Activator Explain why you put subscripts in chemical

Activator Explain why you put subscripts in chemical formulas.
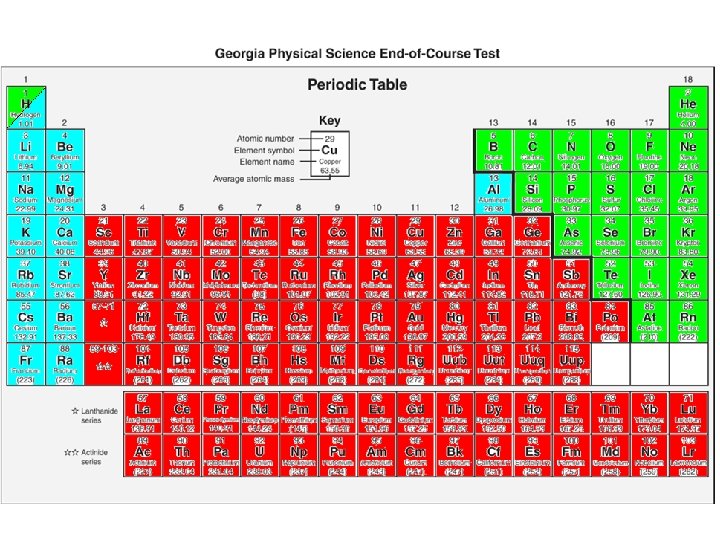
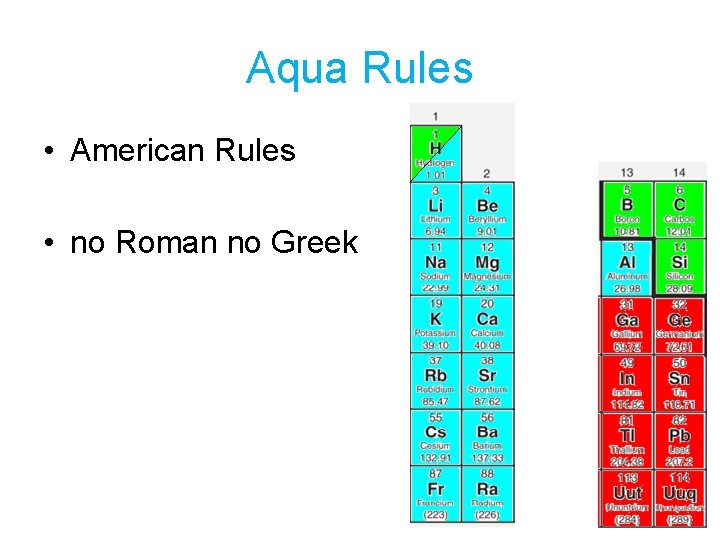
Aqua Rules • American Rules • no Roman no Greek

Aqua Name • Write element names and change ending of second element to –ide. • Example: Na 2 O Sodium oxide

Aqua Formulas • Balance positive and negative charges, then add subscripts • Example: Na 1+ O 2 - Na 2 O It take 2 Na 1+ to balance 1 O 2 -

Red Rules • Roman numerals are the positive oxidation number for the transition metals (“the valley varies”)

Red Name • Write element names and change ending of second element to –ide. • Use the roman grid to find the red oxidation number and write as Roman Numeral.

Red Formulas • Get oxidation number for red element from Roman Numeral, balance charges, and write subscripts. • Example: Iron (III) oxide Oxidation number for Iron (III) = 3+ 2 O Fe 3+ 2 - It takes 2 Fe 3+ and 3 O 2 - to O Fe 3+ O 2 - balance positive and negative charges Fe 2 O 3
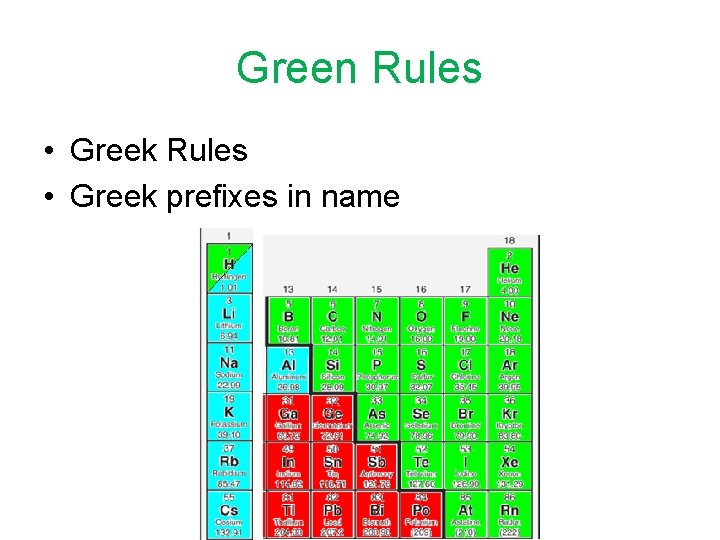
Green Rules • Greek prefixes in name

Greek Prefixes • • • One Two Three Four Five Six Seven Eight Nine Ten mono di tri tetra penta hexa hepta octa nana deca

Green Name • Turn subscripts into Greek prefixes in front of element names (do not use mono- on the first element). Change ending of second element to –ide. • Example: CO 2 Carbon dioxide CO Carbon monoxide

Green Formula • Turn Greek prefixes into formula subscripts. Do not try to balance charges. • Example: Carbon tetrachloride CCl 4

Ticket out the Door Name the following on a sheet of paper: 1. Na. Cl 2. Al. I 3 3. Ni. O 4. Ni 2 O 3 5. Na. F Write the following equations: 1. Iron (II) chloride 2. Sodium chloride

Right or wrong? • If the formula is incorrect, write the correct formula: – Li. F 2 – KBr – Mg. Cl – Al. O 2

Naming Ionic Compound • Bond between a metal and nonmetal 1. Name the metal 2. Name the nonmetal and change the ending to –ide.

