Acute and Chronic Myeloid Leukemia Patrick Brown MD

Acute and Chronic Myeloid Leukemia Patrick Brown, MD

Classification of Myeloid Neoplasms n WHO, 4 th edition, 2008 Category MPN MDS/MPN AML Diseases CML, PV, PMF, ET, mastocytosis refractory cytopenias (RA, RAEB, RARS, etc. ) CMML, a. CML, JMML With/without recurrent genetic abnormalities (t(8; 21), inv(16), MLL, t(15; 17), etc. ), MDS-related, therapyrelated, DS-related, NOS (FAB)

Epidemiology • Know the incidence of ALL, AML and CML, and the peak age at which each of these occur
Epidemiology CML
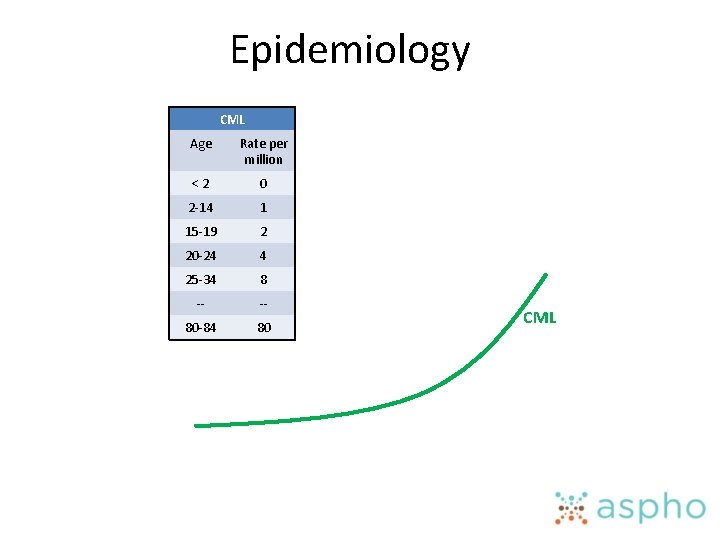
Epidemiology CML Age Rate per million <2 0 2 -14 1 15 -19 2 20 -24 4 25 -34 8 -- -- 80 -84 80 CML

Epidemiology • Know the concordance rate of ALL and AML in identical twins

Monozygotic twins • Index case < 1 yo • Very high (close to 100%) concordance with short latency (weeks) • Prenatal preleukemic event (MLL-r, e. g), intrauterine transfer • MLL-r promotes rapid development of cooperating “hits” • Index case 1 -6 yo • ~10 -20% concordance (most clonal), longer latency • Prenatal preleukemic event (TEL-AML 1, AML 1 -ETO, e. g), intrauterine transfer • Compared to MLL-r, relatively low risk of cooperating “hits” • Index case > 6 yo • Minimal increased risk • No prenatal preleukemic event

Leukemogenesis • Know which constitutional and genetic conditions predispose to the development of leukemia

Major Leukemia Predispositions • • Marrow failure syndromes – MDS, AML JMML-related conditions (NF 1, Noonan) Down syndrome – ALL, TMD/AMKL Others • Ataxia-telangiectasia (ALL, NHL) – ATM mutations; defective DNA repair, immunodeficiency • Bloom syndrome (broad) – BLM mutations; short stature, sun rash, sister chromatid exchanges • Li-Fraumeni syndrome (broad) – TP 53 mutations

Marrow Failure and MDS/AML • Pancytopenia Disorder Clinical features Molecular genetic features Fanconi anemia Characteristic congenital abnormalities (skeletal, renal, mental retardation), progressive bone marrow failure Mutations in one of several components of FA/BRCA multiprotein complex involved in homology-directed DNA repair, resulting in chromosomal instability, increased sensitivity to genotoxic agents (mitomycin C, diepoxybutane), cancer risk Dyskeratosis congenita Abnormal skin pigmentation, nail dystrophy, leukoplakia, progressive bone marrow failure Mutations in one of several components of telomerase complex mutations (DKC 1 (XR), TERC (AD), TINF 2 (AD), others), resulting in defective telomere maintenance

