Acute Leukaemia Ahmad Shihada Silmi Msc FIBMS Staff

Acute Leukaemia Ahmad Shihada Silmi Msc, FIBMS Staff Specialist in Hematology Medical Technology Department Islamic University of Gaza

DEFINITION Leukemia is a malignant disease characterized by unregulated proliferation of one cell type. – It may involve any of the cell lines or a stem cell common to several cell lines.
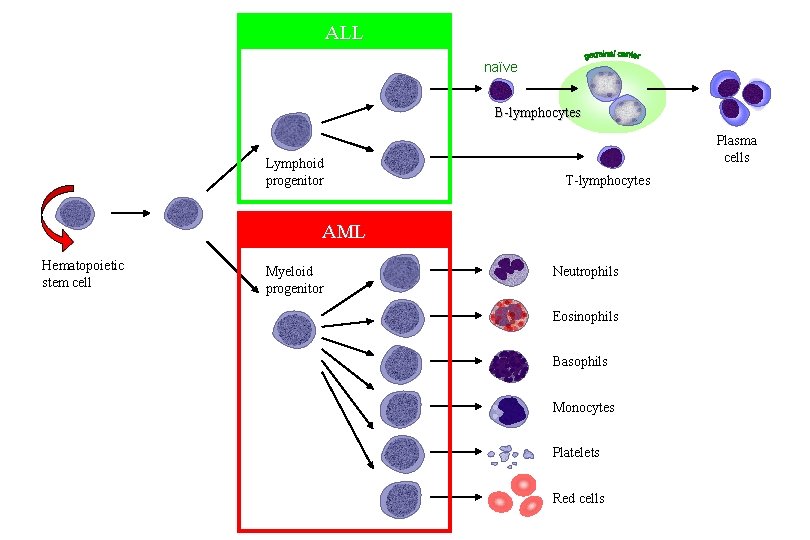
ALL naïve B-lymphocytes Lymphoid progenitor Plasma cells T-lymphocytes AML Hematopoietic stem cell Myeloid progenitor Neutrophils Eosinophils Basophils Monocytes Platelets Red cells

ACUTE vs CHRONIC LEUKAEMIA • Poorly differentiated blast population • Rapidly fatal outcome, if untreated versus • Well differentiated cell population • Associated with longer survival, even if left untreated
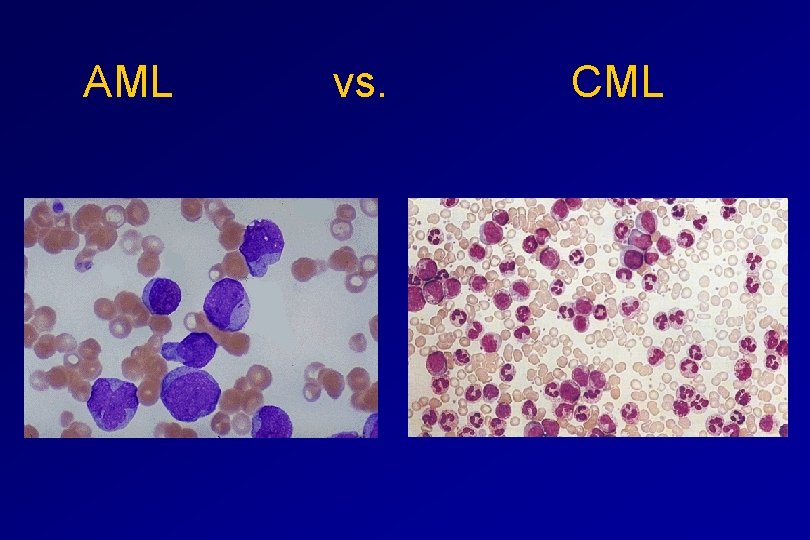
AML vs. CML

ACUTE LEUKAEMIA – Is a result of: Malignant transformation of a stem cell leading to unregulated proliferation and Arrest in maturation at the primitive blast stage. Remember that a blast is the most immature cell that can be recognized as committed to a particular cell line.
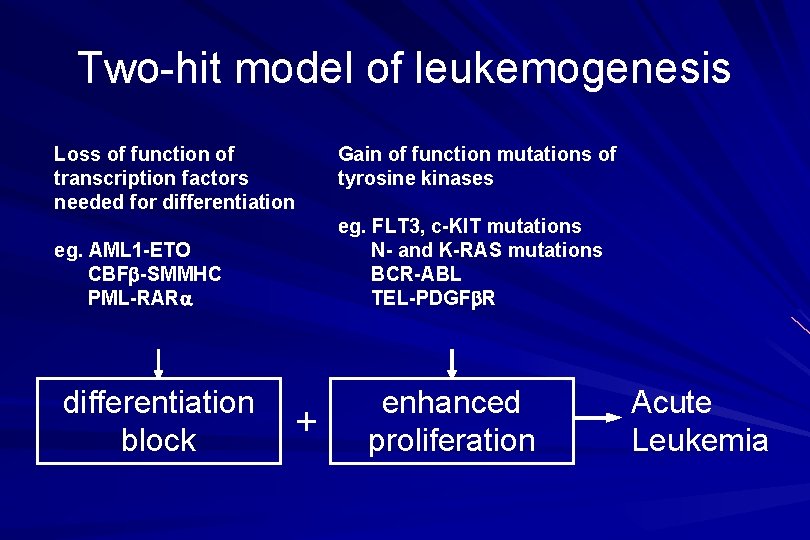
Two-hit model of leukemogenesis Loss of function of transcription factors needed for differentiation eg. FLT 3, c-KIT mutations N- and K-RAS mutations BCR-ABL TEL-PDGFb. R eg. AML 1 -ETO CBFb-SMMHC PML-RARa differentiation block Gain of function mutations of tyrosine kinases + enhanced proliferation Acute Leukemia

Classification of acute leukemias ALL mainly children M > F curable in 70% of children curable in minority of adults AML mainly adults M > F curable in minority of adults

AML Fast Facts n In 2008…(UK) 13, 290 new cases n 8820 deaths n n In comparison… Lung cancer-220, 000 new cases and 160, 000 deaths n Pancreas 42, 000 new cases and 35, 000 deaths n Lymphoma-65, 000 new cases and 19, 000 deaths n n Relatively rare, but devastating disease for which new, less toxic therapies are needed
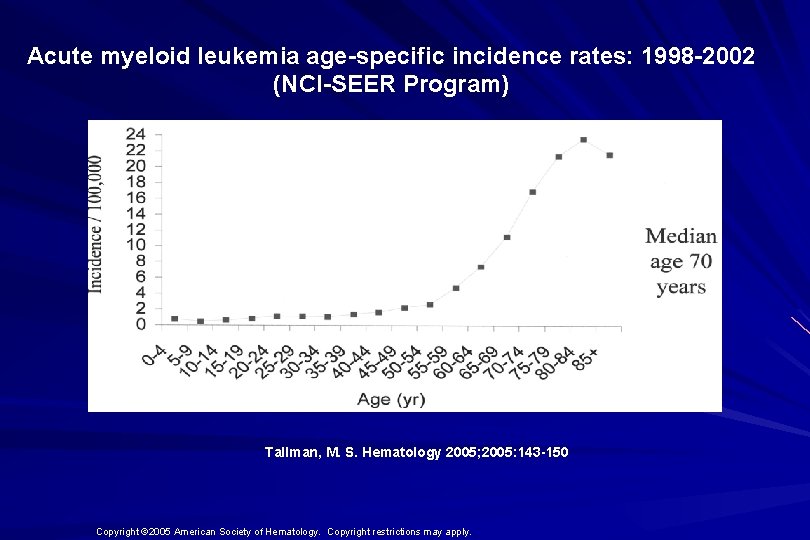
Acute myeloid leukemia age-specific incidence rates: 1998 -2002 (NCI-SEER Program) Tallman, M. S. Hematology 2005; 2005: 143 -150 Copyright © 2005 American Society of Hematology. Copyright restrictions may apply.

Biology of Leukemia Resistance to apoptosis (even when DNA is damaged) De-regulation of growth Differentiation block

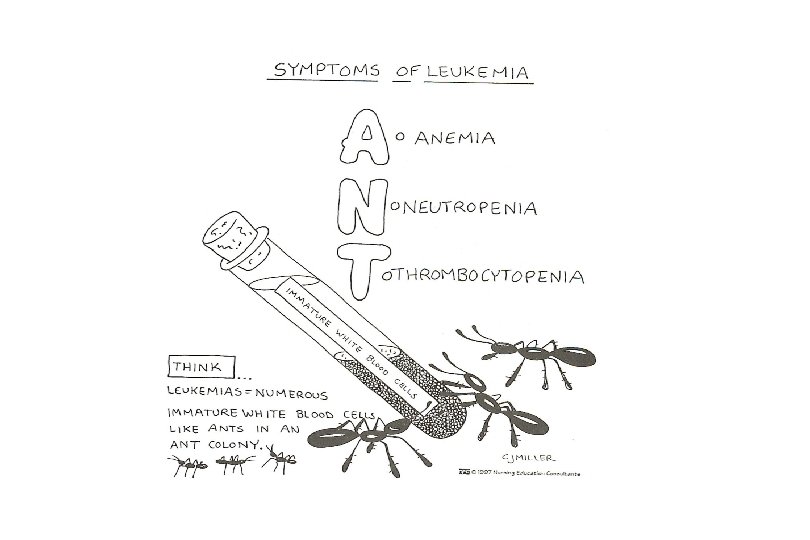
Clinical presentation: Will present with sign or symptoms related to : – Pancytopenia: WBC infection. Hb anemia. platelets bleeding. – Organ infiltration: Lymphadenopathy. More common with ALL than AML. Splenomegally. Hepatomegally. CNS: 5 -10% of patient with ALL

