ACTIVATION OF THE CYTOTOXIC EFFECT OF SOME CYTOSTATICS

















- Slides: 34

ACTIVATION OF THE CYTOTOXIC EFFECT OF SOME CYTOSTATICS THROUGH EXPOSURE AT OPTICAL RADIATION CEEX: nr. 2 -Cex-06 -D 11 -32 Mihail Lucian Pascu 1*, Alexandru Pascu 1, Angela Staicu 1, Madalina Dicu 1, Doina Gazdaru 2, Marieta Costache 3, Jacques Barbe 4, Abdallah Mahamoud 4, Michelle Ferrari 5, Silvia Rao 5, Ligio Liggieri 5, Benone Carstocea 6

ACTIVATION OF THE CYTOTOXIC EFFECT OF SOME CYTOSTATICS THROUGH EXPOSURE AT OPTICAL RADIATION 1 National Institute for Laser, Plasma and Radiation Physics, Bucharest; 2 Physics Faculty, University of Bucharest; 3 Faculty of Biology, University of Bucharest; 4 Faculty of Pharmacy, Universite Mediterrannee of Marseille; 5 CNR- Institute for Energetics and Interphases, Genova 6 Central Military Hospital, Ophthalmology Clinic, Bucharest

APPLICATION Fighting resistance to treatment (microbes, malignant tumours, pseudotumours) by: - modifications induced in molecular structures of medicines following exposure to optical (un-coherent, coherent) radiation. - developing new ways to deliver the medicine to target Medicines: cytostatics, either commercially available or specifically designed

APPLICATION Levels/steps of research development: - exposure of the medicines solutions to optical radiation and study the molecular modifications, if any; - evidence the modifications induced in cell cultures at interaction with solutions containing modified medicines; -evidence action of the modified medicine on pseudotumour tissues produced on eyes; - generation and study of microdroplets which contain medicines in solution

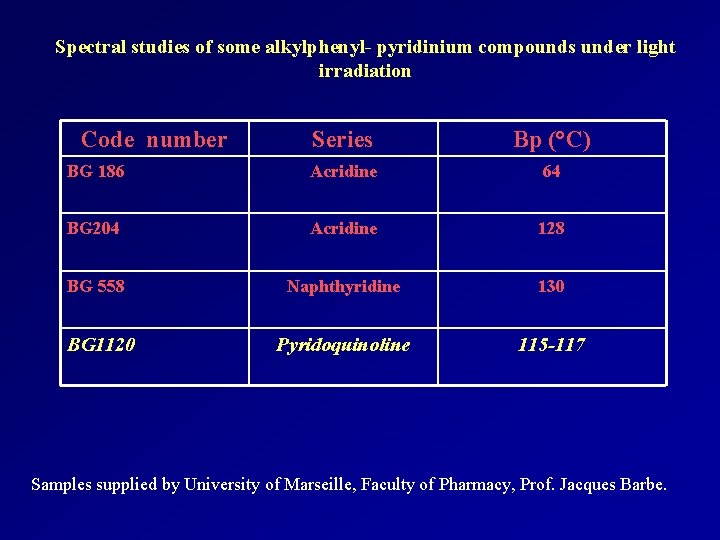
Spectral studies of some alkylphenyl- pyridinium compounds under light irradiation Code number Series Bp (°C) BG 186 Acridine 64 BG 204 Acridine 128 BG 558 Naphthyridine 130 BG 1120 Pyridoquinoline 115 -117 Samples supplied by University of Marseille, Faculty of Pharmacy, Prof. Jacques Barbe.

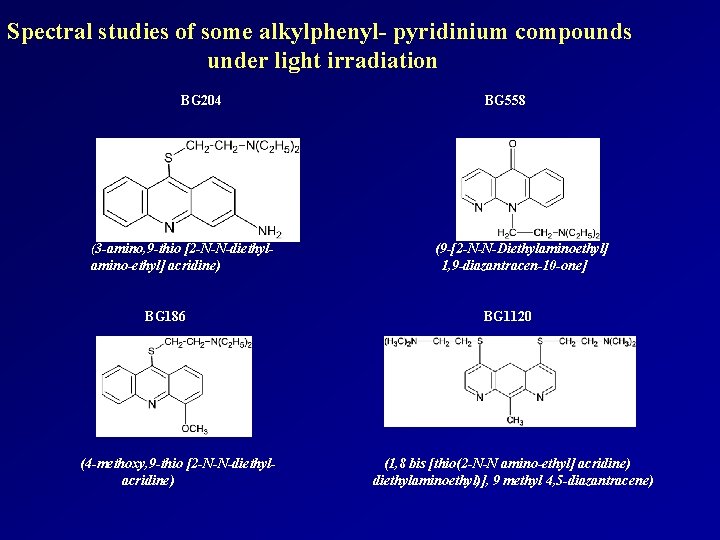
Spectral studies of some alkylphenyl- pyridinium compounds under light irradiation BG 204 (3 -amino, 9 -thio [2 -N-N-diethyl- amino-ethyl] acridine) BG 186 (4 -methoxy, 9 -thio [2 -N-N-diethylacridine) BG 558 (9 -[2 -N-N-Diethylaminoethyl] 1, 9 -diazantracen-10 -one] BG 1120 (1, 8 bis [thio(2 -N-N amino-ethyl] acridine) diethylaminoethyl)], 9 methyl 4, 5 -diazantracene)

