Activation Studies of Gallium Arsenide Photocathodes BY MORGAN

Activation Studies of Gallium Arsenide Photocathodes BY MORGAN DIXON CARLETON COLLEGE; NORTHFIELD, MN MENTORS: DR. IVAN BAZAROV, DR. YULIN LI, DR. XIANGHONG LIU CORNELL UNIVERSITY, SUMMER 2011
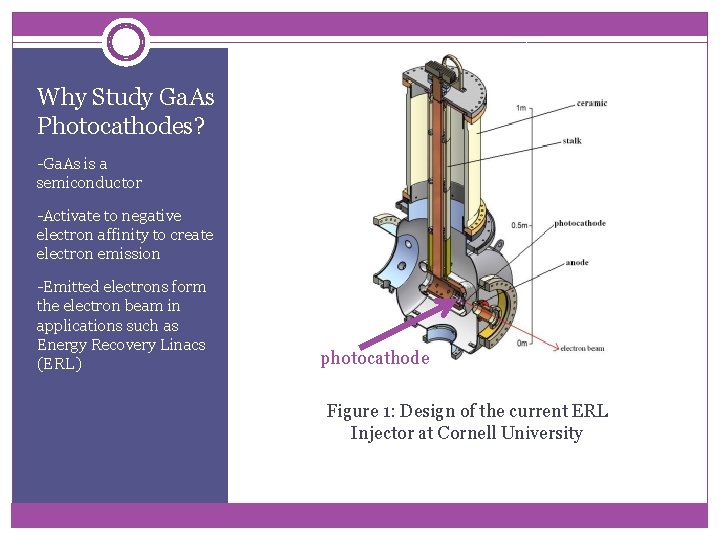
Why Study Ga. As Photocathodes? -Ga. As is a semiconductor -Activate to negative electron affinity to create electron emission -Emitted electrons form the electron beam in applications such as Energy Recovery Linacs (ERL) photocathode Figure 1: Design of the current ERL Injector at Cornell University

Quantum Efficiency (QE) and Lifetime �QE: measure of the number of emitted electrons per incident photon on the cathode �Ip = photocurrent (aka the current of emitted electrons) �Pl = laser power incident upon the cathode �Lifetime: span of time over which a photocathode produces a satisfactory QE Time for QE to reduce to 1/e of it’s original value �Dark Lifetime vs. Operational Lifetime �Want to increase lifetime – currently a maximum of a few days
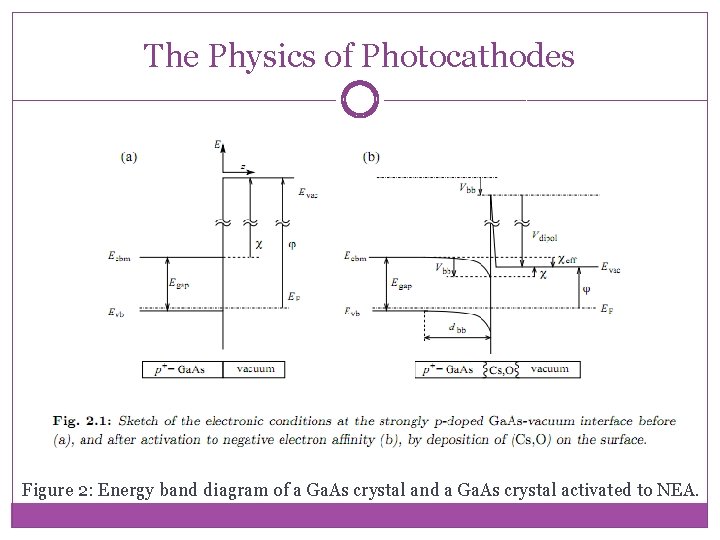
The Physics of Photocathodes Figure 2: Energy band diagram of a Ga. As crystal and a Ga. As crystal activated to NEA.

QE Chamber Figure 4: CAD drawing of the UHV chamber used for all experiments.