Acute Lymphoblastic Leukaemia Cancer of the blood affecting the white blood cell known as LYMPHOCYTES. Commonest in the age 2 -10 years Peak at 3 -4 years. Incidence decreases with age, and a secondary rise after 40 years. In children - most common malignant disease 85% of childhood leukaemia

Acute Lymphoblastic Leukemia Specific manifestation : *bone pain, arthritis *lymphadenopathy *hepatosplenomegaly *mediastinal mass *testicular swelling *meningeal syndrome

Acute Myeloid Leukemia Arise from the malignant transformation of a myeloid precursor Rare in childhood (10%-15%) The incidence increases with age 80% in adults Most frequent leukemia in neonate

Acute Myeloid Leukemia Specific manifestation : - Gum hypertrophy – Hepatosplenomegaly – Skins deposit – Lymphadenopathy – Renal damage – DIVC

On presentation: 1012 leukemia cells (1 liter volume). ~70% of adults treated with induction chemotherapy will achieve remission. – Remission: Normal counts (platelets>100 K, ANC>1000, transfusion independent, blasts < 5%)

AML: Working hypotheses AML is a stem cell disease: – The “bulk” cells circulating in peripheral blood are mostly nondividing, partially differentiated – Leukemia derived from more primitive (more multi-potent) stem cells will be harder to cure. – The features of the bulk cells offer clues as to how primitive the leukemia stem cell is… How much chemotherapy can you give? – Not so much that the normal stem/progenitors in the body (gut, marrow, etc…) can’t recover.