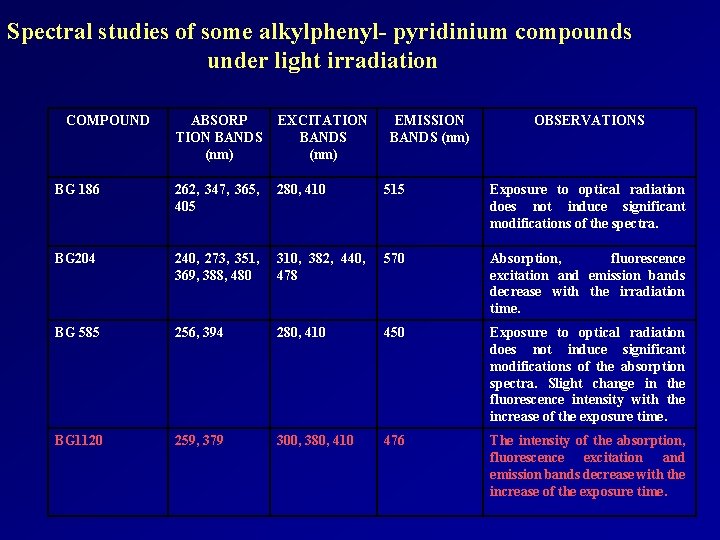
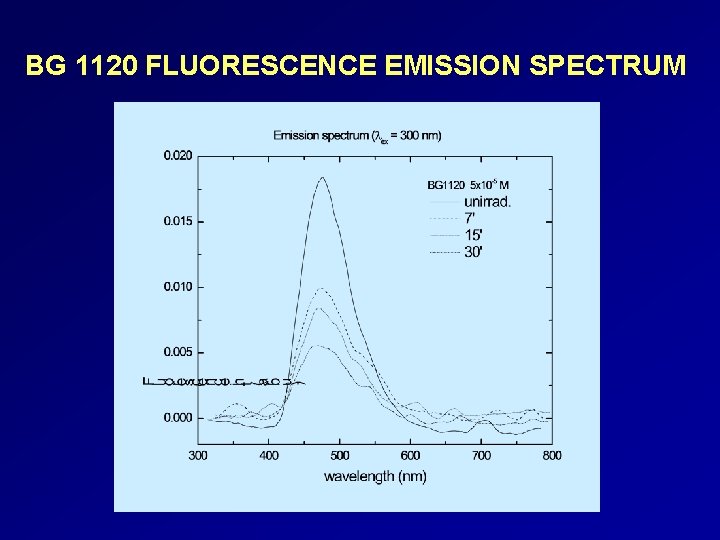
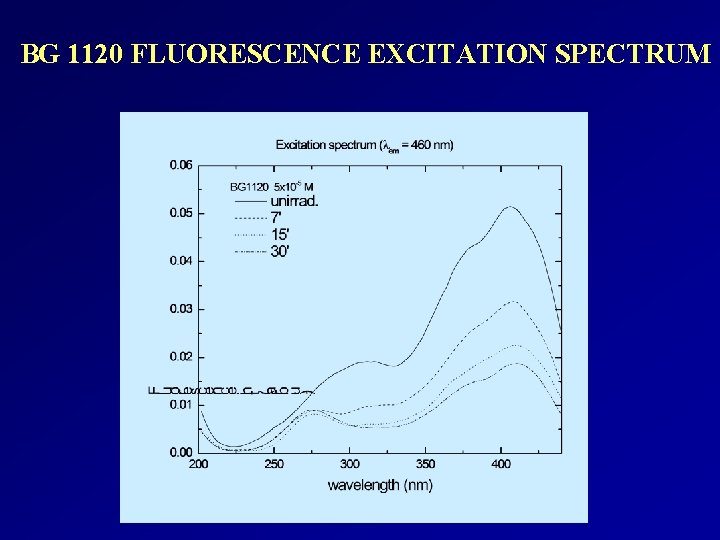
Spectral studies of some alkylphenyl- pyridinium compounds under light irradiation COMPOUND ABSORP TION BANDS (nm) EXCITATION BANDS (nm) EMISSION BANDS (nm) OBSERVATIONS BG 186 262, 347, 365, 405 280, 410 515 Exposure to optical radiation does not induce significant modifications of the spectra. BG 204 240, 273, 351, 369, 388, 480 310, 382, 440, 478 570 Absorption, fluorescence excitation and emission bands decrease with the irradiation time. BG 585 256, 394 280, 410 450 Exposure to optical radiation does not induce significant modifications of the absorption spectra. Slight change in the fluorescence intensity with the increase of the exposure time. BG 1120 259, 379 300, 380, 410 476 The intensity of the absorption, fluorescence excitation and emission bands decrease with the increase of the exposure time.