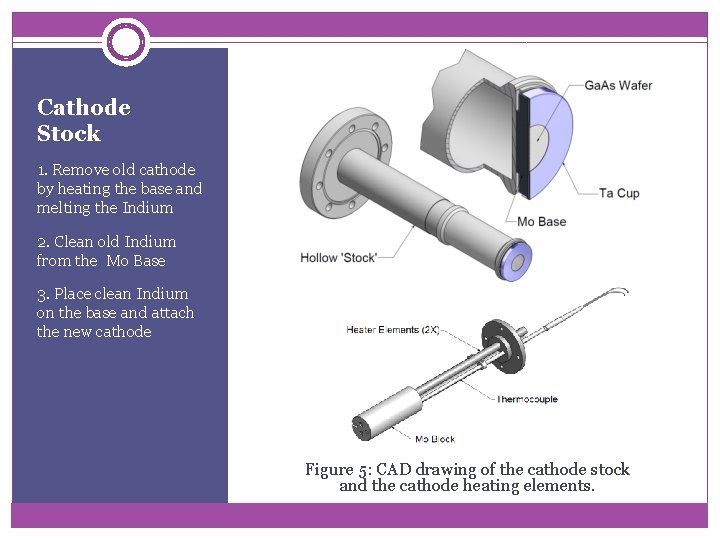
Cathode Stock 1. Remove old cathode by heating the base and melting the Indium 2. Clean old Indium from the Mo Base 3. Place clean Indium on the base and attach the new cathode Figure 5: CAD drawing of the cathode stock and the cathode heating elements.
Lab. VIEW program Figure 6: Screenshots of the lab. VIEW program, GUP. vi

Cathode Preparation Procedure �In house diamond cutting process �Acid etching to remove surface oxides and other contaminants �Attach the clean cathode to the Mo base and place in the chamber �Pump down chamber and bake out to achieve UHV �Vacuum annealing to further remove oxide contaminants on the surface of the cathode Heat cathode to 650 C and hold for two hours
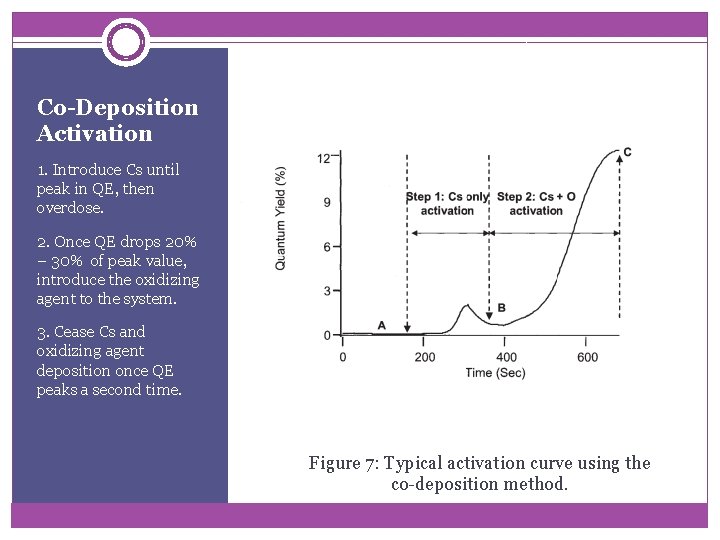
Co-Deposition Activation 1. Introduce Cs until peak in QE, then overdose. 2. Once QE drops 20% – 30% of peak value, introduce the oxidizing agent to the system. 3. Cease Cs and oxidizing agent deposition once QE peaks a second time. Figure 7: Typical activation curve using the co-deposition method.
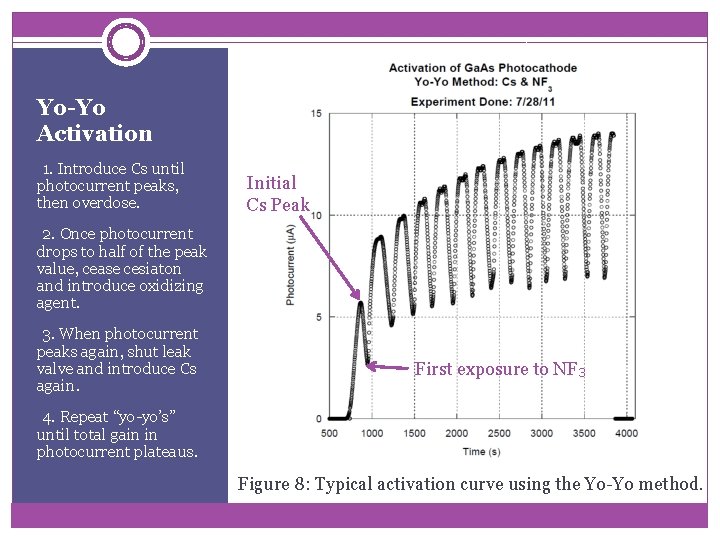
Yo-Yo Activation -1. Introduce Cs until photocurrent peaks, then overdose. Initial Cs Peak -2. Once photocurrent drops to half of the peak value, cease cesiaton and introduce oxidizing agent. -3. When photocurrent peaks again, shut leak valve and introduce Cs again. First exposure to NF 3 -4. Repeat “yo-yo’s” until total gain in photocurrent plateaus. Figure 8: Typical activation curve using the Yo-Yo method.
