Acids Lesson 18 Titration Curves Titration Curves A




- Slides: 10

Acids Lesson 18 Titration Curves


Titration Curves A titration curve is a graph of the p. H versus the volume in a titration. The equivalence point is the end of a titration where the stoichiometry of the reaction is exactly satisfied. moles H+ = moles OHThe transition point is when an indicator changes color and [HInd] = [Ind-].

Choosing an Indicator When you choose an indicator, you must pick one so that the transition point of the indicator matches the equivalence point of the titration. Rule of thumb Salt Equivalence Point Neutral 7 Basic 9 Acidic 5

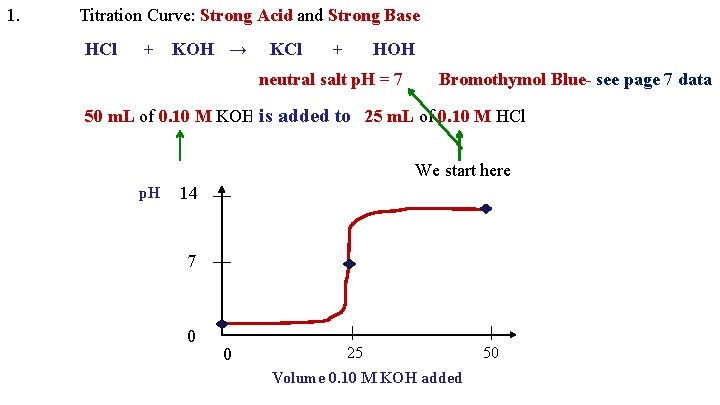
1. Titration Curve: Strong Acid and Strong Base HCl + KOH → KCl + HOH neutral salt p. H = 7 Bromothymol Blue- see page 7 data is added to 50 m. L of 0. 10 M KOH is added to 25 m. L of 0. 10 M HCl We start here p. H 14 7 0 0 25 Volume 0. 10 M KOH added 50

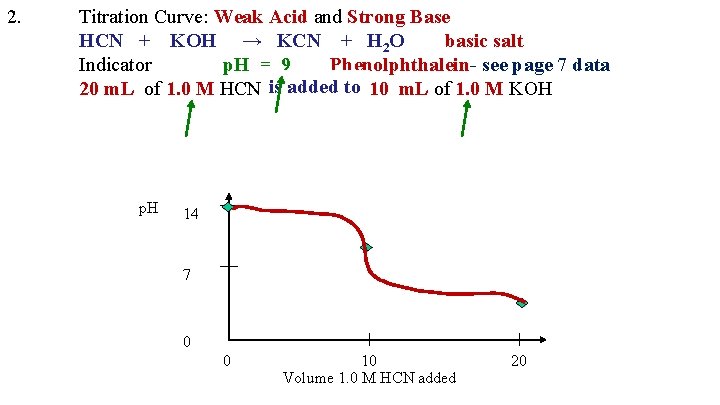
2. Titration Curve: Weak Acid and Strong Base HCN + KOH → KCN + H 2 O basic salt Indicator p. H = 9 Phenolphthalein- see page 7 data is added to 20 m. L of 1. 0 M HCN is added to 10 m. L of 1. 0 M KOH p. H 14 7 0 0 10 Volume 1. 0 M HCN added 20

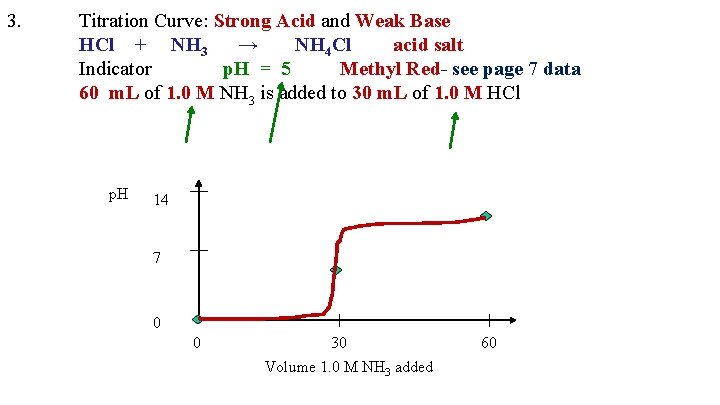
3. Titration Curve: Strong Acid and Weak Base HCl + NH 3 → NH 4 Cl acid salt Indicator p. H = 5 Methyl Red- see page 7 data 60 m. L of 1. 0 M NH 3 is added to 30 m. L of 1. 0 M HCl p. H 14 7 0 0 30 Volume 1. 0 M NH 3 added 60