Marrow Failure and MDS/AML • Anemia Disorder Diamond-Blackfan anemia Clinical features Pure red cell aplasia, characteristic congenital abnormalities (facial, skeletal, genitourinary) Molecular genetic features Mutations in one of several ribosomal protein genes (RPS 19, RPL 5, others)

Marrow Failure and MDS/AML • Neutropenia Disorder Schwachman. Diamond syndrome Severe congenital neutropenia (Kostmann syndrome) Clinical features Molecular genetic features Neutropenia, exocrine pancreatic insufficiency, skeletal abnormalities SBDS mutations, affecting r. RNA processing and 40 S subunit biogenesis Severe congenital neutropenia Mutations in ELA 2 (elastase) most common, others include GFI 1 and HAX 1; result in agranulocytosis; secondary activating mutations of GCSF receptor often responsible for progression to AML

Marrow Failure and MDS/AML • Thrombocytopenia Disorder Congential amegakaryocytic thrombocytopenia Clinical features Congenital thrombocytopenia, progressive bone marrow failure Familial platelet Qualitative and quantitative disorder with platelet defects predisposition to AML Molecular genetic features MPL (thrombopoietin receptor) mutations AML 1(myeloid transcription factor) mutations

Classification • Know the FAB (old) and WHO (new) classifications of AML • Know the characteristic clinical presentations of AML, and associate these with specific AML subtypes § § § DIC/bleeding Hyperleukocytosis CNS leukemia Leukemia cutis Chloromas

FAB: morph/phenotype; 30% blasts AML Subtype M 0 Comments AML without differentiation M 1 Difficult to distinguish from ALL; diagnosis requires expression of surface markers such as CD 13, CD 33 and CD 117 (c -kit) in the absence of lymphoid differentiation AML with minimal differentiation Myeloperoxidase detectable by special stains/flow cytom etry M 2 AML with differentiation M 3 Acute promyelocytic leukemia (APL), hypergranular type M 3 v APL, microgranular variant M 4 Acute myelomonocytic leukemia (AMML) M 4 Eo AMML with eosinophilia M 5 Acute monocytic leukemia M 6 Acute erythroblastic leukemia M 7 Acute megakaryoblastic leukemia Auer rods; common t(8; 21) -> AML 1 -ETO fusion, good prognosis , chloromas Auer rods; DIC/bleeding; ; t(15; 17) -> PML-RAR a fusion, good prognosis with ATRA therapy Cytoplasm of promyelocytes demonstrates a fine granularity, and nuclei are often folded. Same clinical, cytogenetic and therapeutic implications as FAB M 3. Mixture of myeloblasts (at le ast 20%) and monocytic blasts; often with peripheral monocytosis AMML with >5% abnormal eosinophil precursors in marrow (with basophilic granules) , common inv(16), good prognosis >80% of bone marrow non-erythroid cells are monocytic; M 5 a: monoblastic; M 5 b: monocytic (more differentiated); for both M 4 and M 5: infant age, MLL 11 q 23 rearrangements, CNS involvement, chloromas, gingival hyperplasia Rare in children Seen mostly in children with Down syndrome (good prognosis if < 2 years old; GATA 1 mutations ) or mosaicism for trisomy 21; rare in normal children (poor prognosis , t(1; 22) -> OTT-MAL fusion, often infants); myelofibrosis commo n

