Acute Lymphoblastic Leukemia Stephen P Hunger MD Chief

Acute Lymphoblastic Leukemia Stephen P. Hunger, MD Chief, Division of Pediatric Oncology Director, Center for Childhood Cancer Research Children’s Hospital of Philadelphia

Disclosure Slide: Stephen Hunger, MD § Speaker’s Bureau: None • Consultant without compensation: BMS Dasatinib EMEA PIP 2011; Genzyme (Sanofi) Clofarabine ODAC 2011 • Honoraria in Past two years: Jazz Pharmaceuticals; Sigma Tau Pharmaceuticals • Stock ownership: Amgen, Pfizer, Merck • Most therapies used in childhood ALL are offlabel; any therapeutic recommendations I make may include off-label use of drugs

Objective and Methods § To help course attendees pass board certification or recertification exam • Not to review current knowledge about ALL! § Review ABP Pediatric Hematology. Oncology Content Outline § http: //www. aspho. org/uploads/ABP_full_conte nt_outline. pdf

Presentation Format § Content specification statements § Answers § Points of Emphasis

Presentation § Presentation in handout is detailed and covers entire content outline • Too much to review in one hour § For presentation will skip over some slides, indicated with shading in the text box § Recommend that you review all slides in the handout

Presentation § Presentation in handout is detailed and covers entire content outline • Too much to review in one session § For presentation will skip over some slides, indicated with shading in the text box § Recommend that you review all slides in the handout

Incidence and Epidemiology § Know the incidence of ALL and AML, and the peak age at which each of these occur § Know the concordance rate of ALL and AML in identical twins

ALL: Epidemiology § Incidence peaks at 2 -4 years in developed countries • Rate of 80 new cases/million at 3 yrs dropping to 20 new cases/million at 10 yrs in US - 2009 US predicted 2540 ALL <15 yrs old and 2870 <20 yrs old • Age peak absent in low income countries but acquired with industrialization § Incidence in whites almost 2 x higher than in blacks § Modest increase in ALL incidence over

ALL: Genetic Risk Factors § Most ALL cases have no identified underlying genetic predisposition, but new findings in some subsets § Low hypodiploid ALL (30 -39 chromosomes) is a Li Fraumeni syndrome disease § ~50% have TP 53 germline mutations (Holmfeldt, Nature Genetics 2013) § Emerging data on SNPs related to ALL incidence, but relative risk is only 2 -4 x (Perez-Andreu, JNCI 2013) § Rare PAX 5 germline mutations in ALL (Shah, Nature Genetics 2013) § 4 -5% of pediatric leukemias have germline mutations in known cancer pre-disposing genes (Walsh, ASH

Leukemia: Relative Incidence, Subtypes, Mortality § Leukemia: relative proportion of childhood CA • 32% of childhood cancers among those <15 yrs old • 13% of childhood cancers among 15 -19. 99 yrs old § Leukemia: subtype proportions • 0 -5 yrs: ALL 81%, AML 14%, CML 2%, other 3% • 15 -19 yrs: ALL 51%, AML 36%, CML 9%, other 4%

Leukemia: Concordance Between Identical Twins § Concordance rates are close to 100% for leukemia occurring in infants <12 mos. old § Concordance rates are 10% to 15% for typical childhood B-cell precursor ALL occurring after infancy § Concordance is primarily due to prenatal acquisiting of early (initiating) chromosome translocations or other genetic lesions and transplacental transfer of leukemia or preleukemia cells to other twin • Primarily MLL translocations in infants

Leukemogenesis § Know which constitutional and genetic conditions predispose to development of leukemia § Know that Down syndrome is associated with an increased incidence of both ALL and AML § Know the mechanisms by which immunodeficiency states can have increased risk for leukemia

ALL: Predisposing Conditions § Cancer Predisposition syndromes • Li Fraumeni syndrome (SBLA): TP 53 and other genes • Hypodiploid ALL & LFS mutations (Holmfeldt, Nat Gen 2013) • Ataxia telangectasia (T-ALL + T-NHL): ATM • Neurofibromatosis Type I (AML>>ALL): NF 1 • Bloom syndrome (leukemia/lymphoma; AML>ALL): BLM § Congenital anomaly syndromes • Klinefelter's

§ ALL and Down Syndrome Children with DS have 10 -20 x increased risk of leukemia (DS) • Risk period limited to 1 st 3 decades of life • Although relative risk of AML is higher than ALL, DS-ALL is more common than DS-AML except in first year of life § 3% of children with B-precursor ALL have DS • Almost no DS children have T-ALL or infant ALL (<1 yr old) § Decreased rate of common sentinel genetic lesions (Maloney KW et al, Blood 2010) • • MLL translocation or t(9; 22) very rare ETV 6 -RUNX 1 (TEL-AML 1): 2. 5% DS-ALL vs. 24% non DS-ALL Trisomy 4+10: 7. 7% DS-ALL vs. 24% non DS-ALL About half have CRLF 2 -R; P 2 RY 8 -CRLF 2 most common § Overall outcome of DS-ALL inferior to non-DS ALL, but equivalent if limit analysis to cases lacking common sentinel genetic lesions (Maloney KW et al, Blood 2010) § Increased risk of toxic death in DS-ALL

Lymphoid Malignancies and Immunodeficiencies § Increased risk associated with • Congenital immunodeficiencies - Ataxia telangectasia, Wiscott Aldrich syndrome, others • Chronic immunosuppressive therapy • AIDS § B-cell NHL (Burkitt) most common, but BALL and B-precursor ALL (rare) also occur • Often due to EBV driven polyclonal expansion of B-cell clones with acquisition of somatic

Biological Characterization: Immunophenotype § Know the immunophenotypic differences between ALL and AML § Know how to identify lymphoid/myeloid mixed lineage leukemia and biphenotypic leukemia (by immunophenotyping)

Immunophenotype: ALL vs. AML § CD 45 often used to define hematopoietic cells § B-precursor ALL • Typically CD 19+, CD 22+, CD 79 a+ • May express "myeloid antigens" CD 13, CD 33 § T-ALL • c. CD 3+; often s. CD 3+, CD 7+ § AML • MPO+, CD 117+, CD 13+, CD 33+

ALL: Immunophenotype and Myeloid Antigen Expression § Myeloid Ag expression (esp CD 13, CD 33) expression is common in ALL and has no prognostic significance • Particularly common in ETV 6 -RUNX 1+ and Ph+ cases • MLL-R cases often express monocytic markers § Biphenotypic leukemia: Myeloid and lymphoid features present on the same cell (poor prognosis)

