Acids Lesson 4 Acid and Base Definitions Acid

Acids Lesson 4 Acid and Base Definitions
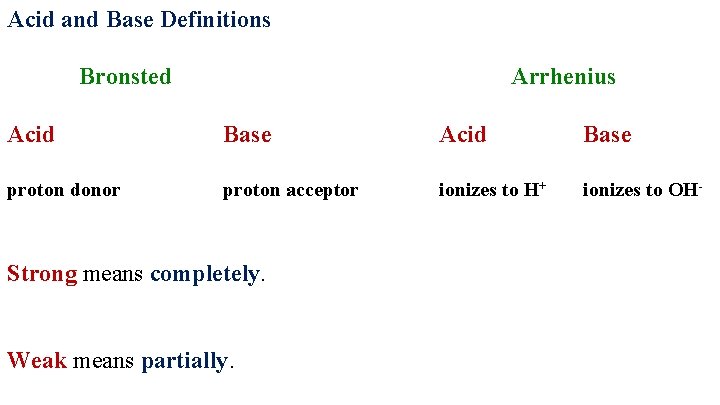
Acid and Base Definitions Bronsted Arrhenius Acid Base proton donor proton acceptor ionizes to H+ ionizes to OH- Strong means completely. Weak means partially.

Definitions 1. Strong Arrhenius Acid- completely ionizes in water to H+ HCl H+ + Cl 2. Strong Bronsted Acid- completely donates a proton to a base HCl + H 2 O H 3 O + + Cl-

3. Strong Arrhenius Base- completely ionizes in water to OH K+ + OH KOH 4. Strong Bronsted Base- completely accepts a proton from an acid NH 2 - + H 2 O NH 3 + OH

5. Weak Arrhenius Acid- partially ionizes in water to H+ + ⇄ H + F HF 6. Weak Bronsted Acid- partially donates a proton to a base ⇄ H 3 O + + HF + H 2 O F-

7. Weak Arrhenius Base- partially ionizes in water to OH Zn 2+ + 2 OH Zn(OH)2 ⇄ 8. Weak Bronsted Base- partially accepts a proton from an acid NH + H O ⇄ NH 4+ + OH 2 3
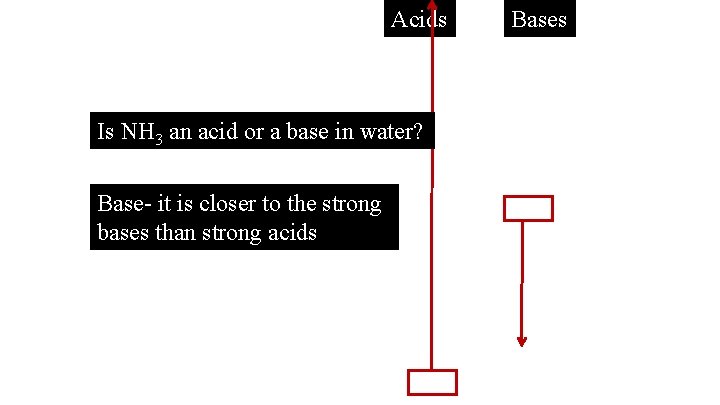
Acids Is NH 3 an acid or a base in water? Base- it is closer to the strong bases than strong acids Bases

Memorize the following NH 3 is a weak base when in water NH 4+ is a weak acid H 2 O is an acid when with a base and a base when with an acid and is amphiprotic H 2 O is always neutral when pure Write a reaction showing water acting as an base H 2 O + HIO 3 ⇄ NH 4+ + OH- Write a reaction showing water acting as a acid H 2 O + NH 3 ⇄ H 3 O+ + IO 3 -
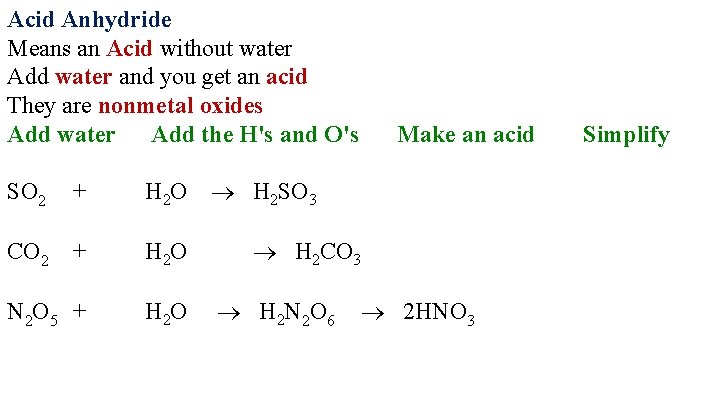
Acid Anhydride Means an Acid without water Add water and you get an acid They are nonmetal oxides Add water Add the H's and O's + H 2 O H 2 SO 3 + H 2 O N 2 O 5 + H 2 O SO 2 CO 2 Make an acid H 2 CO 3 H 2 N 2 O 6 2 HNO 3 Simplify
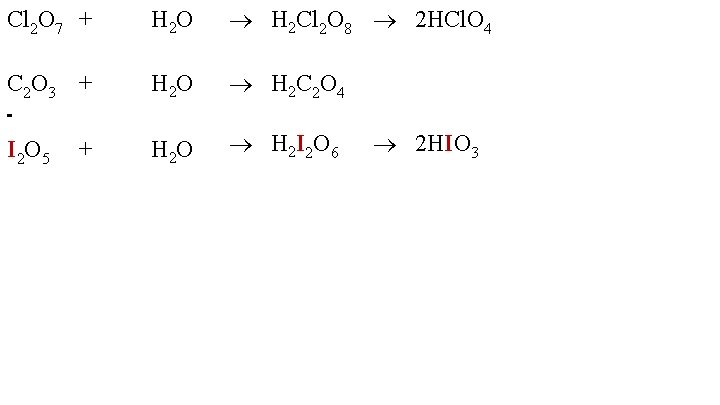
Cl 2 O 7 + C 2 O 3 + I 2 O 5 + H 2 O H 2 Cl 2 O 8 2 HCl. O 4 H 2 O H 2 C 2 O 4 H 2 O H 2 I 2 O 6 2 HIO 3

Basic anhydride Means a base without water Add water and you get a base- OH style They are metal oxides Add water Add the H's and O's Make a base- start with a metal K 2 O + H 2 O 2 KOH Ca. O + H 2 O Ca(OH)2 Na 2 O + H 2 O 2 Na. OH Ba. O + H 2 O Ba(OH)2

Classify each formula and write an equation. strong acid weak acid strong base weak base acid anhydride basic anhydride. HNO 3 Strong acid HNO 3 H+ + NO 3 - CO 2 Acid Anhydride CO 2 + H 2 O H 2 CO 3 ⇄ Mg(OH)2 H 2 C 2 O 4 H+ + HCO 3 - Strong Base Mg(OH)2 Mg 2+ + 2 OH- Weak Acid H 2 C 2 O 4 ⇄ H+ + HC 2 O 4 -

Classify each formula and write an equation. strong acid weak acid strong base weak base acid anhydride basic anhydride. Rb 2 O Basic Anhydride 2 Rb. OH Rb 2 O + H 2 O Rb. OH Rb+ + OH- NH 2 - Strong base NH 2 - + H 2 O NH 3 + OH- CN- Weak base CN- + H 2 O ⇄ HCN + OH- Na. OH Strong base Na. OH Na+ + OH-
- Slides: 13