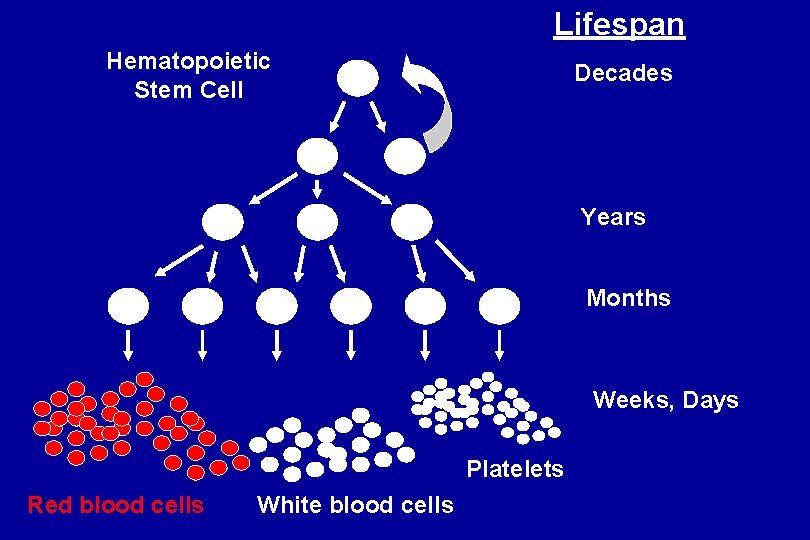
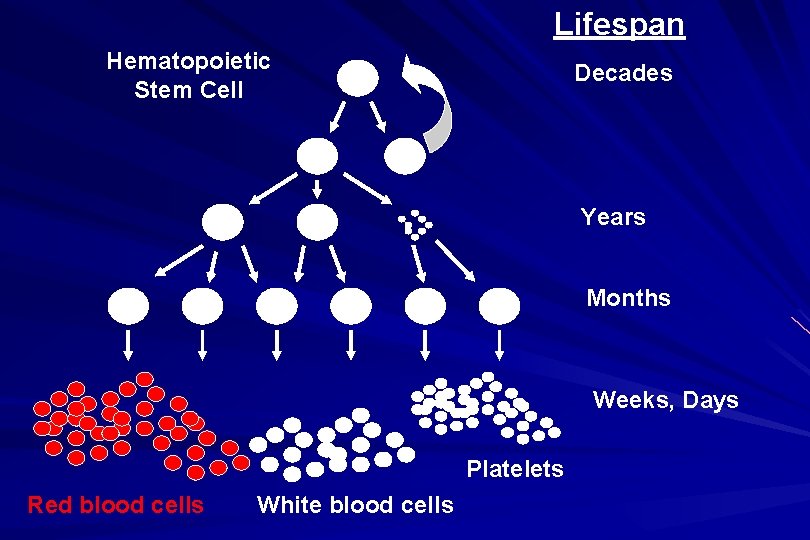
Lifespan Hematopoietic Stem Cell Decades Years Months Weeks, Days Platelets Red blood cells White blood cells
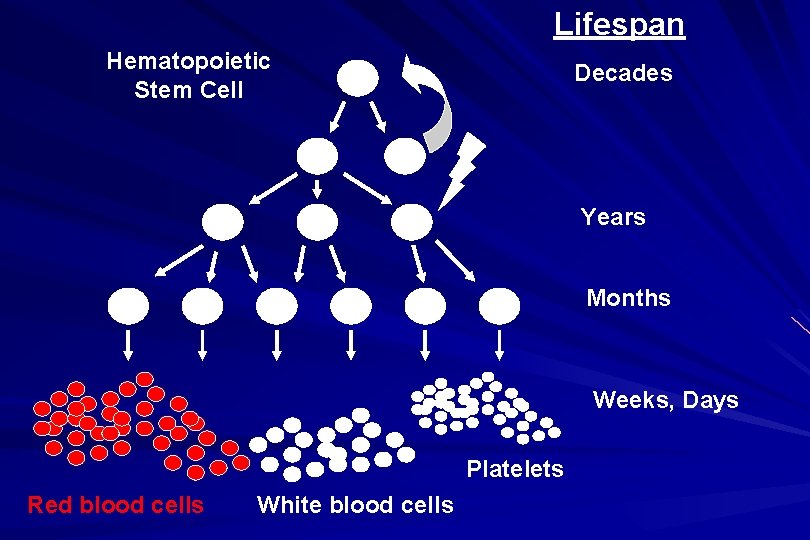
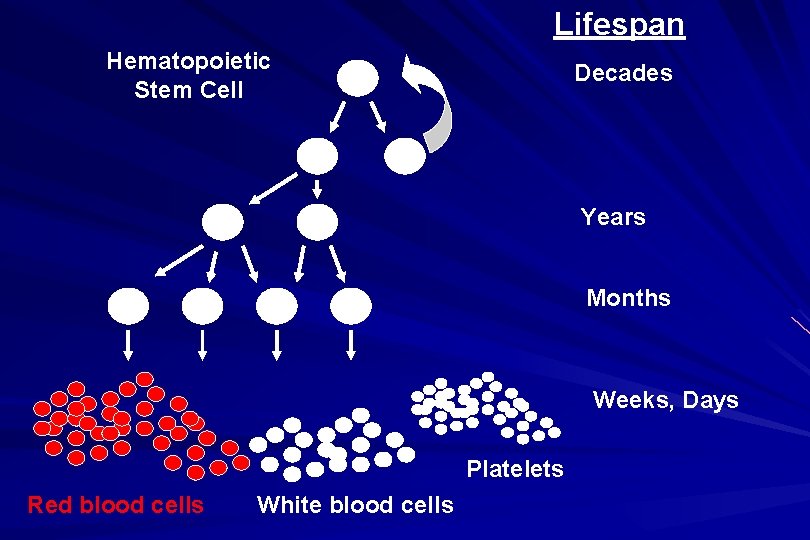
Lifespan Hematopoietic Stem Cell Decades Years Months Weeks, Days Platelets Red blood cells White blood cells
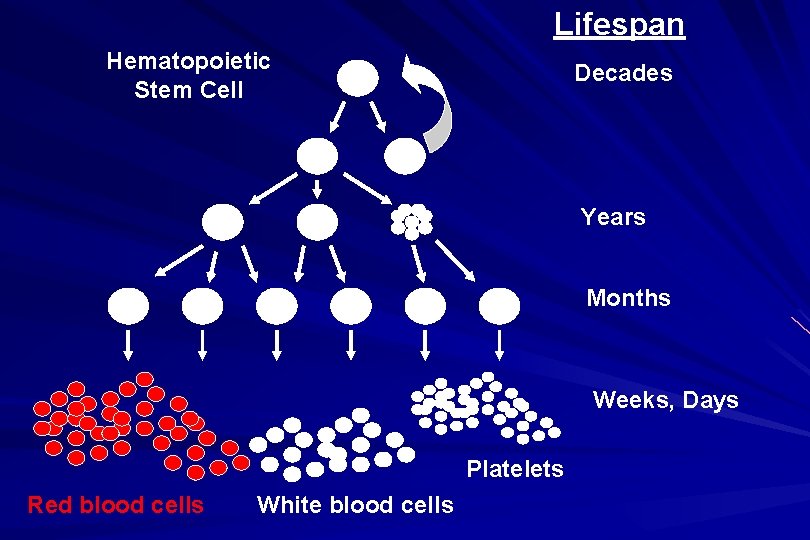
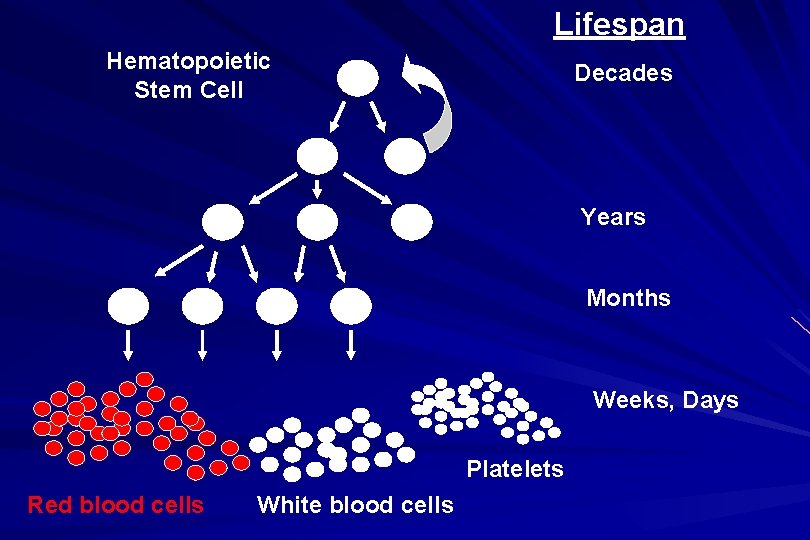
Lifespan Hematopoietic Stem Cell Decades Years Months Weeks, Days Platelets Red blood cells White blood cells
Lifespan Hematopoietic Stem Cell Decades Years Months Weeks, Days Platelets Red blood cells White blood cells
Lifespan Hematopoietic Stem Cell Decades Years Months Weeks, Days Platelets Red blood cells White blood cells
Lifespan Hematopoietic Stem Cell Decades Years Months Weeks, Days Platelets Red blood cells White blood cells
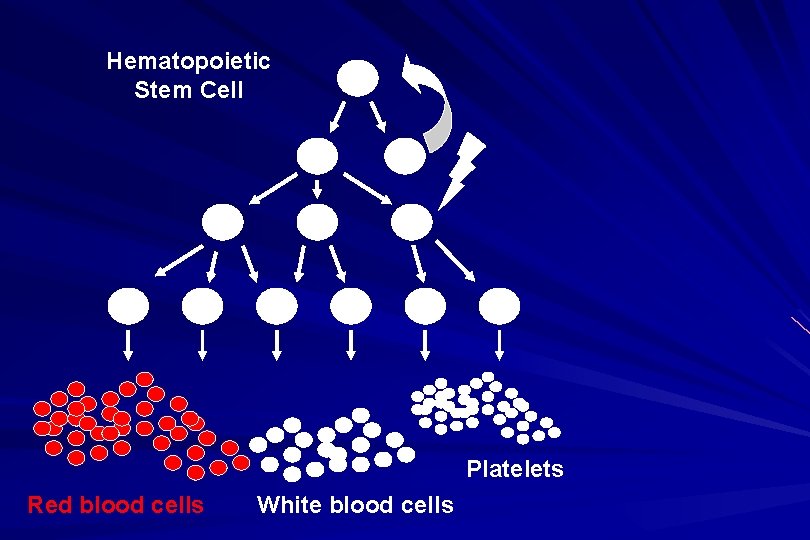
Hematopoietic Stem Cell Platelets Red blood cells White blood cells
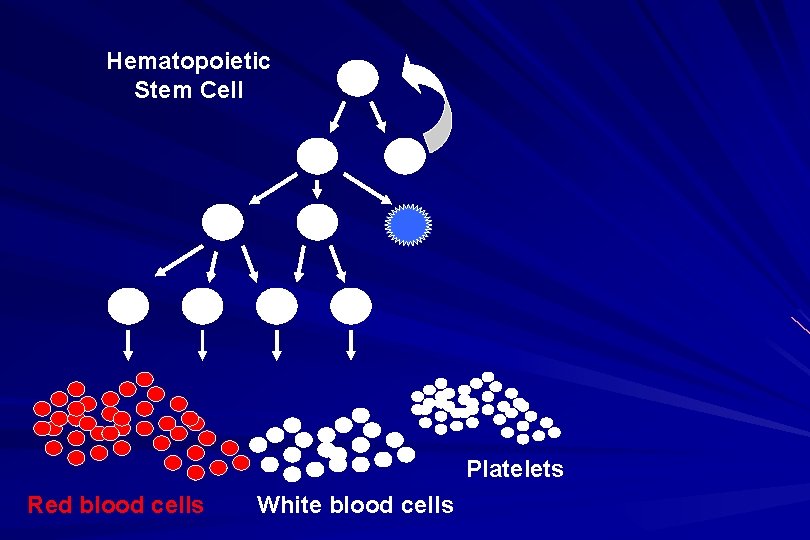
Hematopoietic Stem Cell Platelets Red blood cells White blood cells
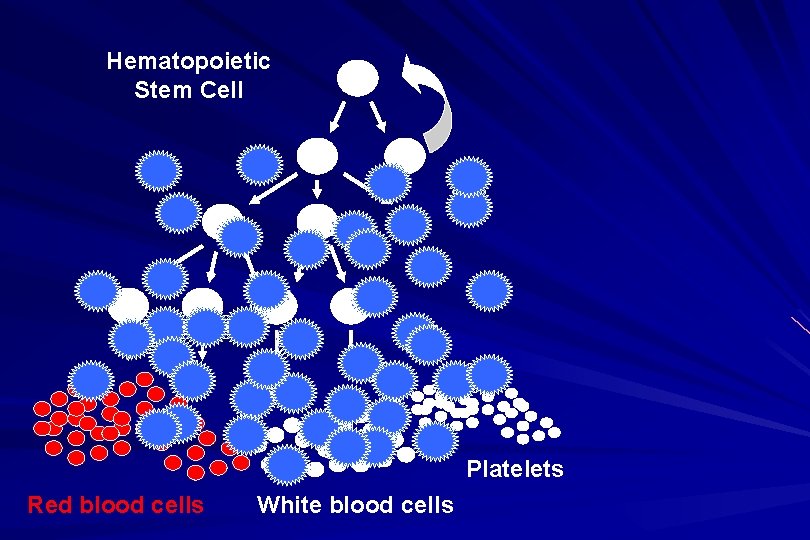
Hematopoietic Stem Cell Platelets Red blood cells White blood cells
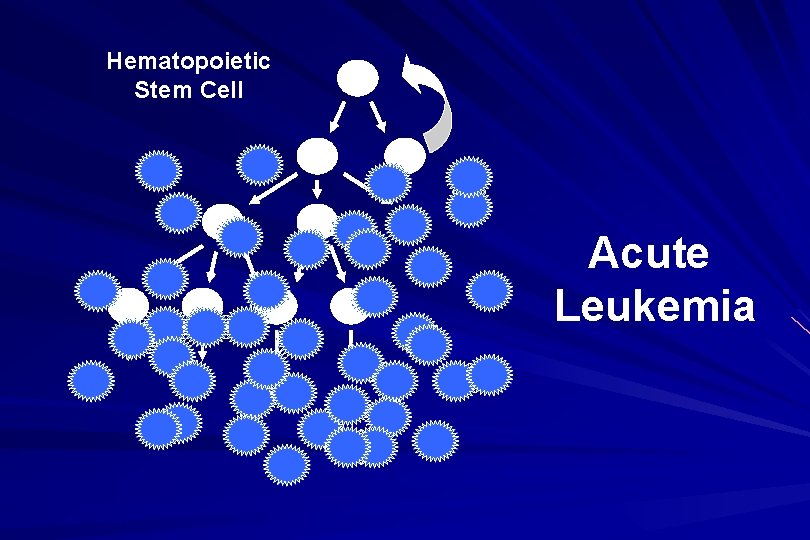
Hematopoietic Stem Cell Acute Leukemia
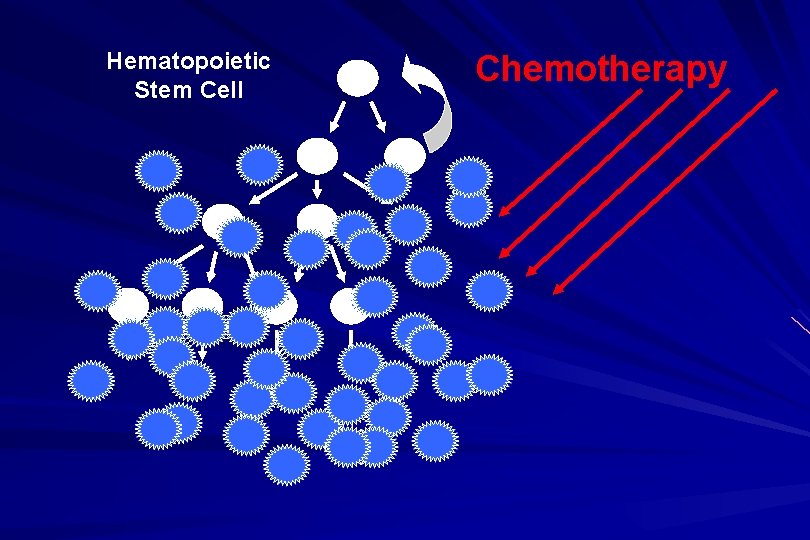
Hematopoietic Stem Cell Chemotherapy
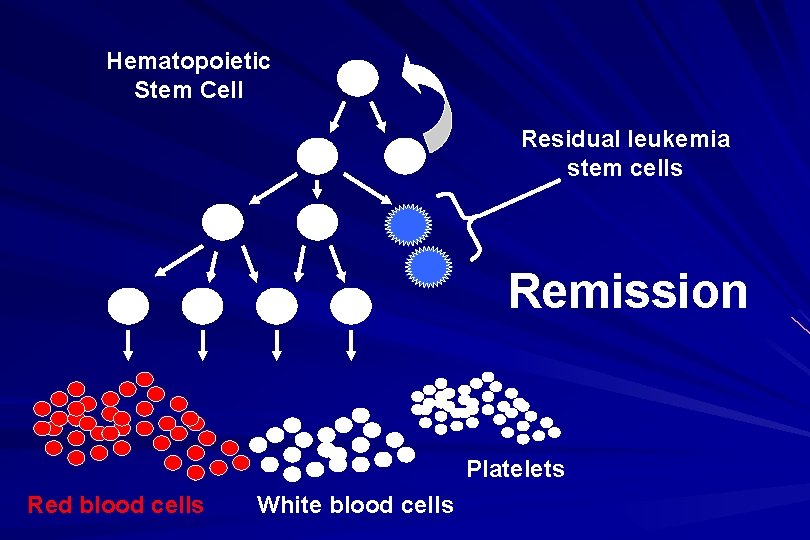
Hematopoietic Stem Cell Residual leukemia stem cells Remission Platelets Red blood cells White blood cells