Samples: Water solutions of BG 204 (2 x 10 -5 M ) , BG 558 and BG 1120 (5 x 10 -5 M) - concentration currently used for clinical applications; Water solutions of BG 186 at 5 x 10 -5 M in 2% methanol Irradiation: Xe lamp, power density 11 m. W/cm 2, spectral range 200 -1100 nm, irradiation time: 4 min ÷ 180 min Spectra: Absorption, fluorescence and FTIR spectra were made; for FTIR, solutions exposed to radiation up to 3 hours, then poured on KBr crystal and let one day to dry. The resulted solid samples were grounded and pressed at 1. 575 x 105 kg cm-2 which allowed to obtain pellets used further to be measured by FTIR.

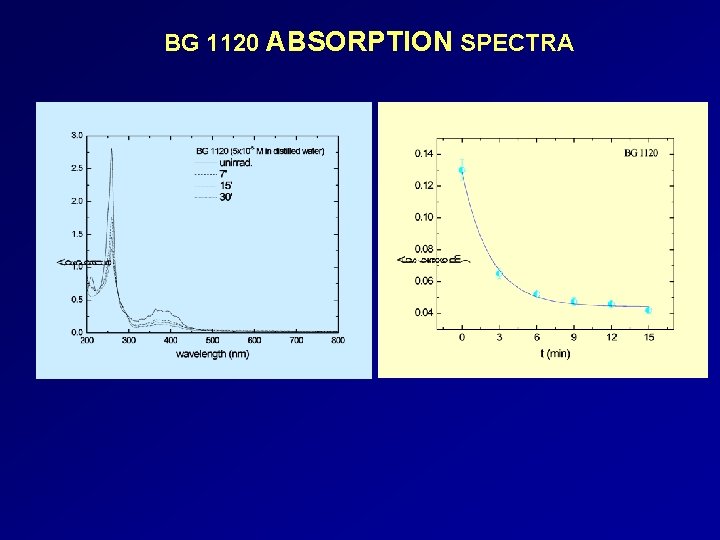
BG 1120 ABSORPTION SPECTRA

BG 1120 FLUORESCENCE EMISSION SPECTRUM

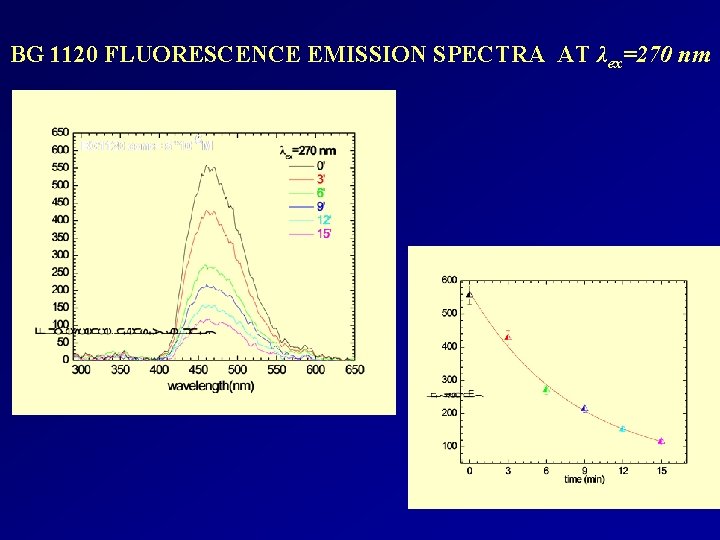
BG 1120 FLUORESCENCE EMISSION SPECTRA AT λex=270 nm