ALL: Clinical and Laboratory Features § Know the epidemiological, clinical and laboratory features that characterize • B-lineage ALL • Mature B-cell (Burkitt's) ALL • T-cell ALL

Clinical and Laboratory Features: BPrecursor ALL § 80 -85% of ALL cases are B-precursor § Almost all cases are CD 19+, CD 22+, CD 79 a+ § Most cases are CD 10+ (c. ALLA+), Td. T+ and HLA-DR+ § About half of MLL-R ALL lack CD 10 expression § Early B-lineage ALLs are c. Ig-negative § Pre-B ALL is c. Ig+, s. Ig-neg

Clinical and Laboratory Features: Mature BCell ALL B-ALL (Burkitt) § <2% of ALL cases are mature § Unique L 3 morphology and express surface Ig (s. Ig+) § Contain translocations that join c-Myc to Ig heavy or light chain locus • t(2; 8), t(8; 14), t(8; 22) § § High frequency of CNS involvement LDH often very elevated High risk of acute tumor lysis syndrome Treat like advanced stage Burkitt’s lymphoma • Extremely poor outcome with typical ALL therapy § May be associated with congenital or acquired

Clinical and Laboratory Features: TCell ALL § ~15% of ALL cases are T-ALLs § Express cytoplasmic CD 3 (c. CD 3+); most s. CD 3+ and Td. T+ § Often express CD 2, 4, 7, 8 § CD 10 expression is variable; most are HLA-DR negative § Higher incidence in males, older teenagers and blacks § Associated with high WBC and mediastinal masses • Increased risk of tumor lysis syndrome § Inferior outcome to B-lineage ALL with less

Recognize the Clinical Complications Related to Hematological Abnormalities in Acute Leukemia § Hematologic abnormalities are due to replacement of normal BM leading to signs/symptoms of BM failure • • White blood count may be low, normal or high Decreased neutrophil # and function: infection Risk of tumor lysis syndrome with high WBC (>50 k) Hyperleukocytosis (WBC >250 -500 k) may be associated with CNS and pulmonary symptoms - Risk of CNS/pulmonary complications much lower than in AML - Controversy over role of leukopheresis in ALL • Decreased platelets: bleeding, bruising and petechiae • Decreased red blood cells: pallor and fatigue

Recognize Hypereosinophilia as a Rare Presenting Feature of ALL § Associated with t(5; 14)(q 31; q 32) that brings IL-3 gene under control of Ig. H promoter • Eosinophil count may be 10, 000100, 000+/mm 3 • Eosinophils are reactive and NOT part of the malignant clone • May have low percentage of marrow blasts - Ig. H FISH can be very useful to identify these blasts - May be associated with increased risk of treatment failure, but data largely anecdotal

ALL: Other Clinical Features § Bone pain due to expansion of marrow cavity • Manifested as general irritability in young children § Adenopathy +/- hepatosplenomegaly § Thymic expansion: mediastinal masses in T-ALL § CNS leukemia: Headache, neck pain, seizures, cranial nerve palsies § Testicular involvement (always exam testes in boys!) • Painless, enlarged, usually “rock hard” testes • Involvement should be confirmed via biopsy

Formulate a Differential Diagnosis for Pancytopenia in Childhood § Leukemia (ALL, AML) § Aplastic anemia • Acquired, congenital, virus associated § § § § Metastatic solid tumor Myelodysplastic syndrome or myelofibrosis Hemophagocytic lymphohistiocytosis Infection or post-infectious (EBV especially) Systemic lupus erythematosus Hypersplenism B 12 or folate deficiency

§ § § § Know the Differential Diagnosis of Hepatosplenomegaly and Pancytopenia Leukemia (ALL, AML) Hepatitis associated aplastic anemia Infection or post-infectious (EBV especially) Metastatic solid tumor Myelodysplastic syndrome or myelofibrosis Hemophagocytic lymphohistiocytosis Systemic lupus erythematosus

Know that ALL can Mimic Juvenile Rheumatoid Arthritis (JRA/JIA) at Presentation § Small subset of ALL pts initially suspected to have JRA; some are treated for JRA before a diagnosis of ALL is established • Strongest factors predictive of ALL are low WBC (<4000), low-nml platelet count (150 -250, 000) and night-time pain • Rash and ANA not helpful - Jones et al, Pediatrics 117: e 840, 2006

Recognize X-ray Findings Associated with ALL • 40. 2% of children had at least 1 radiographic abnormality at ALL presentation – Sinigiglia et al, J Pediatr Orthoped 28: 20, 2008 • • Osteolysis (13. 1%) Metaphyseal bands (9. 8%) Osteopenia (9%) Osteosclerosis (7. 4%) Permeative pattern (5. 7%) Pathological fractures (5. 7%) Periosteal reactions (4. 1%) Mixed lysis-sclerosis lesions (2. 5%)

Know the Differential Diagnosis of Bone Pain in a Child • 38. 3% of children had complaints related to the musculoskeletal system at ALL presentation – Sinigiglia et al, J Pediatr Orthoped 28: 20, 2008 • Pain (34. 4%) • Functional impairment (22. 9%) • Limping (12. 3%) • Swelling (10. 6%) • Joint effusion (5. 7%)

Recognize How the Clinical Presentation of ALL Differs from § Most children with ITPITP are otherwise healthy; most children with ALL have other symptoms § With ITP, parents can often tell exact onset of bruising § A patient with isolated platelet count < 50 k, normal Hgb/Hct and WBC, and no circulating blasts almost never has ALL • Calpin et al (Arch Pediatr Adolesc Med. 1998 Apr; 152(4): 345 -7) reviewed 484 BM aspirates done at Sick Kids to confirm dx of ITP 1984 -1996 • 0/332 that met above criteria had ALL

Know the Differential Diagnosis of Absolute Lymphocytosis § § § Infection (especially pertussis, EBV, CMV) ALL Thyrotoxicosis Neutropenias (relative lymphocytosis) Addison’s disease (relative lymphocytosis)

ALL: Extramedullary Leukemia § Know the criteria for diagnosis of CNS leukemia § Know the clinical manifestations of CNS leukemia § Know the significance of an enlarged testis in ALL § Know the prognostic implications of duration of first remission for a patient with isolated CNS or testicular relapse