AETIOLOGY Ionizing Radiation • Atomic Bombs - Hiroshima and Nagasaki, Utah, Chernobyl, Three Mile Island fall-out - within 1. 5 - 3 years - peak incidence 5 -10 years - dose dependent

AETIOLOGY • Occupational exposure - radiologists - nuclear industry • Diagnostic radiographic procedures - Thorium dioxide - foetal exposure from radiographic pelvimetry • Natural background radiation (EM)

AETIOLOGY • Chemicals - benzene - rubber industry - pesticides • Viruses - EBV - Human retroviruses (HTLV 1&2)

AETIOLOGY Host factors • - familial occurrence - genetic disorders (Down’s syndrome)

AETIOLOGY Medications • - Chemotherapy (Alkylating agents) within 4 -5 years proportionate to dose and duration - Radiotherapy: adds extra risk - Immunosuppression

CLASSIFICATION OF ACUTE LEUKAEMIA Based on cell origin - myeloid: some features of granulocyte and/or monocyte development - lymphoid: from lymphocyte precursors - ambiguous lineage: undifferentiated, bilineal or biphenotypic

CLASSIFICATION OF ACUTE LEUKAEMIA • Standardisation of treatment • Some acute leukaemias have peculiar features - ALL and lymph node, thymus and CNS involvement - APML and bleeding

CLASSIFICATION OF ACUTE LEUKAEMIA • Peripheral blood morphology • Bone marrow morphology • Immunophenotyping • Cytogenetics • Molecular studies

MORPHOLOGY Romanowsky stained smears of peripheral blood or bone marrow Nuclear chromatin pattern is most important
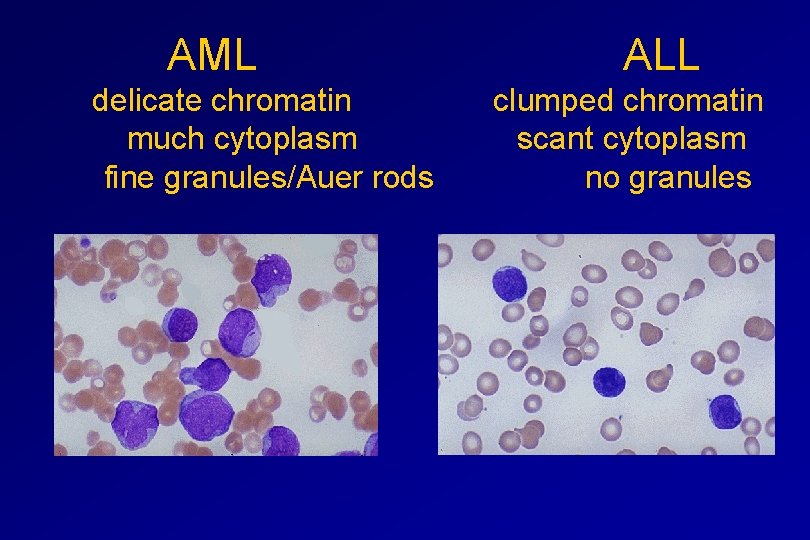
AML ALL delicate chromatin clumped chromatin much cytoplasm scant cytoplasm fine granules/Auer rods no granules

BASIS OF FAB CLASSIFICATION 30 % blast in bone marrow Morphology of blast – Romanowsky stain Cytochemical Stains – Myeloperoxidase – Non specific esterase

CLASSIFICATION OF ACUTE LEUKAEMIAS ALL • ALL/L 1: 80% - small blast cells - high N/C ratio ALL/L 2 : 20% • - larger cells, heterogeneous - lower N/C ratio
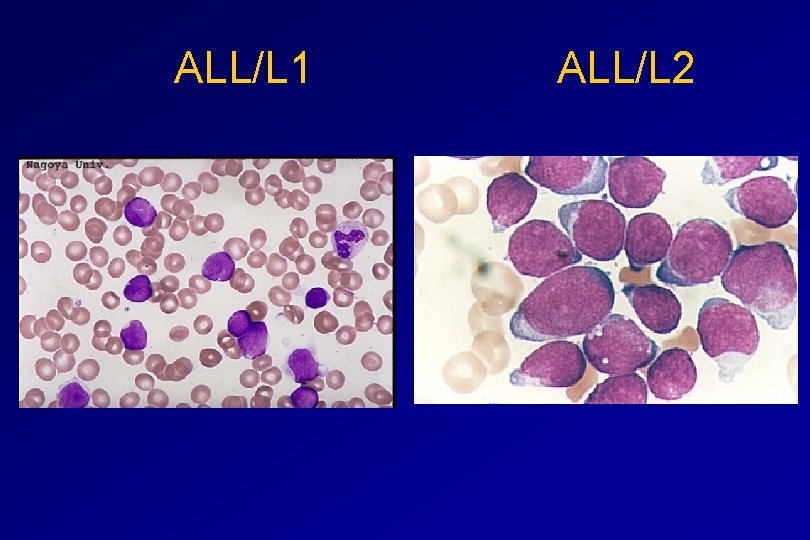
ALL/L 1 ALL/L 2

CLASSIFICATION OF ACUTE LEUKAEMIAS ALL • ALL/L 3 : 3 -5% - large, homogeneous cells - fine chromatin, prominent nucleoli - intensely basophilic cytoplasm, vacuoles
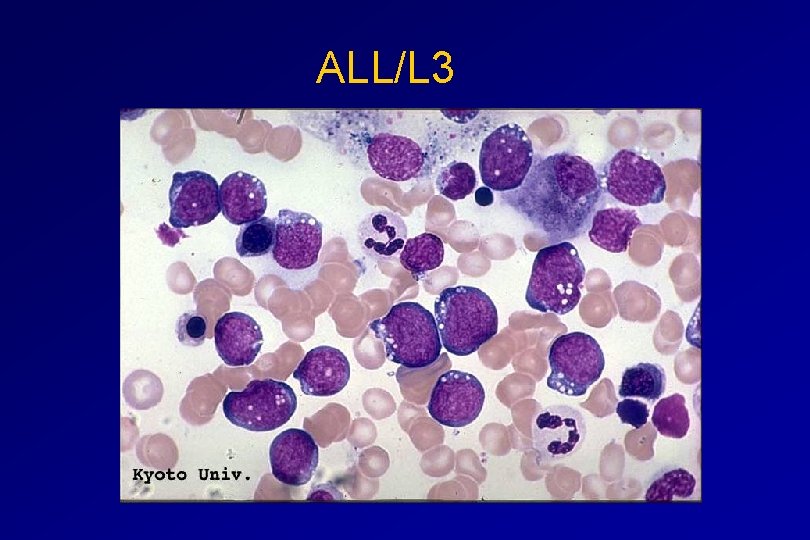
ALL/L 3
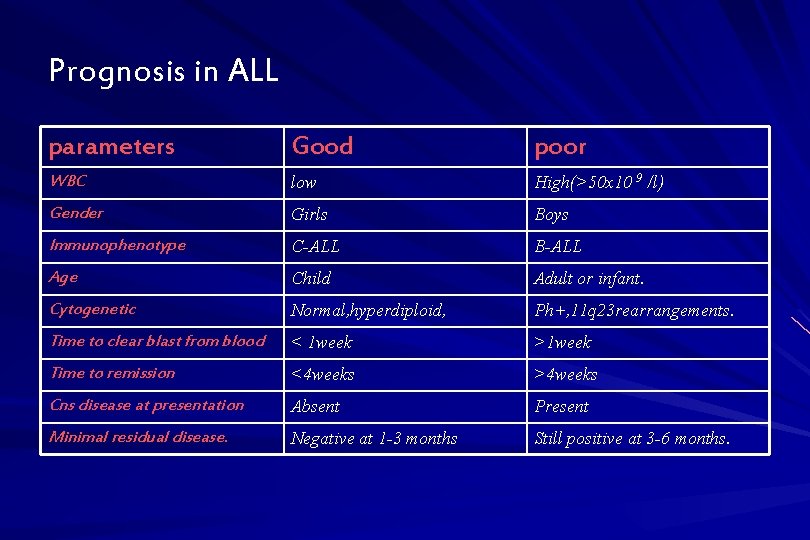
Prognosis in ALL parameters Good poor WBC low High(>50 x 10 9 /l) Gender Girls Boys Immunophenotype C-ALL B-ALL Age Child Adult or infant. Cytogenetic Normal, hyperdiploid, Ph+, 11 q 23 rearrangements. Time to clear blast from blood < 1 week >1 week Time to remission <4 weeks >4 weeks Cns disease at presentation Absent Present Minimal residual disease. Negative at 1 -3 months Still positive at 3 -6 months.
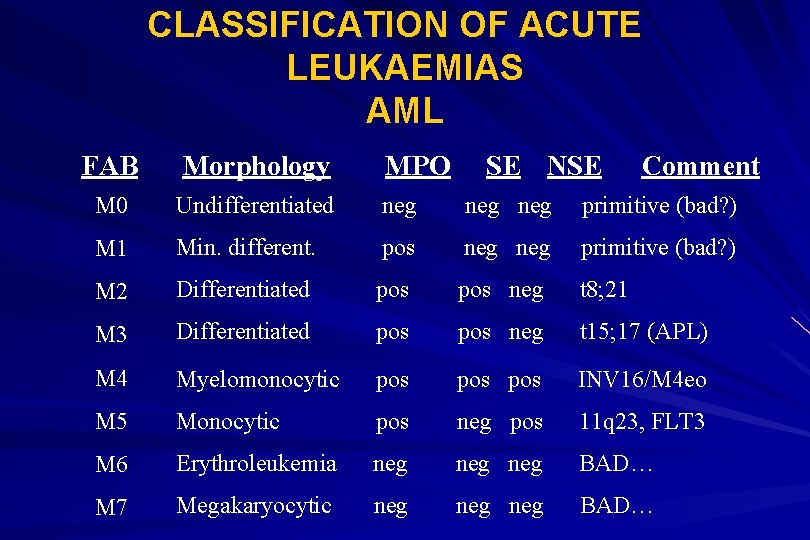
CLASSIFICATION OF ACUTE LEUKAEMIAS AML FAB Morphology MPO SE NSE Comment M 0 Undifferentiated neg neg primitive (bad? ) M 1 Min. different. pos neg primitive (bad? ) M 2 Differentiated pos neg t 8; 21 M 3 Differentiated pos neg t 15; 17 (APL) M 4 Myelomonocytic pos pos INV 16/M 4 eo M 5 Monocytic pos neg pos 11 q 23, FLT 3 M 6 Erythroleukemia neg neg BAD… M 7 Megakaryocytic neg neg BAD…

