ACIDS BASES SALTS A guide for GCSE students

ACIDS, BASES & SALTS A guide for GCSE students KNOCKHARDY PUBLISHING 2010 SPECIFICATIONS

ACIDS, BASES & SALTS INTRODUCTION This Powerpoint show is one of several produced to help students understand selected GCSE Chemistry topics. It is based on the requirements of the AQA specification but is suitable for other examination boards. Individual students may use the material at home for revision purposes and it can also prove useful for classroom teaching with an interactive white board. Accompanying notes on this, and the full range of AS and A 2 Chemistry topics, are available from the KNOCKHARDY WEBSITE at. . . www. knockhardy. org. uk All diagrams and animations in this Powerpoint are original and created by Jonathan Hopton. Permission must be obtained for their use in any commercial work.

ACIDS, BASES & SALTS CONTENTS • Acidity and alkalinity • Indicators • p. H • Acids • General methods for making salts • Making salts from metal oxides • Making salts from metal carbonates • Making salts from metals • Making salts from alkalis – by titration • Making insoluble salts – by precipitation • Questions

ACIDITY AND ALKALINITY • water is a neutral substance • when substances dissolve in water, the solution may become… ACIDIC or ALKALINE or stay NEUTRAL

ACIDITY AND ALKALINITY • water is a neutral substance • when substances dissolve in water, the solution may become… ACIDIC or ALKALINE or stay NEUTRAL • hydrogen ions H+(aq) make solutions acidic • hydroxide ions OH¯(aq) make solutions alkaline • solutions with equal numbers of H+(aq) and OH¯(aq) are neutral

INDICATORS • show by a colour change if a solution is acidic, alkaline or neutral • some can even show acidic or how alkaline a solution is

INDICATORS • show by a colour change if a solution is acidic, alkaline or neutral • some can even show acidic or how alkaline a solution is • well known indicators include…

INDICATORS • show by a colour change if a solution is acidic, alkaline or neutral • some can even show acidic or how alkaline a solution is • well known indicators include… LITMUS acidic RED alkaline BLUE neutral PURPLE
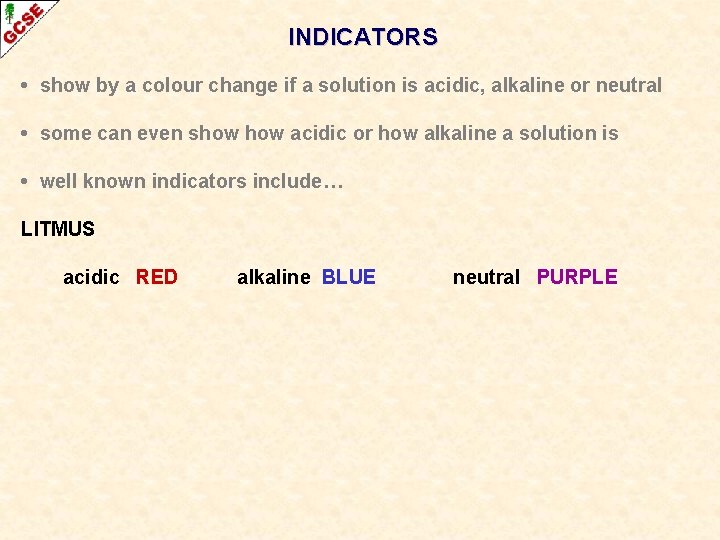
INDICATORS • show by a colour change if a solution is acidic, alkaline or neutral • some can even show acidic or how alkaline a solution is • well known indicators include… LITMUS acidic RED alkaline BLUE neutral PURPLE

INDICATORS • show by a colour change if a solution is acidic, alkaline or neutral • some can even show acidic or how alkaline a solution is • well known indicators include… LITMUS acidic RED alkaline BLUE neutral PURPLE alkaline VIOLET neutral GREEN UNIVERSAL acidic RED

INDICATORS • show by a colour change if a solution is acidic, alkaline or neutral • some can even show acidic or how alkaline a solution is • well known indicators include… LITMUS acidic RED alkaline BLUE neutral PURPLE alkaline VIOLET neutral GREEN UNIVERSAL acidic RED

p. H SCALE

p. H SCALE Used to compare the relative acidity (and alkalinity) of solutions. The value is related to the concentration of H+ions, in solution.