ALL: Definition of CNS Involvement § CNS 1: Absence of blasts on cytospin preparation, regardless of the number of WBCs § CNS 2: Presence < 5/μL WBCs and cytospin positive for blasts, or ≥ 5 /μL WBCs but negative by Steinherz/Bleyer algorithm § CNS 3: Presence of ≥ 5/μL WBCs and cytospin positive for blasts and/or clinical signs of CNS involvement (cranial nerve palsies, etc. )

ALL: CNS Involvement and the Steinherz/Bleyer Algorithm § Designed to help evaluate traumatic LPs • CSF WBC/CSF RBC more than 2 x Blood WBC/Blood RBC § A patient with CSF WBC ≥ 5/μL blasts, whose CSF WBC/RBC is 2 X greater than the blood WBC/RBC ratio, has CNS disease at diagnosis • Example: CSF WBC = 60/μL; CSF RBC = 1500/μL; blood WBC =46000/μL; blood RBC = 3. 0 X 106/μL: - 60/1500 = 0. 04 - 46, 000/3, 000 = 0. 015 - 0. 04 is more than 2 x greater than 0. 015, therefore patient is CNS 3

Clinical Manifestations of CNS Leukemia § Most often asymptomatic § When present, symptoms and exam findings typically reflect increased intracranial pressure (ICP) • Symptoms: headache, vomiting, lethargy, irritability, neck pain • Exam findings: papilledema, seizures, cranial nerve palsies

ALL: CNS Involvement by Subtype Know which ALL immunophenotypes are most likely to have CNS disease at § Common in: infants and Burkitt’s ALL diagnosis (15 -25%) § Intermediate in T-ALL (5 -10%) § Low in non-infant B-lineage ALL (2%)

ALL: Testicular Involvement § Present at diagnosis in <1 -2% of boys § Associated with other high risk features • T-ALL, high WBC § Painless, rock hard, lumpy, enlarged testicle § Involvement should be confirmed by biopsy § Moving away from testicular irradiation if a complete response obtained to induction therapy § Isolated testicular relapse much less
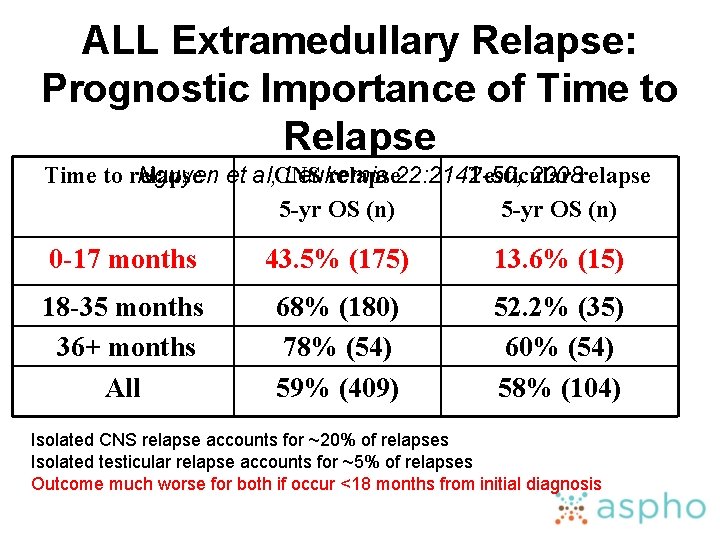
ALL Extramedullary Relapse: Prognostic Importance of Time to Relapse Time to relapse 22: 2142 -50, Testicular Nguyen et al, CNS Leukemia 2008 relapse 5 -yr OS (n) 0 -17 months 43. 5% (175) 13. 6% (15) 18 -35 months 36+ months All 68% (180) 78% (54) 59% (409) 52. 2% (35) 60% (54) 58% (104) Isolated CNS relapse accounts for ~20% of relapses Isolated testicular relapse accounts for ~5% of relapses Outcome much worse for both if occur <18 months from initial diagnosis

ALL: Morphology § Recognize the morphology of ALL, including the unique morphology of L 3 ALL § Know how to distinguish atypical lymphocytes from lymphoblasts § Know how to recognize granular ALL

ALL: FAB L 1 vs. L 2 Blast Morphology § L 2 blasts are • Larger • Have more distinct nucleoli • Often have more cytoplasm
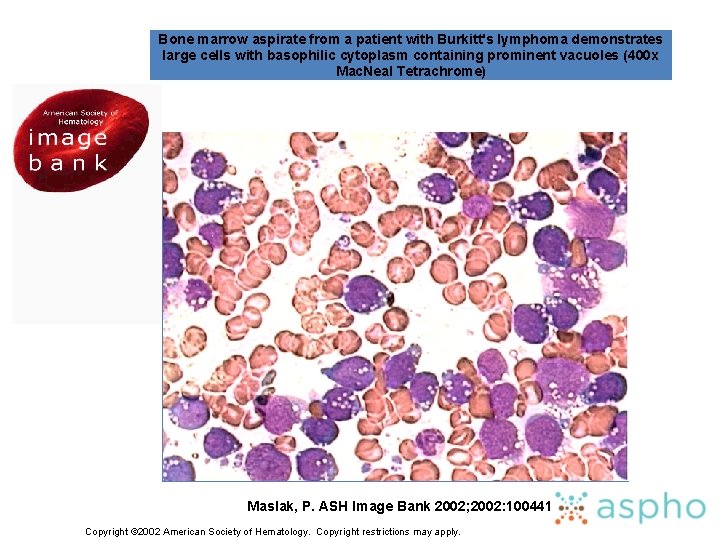
Bone marrow aspirate from a patient with Burkitt's lymphoma demonstrates large cells with basophilic cytoplasm containing prominent vacuoles (400 x Mac. Neal Tetrachrome) Maslak, P. ASH Image Bank 2002; 2002: 100441 Copyright © 2002 American Society of Hematology. Copyright restrictions may apply.
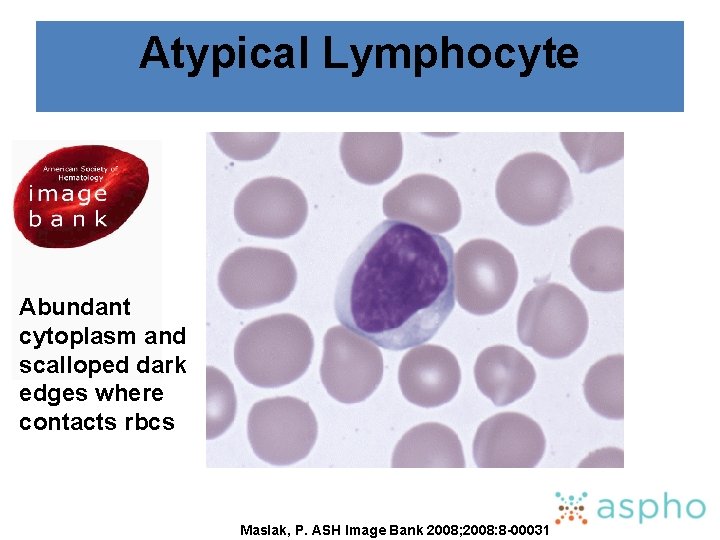
Atypical Lymphocyte Abundant cytoplasm and scalloped dark edges where contacts rbcs Maslak, P. ASH Image Bank 2008; 2008: 8 -00031
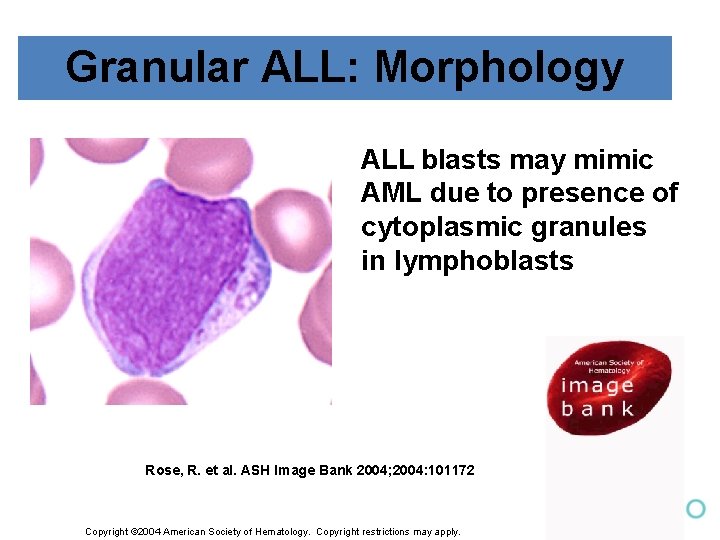
Granular ALL: Morphology ALL blasts may mimic AML due to presence of cytoplasmic granules in lymphoblasts Rose, R. et al. ASH Image Bank 2004; 2004: 101172 Copyright © 2004 American Society of Hematology. Copyright restrictions may apply.