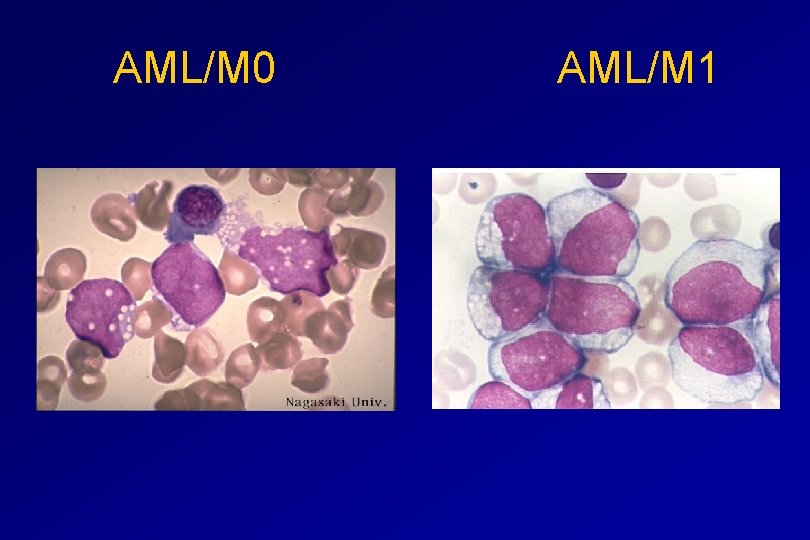
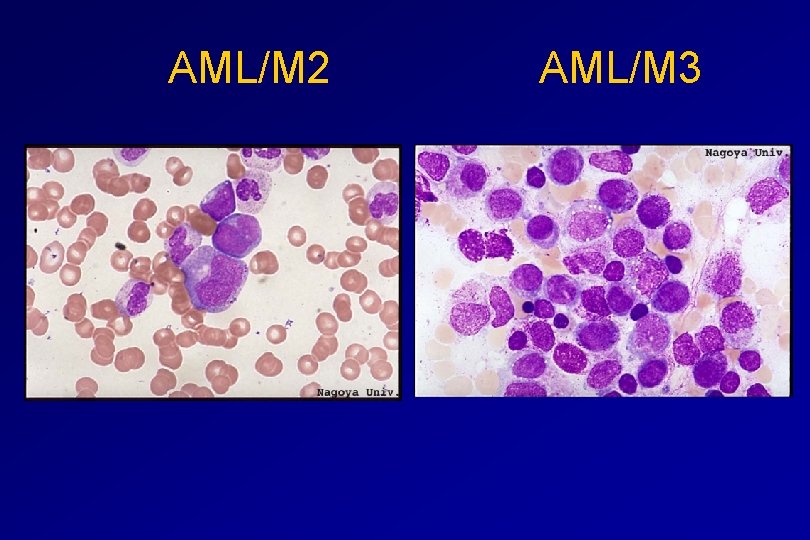
CLASSIFICATION OF ACUTE LEUKAEMIAS AML Granulocytic differentiation 60% - M 0 - Acute myeloblastic leukaemia with minimal differentiation - M 1 - Acute myeloblastic leukaemia with minimal maturation - M 2 - Acute myeloblastic leukaemia with significant maturation - M 3 - Acute promyelocytic leukaemia
AML/M 0 AML/M 1
AML/M 2 AML/M 3

Monocytic differentiation - M 4 - Acute myelomonocytic leukaemia 25% - M 5 - Acute monocytic leukaemia 10% Erythroid differentiation - M 6 - Acute erythroleukaemia <5% Megakaryocytic differentiation - M 7 -Acute megakaryoblastic leukaemia <5%
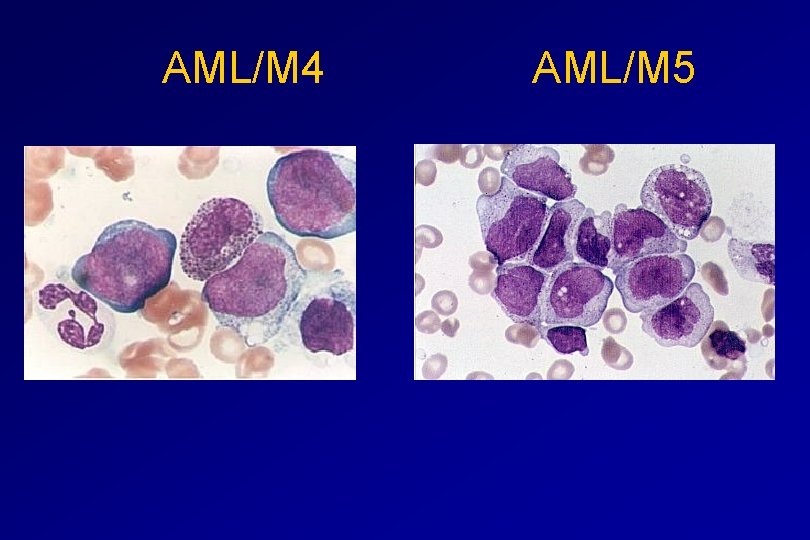
AML/M 4 AML/M 5
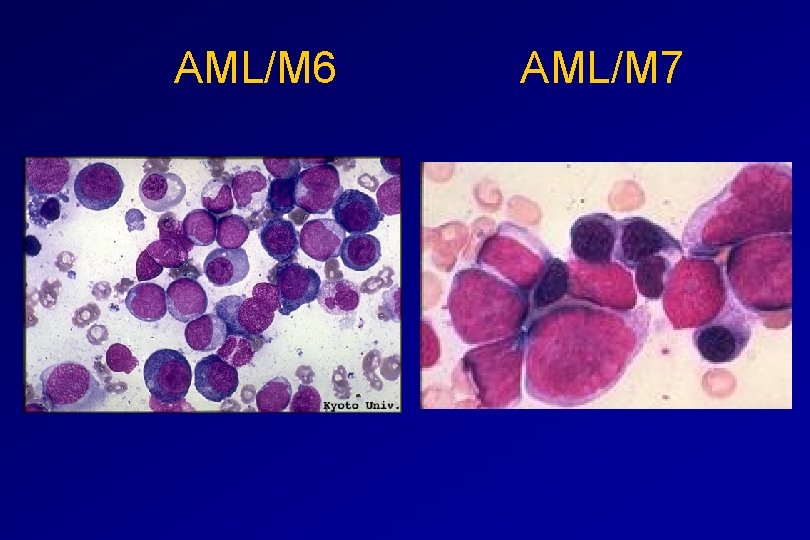
AML/M 6 AML/M 7

IMMUNOPHENOTYPING objective • Measured by flow cytometry as intensity with which a fluorescence - conjugated antibody stains cell surface proteins. • Indicates lineage commitment • Indicates position along the normal haemopoietic pathway at the time of transformation

procedure Flow Cytometry: – Sample of whole blood in green or purple top – RBCs lysed, WBC pellet washed. – Antibodies with fluorochrome labels added. – Stained cells analyzed by FACS – Whole process can be done in two hours.
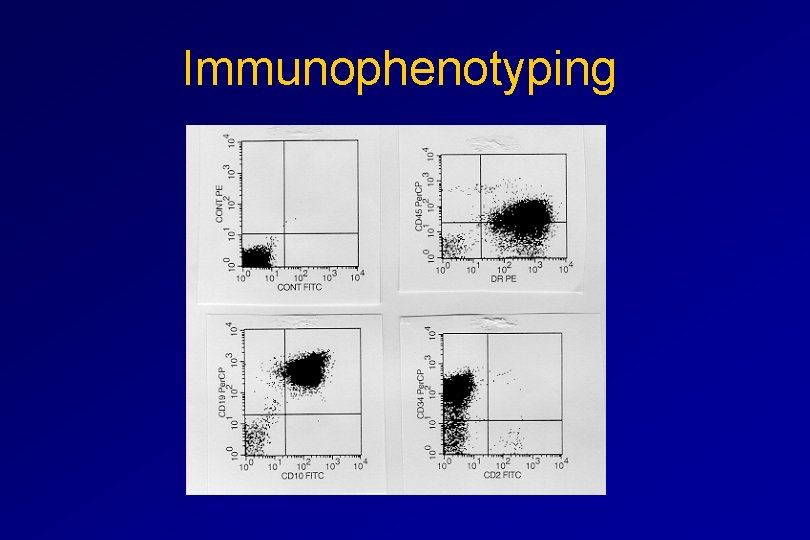
Immunophenotyping

Immunophenotyping MYELOID CD 13 CD 34 LYMPHOID B lineage CD 19 CD 22 CD 10 T lineage CD 7 CD 3 Td. T