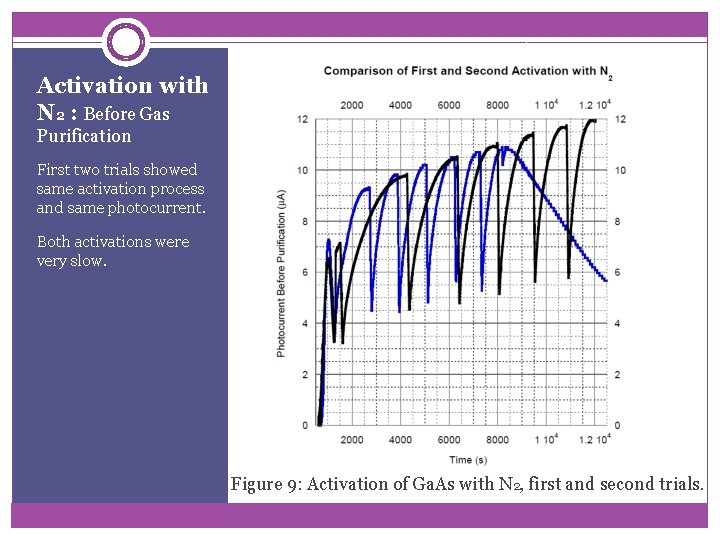
Activation with N 2 : Before Gas Purification First two trials showed same activation process and same photocurrent. Both activations were very slow. Figure 9: Activation of Ga. As with N 2, first and second trials.
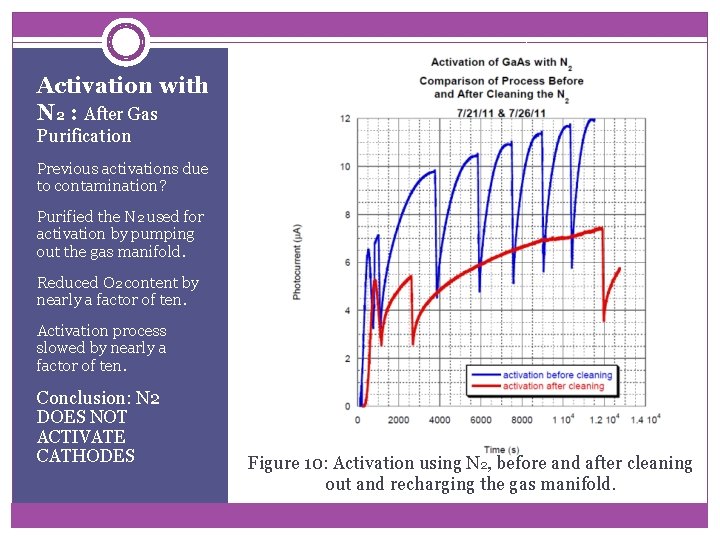
Activation with N 2 : After Gas Purification Previous activations due to contamination? Purified the N 2 used for activation by pumping out the gas manifold. Reduced O 2 content by nearly a factor of ten. Activation process slowed by nearly a factor of ten. Conclusion: N 2 DOES NOT ACTIVATE CATHODES Figure 10: Activation using N 2, before and after cleaning out and recharging the gas manifold.
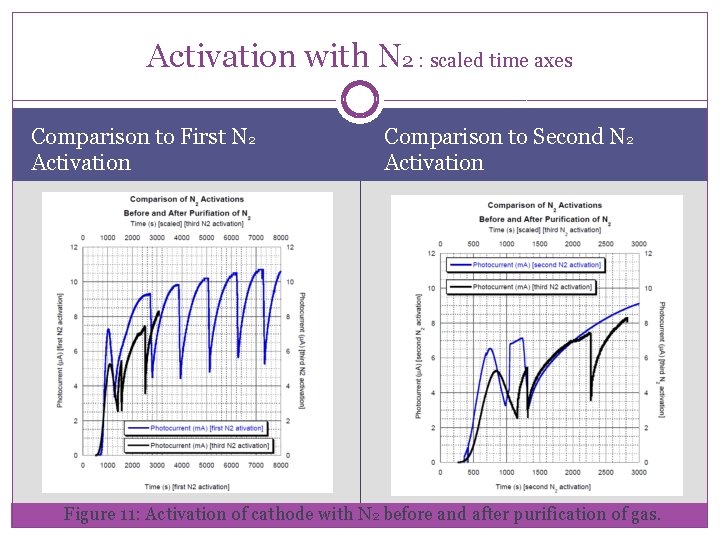
Activation with N 2 : scaled time axes Comparison to First N 2 Activation Comparison to Second N 2 Activation Figure 11: Activation of cathode with N 2 before and after purification of gas.