ALL: Immunophenotypic Features § Know that CD 10+ lymphoblasts and Tlymphoblasts both have terminal deoxynuceotidyl transferase (Td. T) activity • Td. T lost very late in T and B cell development § Know which type of blasts express HLA-DR • Almost all B-precursor ALLs are DR+; most TALLs are DR-neg; some AMLs are DR+ [t(15; 17)]

Know the Relationship Between the Expression of Surface or Cytoplasmic Ig and ALL Subsets (Older Terminology) § Pro-B: CD 19+, CD 10 -neg, c. Ig-neg, s. Ig • Often MLL-R § Pre-pre-B (common): CD 19+, CD 10+, c. Igneg, s. Ig§ Pre-B: CD 19+, CD 10 -neg, c. Ig+, s. Ig-neg • 25% with t(1; 19) and E 2 A-PBX 1 (TCF 3 -PBX 1) § Mature B (Burkitt): CD 19+, CD 10+ or neg, c. Ig+ or neg, s. Ig+ (monoclonal k or l)

Know How to Interpret Cell Surface Markers in ALL, Including CD 10 and HLA-DR § CD 10 expression occurs early in B-cell development and is lost late § Earliest pro-pro B cells are typically CD 10 negative § Lack of CD 10 expression linked to MLL translocations, especially t(4; 11) • Associated with poor outcome, but hard to determine if the adverse risk factor is CD 10 neg status or MLL-R

ALL: Gene Rearrangements § Know the gene rearrangement characteristics of ALL § Know the prognostic significance of the molecular translocations that occur in ALL

ALL: Antigen Receptor Gene Rearrangements § Immuoglobulin heavy (Ig. H) chain rearranges before Ig light chains (kappa before lamba—K comes before L) - DJ rearrangement occurs first, then V region rearranges - Almost all B-lineage ALLs have Ig rearrangements; significant percentage also have T-cell receptor gene (TCR) rearrangements - Infant ALLs may lack Ig rearrangements § Almost all T-ALLs have TCR rearrangements; only

Ig/TCR VDJ Rearrangements are Unique Clonotypic Markers Unique marker of each B cell and its clonal progeny
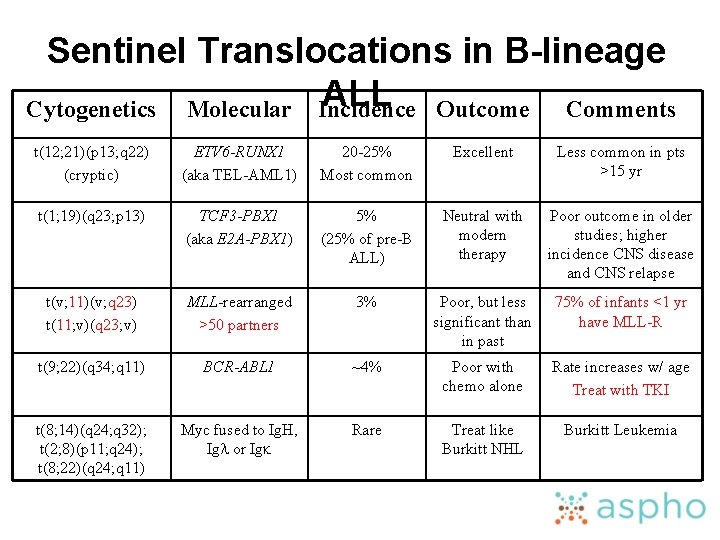
Sentinel Translocations in B-lineage ALL Outcome Comments Cytogenetics Molecular Incidence t(12; 21)(p 13; q 22) (cryptic) ETV 6 -RUNX 1 (aka TEL-AML 1) 20 -25% Most common Excellent Less common in pts >15 yr t(1; 19)(q 23; p 13) TCF 3 -PBX 1 (aka E 2 A-PBX 1) 5% (25% of pre-B ALL) Neutral with modern therapy Poor outcome in older studies; higher incidence CNS disease and CNS relapse t(v; 11)(v; q 23) t(11; v)(q 23; v) MLL-rearranged >50 partners 3% Poor, but less significant than in past 75% of infants <1 yr have MLL-R t(9; 22)(q 34; q 11) BCR-ABL 1 ~4% Poor with chemo alone Rate increases w/ age Treat with TKI t(8; 14)(q 24; q 32); t(2; 8)(p 11; q 24); t(8; 22)(q 24; q 11) Myc fused to Ig. H, Igl or Igk Rare Treat like Burkitt NHL Burkitt Leukemia