WHO: clinical/molecular; 20% blasts • Is the AML due to prior XRT/chemo? • If yes: Dx is Therapy-related AML (t-AML) • Is the AML in a child with Down syndrome? – If yes: Dx is DS-related AML • Is major (“Big 4”) recurring abnormality present? • If yes: Dx is AML w/ t(8; 21); inv(16); t(15; 17); MLL-r • NOTE: No minimum blast % needed • Is there dysplasia, prior MDS and/or MDS-related mutation (-7, del(5 q), etc. )? • If yes, Dx is AML with MDS-related changes – If no to all: Dx is AML, NOS - use FAB to subclassify

t-AML • Know the characteristic chromosomal abnormalities and clinical characteristics in secondary acute myeloid leukemia resulting from topoisomerase II inhibitors and from alkylators, respectively

t-AML • Alkylating agents and radiation • Long latency (5 -7 year) from exposure, common preceding MDS • Cytogenetics: -7, del(7 q), -5, del(5 q); complex cytogenetics • Topoisomerase II inhibitors (etoposide > anthracyclines) • Short latency (1 -2 years) from exposure, rare preceding MDS • Cytogenetics: 11 q 23 rearrangements most common, but t(8; 21), t(15; 17), t(9; 22) and inv(16) can also occur • Regardless of age at presentation or etiology, t-MDS/t. AML tends to be more refractory to therapy than de novo MDS or AML

Recurring Cytogenetic Lesions • Correlate clinical characteristics with chromosomal abnormalities in acute myeloid leukemia • Understand the significance of rearrangements of the ATRA receptor gene in M 3 acute myeloid leukemia • Recognize specific clinical syndromes associated with t(8; 21), inv(16), t(9; 11), t(15; 17), and monosomy 7 or 7 q- in acute myeloid leukemia • Know the molecular abnormalities with which specific and recurring chromosomal abnormalities are associated in acute nonlymphoblastic leukemia
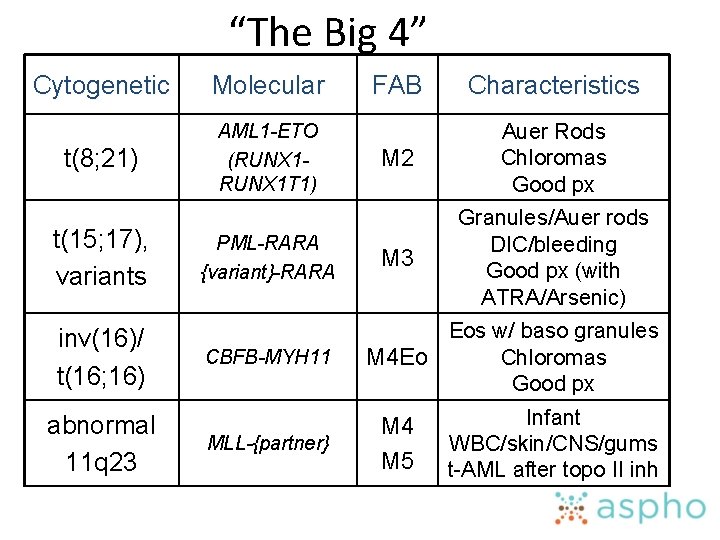
“The Big 4” Cytogenetic Molecular t(8; 21) AML 1 -ETO (RUNX 1 T 1) t(15; 17), variants inv(16)/ t(16; 16) abnormal 11 q 23 PML-RARA {variant}-RARA CBFB-MYH 11 MLL-{partner} FAB Characteristics M 2 Auer Rods Chloromas Good px M 3 Granules/Auer rods DIC/bleeding Good px (with ATRA/Arsenic) Eos w/ baso granules Chloromas M 4 Eo Good px M 4 M 5 Infant WBC/skin/CNS/gums t-AML after topo II inh
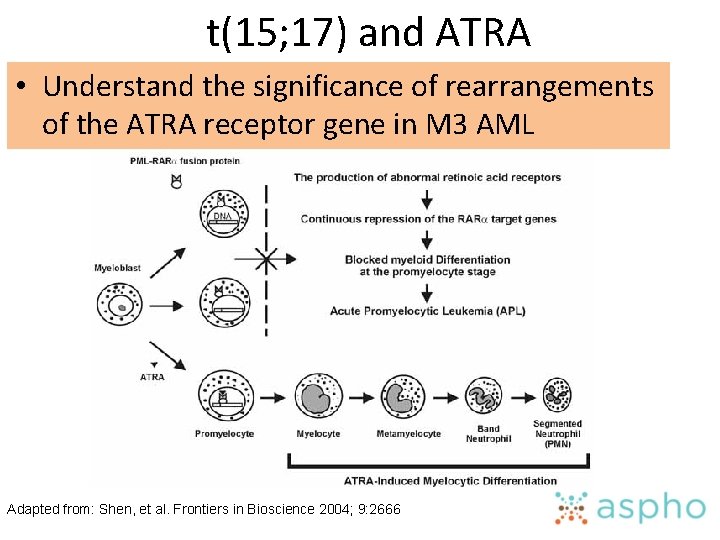
t(15; 17) and ATRA • Understand the significance of rearrangements of the ATRA receptor gene in M 3 AML Adapted from: Shen, et al. Frontiers in Bioscience 2004; 9: 2666

