Acute Myeloid Leukemia By Ahmed Refaat Abd Elzaher

Acute Myeloid Leukemia By Ahmed Refaat Abd Elzaher Assistant lecturer of Medical Oncology South Egypt Cancer Institute Assuit University

Definition & epidemiology v Heterogeneous clonal stem cell malignancy v Immature hematopoietic cells proliferate and accumulate in BM, PB & other tissues features of BM failure. v PB and/or BM blasts > 20 % of nucleated cell (NC) count v If the blasts in the BM are > 5% & < 20% v Recently, certain Cy. G abnormalities MDS classified as AML irrespective of blast count : t(8; 21), inv(16) and t(15; 17) Roboz G & Sung L. ASH-SAP 2010; PP 475

Acute myeloid leukemia • Incidence – 2. 7 per 100, 000 • – median age of presentation : 65 yo AML comprises 90% of all acute leukemias in adults • More prevalent: – Males – European – Hispanic (promyelocytic leukemia (AML M 3)

AML - Etiology • Primary AML – Increased incidence • Genetic fragility – Faconi anemia – Down syndrome • tobacco use? • herbicides? , pesticides? • benzene exposure • Secondary AML – XRT – Topoisomerase II inhibitors (e. g etopisode), alkylating agents – MDS – other cell proliferation disorders • CML, polycythemia vera, primary thrombocytosis, PNH

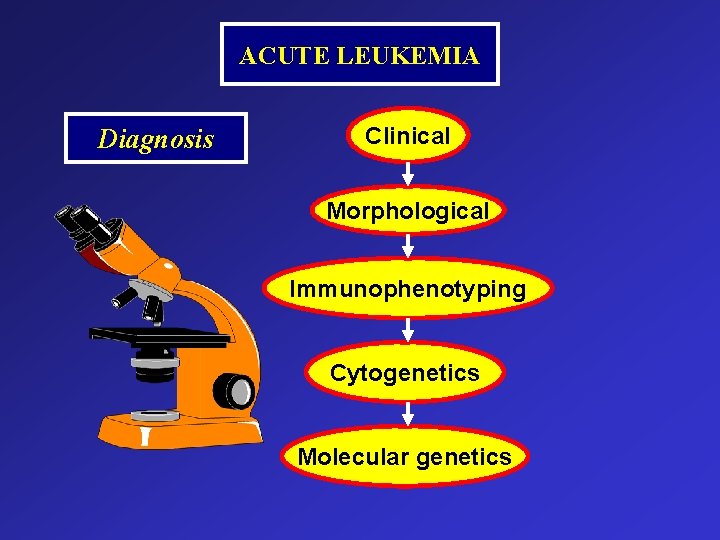
ACUTE LEUKEMIA Diagnosis Clinical Morphological Immunophenotyping Cytogenetics Molecular genetics

Clinical presentation Ø Sign or symptoms related to pancytopenia: • WBC infection. • Hb anemia. • platelets bleeding Ø Approximately 25% of AML patients present with white blood cell (WBC) counts > 50, 000/mm³ Ø Monocytic variants (M 4 or M 5) commonly display infiltration of gingivae, skin, soft tissues or meninges Ø DIC is a common presentation in AML M 3 Ø M 2 : Chloroma: -presents as a mass lesion ‘tumor of leukemic cells’