ALL: Prognostic Factors § Know the clinical and laboratory findings that influence prognosis § Know the NCI consensus risk stratification of ALL § Know that the significance of prognostic factors varies among different treatment regimens

Clinical and Laboratory Findings that Influence Prognosis § Baseline characteristics • Age, WBC, immunophenotype collectively define NCI risk group • Presence/absence of extramedullary disease influences prognosis and dictates need for specific therapies • Sex, race, ethnicity important but not used for treatment stratification • ALL genotype strongly influences prognosis, treatment stratification and sometimes use of targeted therapies; e. g. TKI treatment for Ph+ ALL • Response to therapy is the strongest prognostic factor
![NCI/Rome Risk Groups in Childhood ALL [Applies only to B-lineage ALL!] § Standard risk NCI/Rome Risk Groups in Childhood ALL [Applies only to B-lineage ALL!] § Standard risk](http://slidetodoc.com/presentation_image/2bba1ffc57a369f12134177eb8f8cad6/image-54.jpg)
NCI/Rome Risk Groups in Childhood ALL [Applies only to B-lineage ALL!] § Standard risk (~65% patients) • Age 1. 00 -9. 99 years • Initial white blood count <50, 000/microliter • EFS ~90% § High risk (~35% patients) • Age <1 yr or 10+ yrs OR WBC ≥ 50, 000/ml • Current 5 year EFS 75 -80% for non-infants - Achieved with more intensive therapy • Outcome still poor for infants < 1 year old - ~3% of patients; generally treated on infant specific trials

ALL: Risk-Adapted Therapy • High-risk subgroups fare poorly with less intensive therapies that have high cure rates in lower risk subgroups • Outcome of high-risk subgroups can be improved with more intensive therapy that may not be needed for LR subgroups • Prognostic factors disappear as therapy improves; e. g. t(1; 19)

Prognostic Factors: Initial WBC Count and Age § Recognize initial WBC as a significant risk factor • Higher is worse • ≥ 50, 000/ml used as cutoff but WBC is a continuous variable with best prognosis seen with WBC < 10, 000/ml § Recognize age as a significant risk factor; recognize age subgroups as a prognostic factor for infants <12 months old • Older is worse (or less than 1 yr); 10 yrs used as cutoff but is a continuous variable • 2 -6 yrs is best subgroup; 1 -2 years or 6 -9. 99 yrs is worse

ALL: Response to Therapy § Know that rapidity of response to chemotherapy in ALL is predictive of outcome § Know the methods that can be used to measure rapidity of response, including day 8 and day 15 BM aspirates and peripheral blood assessments § Know the methods for quantitating minimal residual disease (MRD) § Know the rationale for using MRD in predicting outcome in ALL

ALL: Morphological Measures of Early Treatment Response § BFM: PB response to 7 days of steroids + 1 dose IT MTX (prophase or prephase) • Good (PGR): <1000 blasts/ml in PB [90% of pts with EFS 80%] • Poor (PPR): >1000 blasts/ml in PB [10% of pts with EFS 30%] § CCG: day 8 (HR) or day 15 (SR) BM response - M 1: <5% blasts, M 2: 5 -25% blasts, M 3: >25% blasts - Outcome: M 1> M 2 > M 3

§ Minimal Residual Disease (MRD) Morphologic assessment is a crude, but accurate and reproducible way to identify patients likely to have good or bad outcomes § MRD is the presence of cells following chemotherapy below the level of morphologic detection • Techniques to assess MRD should achieve a sensitivity of at least 1/10000 (0. 01%) § More sensitive measures should be a more accurate way to identify groups for risk-adapted therapy § MRD response is most powerful prognostic factor

MRD: Technologies § Detection of leukemia associated phenotypes via flow cytometry • Applicable in almost all cases • Fast, relatively inexpensive • Less sensitive than molecular methods § PCR amplification of antigen receptor loci (Ig or TCR) • Applicable to ~85 -90% of cases • Laborious and expensive (10 x flow), but very sensitive • Now using next generation sequencing (NGS) § Parallel studies of flow and PCR show very similar results § PCR of translocation-derived fusion transcripts • Only suitable for defined subgroups such as Ph+ ALL

Ig/TCR Rearrangements are Unique Clonotypic Markers and MRD Targets Unique marker of each B cell and its clonal progeny
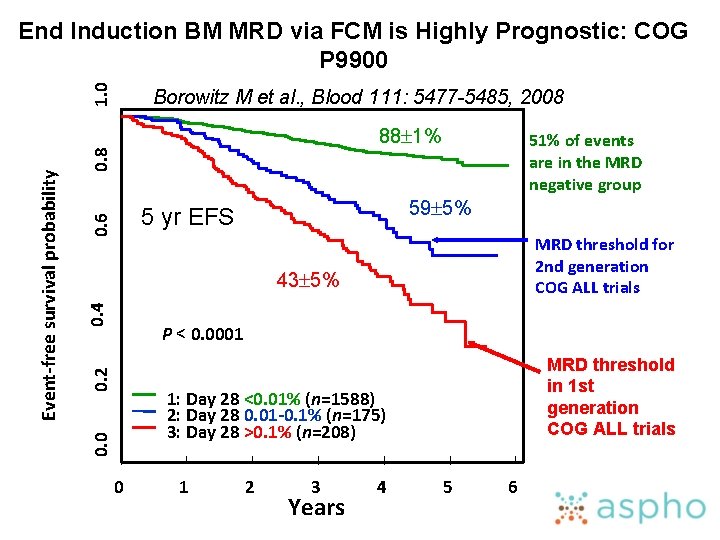
1. 0 End Induction BM MRD via FCM is Highly Prognostic: COG P 9900 Borowitz M et al. , Blood 111: 5477 -5485, 2008 0. 8 51% of events are in the MRD negative group 59 5% 0. 6 5 yr EFS MRD threshold for 2 nd generation COG ALL trials 0. 4 43 5% 0. 2 P < 0. 0001 MRD threshold in 1 st generation COG ALL trials 1: Day 28 <0. 01% (n=1588) 2: Day 28 0. 01 -0. 1% (n=175) 3: Day 28 >0. 1% (n=208) 0. 0 Event-free survival probability 88 1% 0 1 2 3 Years 4 5 6