Clinical aspects of APL • High risk of life-threatening bleeds (~10% mortality) due to DIC/fibrinolysis + low plts – Basics: close monitoring of PT, a. PTT, fibrinogen, D-dimer, plts with liberal use of products: plts, cryo, FFP – Start ATRA immediately with suspected APL: rapidly improves coag parameters and reduces bleeding risk – Other interventions controversial (heparin, amicar, r. VIIa, …) • Differentiation syndrome – Fever, edema, pulm infiltrates, hypoxia, resp distress, hypotension, renal/hepatic dysfunction, effusions, rash – Risk correlates with high WBC; can occur in absence of ATRA – Rx: Prompt recognition and use of dexamethasone, hold ATRA until resolving • Arsenic Trioxide – Increasingly incorporated into consolidation for APL in effort to reduce cumulative anthracycline exposure - recent data suggests that “low risk” APL (<10 k WBC) is curable with ATRA and Arsenic only!

MDS vs. AML • Know the relationship between myelodysplastic syndromes and acute myeloid leukemia • In children (unlike adults), the vast majority of AML cases are “de novo” (i. e. , not preceded by MDS) • MDS in children commonly arises in setting of underlying constitutional disorder (DS, marrow failure syndrome, etc. ) • MDS in children has similar implications as in adults • Eventual progression to AML is the general rule; pace of progression is variable • Generally requires HSCT for cure • Questionable role for pre-HSCT chemotherapy

Hyperleukocytosis • Recognize the potential complications of hyperleukocytosis in acute myeloid leukemia • Plan the management of hyperleukocytosis in acute myeloid leukemia
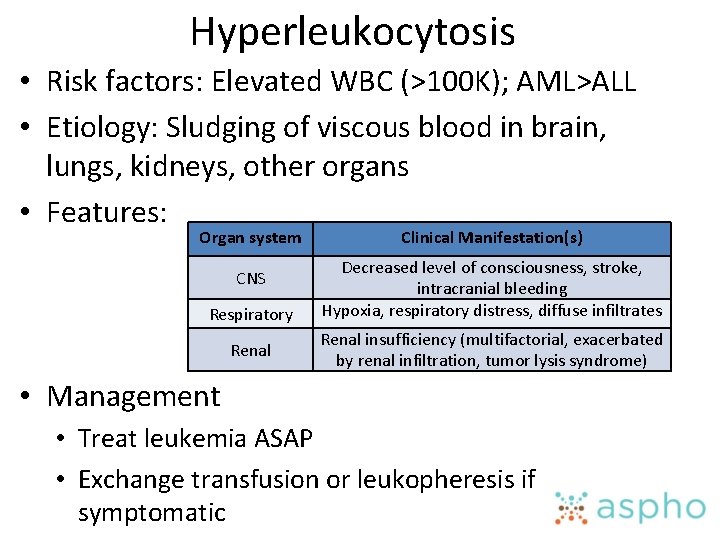
Hyperleukocytosis • Risk factors: Elevated WBC (>100 K); AML>ALL • Etiology: Sludging of viscous blood in brain, lungs, kidneys, other organs • Features: Organ system CNS Respiratory Renal Clinical Manifestation(s) Decreased level of consciousness, stroke, intracranial bleeding Hypoxia, respiratory distress, diffuse infiltrates Renal insufficiency (multifactorial, exacerbated by renal infiltration, tumor lysis syndrome) • Management • Treat leukemia ASAP • Exchange transfusion or leukopheresis if symptomatic