Leukostasis • Leukostasis – predominantly in those with WBC counts > 100, 000 (10% of patients); can also be seen in patients with WBC > 50, 000 – Most common in those with M 4 or M 5 leukemia • Common symptoms – Pulmonary: dyspnea, chest pain – CNS: headaches, altered mentation, CN palsies, ocular symptoms – Priapism – Myocardial Infarction
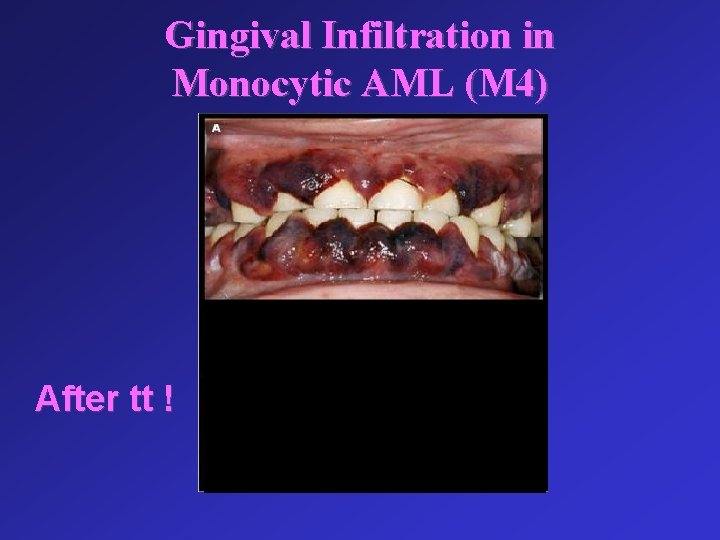
Gingival Infiltration in Monocytic AML (M 4) After tt !

Diagnosis Ø Previously >30% blasts on BM aspirate (per FAB criteria)¹ Ø Recently changed to > 20% blasts in the peripheral blood or BM (per WHO criteria)² Ø Patients with certain cytogenic abnormalities are considered to have AML regardless of blast percentage² • t(8; 21) (q 22; q 22), inversion (16) (p 13 q 22) • t(16; 16) (p 13; q 22), and t(15; 17) (q 22; q 12) 1 - Bennett et al, Br J Haematol 1976 2 -Vardiman et al, blood 2002
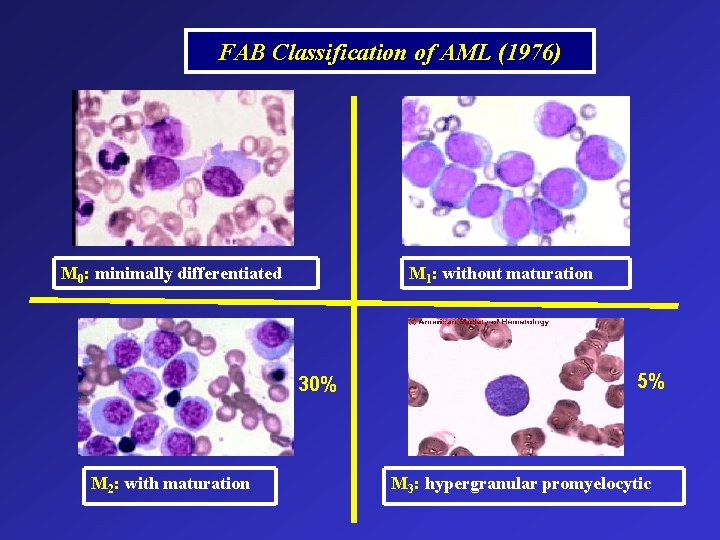
FAB Classification of AML (1976) M 0: minimally differentiated M 1: without maturation 30% M 2: with maturation 5% M 3: hypergranular promyelocytic
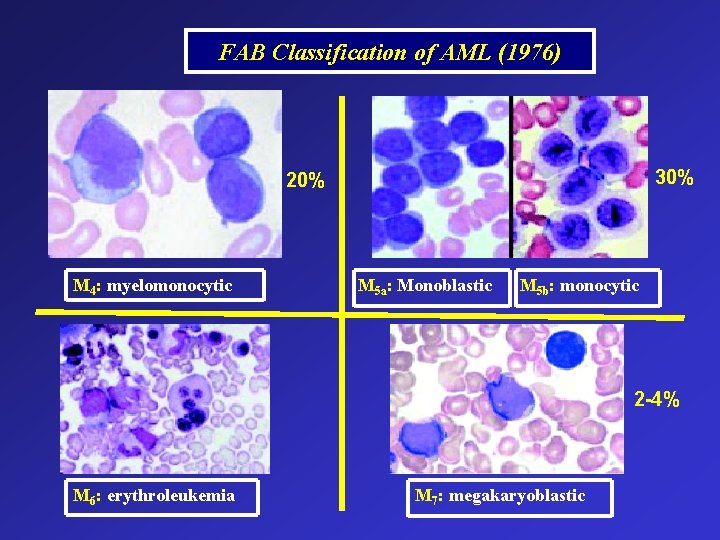
FAB Classification of AML (1976) 30% 20% M 4: myelomonocytic M 5 a: Monoblastic M 5 b: monocytic 2 -4% M 6: erythroleukemia M 7: megakaryoblastic