Cytogenetics and Oncogenes § Know the prognostic significance of cytogenetic and molecular diagnostic findings in ALL § Know that a “normal” ALL karyotype may reflect inadequate growth of ALL cells +/occult translocation

ALL: Cytogenetics § Know the prognostic significance of cytogenetic and molecular diagnostic findings including • Hyperdiploidy, hypodiploid and relation between chromosome number and DNA index (DI) • Philadelphia chromosome (Ph+) • TEL-AML 1 (ETV 6 -RUNX 1), molecular counterpart to t(12; 21) - Most common cytogenetic abnormality in Bprecursor ALL § Know the cytogenetic/molecular abnormalities most common in infant ALL < 12 months § MLL (11 q 23) translocations in 75%, most common is

Chromosome Number in ALL Cytogenetics Incidence Outcome Comments Hyperdiploidy DNA Index >1. 16 or 53+ chromosomes 20 -25% Excellent Trisomy 4, 10 and 17 best Hypodiploidy <44 chromosomes or DNA index <0. 81 1 -2% Poor Worsening prognosis with fewer chromosomes; Near haploid is worst

Philadelphia Chromosome Positive (Ph+) ALL Know the prognostic significance of Ph+ ALL § 3 -4% of childhood ALLs are Ph+ • Increases to 25% by age 30 -40 and 50% in elderly § Age, WBC, and early response are important prognostic factors in Ph+ ALL § Dismal outcome for Ph+ ALL in pre-imatinib era • SCT used commonly but outcomes still poor
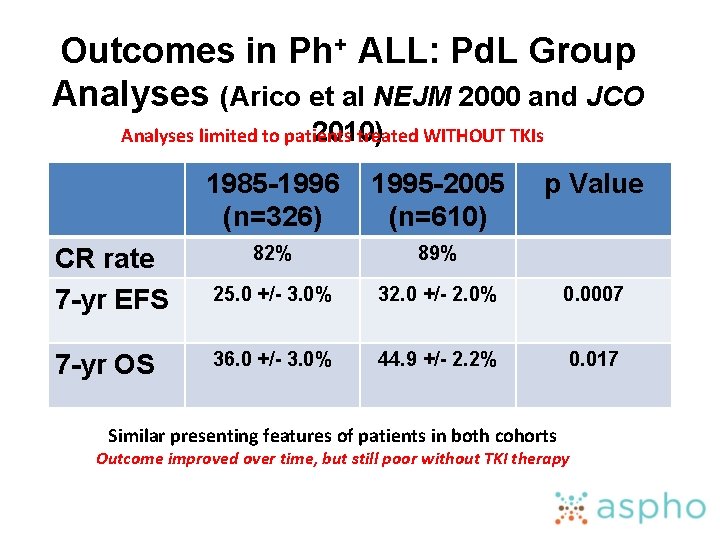
Outcomes in Ph+ ALL: Pd. L Group Analyses (Arico et al NEJM 2000 and JCO 2010) Analyses limited to patients treated WITHOUT TKIs 1985 -1996 (n=326) 1995 -2005 (n=610) p Value CR rate 7 -yr EFS 82% 89% 25. 0 +/- 3. 0% 32. 0 +/- 2. 0% 0. 0007 7 -yr OS 36. 0 +/- 3. 0% 44. 9 +/- 2. 2% 0. 017 Similar presenting features of patients in both cohorts Outcome improved over time, but still poor without TKI therapy

COG AALL 0031 Cohort 5 vs. Historical Controls 7 -yr EFS AALL 0031 c 5 72% POG ALinc 14 -16 27% Led to 2013 FDA approval of imatinib for Ph+ ALL in pediatrics Continuous imatinib plus intensive chemotherapy greatly improved EFS compared with historical controls from pre-imatinib era Schultz KR et al, JCO 2009 and Leukemia 2014

T-ALL: Sentinel Chromosome Translocations § Unlike B-precursor ALL, fusion genes are very rare § Hyper- and hypdodiploidy uncommon in T-ALL § Few lesions have major prognostic significance in T-ALL § Translocations with a TCR more common in TALL • TCRd on 14 q 11; TCRb on 7 q 35 § A number of translocations occur at low frequency • TTG 2/RHOM 2 (11 p 13), TTG 1/RHOM 1 (11 p 15), HOX 11/TCL 1 (10 q 24), TAL/SCL (1 p 32), MYC (8 q 24), TAL 2 (9 q 32), LYL (19 p 13), LCK (1 p 34) § Most common lesion is small, cryptic deletion of

ALL: Chemotherapy § Identify drugs most valuable for remission induction • Corticosteroid (Dex, Pred), VCR, ASNase +/anthracycline § Recognize delayed intensification as a useful strategy for standard and high risk ALL • Benefit established in multiple randomized trials • Two DI phases no better than one (CCG 1961 and 1991) § Recognize the importance of risk-adjusted

