ACIDS y BASES Characteristics of Acids Taste Sour

ACIDS y BASES

Characteristics of Acids • Taste Sour • Affect indicators (red=acid) • Neutralize Bases • Often produce hydrogen gas • p. H between 0 and <7

Characteristics of Bases • Taste Bitter • Feel Slippery • Neutralize Acids (Antacids) • Affect indicators (base=blue) • p. H between >7 and 14 • Dissolve grease (Drano, Windex)

Examples of Acids • HCl • H 2 SO 4 • HNO 3 • HF • Juices

Examples of Bases • Na. OH • Ca(OH)2 • KOH • Soap, Ammonia, Lye, Baking Soda

There are 3 definitions for acids and bases • Arrhenius Theory • Brønsted-Lowry Theory • Lewis Theory
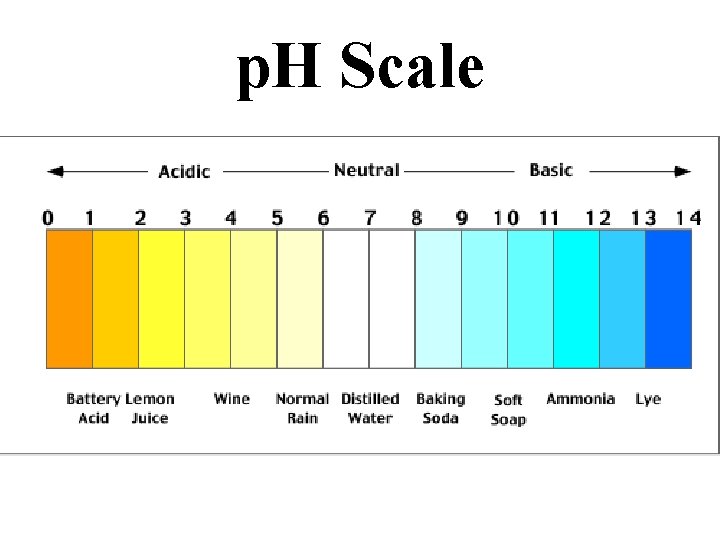
p. H Scale
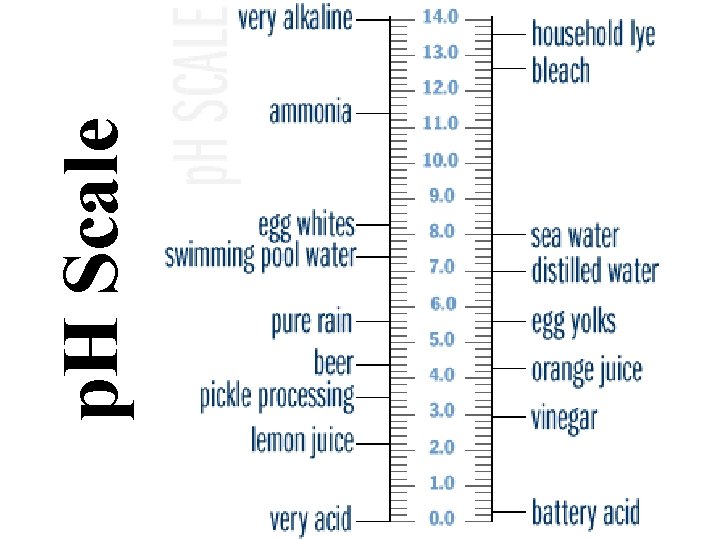
p. H Scale
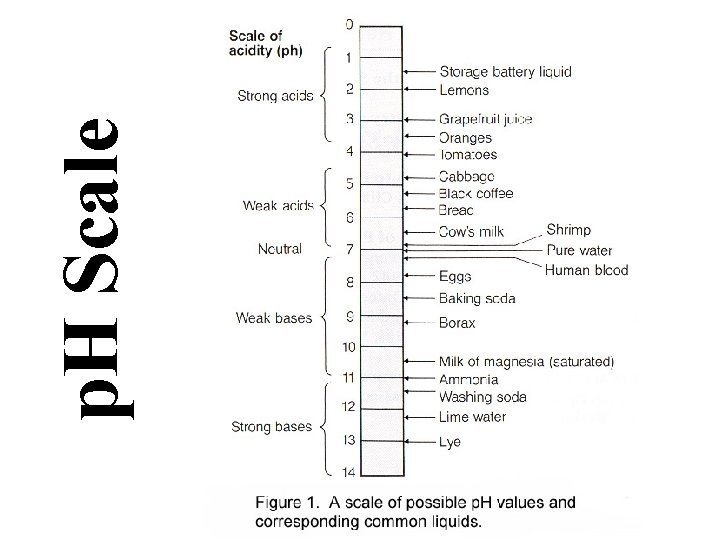
p. H Scale
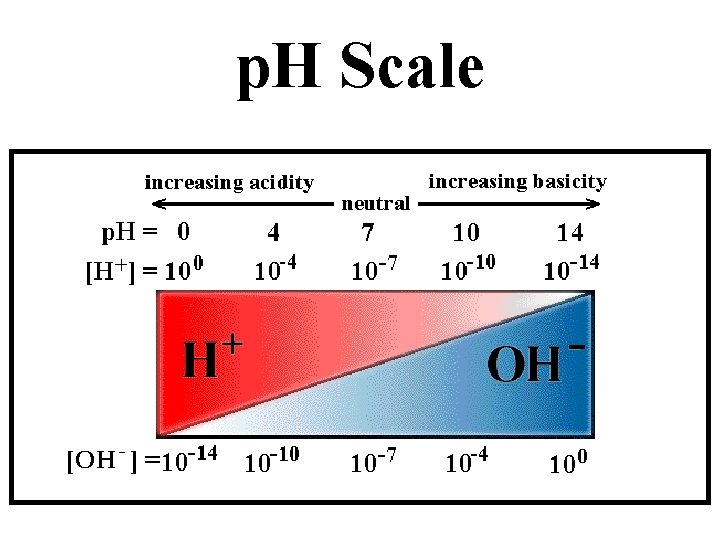
p. H Scale