AML (WHO 2008) Cytogenetics & Moleculars EXTREMELY IMPORTANT More Easily… FAB ! Excluding APL (M 3)

WHO classification (2008) Poor prognosis… BAD cytogenetics AML with MDS Therapy-related AML …. (eg alklating agents, topoisom. II inhib. , RT…) (FAB excluding APL)

“Therapy-related AML: AML MM Alkylating agents and radiotherapy Topoisomerase II inhibitors Onset 5 -6 years postexposure 1 -5 years postexposure Ch abn. Of MDS Rearrangements of ch 11

Cytochemistry ___________ALL______AML_____ • • • Myeloperoxidase Suddan black Non-specific esterase Periodic acid Schiff (PAS) Acid phosphatase + in B-ALL + in T-ALL + + + in M 4, M 5 + in M 6 Bennett et al, Br J Haematol 1976
Immunophenotype • Based on blast surface antigen expression • CD 34 – Stem cell marker • CD 117, CD 33, CD 13, MPO – Myeloid markers • CD 14, CD 64 – Monocytic marker • Glycophorin A – Erythroid marker • CD 41, CD 61 – Megakaryocytic markers
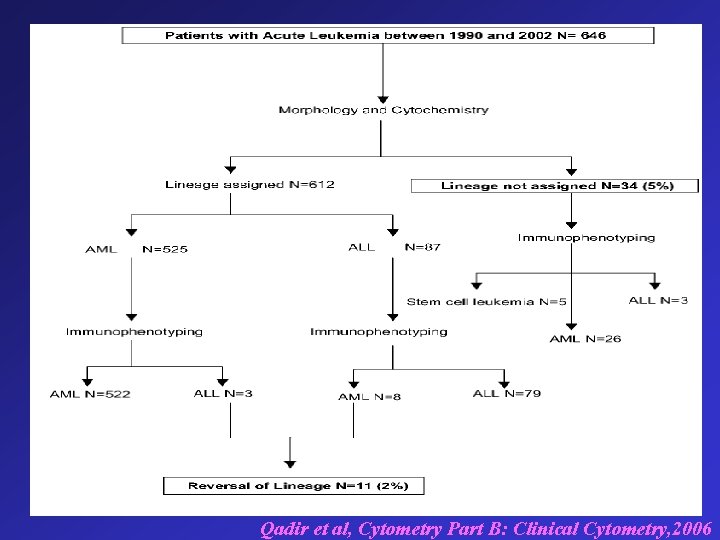
Qadir et al, Cytometry Part B: Clinical Cytometry, 2006