CCG 106: BFM Therapy is Superior to CCG Therapy for HR-ALL [Gaynon PS et al JCO 11: 2234, 1993] CCG 106 enrolled patients from 1983 -87 7 -Year EFS + S Reg A (BFM): 63% + 72% Reg B (NYII): 61% + 72% Reg C (CCG): 40% + 56% Regimen B had increased morbidity compared to A Regimen A, with DI phase based on BFM 76, was adapted as standard CCG therapy for HR ALL
![CCG 105: Long Term Outcome [1983 -1989] • Tested components of BFM therapy by CCG 105: Long Term Outcome [1983 -1989] • Tested components of BFM therapy by](http://slidetodoc.com/presentation_image/2bba1ffc57a369f12134177eb8f8cad6/image-72.jpg)
CCG 105: Long Term Outcome [1983 -1989] • Tested components of BFM therapy by randomizing IR pts to std CCG therapy +/- BFM intensifications: – – • • • Tubergen DG et al J Clin Oncol 11: 527 -37, 1993 Std CCG IIC: BFM protocol Ia + Ib DI: Added DI (protocol IIa + IIb to std) BFM: Used protocol I and II DI improved outcome No added advantage to Ia (4 drug induction), or Ib (intensive consolidation) for IR pts if DI given Based on these results, all subsequent CCG trials included DI, but SR trials did not have Ia (4 -drug induction) or Ib (intensive consolidation) Hunger SP et al Pediatr Blood Cancer 2005 45: 87680, 2005
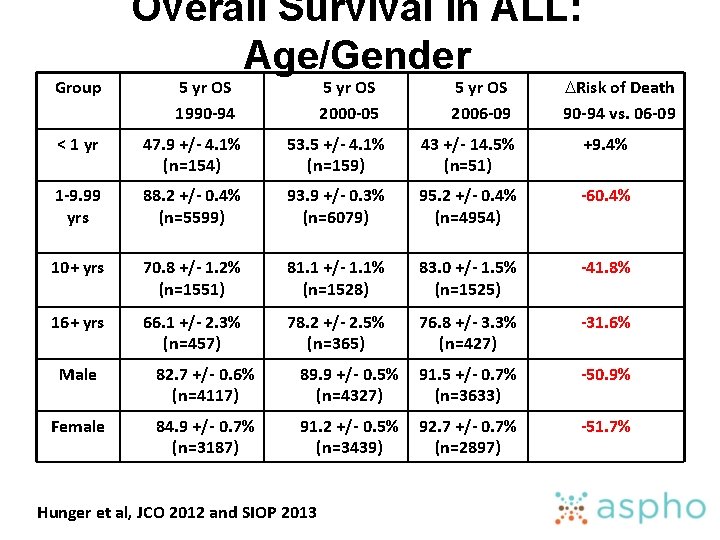
Group Overall Survival in ALL: Age/Gender 5 yr OS 1990 -94 5 yr OS 2000 -05 5 yr OS 2006 -09 DRisk of Death 90 -94 vs. 06 -09 < 1 yr 47. 9 +/- 4. 1% (n=154) 53. 5 +/- 4. 1% (n=159) 43 +/- 14. 5% (n=51) +9. 4% 1 -9. 99 yrs 88. 2 +/- 0. 4% (n=5599) 93. 9 +/- 0. 3% (n=6079) 95. 2 +/- 0. 4% (n=4954) -60. 4% 10+ yrs 70. 8 +/- 1. 2% (n=1551) 81. 1 +/- 1. 1% (n=1528) 83. 0 +/- 1. 5% (n=1525) -41. 8% 16+ yrs 66. 1 +/- 2. 3% (n=457) 78. 2 +/- 2. 5% (n=365) 76. 8 +/- 3. 3% (n=427) -31. 6% Male 82. 7 +/- 0. 6% (n=4117) 89. 9 +/- 0. 5% (n=4327) 91. 5 +/- 0. 7% (n=3633) -50. 9% Female 84. 9 +/- 0. 7% (n=3187) 91. 2 +/- 0. 5% (n=3439) 92. 7 +/- 0. 7% (n=2897) -51. 7% Hunger et al, JCO 2012 and SIOP 2013

Know the Principles of Treatment for Different Risk Groups of ALL (COG Approach) • Standard Risk ALL • High Risk ALL (and TALL) – 4 -week 3 -drug (Dex) induction – 4 -week oral consolidation with intensive IT – 8 -week Int Maint (IM) with Capizzi IV MTX w/o rescue – 8 -week DI phase with standard re-consolidation – 2 nd Capizzi IM (sometimes) – Maint with q 4 week – 4 -week 4 -drug (Pred 28 or Dex 14) induction – 8 -week intensive IV consolidation with intensive IT – 8 -week Int Maint with HD MTX or Capizzi IV MTX + ASNase – 8 -week DI phase with intensive re-consolidation – 2 nd Capizzi IM

Prevention of CNS Leukemia Dramatically Improved ALL Outcome § 1960 s: chemotherapy induced remissions in most patients, but very high rate of early CNS relapses • 50 -75% had CNS relapse within 6 -12 months § Most chemo agents have poor CNS penetration and prophylactic CNS treatment is essential • Dramatic increase in cure rates in early 70 s with universal use of cranial (c. XRT) or craniospinal

ALL: Prophylactic or Presymptomatic Treatment of thecan. CNS § Know that irradiation be avoided in many pts if effective systemic and intrathecal chemotherapy are given • Greatly decreased use of c. XRT over time; some groups use only for CNS 3 or not at all (SJCRH, DCOG, MRC, Co. ALL) • Other groups still use c. XRT for some patient subsets (COG, DFCI, BFM, others) - Typically includes some/all T-ALL especially with high WBC, some subsets with poor MRD response, and all CNS 3 § Plan the radiation field for prophylactic CNS irradiation • Current presymptomatic dose usually 1200 c. Gy c. XRT,

ALL: CNS Therapy § Plan the management of CNS leukemia detected at the time of initial diagnosis • Remember CNS 3 definitions • Most (not all) groups treat with HR-ALL chemotherapy that includes 1800 c. Gy cranial irradiation plus extended IT therapy § Know the management of CNS leukemia that develops after prophylaxis • Time to relapse is a strong predictor of outcome (<18 mos < 18 -36 mos < 36+ months • Effective systemic therapy is critical as the biggest risk is subsequent BM relapse • 1800 c. Gy is “standard”; COG AALL 02 P 2 tested 1200 c. Gy and had higher failure rates (reasons not certain)

ALL: CNS Therapy and Adverse Effects Thisthe Seems Important Given of Content § Know complications of. Number intrathecal MTX Specifications! and CNS XRT in pts with ALL § Recognize the late complications of c. XRT § Know that a brain tumor is a late complication of c. XRT § Know the relationship between treatments for ALL and late CNS complications § Know the effect of age on neuropsychologic function after c. XRT in ALL

CNS-Directed Therapy: Acute Complications § Intrathecal chemotherapy • Arachnoiditis (headache, nausea, vomiting, meningismus) • Seizures (occur in 1 -2%, particularly with repeats ITs) • Transient stroke-like symptoms • Typically occur 7 -11 days post IT • Rare occurrence of subacute encephelopathy, myelopathy, and weakness/paraplegia linked to IT MTX § Irradiation • Somnolence syndrome 5 -7 weeks post c. XRT § Administration of high dose IV MTX after

CNS-Directed Therapy: Late Complications § Changes on neuroimaging studies • Cortical atrophy, necrotizing leukocencephalopathy, subacute leukocencephalopathy, mineralizing leukocencephalopathy (HD MTX may increase risk; uncertain) - Calcifications may be most significant finding § Impaired intellectual function and school performance • Linked to dose and age at time of c. XRT (<8 yrs is worse) § Development of secondary brain tumors (10+ yrs post XRT) § Meningioma (15 -20 yrs post XRT) highly curable; others not § Neuroendocrine changes • Impaired growth and GH responses; much lower risk if <2400 c. Gy