Arrehenius Theory ON ACIDS… An acid is a substance that ionizes in water to + give hydrogen ions (H )

Arrehenius Theory ON BASES… A base is a substance that ionizes in water to give hydroxide ions (OH )
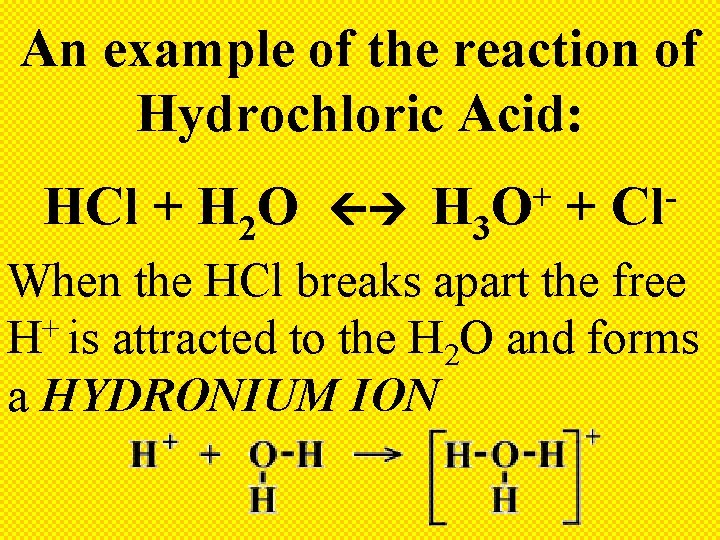
An example of the reaction of Hydrochloric Acid: HCl + H 2 O H 3 + O + Cl When the HCl breaks apart the free + H is attracted to the H 2 O and forms a HYDRONIUM ION

So who is the Arrhenius Acid given: HCl + H 2 O + H 3 O HCl + Cl Why? + Because it produces H

An example of the reaction of Potassium Hydroxide: KOH + H 2 O + K + 2 OH When the KOH breaks apart it forms the HYDROXIDE ION (OH )