p. H SCALE Used to compare the relative acidity (and alkalinity) of solutions. The value is related to the concentration of H+ions, in solution. “The greater the concentration of hydrogen ions in solution, the lower the p. H”

p. H SCALE Used to compare the relative acidity (and alkalinity) of solutions. The value is related to the concentration of H+ions, in solution. “The greater the concentration of hydrogen ions in solution, the lower the p. H” <— 0 1 2 3 4 5 6 strongly acidic weakly acidic 7 neutral 8 9 10 11 12 13 14 —> weakly alkaline strongly alkaline

p. H SCALE Used to compare the relative acidity (and alkalinity) of solutions. The value is related to the concentration of H+ions, in solution. “The greater the concentration of hydrogen ions in solution, the lower the p. H” <— 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 —> strongly acidic weakly acidic neutral weakly alkaline strongly alkaline HCl CH 3 COOH H 2 O NH 3 Na. OH AMMONIA SODIUM HYDROXIDE HYDROCHLORIC ACID ETHANOIC ACID

p. H SCALE Used to compare the relative acidity (and alkalinity) of solutions. The value is related to the concentration of H+ions, in solution. “The greater the concentration of hydrogen ions in solution, the lower the p. H” <— 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 —> strongly acidic weakly acidic neutral weakly alkaline strongly alkaline HCl CH 3 COOH H 2 O NH 3 Na. OH AMMONIA SODIUM HYDROXIDE HYDROCHLORIC ACID ETHANOIC ACID Acidic solutions Alkaline solutions Neutral solutions p. H < 7 p. H > 7 p. H = 7

p. H SCALE Despite what some people think, the p. H scale is not restricted to between 0 and 14. It extends beyond those values, but is not as relevant; e. g. the p. H of the dilute HCl and H 2 SO 4 found on the laboratory shelves is -0. 3 and -0. 6 respectively.

p. H SCALE Despite what some people think, the p. H scale is not restricted to between 0 and 14. It extends beyond those values, but is not as relevant; e. g. the p. H of the dilute HCl and H 2 SO 4 found on the laboratory shelves is -0. 3 and -0. 6 respectively. Weak acids have higher p. H values than stronger ones of the same concentration. Ethanoic acid CH 3 COOH has a larger p. H than hydrochloric acid HCl of the same concentration.

MEASURING p. H Indicator Add a few drops of universal (full-range) indicator to the solution and compare the colour against a chart. The colour you “see” depends not only on how many drops of indicator you add but also on your perception of colour.

MEASURING p. H Indicator Add a few drops of universal (full-range) indicator to the solution and compare the colour against a chart. The colour you “see” depends not only on how many drops of indicator you add but also on your perception of colour. p. H meters Electrical/electronic devices which measure the conductivity of the solution. Having calibrated the apparatus against a solution of known p. H, you place the probe in any solution and read off the p. H value on a scale or digital read-out.

ACIDS

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can be arranged into groups according to how many H+ ions they produce from each molecule

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can be arranged into groups according to how many H+ ions they produce from each molecule ACID hydrochloric nitric ethanoic HCl HNO 3 CH 3 COOH SALTS ION EXAMPLE chloride nitrate ethanoate Cl¯ NO 3¯ CH 3 COO¯ Na. Cl Na. NO 3 CH 3 COONa THE ABOVE ARE MONOPROTIC (MONOBASIC) ACIDS

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can be arranged into groups according to how many H+ ions they produce from each molecule ACID hydrochloric nitric ethanoic HCl HNO 3 CH 3 COOH SALTS ION EXAMPLE chloride nitrate ethanoate Cl¯ NO 3¯ CH 3 COO¯ Na. Cl Na. NO 3 CH 3 COONa THE ABOVE ARE MONOPROTIC (MONOBASIC)ACIDS sulphuric H 2 SO 4 “carbonic” H 2 CO 3 hydrogensulphate hydrogencarbonate HSO 4¯ SO 42 HCO 3¯ CO 32 - THE ABOVE ARE DIPROTIC (DIBASIC)ACIDS Na. HSO 4 Na 2 SO 4 Na. HCO 3 Na 2 CO 3

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can also arranged into groups according to the extent they split into ions when put in water.

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can also arranged into groups according to the extent they split into ions when put in water. STRONG ACIDS COMPLETELY dissociate (split up) into ions e. g. sulphuric, hydrochloric, nitric

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can also arranged into groups according to the extent they split into ions when put in water. STRONG ACIDS COMPLETELY dissociate (split up) into ions e. g. sulphuric, hydrochloric, nitric WEAK ACIDS PARTIALLY dissociate (split up) into ions e. g. ethanoic, citric, carbonic

ACIDS Definition ACIDS ARE PROTON (H+ ion) DONORS Categories Acids can also arranged into groups according to the extent they split into ions when put in water. STRONG ACIDS COMPLETELY dissociate (split up) into ions e. g. sulphuric, hydrochloric, nitric WEAK ACIDS PARTIALLY dissociate (split up) into ions e. g. ethanoic, citric, carbonic This means that if you take similar concentrations of HCl and CH 3 COOH, the concentration of H+ ions in the solution of CH 3 COOH will be less as only a few of its molecules will have split up into ions. Its p. H will consequently be higher.