Cytogenetic risk groups Roboz G & Sung L. ASH-SAP 2010; 475 -487 Jabbour E et al. Mayo Clin Proc. 2006; 81: 247 (CBF) Normal / t(9; 11) (Trisomy 8) So, practically… Others: Complex (≥ 3 abnorm. ), monosomal karyotype 5 or 7 (MK 5 / 7 seems to be of the worst !) t(9; 22), t(6; 11), abn(17).
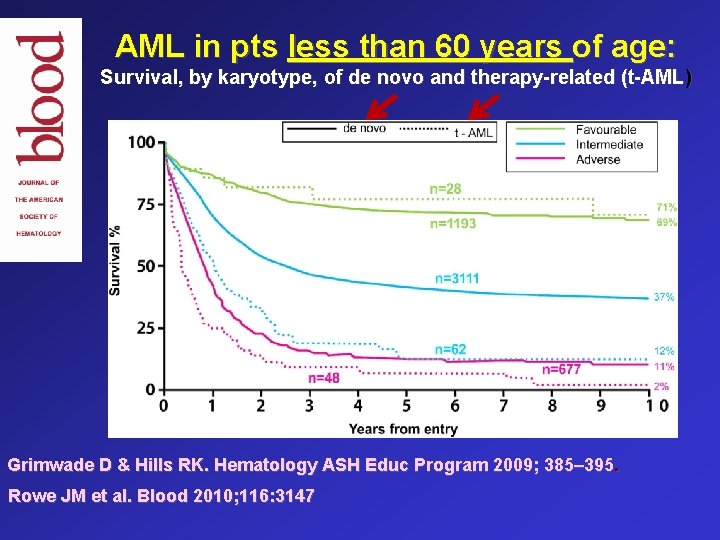
AML in pts less than 60 years of age: Survival, by karyotype, of de novo and therapy-related (t-AML) Grimwade D & Hills RK. Hematology ASH Educ Program 2009; 385– 395. Rowe JM et al. Blood 2010; 116: 3147
Molecular genetics: The field of biology that studies the structure and function of genes at a molecular (atomic) level Moleculars Cytogenetics (Cy. Gs)

“The most important prognostic indicators in AML are age, cytogenetics, and molecular genetics…. ”

Molecular mutations and prognosis (Normal & good cytogenetics… BAD is bad !!!) (Intermediate risk) GOOD The WORST (BAD) 20 -30% of t(8; 21) & 30 -40% of inv 16 & inv 16
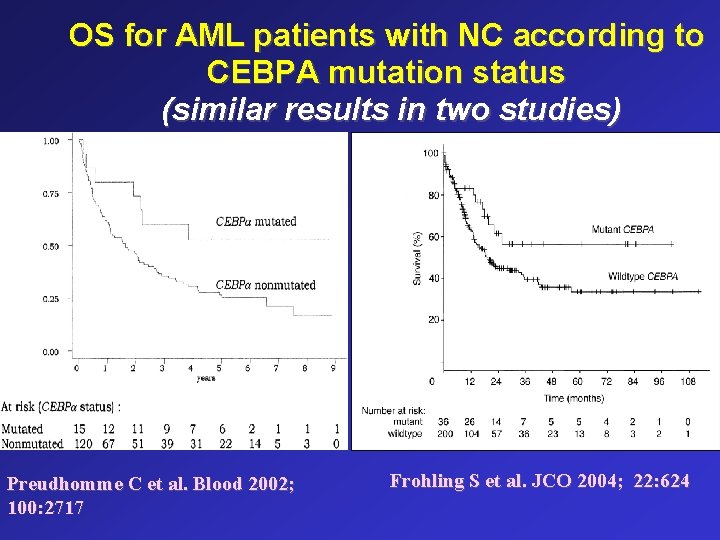
OS for AML patients with NC according to CEBPA mutation status (similar results in two studies) Preudhomme C et al. Blood 2002; 100: 2717 Frohling S et al. JCO 2004; 22: 624

Remember… v cytogenetic markers revolutionized therapeutic decisions favorable, intermediate & unfavorable. v However, patients tend to respond differently to a specific therapeutic approach within the same cytogenetic risk group`. Molecular markers of AML. v Both the NCCN and the ELN guidelines recommend: molecular genetics (mainly FLT 3, NPM 1, C-Kit and CEBPA) to further categorize cytogenetically normal patients…. More individualized decision making !

Cytogenetics and Molecular Abnormalities


DNMT 3 A Mutations Common in Intermediate-Risk AML • Mutations in the DNA methyltransferase gene, DNMT 3 A, are also associated with poor prognosis and are highly recurrent in patients with intermediate-risk AML. Presence of any DNMT 3 A mutation, either alone or in combination with the FLT 3 internal tandem duplication (ITD) mutation, is associated with significantly shorter overall survival Ley TJ, et al. ASH 2010. Abstract 99.