BG 1120 FLUORESCENCE EXCITATION SPECTRUM

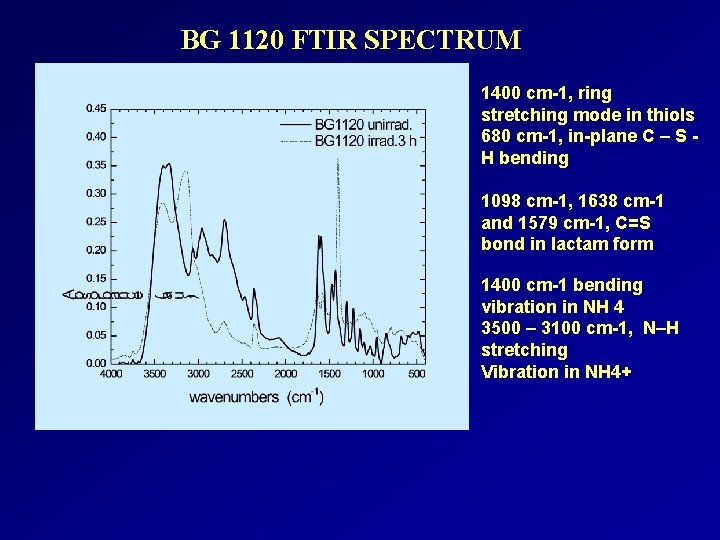
BG 1120 FTIR SPECTRUM 1400 cm-1, ring stretching mode in thiols 680 cm-1, in-plane C – S H bending 1098 cm-1, 1638 cm-1 and 1579 cm-1, C=S bond in lactam form 1400 cm-1 bending vibration in NH 4 3500 – 3100 cm-1, N–H stretching Vibration in NH 4+

BG 1120: POSSIBLE PHOTOCHEMICAL PROCESSES

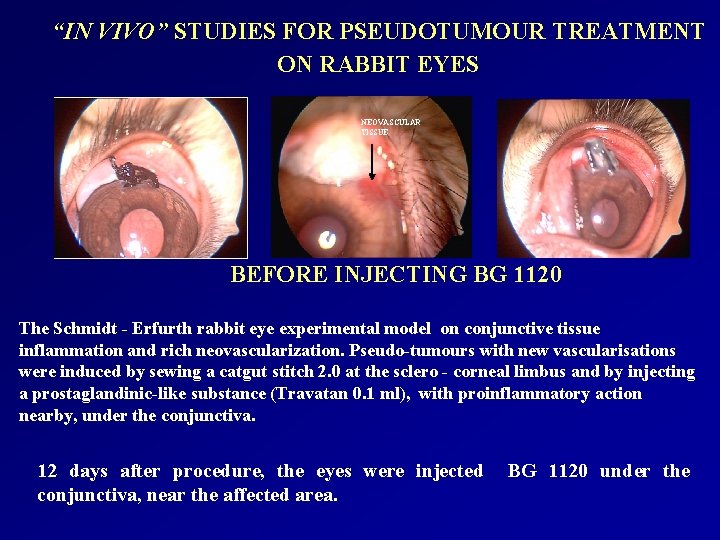
“IN VIVO” STUDIES FOR PSEUDOTUMOUR TREATMENT ON RABBIT EYES NEOVASCULAR TISSUE BEFORE INJECTING BG 1120 The Schmidt - Erfurth rabbit eye experimental model on conjunctive tissue inflammation and rich neovascularization. Pseudo-tumours with new vascularisations were induced by sewing a catgut stitch 2. 0 at the sclero - corneal limbus and by injecting a prostaglandinic-like substance (Travatan 0. 1 ml), with proinflammatory action nearby, under the conjunctiva. 12 days after procedure, the eyes were injected conjunctiva, near the affected area. BG 1120 under the

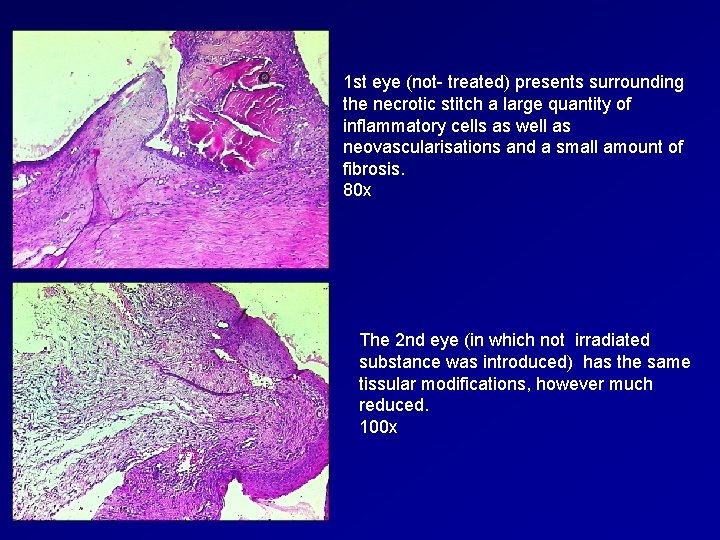
1 st eye (not- treated) presents surrounding the necrotic stitch a large quantity of inflammatory cells as well as neovascularisations and a small amount of fibrosis. 80 x The 2 nd eye (in which not irradiated substance was introduced) has the same tissular modifications, however much reduced. 100 x