Write the formulas and name the compounds 1. Na+1 Cl-1 5. Mg+2 P-3 2. Ba+2 F-1 6. Ca+2 Cl-1 3. K+1 S-2 7. Cs+1 P-3 4. Al+3 I-1 8. Al+3 P-3

Write the formulas and Name the compounds 1. K bonded with Cl 5. Sr bonded with P 2. Ba bonded with P 6. Cs bonded with Cl 3. Fr bonded with O 7. Na bonded with N 4. Al bonded with S 8. Al bonded with Cl

Ticket Out The Door • Write formulas and name the following: 1. Na bonded with P 2. Ca bonded with N 3. Cs bonded with Se 4. Ba bonded with Br 5. Li bonded with I

Name the Compounds (p. 54) 6. Zn. S 11. Fe. O 7. Ag 2 O 12. Fe 2 O 3 9. Ni. O 13. Cr. S 10. Ni 2 O 3 14. Cr 2 S 3

Name the Compounds (p. 54) 15. Cu. Cl 19. Mn. Br 2 16. Cu. Cl 2 20. Mn. Br 4 17. Pb. O 18. Pb. O 2

Ticket Out the Door Name the following: 1. Na. Cl 2. Al. I 3 3. Ni. O 4. Ni 2 O 3 5. Na. F

Write and name the following Ionic Compounds with “regular” metals 1. Na. Cl Sodium chloride 2. Na. F Sodium fluoride 3. Al 2 O 3 Aluminum oxide 4. Ca. I 2 Calcium iodide 5. Al. I 3 Aluminum iodide 6. Li. Cl Lithium chloride 7. KCl Potassium chloride 8. Ca. S Calcium sulfide 9. Be. F Beryllium fluoride 10. K 3 N Potassium nitride

Write and name the following Ionic Compounds with “transition” metals 1. Zn. S Zinc (II) sulfide 2. Ni. O Nickel (II) oxide 3. Ni 2 O 3 Nickel (III) oxide 4. Cr. Cl 2 Chromium (II) chloride 5. Cr. Cl 3 Chromium (III) chloride 6. Mn. O Manganese (II) oxide 7. Mn 2 O 3 Manganese (III) oxide 8. Mn. Br 2 Manganese (II) bromide 9. Mn. Br 4 Manganese (IV) bromide 10. Fe. O Iron (II) oxide

Name the following 1. Ti. Br 3 Titanium (III) bromide 2. Cu 3 P Copper (I) phosphide 3. Ca. Se Calcium selenide 4. Al. As Aluminum arsenide 5. Na. Br Sodium bromide 6. Zn 3 P 2 Zinc (II) phosphide 7. Cu 2 O Copper (I) oxide 8. Sn. S 2 Tin (IV) sulfide 9. Pb 3 N 2 Lead (II) nitride 10. Cs 2 O Cesium oxide

Write the following formulas 1. Magnesium phosphide 6. Sodium fluoride 2. Aluminum iodide 7. Barium chloride 3. Lithium bromide 8. Sodium chloride 4. Potassium sulfide 9. Calcium oxide 5. Barium fluoride 10. Beryllium sulfide

Write the following formulas 1. Silver (I) oxide Ag 2 O 2. Copper (I) bromide Cu. Br 3. Copper (II) bromide Cu. Br 2 4. Iron (II) oxide Fe. O 5. Iron (III) oxide Fe 2 O 3 6. Chromium (II) chloride Cr. Cl 2 7. Chromium (III) chloride Cr. Cl 3 8. Zinc (II) sulfide Zn. S 9. Manganese (II) oxide Mn. O 10. Manganese (III) oxide Mn 2 O 3

Write the following formulas 1. Lithium oxide Li 2 O 2. Magnesium oxide Mg. O 3. Potassium sulfide K 2 S 4. Calcium sulfide 6. Copper (II) chloride Cu. Cl 2 Ca. S 5. Aluminum chloride Mn. Br 2 10. Manganese (IV) bromide Al. Cl 3 7. Lead (II) oxide Pb. O 8. Lead (IV) oxide Pb. O 2 9. Manganese (II) bromide Mn. Br 4

Warm Up What is wrong with the following? 1. Aluminum (III) trichloride 2. Iron chloride 3. Carbon (IV) oxide 4. Monocarbon dioxide 5. Magnesium monoxide

Write the names for. . . 1. Na 3 N Sodium nitride 2. NO 2 Nitrogen dioxide 3. Fe. O Iron (II) oxide 4. Fe 2 O 3 Iron (III) oxide

Write the names for. . . 5. NO Nitrogen monoxide 6. N 2 O Dinitrogen monoxide 7. Al. Cl 3 Aluminum chloride 8. Al 2 O 3 Aluminum oxide

Write the formulas for. . . 1. Dinitrogen pentaoxide N 2 O 5 2. Nitrogen dioxide NO 2 3. Carbon tetrachloride CCl 4 4. Aluminum chloride Al. Cl 3

Write the formulas for. . . 1. Chromium (II) sulfide Cr. S 2. Chromium (III) sulfide Cr 2 S 3 3. Manganese (IV) bromide Mn. Br 4 4. Aluminum oxide Al 2 O 3
- Slides: 33