CYTOGENETICS IN ACUTE LEUKAEMIA • Translocations, deletions, inversions etc • Diagnosis • Prognosis • Minimal Residual Disease (MRD)
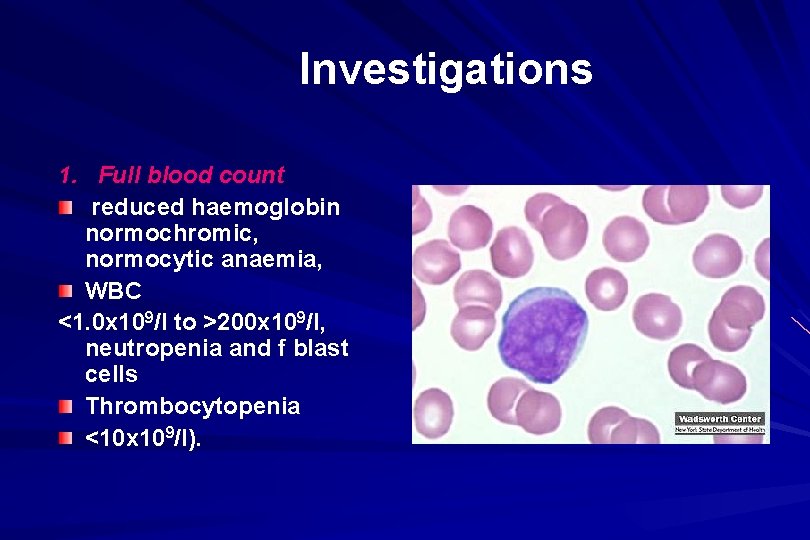
Investigations 1. Full blood count reduced haemoglobin normochromic, normocytic anaemia, WBC <1. 0 x 109/l to >200 x 109/l, neutropenia and f blast cells Thrombocytopenia <10 x 109/l).
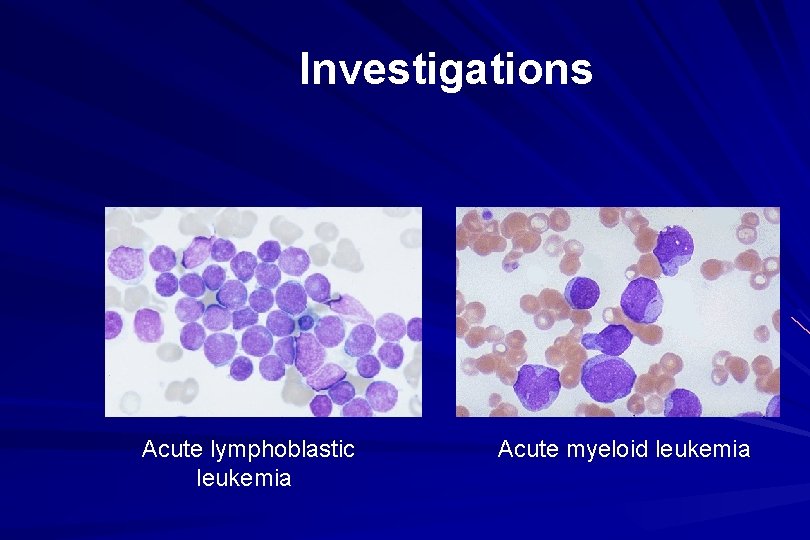
Investigations Acute lymphoblastic leukemia Acute myeloid leukemia

Investigations ALL(Lymphoblast) – Blast size : small – Cytoplasm: Scant – Chromatin: Dense – Nucleoli : Indistinct – Auer-rods: Never present AML (Myeloblast) – Large – Moderate – Fine, Lacy – Prominent – Present in 50%
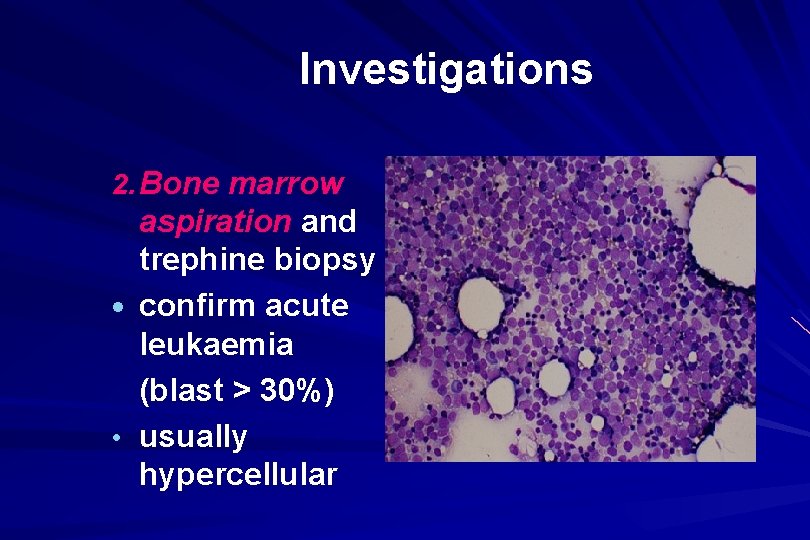
Investigations 2. Bone marrow aspiration and trephine biopsy · confirm acute leukaemia (blast > 30%) • usually hypercellular

Investigations 3. Cytochemical staining ) Peroxidase : * negative ALL * positive AML Positive for myeloblast

Sudan black n Sudan black stains phospholipids, neutral fats and sterols found in primary and secondary granules of granulocytic cells and to a lesser extent in monocytic lysosomes. Rare positives occur in lymphoid cells

Nonspecific Esterase n Nonspecific esterase – is used to identify monocytic cells which are diffusely positive. T lymphocytes may have focal staining

Leukocyte Alkaline phosphatase n Leukocyte alkaline phosphatase – is located in the tertiary granules of segmented neutrophils, bands and metamyelocytes. The LAP score is determined by counting 100 mature neutrophils and bands. Each cell is graded from 0 to 5. The total LAP score is calculated by adding up the scores for each cell.
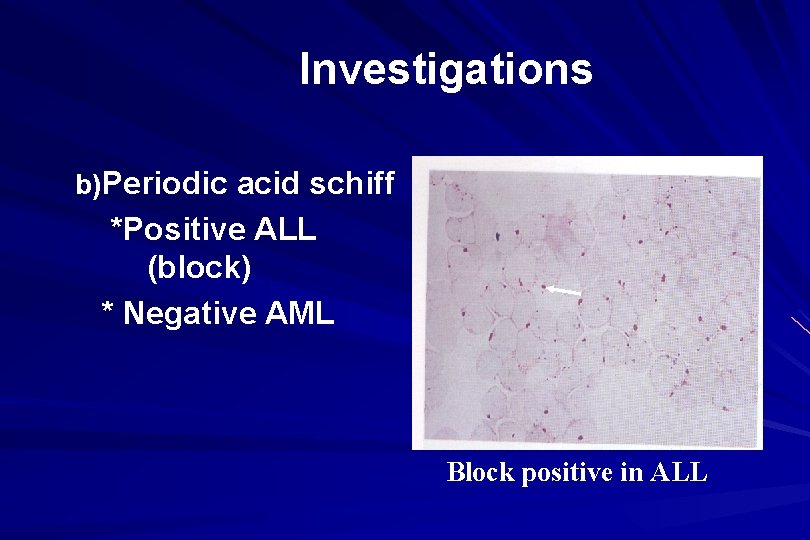
Investigations b)Periodic acid schiff *Positive ALL (block) * Negative AML Block positive in ALL
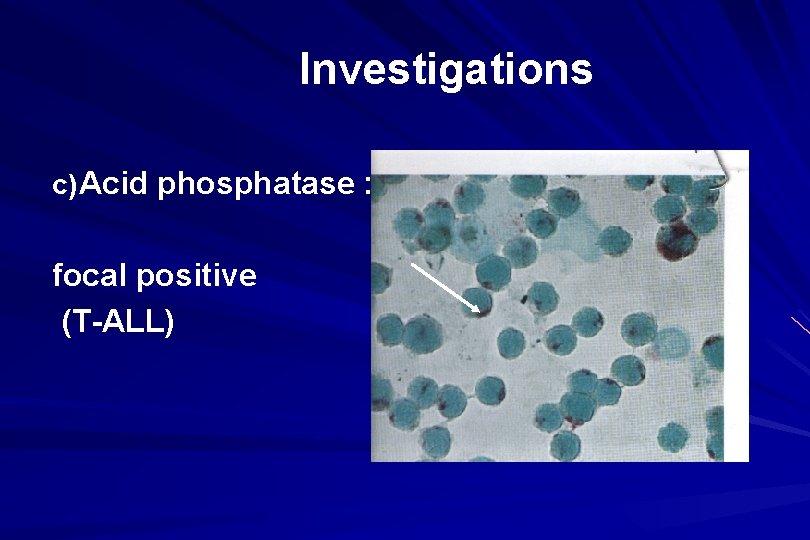
Investigations c) Acid phosphatase : focal positive (T-ALL)

Investigations 4. Immunophenotyping · identify antigens present on the blast cells · determine whether the leukaemia is lymphoid or myeloid(especially important when cytochemical markers are negative or equivocal. E. g : AML-M 0) · differentiate T-ALL and B-ALL

Monoclonal antibodies(Mc. Ab) are group based on antigen on the leucocytes and are recognised under a cluster of differentiation(CD).