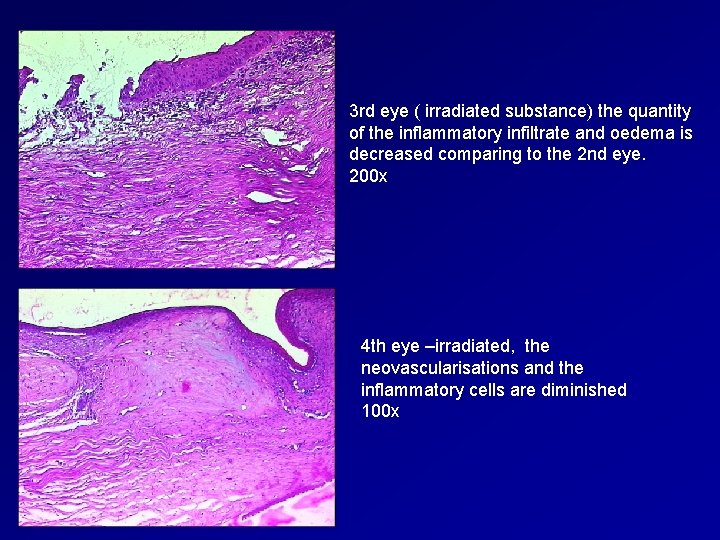
3 rd eye ( irradiated substance) the quantity of the inflammatory infiltrate and oedema is decreased comparing to the 2 nd eye. 200 x 4 th eye –irradiated, the neovascularisations and the inflammatory cells are diminished 100 x

CONCLUSIONS q The photo-transformation of the BG 1120 analysed samples shows that the molecules are photo-reactive and their therapeutic effect may be improved by exposure to UV-VIS radiation. q BG 1120 decreased inflammation and neovascularisations in the conjunctival tissue, especially after being irradiated. q Two mechanisms could be involved in the process: • the cytostatic mechanism: interference with the DNA synthesis and the cellular replication in the tissues with active proliferation. • the photosensitising mechanism that leads to the singlet oxygen and free radicals and finally to necrosis of the “treated” tissue. q Direct exposure of the eye injected not recommended.

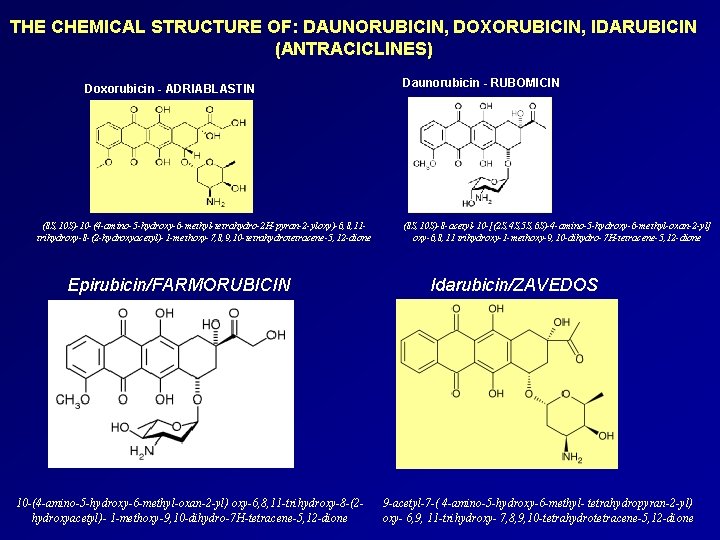
THE CHEMICAL STRUCTURE OF: DAUNORUBICIN, DOXORUBICIN, IDARUBICIN (ANTRACICLINES) Doxorubicin - ADRIABLASTIN (8 S, 10 S)-10 -(4 -amino-5 -hydroxy-6 -methyl-tetrahydro-2 H-pyran-2 -yloxy)-6, 8, 11 trihydroxy-8 -(2 -hydroxyacetyl)-1 -methoxy-7, 8, 9, 10 -tetrahydrotetracene-5, 12 -dione Epirubicin/FARMORUBICIN 10 -(4 -amino-5 -hydroxy-6 -methyl-oxan-2 -yl) oxy-6, 8, 11 -trihydroxy-8 -(2 hydroxyacetyl)- 1 -methoxy-9, 10 -dihydro-7 H-tetracene-5, 12 -dione Daunorubicin - RUBOMICIN (8 S, 10 S)-8 -acetyl-10 -[(2 S, 4 S, 5 S, 6 S)-4 -amino-5 -hydroxy-6 -methyl-oxan-2 -yl] oxy-6, 8, 11 trihydroxy-1 -methoxy-9, 10 -dihydro-7 H-tetracene-5, 12 -dione Idarubicin/ZAVEDOS 9 -acetyl-7 -( 4 -amino-5 -hydroxy-6 -methyl- tetrahydropyran-2 -yl) oxy- 6, 9, 11 -trihydroxy- 7, 8, 9, 10 -tetrahydrotetracene-5, 12 -dione

DAUNORUBICIN (DNR), DOXORUBICIN (DOX), IDARUBICIN (ANTRACICLINES) -2000 analogues of DOX and DNR synthesised to reduce the negative effects on organisms; - Only some of them, exhibiting a reduced degree of toxivity were clinically studied: Idarubicin, Epirubicin -Antibiotics in the antraciclin class which act as “intercalator” and may generate free radicals.