Immunophenotype • Know the immunophenotypic differences between acute lymphoid and acute myeloid leukemia • Know how to identify lymphoid/myeloid mixed lineage acute lymphoblastic leukemia and biphenotypic leukemia (by immunophenotyping)
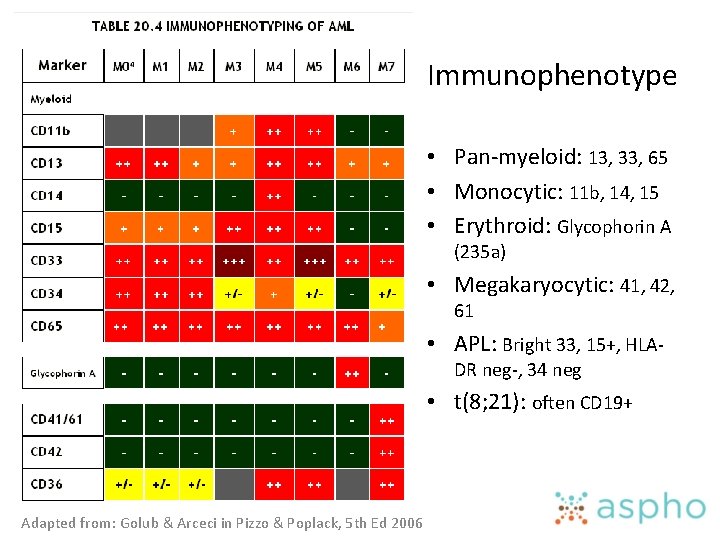
Immunophenotype • Pan-myeloid: 13, 33, 65 • Monocytic: 11 b, 14, 15 • Erythroid: Glycophorin A (235 a) • Megakaryocytic: 41, 42, 61 • APL: Bright 33, 15+, HLADR neg-, 34 neg • t(8; 21): often CD 19+ Adapted from: Golub & Arceci in Pizzo & Poplack, 5 th Ed 2006

Acute Leukemia of Ambiguous Lineage • Acute Undifferentiated Leukemia (AUL) • Express no lineage-specific markers • Mixed Phenotype Acute Leukemia (MPAL) • Distinct populations of blasts of different lineages; and/or single population of blasts co-expressing antigens of multiple lineages • Often MLL-rearranged • T/myeloid, B/myeloid most common

Prognostic Factors & Risk Stratification • Know prognosis of various sub-types of acute myeloid leukemia • Know the prognostic significance of the non-random cytogenetic abnormalities in acute myeloid leukemia • Know the prognostic importance of Down syndrome in acute nonlymphoblastic leukemia Risk Stratification is the prospective use of prognostic factors to assign patients to specific treatments. Only a subset of prognostic factors are used in risk stratification

Prognostic Factors not used • Host characteristics • Age and sex: not independent on most recent studies • Race: non-whites - inferior survival • BMI: under- or over-weight - inferior survival • Pharmacogenomics • Disease characteristics • High WBC – no longer used, except in APL (> 10 K is high risk – use anthracycline immediately) • FAB – supplanted by cyto/molecular