Certain antigens have prognostic significance Rare cases of biphenotypic where both myeloid and lymphoid antigen are expressed Able to identify the subtype of leukemia. E. g : AML-M 7 has a specific surface marker of CD 61 etc

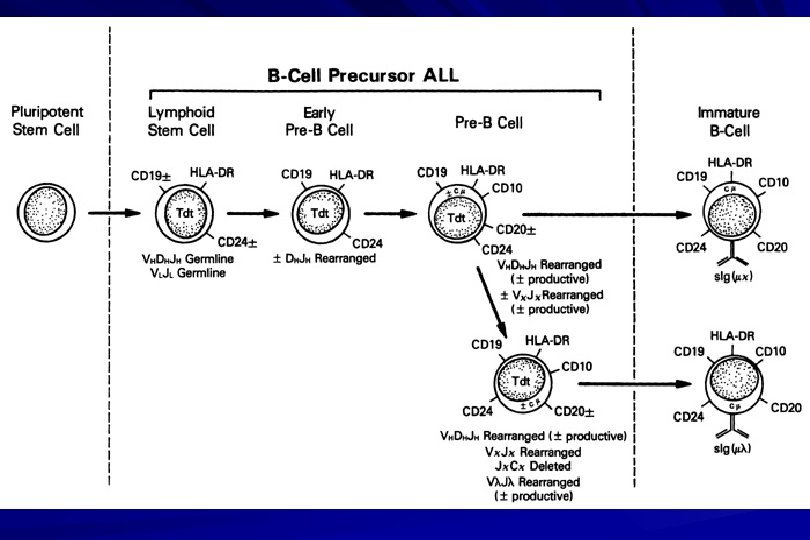


Monoclonal antibodies(Mc. Ab) are recognised under a cluster of differentiation(CD). MONOCLONAL ANTIBODIES USED FOR CHARACTERISATION OF ALL AND AML. Monoclonal antibodies AML : CD 13, CD 33 ALL : B-ALL CD 10, CD 19, CD 22 T-ALL CD 3, CD 7

Investigations 5. Cytogenetics and molecular studies detect abnormalities within the leukaemic clone diagnostic or prognostic value E. g : the Philadelphia chromosome : the product of a translocation between chromosomes 9 and 22 confers a very poor prognosis in ALL

Investigations COMMON CHROMOSOME ABNORMALITIES ASSOCIATED WITH ACUTE LEUKEMIA t(8; 21) AML with maturation (M 2) t(15; 17) AML-M 3(APML) Inv 16 AML-M 4 t(9; 22) Chronic granulocytic leukemia t(8; 14) B-ALL

Others Invx 6. Biochemical screening leucocyte count very high - renal impairment and hyperuricaemia 7. Chest radiography · mediastinal mass - present in up to 70% of patients with T -ALL In childhood ALL bone lesions may also seen.

Others Invx 8. Lumbar puncture initial staging inv. to detect leukaemic cells in the cerebrospinal fluid, indicating involvement of the CNS Done in acute lymphoblastic leukemia
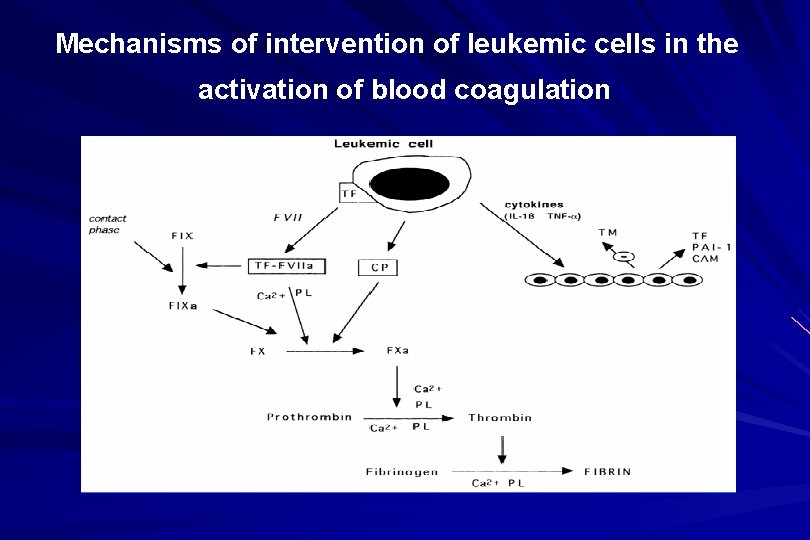
Mechanisms of intervention of leukemic cells in the activation of blood coagulation

GENETICS Leukemic mutations interfere with transcription factor functions, abrogate cell differentiation, and support proliferation. As a consequence, the blood is flooded with immature, nonfunctional cell types.
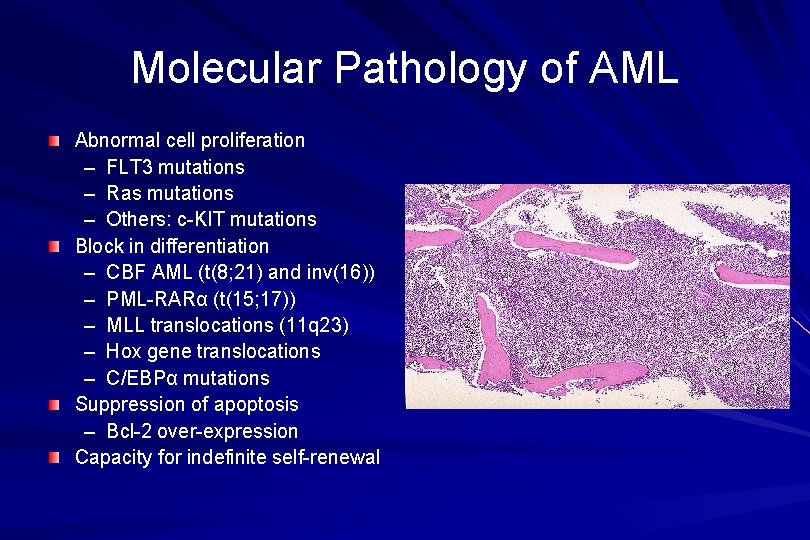
Molecular Pathology of AML Abnormal cell proliferation – FLT 3 mutations – Ras mutations – Others: c-KIT mutations Block in differentiation – CBF AML (t(8; 21) and inv(16)) – PML-RARα (t(15; 17)) – MLL translocations (11 q 23) – Hox gene translocations – C/EBPα mutations Suppression of apoptosis – Bcl-2 over-expression Capacity for indefinite self-renewal

ACUTE LEUKAEMIA EXAMPLES of SOME CYTOGENETIC and MOLECULAR ABNORMALITIES
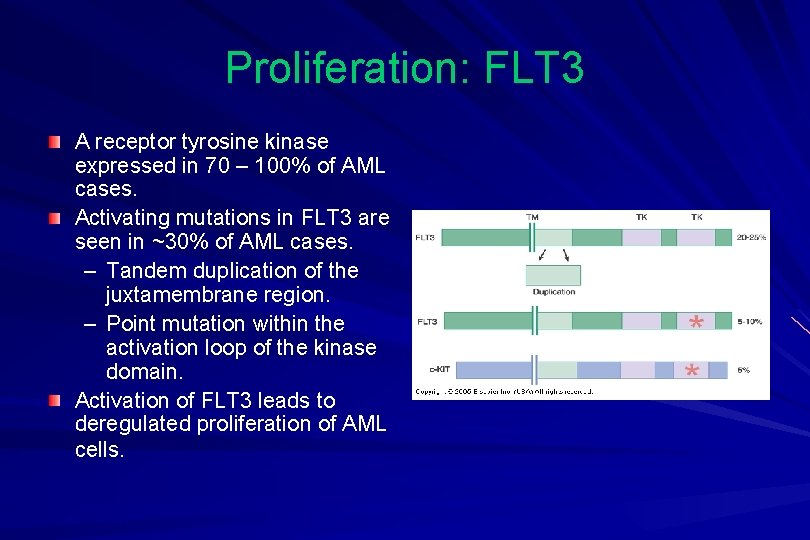
Proliferation: FLT 3 A receptor tyrosine kinase expressed in 70 – 100% of AML cases. Activating mutations in FLT 3 are seen in ~30% of AML cases. – Tandem duplication of the juxtamembrane region. – Point mutation within the activation loop of the kinase domain. Activation of FLT 3 leads to deregulated proliferation of AML cells.

Proliferation: Ras Small guanine nucleotide binding proteins that play a role in several cell signaling pathways. Activating point mutations seen in ~25% of AML cases. – N-Ras: 10 – 25% – K-Ras: 5 – 15% – H-Ras: Rare Activation of Ras leads to deregulated cell proliferation.

