Acids Sour taste Change the color of acidbase

Acids Sour taste • Change the color of acid-base indicators • (ex. turn litmus to red) Some react with metals and release hydrogen gas • React with bases to produce salts and water • (salt = cation from a base and anion from an acid) Contain H+ when dissolved in aqueous solutions • p. H less than 7 • Binary acids, oxyacids and organic acids •

Bases Taste bitter • Change the color of acid-base indicators • (ex. turn litmus to blue) Dilute solutions feel slippery • React with acids to produce salts and water • p. H more than 7 • Many soluble bases (alkali) contain hydroxide ions (OH-) •

Definitions of Acids and Bases 1. Arrhenius ◦ Acid : compound that creates H+ in a solution ◦ Base : compound that creates OH- in a solution �The H+ ion attracts other molecules so strongly that it normally exists as the hydronium ion (H 3 O+) when in water

Definitions of Acids and Bases (con’t) 2. Brønsted-Lowry • Most commonly used definition for acids and bases • Created by two scientists around the same time (1923) • A Brønsted-Lowry acid is a molecule or ion that is a proton donor (hydrogen ion, H+). • Ex. Hydrogen chloride acts as a Brønsted-Lowry acid when it reacts with ammonia. HCl + NH 3 → NH 4+ + Cl- • Water can act as a Brønsted-Lowry acid NH 3(aq) + H 2 O(l) → NH 4+ + OH-
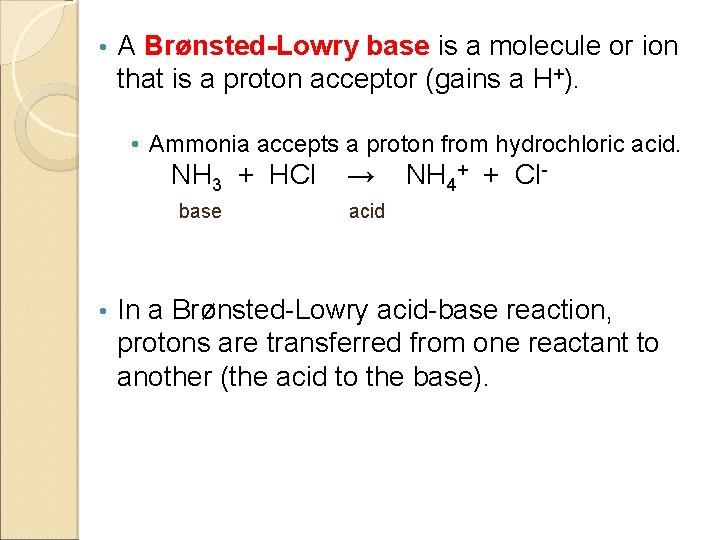
• A Brønsted-Lowry base is a molecule or ion that is a proton acceptor (gains a H+). • Ammonia accepts a proton from hydrochloric acid. NH 3 + HCl base • → NH 4+ + Cl- acid In a Brønsted-Lowry acid-base reaction, protons are transferred from one reactant to another (the acid to the base).

Conjugate acids and bases • The substance that remains after a Brønsted-Lowry acid has given up a proton is the conjugate base. • The substance that is formed when a base accepts a proton is the conjugate acid. HF(aq) + H 2 O(l) → acid base conjugate F-(aq) + H 3 O+(aq) conjugate base acid

• Monoprotic acids • Only have one acidic proton (one hydrogen ion) • ex. HCl. O 4, HCl, HNO 3 • Polyprotic acids (diprotic, triprotic) • Can donate more than one proton per molecule • ex. H 2 SO 4, H 3 PO 4

Interesting fact… Water can act as an acid or a base!!! Amphoteric compound: Any compound that can react as either an acid or a base