ABSORPTION SPECTRA: DAUNORUBICIN, DOXORUBICIN, IDARUBICIN (ANTRACICLINES)

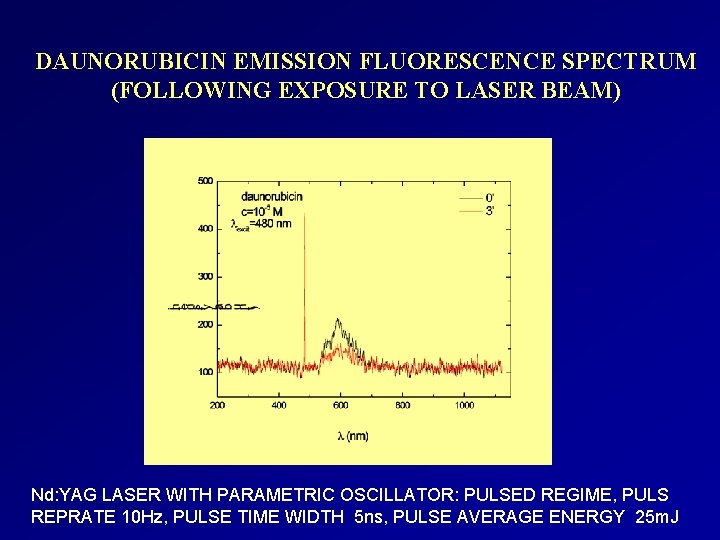
DAUNORUBICIN EMISSION FLUORESCENCE SPECTRUM (FOLLOWING EXPOSURE TO LASER BEAM) Nd: YAG LASER WITH PARAMETRIC OSCILLATOR: PULSED REGIME, PULS REPRATE 10 Hz, PULSE TIME WIDTH 5 ns, PULSE AVERAGE ENERGY 25 m. J

MICRODROPLETS STUDIES OBJECTIVES The characterization of wetting properties of generated droplets, on hydrophobic and superhydrophobic surfaces, considering the specificity of the droplets content and their potential use in malignant tumours treatment. MEASUREMENTS -Surface Tensions (ST) -Contact Angle The studies of the drug molecules adsorption performed at liquid-air and liquid-solid interface (surfaces with water contact angle above 90° - hydrophobic and above 150° superhydrophobic). MATERIALS Solutions containing: * Anthracene derivative - 4, 5 bis[thio(2 NNdimethylaminoethyl)], 9 methyl 1, 8 diazaanthracene, code BG 1120; * Doxorubicin – pure substance commercially available; at 10 -3 M, 10 -4 M and 10 -5 M concentrations in water METHOD Drop Shape Analysis MEASUREMENTS SYSTEMS: - Profile Analysis Tensiometer (Sinterface, PAT 1) - Automatic Surface Tension Real-time Acquisition – ASTRA

Surface tension measurements Capillary flow σAW σSW σAS Air - water /medicines water solutions - solid interface Contact angle water drop/solid surface θ < 90º: hydrophilic surface good wetting Contact angle water drop/solid surface θ > 90º : hydrophobic surface poor wetting Contact angle water drop/solid surface θ > 150º : super hydrophobic or ultraphobic surface – even poorer wetting The drop shape is governed by the action of forces pulling at the three-phase contact line of the drop in the plane of solid. σAS = σSW + σAW cos θ (Young equation, 1807) Surface tension (N/m) = Surface energy (J/m 2 )

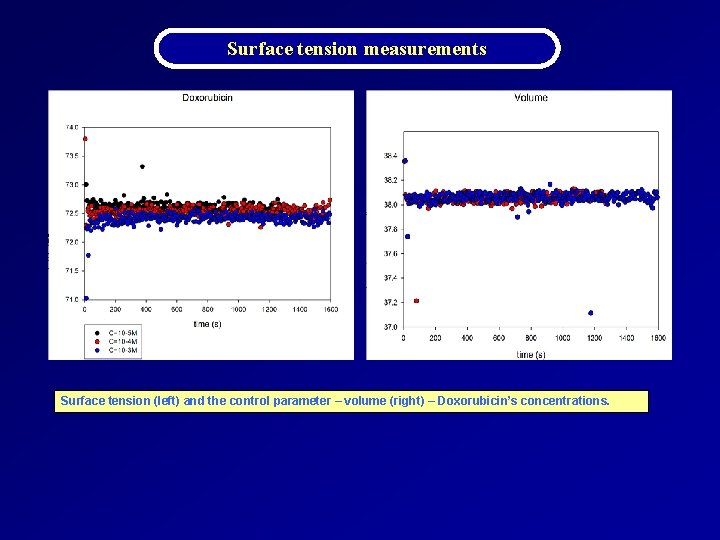
Surface tension measurements Concentration BG 1120 γ (m. N/m) Doxorubicin γ (m. N/m) C = 1 x 10 -5 M 72. 3 (± 0. 8) 72. 5 (± 0. 6) C = 1 x 10 -4 M 71. 1 (± 0. 6) 72. 3 (± 0. 9) C = 1 x 10 -3 M 71. 0 (± 0. 9) 72. 3 (± 0. 4) C = 1 x 10 -3 M - RO 69. 6 (± 1. 2) - Water γ (m. N/m) Solvent Ultrapure water 72. 4 (± 0. 6) Distilled water Measurements concerning Dynamic Surface Tension (DST) showed that in BG 1120 case, the obtained values for the solutions prepared in laboratory and for the solution prepared four months ago, are closed; within the limits of the errors of measurement are close to the pure water value. The same holds for Doxorubicin.