Molecular markers of AML Isocitrate dehydrogenase (IDH ) : IDH 1 and IDH 2 mutations occur in 25– 30% of patients with CNAML, and in general predict for worse outcome in certain molecular (NPM 1 wild-type) and clinical (older age) subsets of patients

Evaluation ● Cytogenetics are critical. ● Molecular diagnostics mandatory for all patients including the elderly: at least FLT 3 -ITD, NPM 1, CEBPA; also consider C-KIT in patients with corebinding factor AML. ● Post-remission monitoring of minimal residual disease using cytogenetics, PCR, and multicolor flow cytometry. ● Send HLA typing of patient and siblings at time of initial diagnosis.

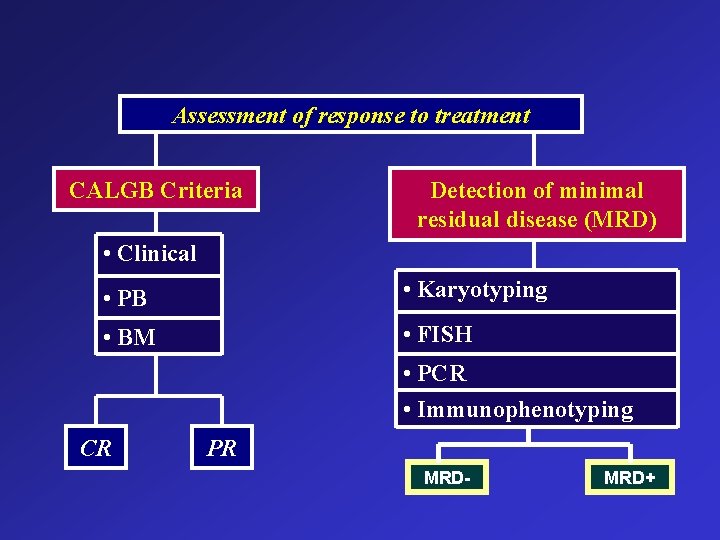
Assessment of response to treatment CALGB Criteria Detection of minimal residual disease (MRD) • Clinical • PB • Karyotyping • BM • FISH • PCR • Immunophenotyping CR PR MRD- MRD+

Criteria of response to treatment (CALGB) Definition of CR Clinical • P. S. = 100% • No organomegaly Peripheral blood Bone marrow • TLC= 2 -10 X 109/L • Normocelluar – ANC > 1000 – No blasts • HB > 11 (females) > 12 (males) • PLT > 100 000/m 3 • Blasts <5% The presence of Auer rods is not allowed in CR marrow NCCN Guidelines Version 2. 2011

…previously !! Essentially, routine practice nowadays for all AML pts !

Treatment of AML 1) Supportive treatment (for all pts) 2) Induction therapy 3) Post-remission (consolidation) therapy 4) Treatment of Relapsed AML 5) Special considerations § Elderly AML § Concept of targeted therapy in AML

Supportive treatment v Hydration and allopurinol to prevent tumor lysis syndrome… esp initial cyto-reduction with HU (if TLC) v Availability of “full” neutropenic precautions v Insertion of a central line to deliver chemotherapy. v Blood and platelets transfusion v Oral care and nutritional advice v Antimicrobials (prophylactic / therapeutic) v Sperm banking v Hormonal therapy to control menses during time of thrombocytopenia.

To begin with, till this very moment…. It is still the: “ 3 + 7” regimen (from the 1970 s …. . !!) Anthracycline X 3 d (usually Daunorubicin) Ara. C (Cytarabine) X 7 d Although AML…heterogeneous Same tt approach (except APL)… !