So who is the Arrhenius Base given: KOH + H 2 O Why? + K + 2 OH KOH Because it produces OH

The world was cool with the Arrehenius definition until NH 3 (Ammonia) came along. When combined with water it increased the number of OH ions…looks like we need a new definition

Brønsted-Lowry Theory ON ACIDS… Acids are substances that are capable of donating a proton

Brønsted-Lowry Theory ON BASES… Bases are substances capable of accepting a proton

Example Brønsted Acids and Bases: NH 3 + H 2 O NH 4 + + OH Here, H 2 O acts as a Brønsted acid by donating a proton to NH 3 which acts as a Brønsted base.
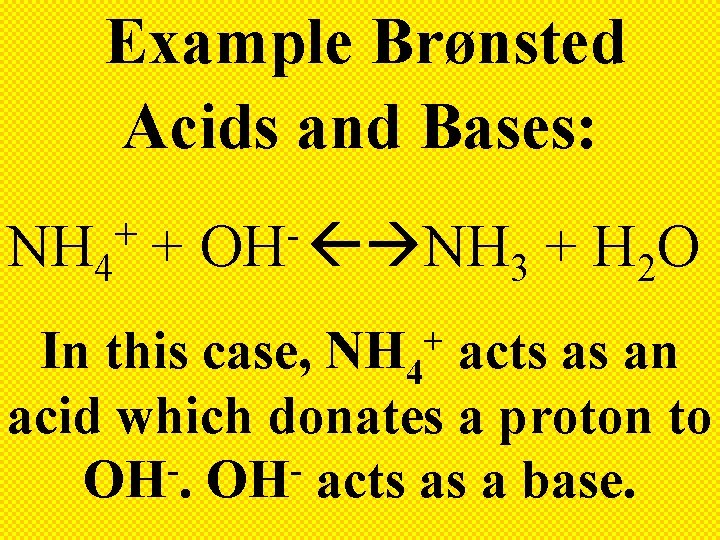
Example Brønsted Acids and Bases: + NH 4 + OH NH + H O 3 2 In this case, NH 4 acts as an acid which donates a proton to OH. OH acts as a base. +

Lewis Theory ON ACIDS… Acids are substances that are electron-pair receptors

Lewis Theory ON BASES… Bases are substances capable of donating electron-pairs

Conjugate Base The substance that remains after an acid has donated a + H ion

Conjugate Acid The substance formed when a base + accepts a H ion
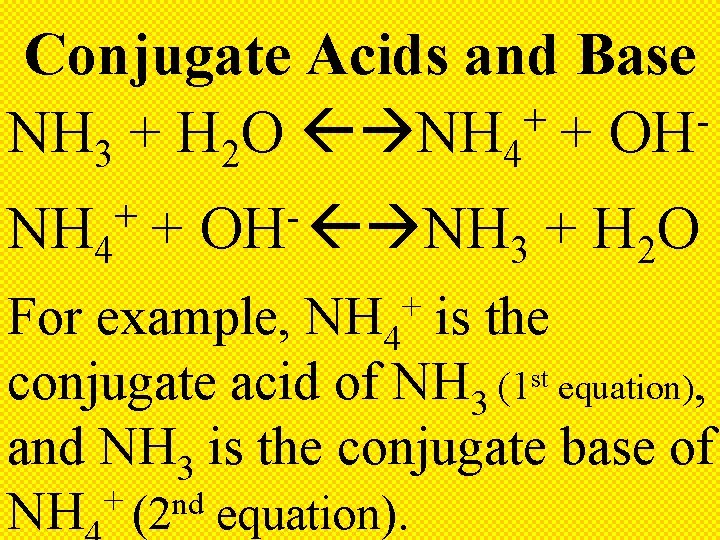
Conjugate Acids and Base + NH 3 + H 2 O NH 4 + OH + NH 4 + OH NH + H O 3 2 For example, NH 4 is the conjugate acid of NH 3 (1 st equation), and NH 3 is the conjugate base of + nd NH (2 equation). +

Interesting fact… Water can act as an acid or a base!!!

Amphoteric A substance that can act as either an acid or a base.
- Slides: 28