Recent Risk Stratification • Favorable • • Down syndrome-related (esp. if < 4 y. o. ) APL with t(15; 17) or variant Cytogenetics: inv(16), t(8; 21) Molecular: NPM 1, CEBPA Separate protocols • Unfavorable • • t-AML, MDS-related AML Cytogenetics: -7, 5 q-, abn(3 q) Molecular: FLT 3/ITD (high allelic ratio) Primary induction failure (>5% blasts after course 2) • Intermediate: everything else
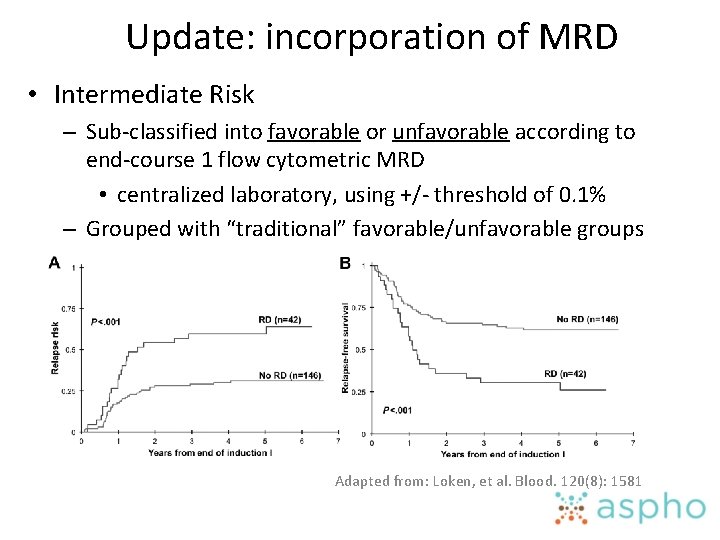
Update: incorporation of MRD • Intermediate Risk – Sub-classified into favorable or unfavorable according to end-course 1 flow cytometric MRD • centralized laboratory, using +/- threshold of 0. 1% – Grouped with “traditional” favorable/unfavorable groups Adapted from: Loken, et al. Blood. 120(8): 1581

Current Risk Stratification • Favorable (Low Risk, LR) • • • Down syndrome-related (esp. if < 4 y. o. ) Separate protocols APL with t(15; 17) or variant Cytogenetics: inv(16), t(8; 21) Molecular: NPM 1, CEBPA Intermediate with end course 1 MRD negative • Unfavorable (High Risk, HR) • • • t-AML, MDS-related AML Cytogenetics: -7, 5 q-, abn(3 q) Molecular: FLT 3/ITD (high allelic ratio) Primary induction failure (>5% blasts after course 2) Intermediate with end course 1 MRD positive

Therapy • Know which drug combinations are most effective in the treatment of acute myeloid leukemia • Know that high-dose cytarabine is effective in the treatment of acute myeloid leukemia • Know the role of CNS prophylaxis in the treatment of acute myeloid leukemia • Know the evidence against the use of extended maintenance therapy for AML • Know the indications for allogeneic HSCT in AML • Know the various components of prophylactic and acute supportive care for children with acute myeloid leukemia receiving treatment

§ Current Standard Therapy Remission induction • 2 courses of intense chemotherapy (Ara-C, Doxo, Etoposide); high risk of invasive infection; CR rate ~ 7580% • Current protocol “switches” 2 nd induction course to mitoxantrone/ HD Ara-C for HR patients § Post-remission consolidation • BMT • All HR with best donor (matched sib preferred, MUD/UCB donors acceptable) • No LR (even with matched sib) • Additional chemo “intensification” courses (2) • HR w/o any donor, and all LR • Usually HD Ara-C combined with drugs not used in induction (mitoxantrone, L-asp, e. g. )

Current Standard Therapy § CNS Prophylaxis • Much less intense than in ALL due to low rates of CNS relapse with standard therapy • For CNS+ at diagnosis, weekly IT Ara-C until CSF clears, then once per course; no cranial XRT § Maintenance • Unlike ALL, there is no role for extended maintenance (a survival benefit has never been demonstrated)

Current Standard Therapy § Supportive care • Admission during periods of neutropenia recommended, due to significant risk of lifethreatening infections • Most infections are episodes of bacteremia and sepsis • Strep. Viridans • Gram negative enterics (E. coli, Klebsiella, Pseudomonas, etc. ) • Aggressive empiric treatment with broad spectrum antibiotics to cover these organisms is standard • Bacterial prophylaxis is controversial and not considered standard due to concerns re: antibiotic resistance • Fungal and pneumocystis prophylaxis are commonly used