Induction Treatment Of AML Standard DA (3+7) BUT … Initially • Dauno 45 mg/m 2 D 1 -3 I. V • Ara. C 100 / 200 mg/m 2 D 1 -7 C. I No Difference in OS
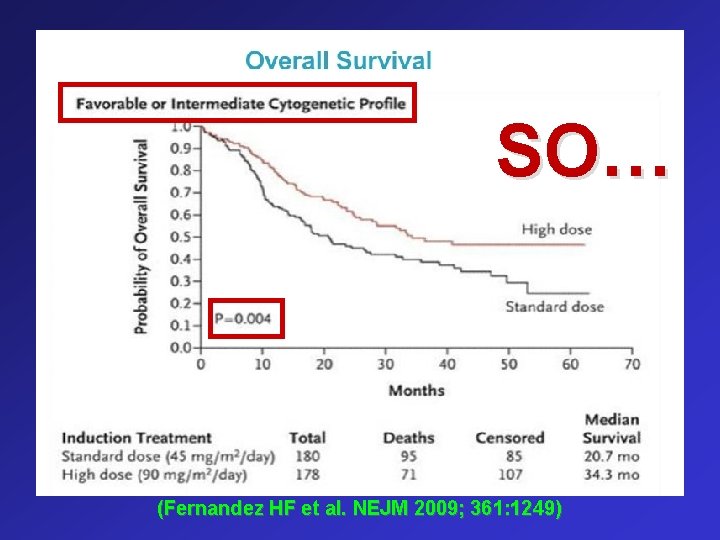
SO… (Fernandez HF et al. NEJM 2009; 361: 1249)

Which anthracycline ? Idarubicin (10 or 12 mg/m 2 X 3 d) OR Mitoxantrone 12 mg/m 2 X 3 d NO advantage over the higher doses of Dauno (i. e 60 or 90)

Ok, 3+7…. then what ? !

Still with remission induction Day 14 BM Blasts <5% wait till CBC recovery (Nadir) Blasts >5% & < 50% of initial blasts 2 nd 3+7 (if tolerable & NO active infection) Blasts > 50% of initial blasts Salvage (eg HAM / FLAG-M) Upon CBC recovery BM

Treatment of AML 1) Supportive treatment (for all pts) 2) Induction therapy 3) Post-remission (consolidation) therapy 4) Treatment of Relapsed AML 5) Special considerations § Elderly AML § Concept of targeted therapy in AML

Consolidation therapy: “EXTREMELY” crucial…. . • Rational: eradicate residual Leukemic clone Thus, traditionally…

Consolidation after CR (exclusively… cytogenetics) BUT, currently…. AML (1 st CR) Good risk Hi. DAC x 3 -4 Intermed. risk Allo-HSCT OR Hi. DAC Poor risk Allo-HSCT OR clinical trial Two meta analyses superiority of Allo in these risk groups: 1)Cornelissen J et al. Blood 2007; 109: 3658 2)Koreth A. et al. JAMA 2009; 301: 2349

Current facts The new era of molecular incorporation…. therapeutic map is changing (Few grey zones. . . !)

Favorable No role for HSCT, EXCEPT: Those with a mutation in c. KIT (eg with CBF t 8; 21) or FLT 3 Intermediate 45% of AML, mostly cytogenetically normal (CN) Allo-HSCT for all EXCEPT mutant NPM 1 but FLT 3 ITD -ve OR CEBPA+ve Unfavorable Allo-HSCT for all
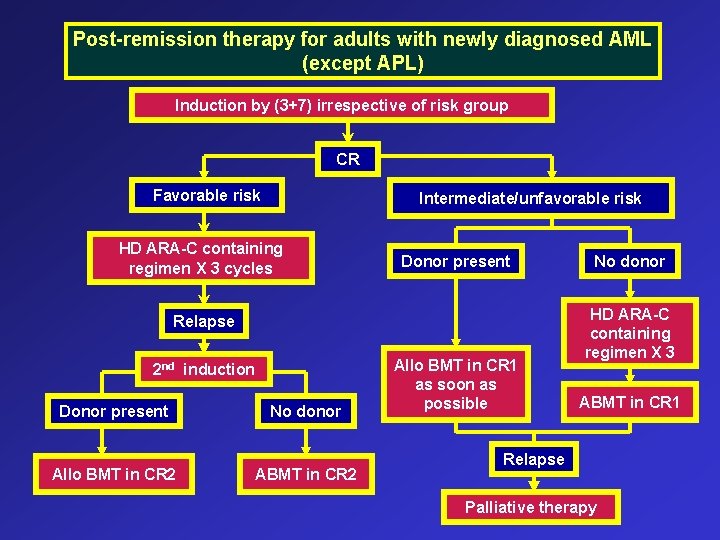
Post-remission therapy for adults with newly diagnosed AML (except APL) Induction by (3+7) irrespective of risk group CR Favorable risk Intermediate/unfavorable risk HD ARA-C containing regimen X 3 cycles Donor present Relapse 2 nd induction Donor present No donor Allo BMT in CR 2 Allo BMT in CR 1 as soon as possible No donor HD ARA-C containing regimen X 3 ABMT in CR 1 Relapse Palliative therapy