Measuring acidity or basicity p. H is a measure of how acidic or basic a solution is. (“p” stands for potential and “H” stands for hydrogen; therefore, the potential of a substance to attract hydrogen ions) p. H ranges from 0 to 14. • Acidic solutions have p. H values < 7 • A p. H of 7 is neutral. • Basic solutions have p. H values >7
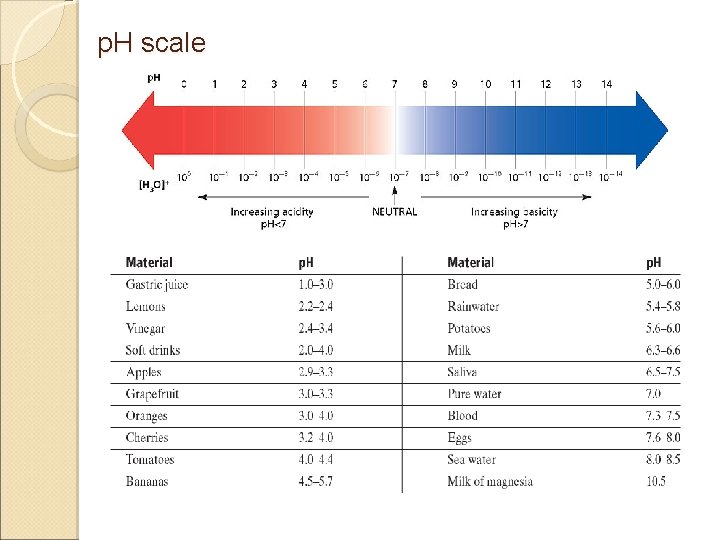
p. H scale
![p. H Concentration of pure water at 25°C: [H+] = 1. 0 × 10− p. H Concentration of pure water at 25°C: [H+] = 1. 0 × 10−](http://slidetodoc.com/presentation_image_h2/d0d293987a116d79e5827b13aa5fb578/image-11.jpg)
p. H Concentration of pure water at 25°C: [H+] = 1. 0 × 10− 7 M [OH−] = 1. 0 × 10− 7 M Brackets, [ ], symbolize the molarity concentration

Calculating p. H �The p. H of a solution is the negative logarithm of the hydrogen ion concentration, [H+]. p. H = −log [H+] ◦ example: for a neutral solution [H+] = 1× 10− 7 p. H = −log [H+] = −log(1 × 10− 7) = −(− 7. 0) = 7. 0 �The p. OH of a solution is defined as the negative logarithm of the hydroxide ion concentration, [OH−]. p. OH = −log [OH–] �p. H + p. OH = 14. 0
![Practice problems 1. What is the p. H of coffee if the [H+] is Practice problems 1. What is the p. H of coffee if the [H+] is](http://slidetodoc.com/presentation_image_h2/d0d293987a116d79e5827b13aa5fb578/image-13.jpg)
Practice problems 1. What is the p. H of coffee if the [H+] is 1 x 10− 5 M? STEP 1 Enter the [H+] value: Enter 1 x 10 5 (press 1 EE 5, then change sign ) STEP 2 Press log key and change the sign: log (1 x 10− 5) = [ 5] STEP 3 Make the number of digits after the decimal point equal to the number of significant figures in the coefficient: [H+]= 1 x 10− 5 Answer: p. H is 5. 0 13

Stomach acid (HCl) has a concentration of approximately 2 x 10− 4 M. What is the p. H of the solution? STEP 1 Enter the [H+] value: 2 x 10 4 (press 2 EE 4, then change sign) STEP 2 Press log key and change the sign: log (2 x 10− 4) = [ 3. 7] STEP 3 Make the number of digits after the decimal point equal to the number of significant figures in the coefficient [H+] = 2 x 10− 4 Answer: p. H is = 3. 7
![Calculating [H+] from p. H What is the concentration of nitric acid (HNO 3) Calculating [H+] from p. H What is the concentration of nitric acid (HNO 3)](http://slidetodoc.com/presentation_image_h2/d0d293987a116d79e5827b13aa5fb578/image-15.jpg)
Calculating [H+] from p. H What is the concentration of nitric acid (HNO 3) that has a p. H of 3. 80? STEP 1 Enter the p. H value, change sign: – 3. 80 STEP 2 Convert p. H to concentration: Use 2 nd function key and then 10 x key or inverse key and then log key answer = 1. 584893 − 06 STEP 3 Adjust the significant figures in the coefficient (2 digit following decimal point = 2 digit in the coefficient): Answer: [HNO 3] = 1. 6 x 10− 6 M
- Slides: 15