Surface tension measurements Surface tension (left) and the control parameter – volume (right) – Doxorubicin’s concentrations.

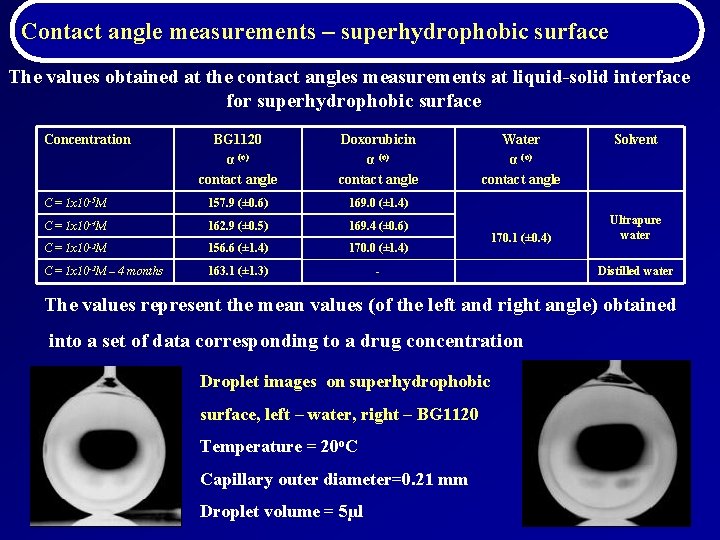
Contact angle measurements – superhydrophobic surface The values obtained at the contact angles measurements at liquid-solid interface for superhydrophobic surface Concentration BG 1120 α (o) contact angle Doxorubicin α (o) contact angle C = 1 x 10 -5 M 157. 9 (± 0. 6) 169. 0 (± 1. 4) C = 1 x 10 -4 M 162. 9 (± 0. 5) 169. 4 (± 0. 6) C = 1 x 10 -3 M 156. 6 (± (± 1. 4) 170. 0 (± 1. 4) C = 1 x 10 -3 M – 4 months 163. 1 (± 1. 3) - Water α (o) contact angle 170. 1 (± 0. 4) Solvent Ultrapure water Distilled water The values represent the mean values (of the left and right angle) obtained into a set of data corresponding to a drug concentration Droplet images on superhydrophobic surface, left – water, right – BG 1120 Temperature = 20 o. C Capillary outer diameter=0. 21 mm Droplet volume = 5µl

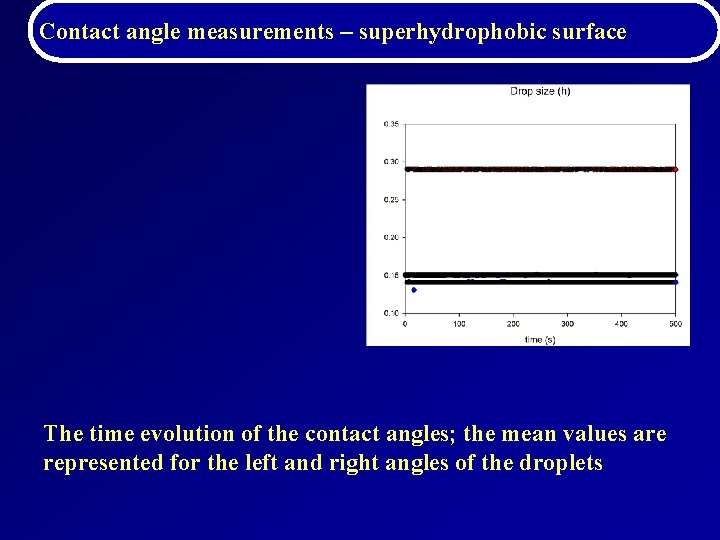
Contact angle measurements – superhydrophobic surface The time evolution of the contact angles; the mean values are represented for the left and right angles of the droplets (generated on the same local superhydrophobic surface) for a set of data corresponding to BG 1120 concentrations; the control parameter was represented by the drop size (h)