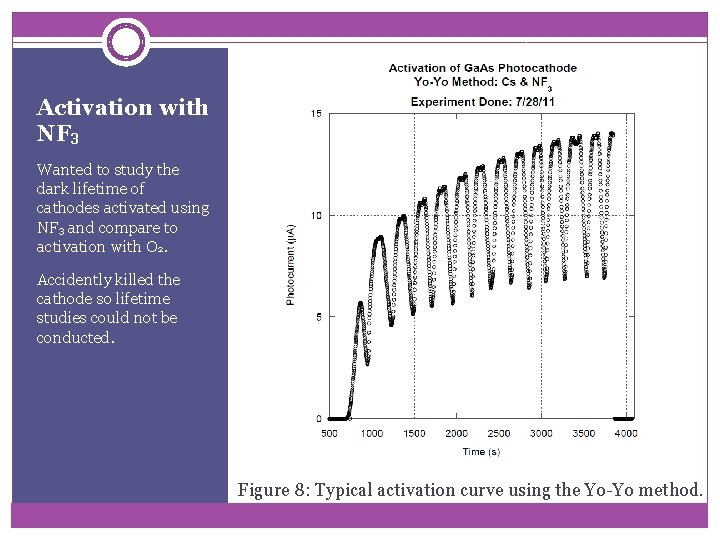
Activation with NF 3 Wanted to study the dark lifetime of cathodes activated using NF 3 and compare to activation with O 2. Accidently killed the cathode so lifetime studies could not be conducted. Figure 8: Typical activation curve using the Yo-Yo method.
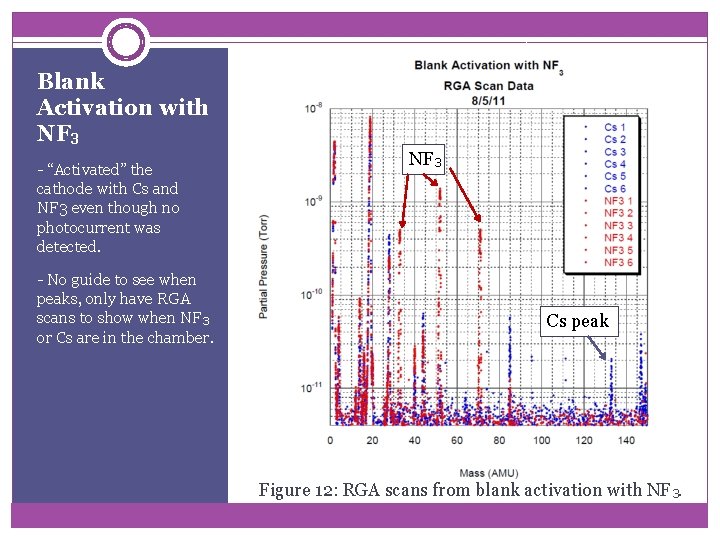
Blank Activation with NF 3 - “Activated” the cathode with Cs and NF 3 even though no photocurrent was detected. - No guide to see when peaks, only have RGA scans to show when NF 3 or Cs are in the chamber. NF 3 Cs peak Figure 12: RGA scans from blank activation with NF 3.
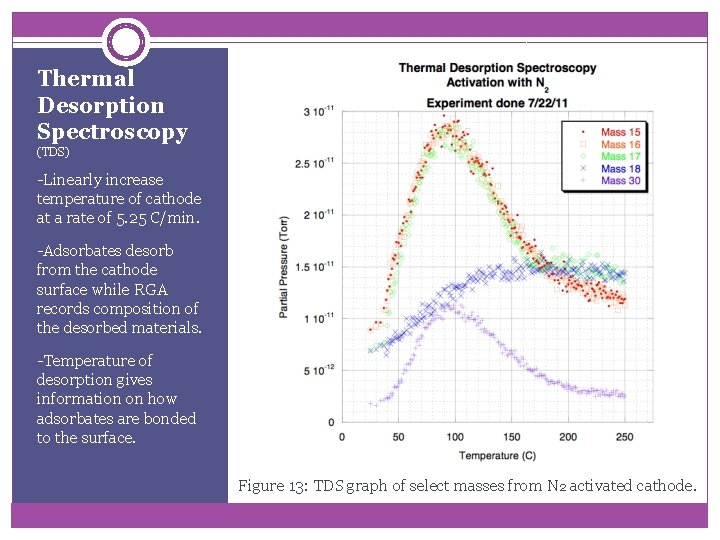
Thermal Desorption Spectroscopy (TDS) -Linearly increase temperature of cathode at a rate of 5. 25 C/min. -Adsorbates desorb from the cathode surface while RGA records composition of the desorbed materials. -Temperature of desorption gives information on how adsorbates are bonded to the surface. Figure 13: TDS graph of select masses from N 2 activated cathode.