Late Effects in AML § Recognize the late complications of therapy for AML • After chemotherapy only – Anthracyclines: cumulative exposure ~ 450 mg/m 2 doxorubicin equivalents – cardiotoxicity – Etoposide: t-MDS/AML – High dose Ara-C: neurotoxicity • After HSCT – Above, plus HSCT-related risks

Radiation in AML § Know the indications for radiotherapy in AML • Generally limited to emergent treatment of lifethreatening complications of chloromas (e. g. , spinal cord compression) • TBI is not a typical component of HSCT prep regimens for AML • Cranial XRT is generally not required to control CNS disease in AML

CML: Biology • Know the clinical, laboratory, and prognostic features of CML – Recognize priapism as a presenting feature of chronic myeloid leukemia (? ) • Recognize the hematologic changes associated with a blast crisis in chronic myeloid leukemia • Know that a blast crisis in chronic myeloid leukemia can involve other cell lines • Know the association of BCR-ABL 1 oncogene with chronic myeloid leukemia • Know the clinical, laboratory and molecular characteristics that differentiate Ph+ chronic myeloid leukemia from Ph+ acute lymphocytic leukemia

CML: Definition • • Myeloproliferative neoplasm (MPN) Origin: abnormal pluripotent HSC Ph+; t(9; 22)(q 34; q 11. 2); BCR-ABL 1 Triphasic natural history: – Chronic Phase (CP) – Accelerated Phase (AP) – Blast Crisis (BC)

CML: Clinical Features • Most pts diagnosed in Chronic Phase (CP) • 20 -40% asx, dx’d due to abnl CBC (incidental findings of leukocytosis with left shift, +/- anemia, +/- thrombocytosis, basophilia, +/- eosinophilia) • Sx: abd pain, dysphagia, inc abd girth (all from splenomegaly), fatigue, wt loss, night sweats, bleeding, (rarely) leukostasis/priapism(? ) • Natural history: progression (widely variable latency) – Accelerated phase (AP): decreased response to treatment, clonal evolution, inc blasts (<20%), inc basophilia – Blast crisis (BC): acute leukemia (2/3 AML, 1/3 ALL) Distinguishing CML-CP or CML-AP from Ph+ ALL is simple due to obvious differences in CBC and PB smear; CML presenting with lymphoid BC vs. Ph+ ALL is a more challenging distinction – molecular studies are crucial.

CML: Diagnosis • Demonstration of p 210 BCR-ABL 1 fusion (PCR) in blood in proper clinical context is likely sufficient • Marrow typically included to definitively rule out accelerated phase and improve karyotyping Adapted from: http: //www. cincinnatichildrens. org/svc/alpha/m/molecular-genetics/bcrtesting. htm
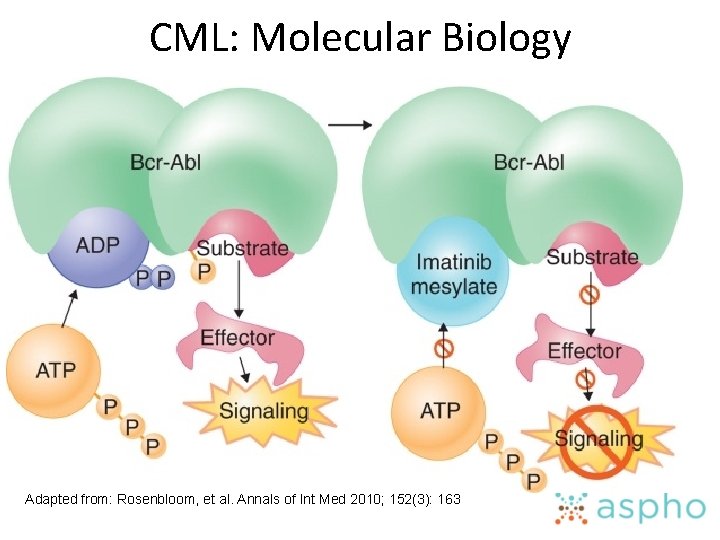
CML: Molecular Biology Adapted from: Rosenbloom, et al. Annals of Int Med 2010; 152(3): 163