Contact angle measurements – superhydrophobic surface The time evolution of the contact angles; the mean values are represented for the left and right angles of the droplets

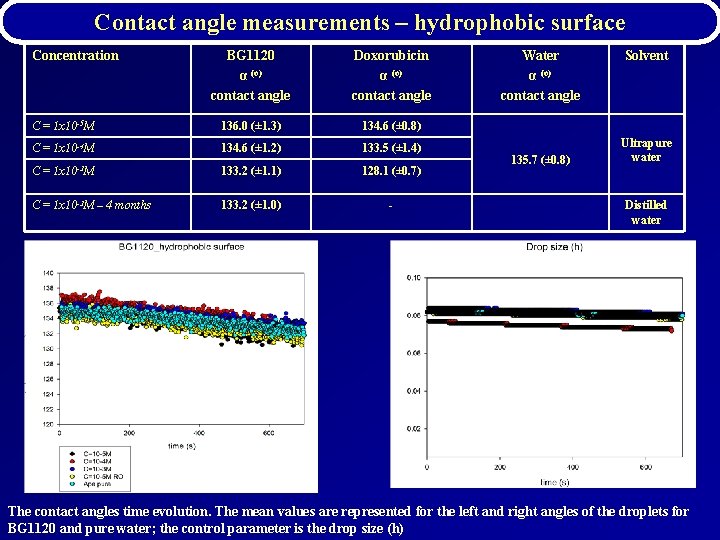
Contact angle measurements – hydrophobic surface Concentration BG 1120 α (o) contact angle Doxorubicin α (o) contact angle C = 1 x 10 -5 M 136. 0 (± 1. 3) 134. 6 (± 0. 8) C = 1 x 10 -4 M 134. 6 (± 1. 2) 133. 5 (± 1. 4) C = 1 x 10 -3 M 133. 2 (± 1. 1) 128. 1 (± 0. 7) C = 1 x 10 -3 M – 4 months 133. 2 (± 1. 0) - Water α (o) contact angle 135. 7 (± 0. 8) Solvent Ultrapure water Distilled water The contact angles time evolution. The mean values are represented for the left and right angles of the droplets for BG 1120 and pure water; the control parameter is the drop size (h)

MOVIE H 2 O on superhydrofobic surface BG 1120 on superhydrofobic surface Droplet volume = 5 µl = ct. ; Dcapillary = 0. 21 mm

Conclusions w. The measurements show that the equilibrium values of the ST are constant in time and equal for all drugs concentrations - the ST values do not depend on the drug concentrations in the solutions. w. The values are close to pure water ones – i. e. at drug concentrations which were used the adsorption effects at water-air interface are absent or very low and the distribution of drug molecules in droplet is uniform and remains constant in time w. The equilibrium values of contact angles of droplets on superhydrophobic surfaces do not depend on the solution concentrations - the droplets do not wet the surface; they have a good compatibility with it, from the point of view of the droplet instrumentation. w. The equilibrium values of the contact angles of the droplets on hydrophobic surfaces do not depend on the solution concentrations and are nearly as high as for the pure water.

Conclusions Staff: 12 people; two young scientists hired w Equipment aquisition, such as : Nd: YAG/OPO laser, computing equipment, optics equipment/components to measure LIF, etc. w Oportunity to cooperate/continue cooperation in Romania and abroad. -In Romania: * Continuing: +University Bucharest , Faculty of Physics + Central Military Hospital, Ophthalmology Clinic, Bucharest * New: -Abroad: * New: + University Bucharest, Faculty of Biology + Faculty of Pharmacy, Universite Mediterrannee of Marseille; + CNR- Institute for Energetics and Interphases, Genova

Conclusions International cooperation: -European COST networks: +Network P 21 Physics of Droplets + Networks B 16, BM 0701 Resistance to treatment + Romanians in foreign laboratories: 2 > STSM at Genova; in preparation at Berlin/Max Planck Institute > working stage at Faculty of Biology, University of Becancon + Foreign scientists 1: from Genova to Bucharest, end 2008 w Published papers in ISI journals: 5, at the moment w Communications: 22 + In Romania at National conferences: 8 (CNF, Annual Conference of the Physics Faculty, University of Bucharest, National Conferences of opthalmology, INDLAS) + INVITED PAPERS at International conferences: 14 (4 LASER FLORENCE, Italy; 3 COST medicine/biology conferences – Istanbul, Copenhagen, Budapest; 1 AMAA - Bucharest; 1 SIS 2008 – Berlin; 1 COST physics conference – Erlangen; 3 International opthalmology conferences- Sinaia, Viena, Bucharest; 1 accepted at the Gordon Conference 2009, Galvestone USA )