Treatment of special forms of AML M 5 High Risk Of CNS Disease After reaching CR by induction chemotherapy, give triple intrathecal prophylaxis: • MTX 15 mg • ARA-c 40 mg • Dexamethazone 4 mg Every 8 weeks for a total of 6 injections.
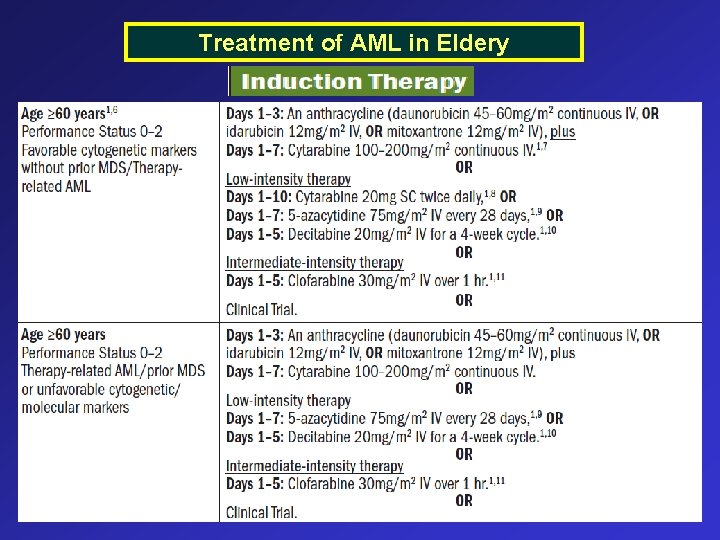
Treatment of AML in Eldery
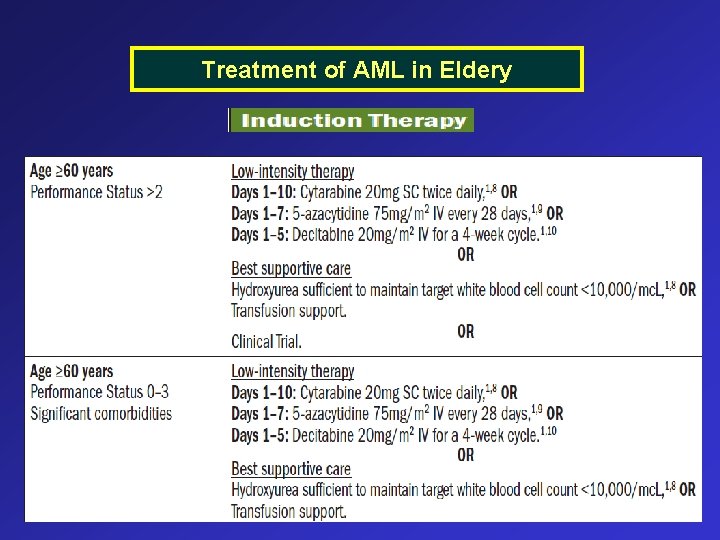
Treatment of AML in Eldery
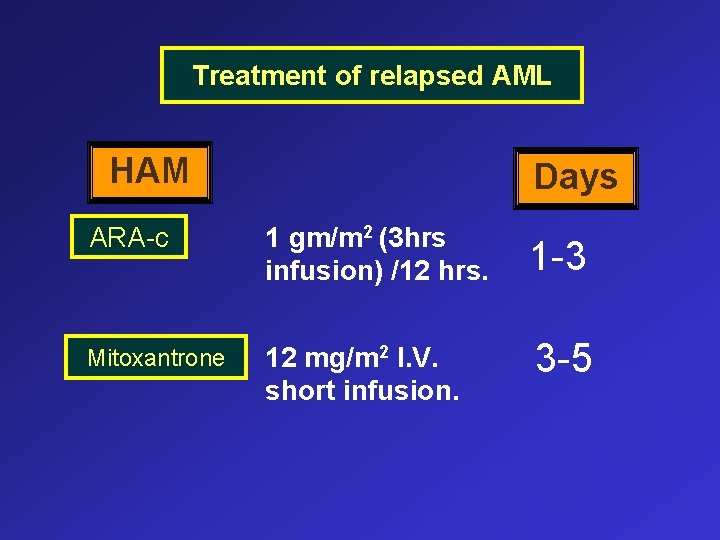
Treatment of relapsed AML HAM Days ARA-c 1 gm/m 2 (3 hrs infusion) /12 hrs. 1 -3 Mitoxantrone 12 mg/m 2 I. V. short infusion. 3 -5