CML: Treatment • Know the principles of using targeted therapy (such as imatinib) in patients with chronic myeloid leukemia • Plan the treatment of a blast crisis in a patient with chronic myeloid leukemia • Know the indications for and timing of HSCT in a patient with chronic myeloid leukemia • Know therapeutic options for a patient who has a recurrence of chronic myeloid leukemia after HSCT

CML: Impact of Tyrosine Kinase Inhibitors • Pre-TKI Era – The only curative long term curative therapy is allogeneic stem cell transplant, so essentially all children receive HSCT with best available donor – Hydroxyurea and interferon used to control disease prior to SCT • Post-TKI Era – Begin TKI, continue indefinitely – Consider HSCT in chronic phase only for patients with HLA-matched family donor (even then controversial) – For recurrent or refractory chronic phase, try higher dose TKI and/or new TKI first (guided by mutation analysis), then HSCT for treatment failure – For accelerated phase or blast crisis, attempt to induce remission with chemotherapy (AML or ALL, depending on blast phenotype) + TKI, and then take to HSCT

CML: Treatment of CML-CP • ABL tyrosine kinase inhibitors (TKI) – Imatinib (Gleevec) – Novartis • Kids: 340 mg/m 2 qd (600 mg max) • Adol/adult: 400 mg qd (may increase to 600 mg and 800 mg as needed for response) – Dasatinib (Sprycel) – BMS (100 mg qd) More rapid and sustained – Nilotinib (Tasigna) – Novartis (400 mg bid) cytogenetic/molecular responses – *Ponatinib (Iclusig) – Ariad (45 mg qd) – active against T 315 I mutant • HSCT – controversial – HSCT remains the only proven cure for CML – TKI’s achieve sustained disease control, but recurrence likely upon discontinuation – Major prognostic factor for HSCT: disease phase (CP>AP>BC) – CML is very sensitive to graft vs. leukemia (GVL) effect – relapses post-HSCT can be effectively treated with donor lymphocyte infusions (DLI), especially if no GVH with first HSCT – When HSCT is used, TKI “maintenance” is typically added post-engraftment in effort to prevent relapse

CML: Response • There are three principal strategies to assess response – Hematologic response (HR; determined with PB and exam) • Complete (CHR): normal counts, no HSM – Cytogenetic response (Cy. R; determined with BCR-ABL FISH on BMA) • Complete (CCy. R): no Ph+ • Partial (PCy. R): major <= 35%; minor 36 -95% – Molecular response (MR; determined with BCR-ABL q. PCR on PB) • Complete (CMR): PCR neg • Major (MMR): BCR-ABL: ABL ratio <0. 1% (international scale), or 3 log reduction in copy# • Most recent guidelines – BMA for response not needed if validated q. PCR testing available – Every 3 month testing until MMR, then every 3 -6 months

CML: Response Timepoint Suboptimal Failure 3 mo No PCy. R (major) and/or BCR-ABL 1 q. PCR >10% No CHR and/or no PCy. R (minor) 6 mo No CCy. R and/or BCR-ABL q. PCR >1% No PCy. R (major) and/or BCR-ABL 1 q. PCR >10% 12 mo No MMR (q. PCR > 0. 1%) No CCy. R and/or BCR-ABL q. PCR >1% 18 mo - No MMR (q. PCR > 0. 1%)

CML: Response • Response to TKI treatment is the most important prognostic factor in CML • Patients with a rapid and sustained response to TKI have an excellent outcome • TKI resistance – Primary – treatment failure or suboptimal response with TKI – Secondary - loss of response (rising q. PCR, recurrence of FISH+, development of CML-AP, etc. ) – Both types are most commonly caused by acquired mutations in ATPbinding pocket of ABL – T 315 I mutations have been most problematic, since they are also resistant to 2 nd and 3 rd line TKI’s – recently approved ponatinib has T 315 I activity • TKI-resistant patients generally require HSCT for cure
- Slides: 50