Acids Give foods a sour or tart taste





![Ion Product Constant for Water When [H+] increases, [OH-] decreases and vice versa For Ion Product Constant for Water When [H+] increases, [OH-] decreases and vice versa For](https://slidetodoc.com/presentation_image_h2/641f1e144060bb47d599ebdaa4cf601f/image-6.jpg)










- Slides: 17

Acids Give foods a sour or tart taste Vinegar (acetic acid) Lemons (citric acid) Aqueous solutions of acids are electrolytes Conduct electricity Causes indicators to change color Metals react with acids to produce hydrogen gas

Bases Have a bitter taste Soap Feel slippery Aqueous solutions of bases are electrolytes Cause indicators to change color Neutralize an acid

Arrhenius Acids Hydrogen containing compounds that ionize to yield hydrogen ions (H+) in aqueous solutions Only hydrogens in very polar bonds are ionizable When hydrogen is joined to a very electronegaive element Acids that produce one ionizable hydrogen are known as monoprotic acids (ex: HNO 3) Acids that produce 2 ionizable hydrogens are known as diprotic acids (ex: H 2 SO 4) Acids that produce 3 ionizable hydrogens are known as triprotic acids (ex: H 3 PO 4)

Arrhenius Bases Compounds that ionize to yield hydroxide ions (OH-) in aqueous solutions Alkali metals react with water to produce solutions that are basic Na + H 2 O Na. OH + H 2

Hydrogen Ions from Water Reactions in which water molecules produce ions is called the self-ionization of water H 2 O H+ + OH- Hydrogen ions will join with water molecules to form hydronium ions (H 3 O+) In pure water, the equilibrium concentrations of H+ and OH- are each only 1. 0× 10 -7 M This is considered to be a neutral solution
![Ion Product Constant for Water When H increases OH decreases and vice versa For Ion Product Constant for Water When [H+] increases, [OH-] decreases and vice versa For](https://slidetodoc.com/presentation_image_h2/641f1e144060bb47d599ebdaa4cf601f/image-6.jpg)
Ion Product Constant for Water When [H+] increases, [OH-] decreases and vice versa For aqueous solutions, the product of [H+] and [OH-] equals 1. 0× 10 -14 M This is called the ion product constant of water (Kw) Kw = [H+] × [OH-] = 1. 0× 10 -14 M When [H+] is greater than [OH-] --- acidic solution [H+] is greater than 1. 0 × 10 -7 M When [H+] [OH-] is greater than [H+] --- basic solution is less than 1. 0 × 10 -7 M

Problems The concentration of hydroxide ion is found to be 1. 0× 10 -11 M. Find the concentration of hydrogen ion and determine if the solution is acidic, basic, or neutral.

Problems Calculate the concentration of the other ion and classify the solution as acidic, basic, or neutral. a. b. c. [OH-] = 1. 0× 10 -6 [H+] = 1. 0× 10 -7 [H+] = 8. 1× 10 -3

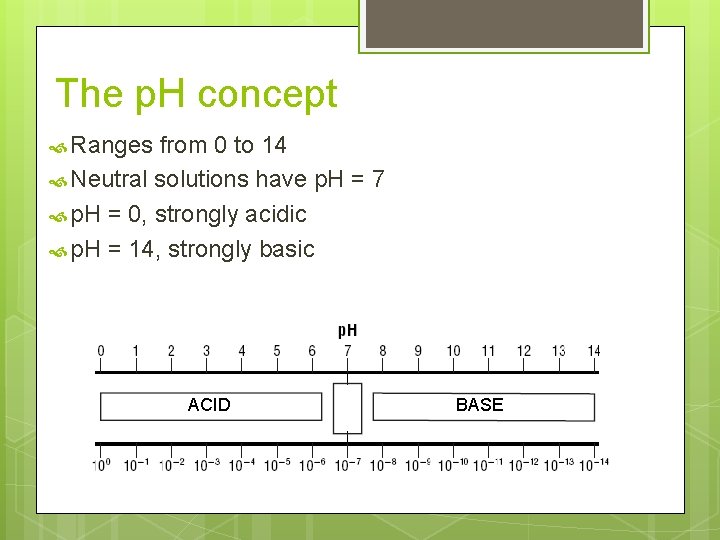
The p. H concept Ranges from 0 to 14 Neutral solutions have p. H = 7 p. H = 0, strongly acidic p. H = 14, strongly basic ACID BASE

Calculating p. H of a solution is the negative logarithm of the hydrogen ion concentration p. H = -log [H+] = 1. 0 × 10 -7 p. H = 7 p. H > 7, basic p. H < 7, acidic If [H+] is written in scientific notation and has a coefficient of 1, then the p. H of the solution equals the exponent If [H+] = 1. 0 × 10 -2 p. H = 2 (acid) p. H is an integer number, you can directly write the [H+] p. H = 9 [H+] = 1. 0 × 10 -9

Calculating p. OH of a solution is the negative logarithm of the hydroxide ion concentration p. OH = -log [OH-] p. OH = 7, neutral p. OH < 7, basic p. OH > 7, acidic p. H + p. OH = 14

Problems A carbonated soft drink is found to have a hydrogen ion concentration of 7. 3 × 10 -7 M, what is the p. H and p. OH of the drink?

Problems Calculate a) b) c) d) the p. H and p. OH of the solutions below and classify as acid, base, or neutral. [H+] = 1. 0 × 10 -2 [OH-] = 5. 6 × 10 -8 [H+] = 2. 7 × 10 -3 [OH-] = 0. 61 M

Neutralization Reactions in which an acid and a base react in aqueous solution to produce a salt and water Salts are compounds that consist of an anion from an acid and a cation from a base

Titration Process of adding a known amount of solution with a known concentration to determine the concentration of another solution Solution of known concentration is the standard solution Point at which the indicator changes color is the end point Only have water and salt remaining, plus a trace of indicator p. H = 7 When moles of hydrogen ions = moles of hydroxide ions you have reached the equivalence point

Problems In a titration, 53. 7 m. L of a 0. 100 M HCl solution is needed to neutralize 80 m. L of KOH solution. What is the molarity of the KOH solution?

A 45 m. L sample of nitric acid solution is neutralized by 119. 4 m. L of 0. 200 M Na. OH solution. What is the molarity of the nitric acid solution? A 70 m. L sample of sulfuric acid solution is neutralized by 256. 3 m. L of 0. 100 M Na. OH solution. What is the molarity of the sulfuric acid solution?