Differentiation: t(15, 17)and the PML/RAR gene Acute Promyelocytic Leukaemia - gene: retinoic acid receptor (RAR ) gene fused with PML gene (PML/RAR gene) This fusion PML-RAR protein is responsible for preventing immature myeloid cells from differentiating into more mature cells.
![t(8; 21) - [AML 1 -ETO] The AML 1 gene encodes the DNA-binding t(8; 21) - [AML 1 -ETO] The AML 1 gene encodes the DNA-binding](http://slidetodoc.com/presentation_image/13dbf0167c8fe2d7775fa5349ab1f58f/image-88.jpg)
t(8; 21) - [AML 1 -ETO] The AML 1 gene encodes the DNA-binding subunit of the AML 1/CBFb core binding factor transcription complex, whereas ETO encodes the mammalian homologue of the Drosophila protein Nervy. AML 1 and ETO are both involved in transcriptional regulation of genes in hematopoietic precursor cells. AML 1 -ETO fusion protein represses genes whose transcription is normally activated by AML/CBFb.
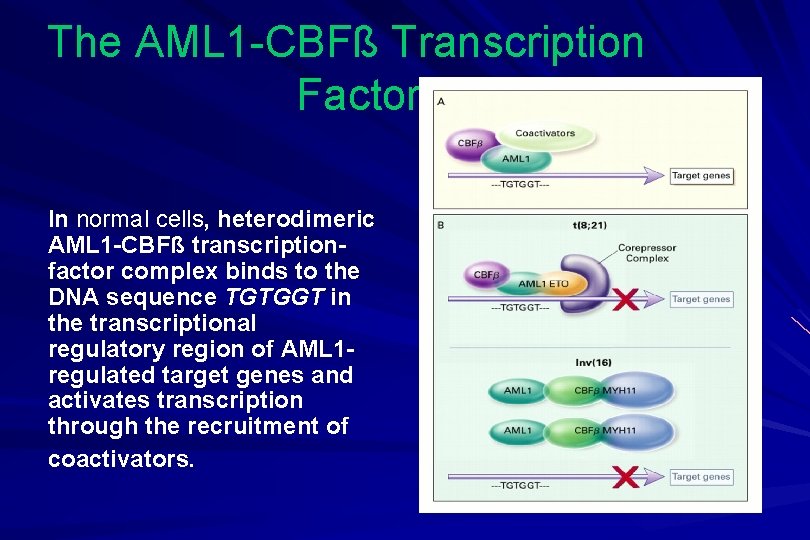
The AML 1 -CBFß Transcription Factor In normal cells, heterodimeric AML 1 -CBFß transcriptionfactor complex binds to the DNA sequence TGTGGT in the transcriptional regulatory region of AML 1 regulated target genes and activates transcription through the recruitment of coactivators.
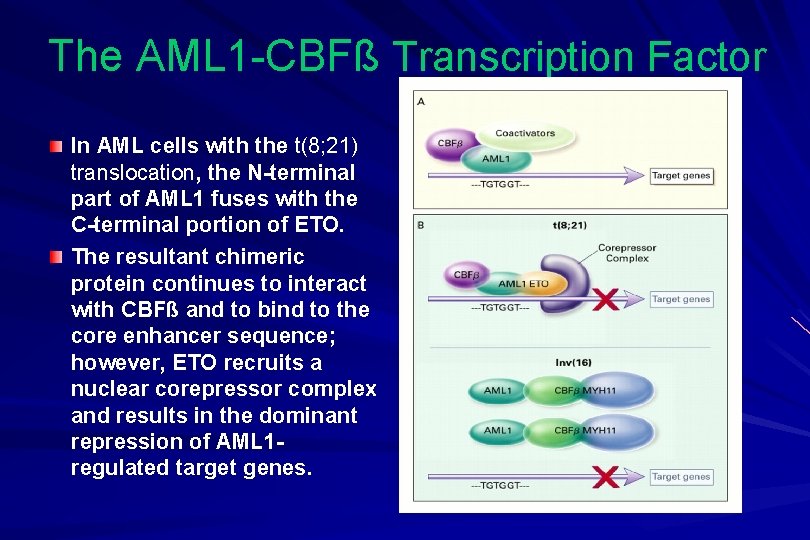
The AML 1 -CBFß Transcription Factor In AML cells with the t(8; 21) translocation, the N-terminal part of AML 1 fuses with the C-terminal portion of ETO. The resultant chimeric protein continues to interact with CBFß and to bind to the core enhancer sequence; however, ETO recruits a nuclear corepressor complex and results in the dominant repression of AML 1 regulated target genes.
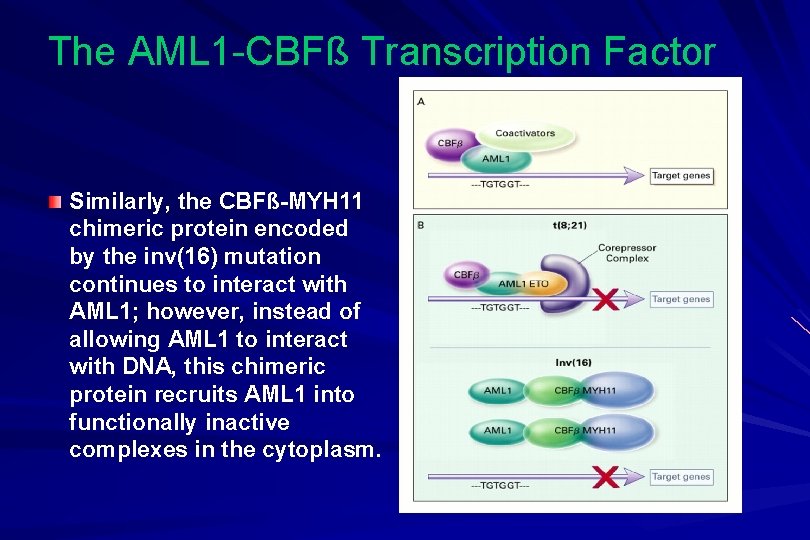
The AML 1 -CBFß Transcription Factor Similarly, the CBFß-MYH 11 chimeric protein encoded by the inv(16) mutation continues to interact with AML 1; however, instead of allowing AML 1 to interact with DNA, this chimeric protein recruits AML 1 into functionally inactive complexes in the cytoplasm.

In summary Differentiation: Core Binding Factor-β CBFβ is located at 16 q 22. Inv(16)(p 13 q 22) and t(16; 16)(p 13; q 22): CBFβMYH 11 fusion gene that functions as a dominant negative inhibitor of CBF. The result is the suppression of transcription of genes crucial for hematopoiesis / differentiation.
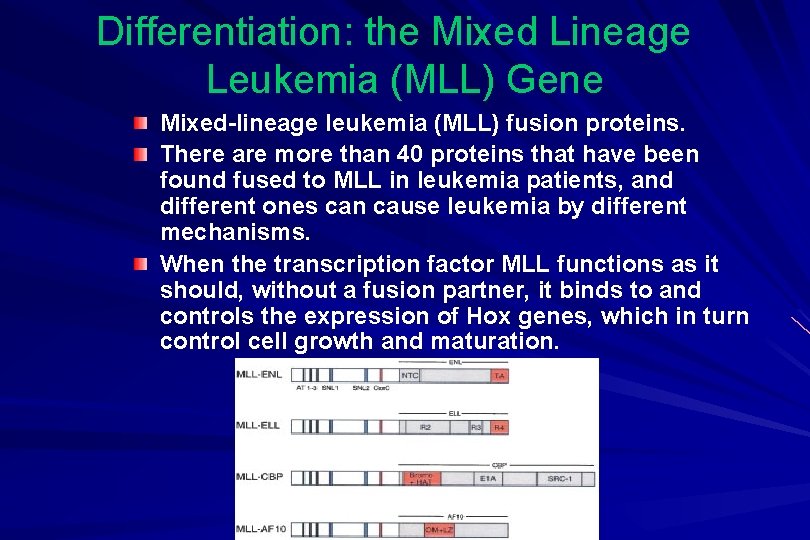
Differentiation: the Mixed Lineage Leukemia (MLL) Gene Mixed-lineage leukemia (MLL) fusion proteins. There are more than 40 proteins that have been found fused to MLL in leukemia patients, and different ones can cause leukemia by different mechanisms. When the transcription factor MLL functions as it should, without a fusion partner, it binds to and controls the expression of Hox genes, which in turn control cell growth and maturation.

Differentiation: the Mixed Lineage Leukemia (MLL) Gene MLL is a protein that regulates Hox gene expression. Hox family members play critical roles in transcription of genes involved in hematopoiesis. Normal MLL requires cleavage by taspase into its mature form → dysregulated Hox gene expression → block in differentiation.

t (12, 21) - most common in childhood ALL - gene: AML 1 and TEL - “extremely” high long-term survival

Pathogenesis: Summary Strategies that interfere with over-activation of signaling pathways and the block in cellular differentiation have the potential to improve patient outcomes in AML.

Approach to acute leukaemia • In all, but the elderly or medically unfit, intensive initial treatment with combinations of chemotherapeutic drugs is indicated • The aim is to rapidly clear leukaemic cells from the marrow and to restore normal bone marrow function, ie achieve REMISSION • CURE is the goal

Principles of treatment combination chemotherapy – first goal is complete remission – further Rx to prevent relapse supportive medical care – transfusions, antibiotics, nutrition psychosocial support – patient and family

Response to treatment • Childhood ALL: >98% remission 80% cure • Adult AML and ALL: 60 -75% remission 25 -35% cure
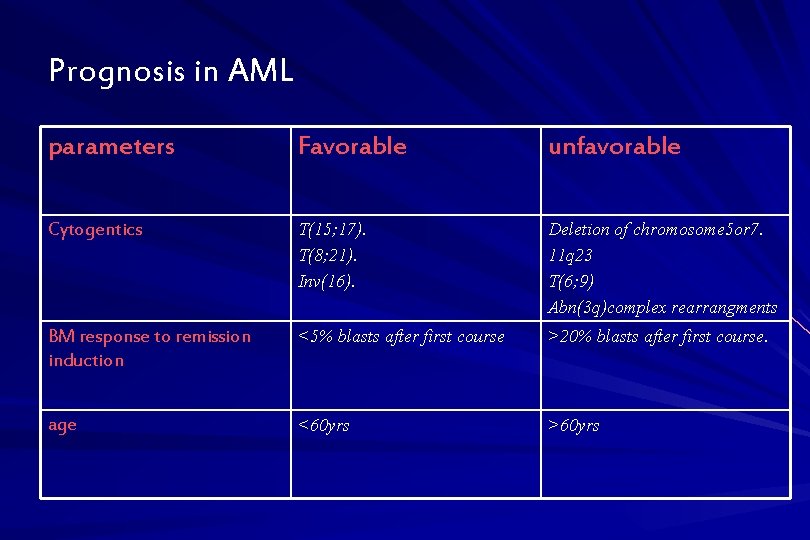
Prognosis in AML parameters Favorable unfavorable Cytogentics T(15; 17). T(8; 21). Inv(16). Deletion of chromosome 5 or 7. 11 q 23 T(6; 9) Abn(3 q)complex rearrangments BM response to remission induction <5% blasts after first course >20% blasts after first course. age <60 yrs >60 yrs

Current Research Inhibition of the products of genetic mutations found in AML cells Inhibition of proteins that cause chemotherapy resistance Use of antibody therapy against AML cells Use of new or existing drugs given in different doses and schedules Techniques to make stem cell transplantation safer, easier, and more effective Evaluation of drugs called hypomethylating therapy
- Slides: 101