Plan the Diagnostic Evaluation and Management of ALL Patient with Seizures § Differential diagnosis includes leukemia involvement, infection, metabolic abnormalities, CVA, drug toxicity (especially MTX or IT chemo) • Imaging studies • LP with cytospin for blasts and culture • Electrolytes and glucose § Different causes predominate at different times in Rx

Know the Complications of ALL Induction Therapy § Hyperleukocytosis • May cause CNS or pulmonary symptoms • Leukopheresis rarely used in ALL § Acute tumor lysis syndrome § Infection § Hyperglycemia • Treat with insulin; don't change steroid dose § Thrombosis or bleeding (ASNase)

Plan the Management of a Patient with Pancytopenia During ALL Induction § Treat with antibiotics until signs of count recovery • Many different definitions for "count recovery" § Steroid use increases risk of fungal infection; hyperglycemia may also increase risk • Early empiric antifungal therapy if fever is prolonged • Control blood glucose § Infection risk high due to BM replacement

Know the Clinical & Lab Features of Tumor Lysis Syndrome (TLS) and its Prophylaxis & Management Howard NEJM 364: 1844, 2011) § Clinical and(see Lab Features § Laboratory TLS: triad of increased uric acid, PO 4, K+, +/hypocalcemia § Clinical TLS: above + increased Cr, seizures, cardiac dysrhythmias or death § Due to inability to handle byproducts of leukemia cell death § May occur spontaneously or after therapy is started § Most problematic in leukemias with high growth fraction and/or high tumor burden § Burkitt’s; T-ALL; infant ALL; high WBC; bulky extramedullary disease

§ • • Prophylaxis & Management and Tumor Lysis Syndrome May need to start ALL therapy gradually in some cases Classic approach is hydration at ~2 x maintenance, alkalinization of urine (low urine p. H promotes ppt of uric acid crystals), and allopurinol to inhibit xanthine oxidase Use diuretics (furosemide) if urine output low May use rasburicase (recombinant urate oxidase) in those at very high risk • Usually based on WBC, tumor bulk, and growth fraction (Burkitt’s, T-ALL) Alkaline urine decreases calcium phosphate solubility so should not alkalinize urine if TLS

ALL Relapse Content § Know the importance of CR 1 duration as a prognostic factor after relapse § Plan the management of testicular relapse § Know the role of various types of stem cell transplantation (SCT) in management of relapsed ALL

Prognostic Factors Following ALL Relapse § Site of relapse • Bone marrow worse than extramedullary § Time to relapse • Earlier much worse than later § Age at initial diagnosis • Very poor outcome for teenagers that relapse § Immunophenotypic and genetic features • BM relapse of T-ALL has very poor outcome • Relapsed TEL-AML 1+ ALL does relatively well
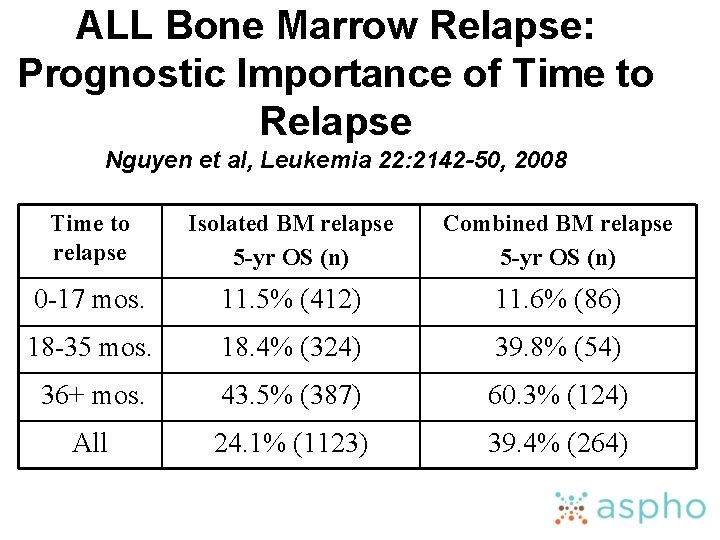
ALL Bone Marrow Relapse: Prognostic Importance of Time to Relapse Nguyen et al, Leukemia 22: 2142 -50, 2008 Time to relapse Isolated BM relapse 5 -yr OS (n) Combined BM relapse 5 -yr OS (n) 0 -17 mos. 11. 5% (412) 11. 6% (86) 18 -35 mos. 18. 4% (324) 39. 8% (54) 36+ mos. 43. 5% (387) 60. 3% (124) All 24. 1% (1123) 39. 4% (264)

ALL: Testicular Relapse § About 2% of boys will have an isolated testicular relapse • 104/5374 boys treated on CCG trials 1988 -2002 (Nguyen et al, Leukemia 2008) § Standard treatment includes intensive systemic therapy plus 2400 c. Gy bilateral testicular irradiation • Risk of contralateral testicular relapse high without irradiation • Leads to sterility and need for hormone replacement § Other studies suggest orchiectomy can obviate

§ Relapsed ALL: Role of Allogeneic Stem Cell Transplantation (SCT)sib and Outcomes now equivalent for matched unrelated donor SCT; use same indications for both § No role of autologous SCT in ALL § SCT indicated for ALL with early (<3 yrs) 1 st marrow relapse § Increasing use of SCT for late marrow relapse with high MRD at end of 1 st course of reinduction therapy § Some believe (disagreement) that SCT indicated for early CNS relapse (<18 mos) nd

Late Effects of ALL Therapy § CNS toxicity (see prior slides) § Anthracycline cardiotoxicity • Related to cumulative dose, which is relatively low for most ALL treatment regimens • DFCI (and COG) data suggest that use of dexrazoxane does not affect treatment efficacy and protects against surrogate markers of cardiotoxicity • Most current chemotherapy regimens use <200 mg/m 2 of anthracyclines so dexrazoxane may be less of an issue than in pediatric solid tumors or

Osteonecrosis (Avascular Necrosis) (see Mattano Lancet Oncology 2012) § A major side effect of modern ALL therapy; 90% occur within 2 years of diagnosis • Weight bearing joints most common sites (hips, knees, ankles) • 90% of pts have hip/knee involvement • 80% of pts have multiple sites involved • 62/143 (43. 4%) pts had operative interventions § Low risk in pts <10 yrs treated with less intensive therapy (<5%) § Much higher risk ≥ 10 yrs and those treated more intensively

GOOD LUCK!
- Slides: 93