Treatment of relapsed AML HAM - Prophylaxis ARA-C induced chemical conjunctivitis (excreted in tears) : Dexamethazone eye drops (e. g. Isoptodex): 2 drops in each eye qid day 1 -7. - High dose ARA-c should not be given over a period shorter than 3 hours : Due to enteritis problem - Antiemetic ttt Zofran 16 -32 mg C. I. or I. V. (2 -4 amp. ).
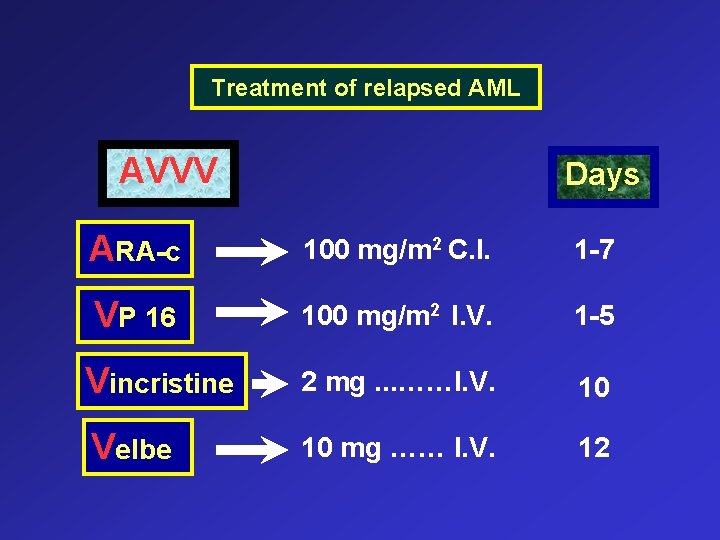
Treatment of relapsed AML AVVV Days ARA-c 100 mg/m 2 C. I. 1 -7 VP 16 100 mg/m 2 I. V. 1 -5 Vincristine 2 mg. . . ……I. V. 10 Velbe 10 mg …… I. V. 12

Treatment of relapsed AML FLAG Fludarabine ARA-c* G-CSF** Days 25 mg/m 2 1 hr inf. 1 -5 1 gm/m 2/12 hrs 3 hrs inf. 1 -5 200 gm /m 2 S. C. D 0 ANC >1000 *ARA-c should be given at least 4 hrs after Fludarabine to modulate pharmacokinetics of ARA-c (++ level of ARA-CTP in blast cells). ** Our experience is either to drop G-CSF or to start it on day +6

Epigenetic changes (Heritable modifications in gene function or activity without changes in DNA sequence) X X X • DNA hypermethylation • Histone deacetylation REVERSIBLE ! Gene silencing…”switching off” (tumor suppressor genes) Carcinogenesis Hypomethylating agents… AZA / DEC Histone Deacetylation Inhibitors (HDACi)

It is easy to kill cancer cells, but the challenge is keeping the patient alive at the same time…. . !

Thank You
- Slides: 59