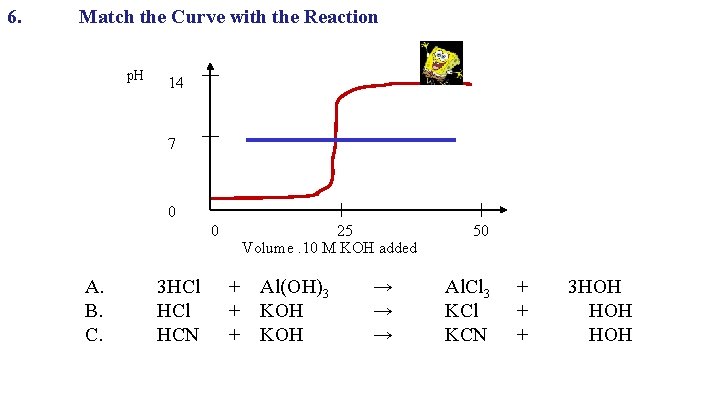
6. Match the Curve with the Reaction p. H 14 7 0 0 A. B. C. 25 Volume. 10 M KOH added 3 HCl + Al(OH)3 HCl + KOH HCN + KOH → → → 50 Al. Cl 3 KCl KCN + 3 HOH + HOH

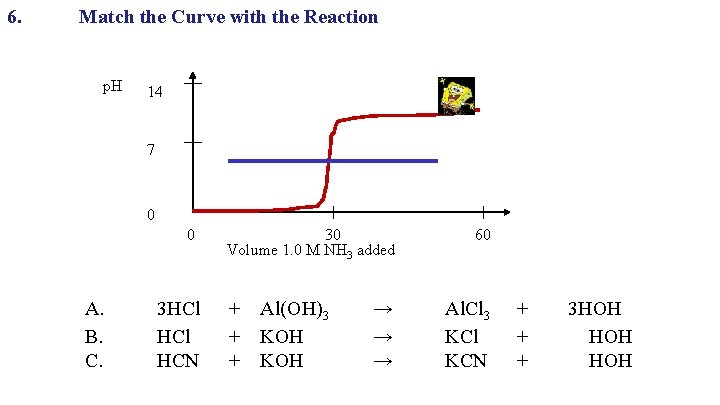
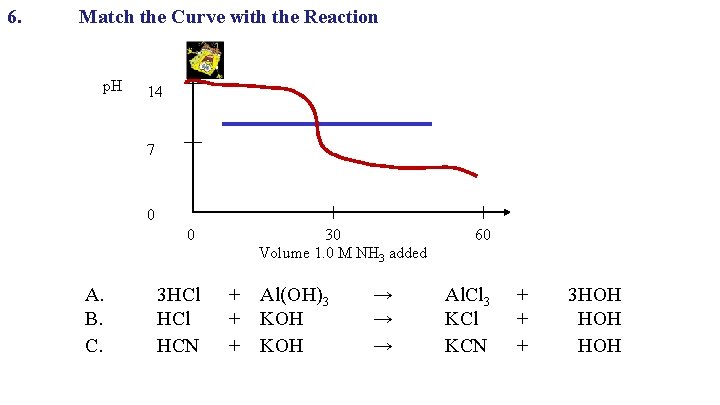
6. Match the Curve with the Reaction p. H 14 7 0 0 A. B. C. 30 Volume 1. 0 M NH 3 added 3 HCl + Al(OH)3 HCl + KOH HCN + KOH → → → 60 Al. Cl 3 KCl KCN + 3 HOH + HOH

6. Match the Curve with the Reaction p. H 14 7 0 0 A. B. C. 30 Volume 1. 0 M NH 3 added 3 HCl + Al(OH)3 HCl + KOH HCN + KOH → → → 60 Al. Cl 3 KCl KCN + 3 HOH + HOH