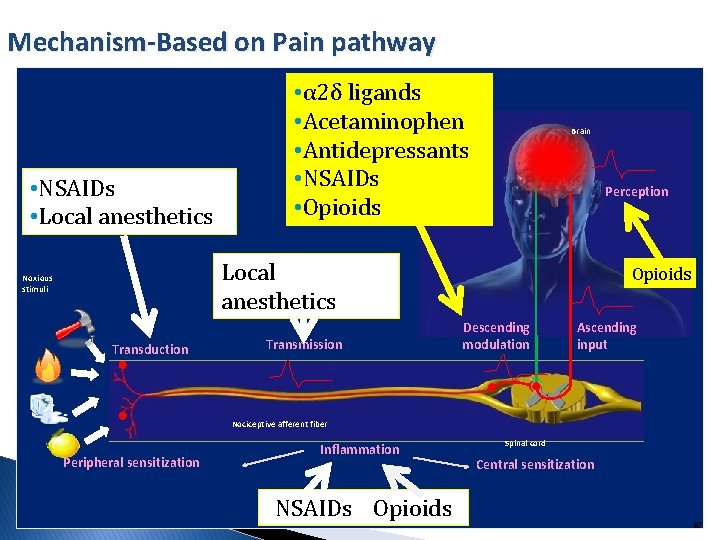
Mechanism-Based on Pain pathway • NSAIDs • Local anesthetics • α 2δ ligands • Acetaminophen • Antidepressants • NSAIDs • Opioids Brain Perception Local anesthetics Noxious stimuli Transduction Transmission Opioids Descending modulation Ascending input Nociceptive afferent fiber Peripheral sensitization Inflammation NSAIDs Opioids Spinal cord Central sensitization 87

Thank you for your attention
- Slides: 88