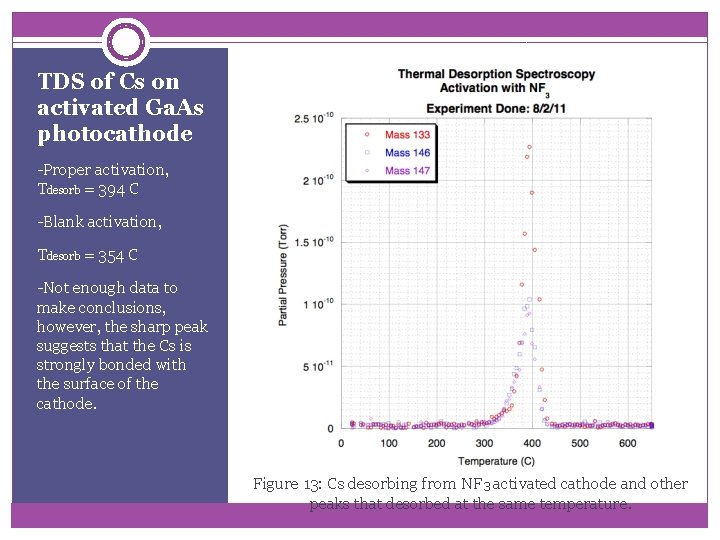
TDS of Cs on activated Ga. As photocathode -Proper activation, Tdesorb = 394 C -Blank activation, Tdesorb = 354 C -Not enough data to make conclusions, however, the sharp peak suggests that the Cs is strongly bonded with the surface of the cathode. Figure 13: Cs desorbing from NF 3 activated cathode and other peaks that desorbed at the same temperature.
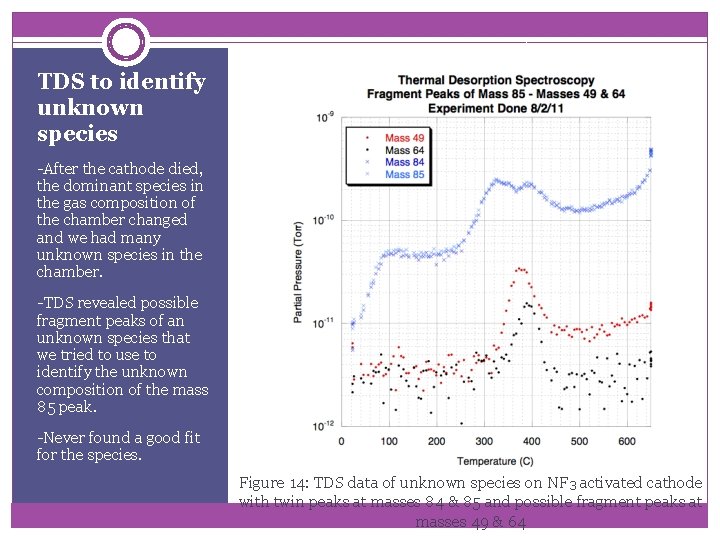
TDS to identify unknown species -After the cathode died, the dominant species in the gas composition of the chamber changed and we had many unknown species in the chamber. -TDS revealed possible fragment peaks of an unknown species that we tried to use to identify the unknown composition of the mass 85 peak. -Never found a good fit for the species. Figure 14: TDS data of unknown species on NF 3 activated cathode with twin peaks at masses 84 & 85 and possible fragment peaks at masses 49 & 64

Conclusions �The equipment and computing resources are set up to begin work with the new QE chamber. �Nitrogen will NOT activate Ga. As photocathodes are very sensitive to contaminants in activating gasses, so gases used in activating processes should be very pure to be sure which species are activating the cathode. �Cs appears to strongly bind with the Ga. As surface when the cathode is activated with NF 3.

Acknowledgements �Thank you very much to my mentors Ivan, Yulin and Xianghong, and all other members of Cornell’s photocathode project, for their wonderful support throughout the summer. �This project was funded by the NSF.
- Slides: 20