ACIDS Reactions The hydrogen ions produced by acids can be replaced by other positive ions (metal ions or ammonium ions) to produce salts. This occurs when dilute acids react with metals, oxides of metals, hydroxides of metals, carbonates, hydrogencarbonates and ammonia.
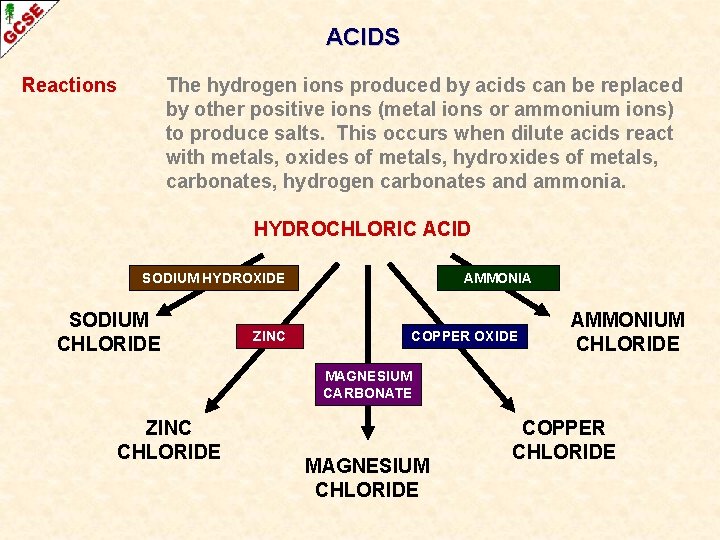
ACIDS Reactions The hydrogen ions produced by acids can be replaced by other positive ions (metal ions or ammonium ions) to produce salts. This occurs when dilute acids react with metals, oxides of metals, hydroxides of metals, carbonates, hydrogen carbonates and ammonia. HYDROCHLORIC ACID SODIUM HYDROXIDE SODIUM CHLORIDE ZINC AMMONIA COPPER OXIDE AMMONIUM CHLORIDE MAGNESIUM CARBONATE ZINC CHLORIDE MAGNESIUM CHLORIDE COPPER CHLORIDE
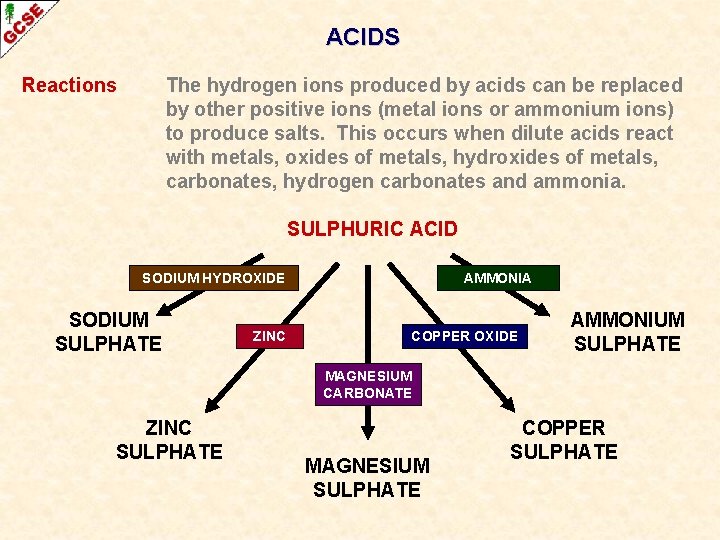
ACIDS Reactions The hydrogen ions produced by acids can be replaced by other positive ions (metal ions or ammonium ions) to produce salts. This occurs when dilute acids react with metals, oxides of metals, hydroxides of metals, carbonates, hydrogen carbonates and ammonia. SULPHURIC ACID SODIUM HYDROXIDE SODIUM SULPHATE ZINC AMMONIA COPPER OXIDE AMMONIUM SULPHATE MAGNESIUM CARBONATE ZINC SULPHATE MAGNESIUM SULPHATE COPPER SULPHATE
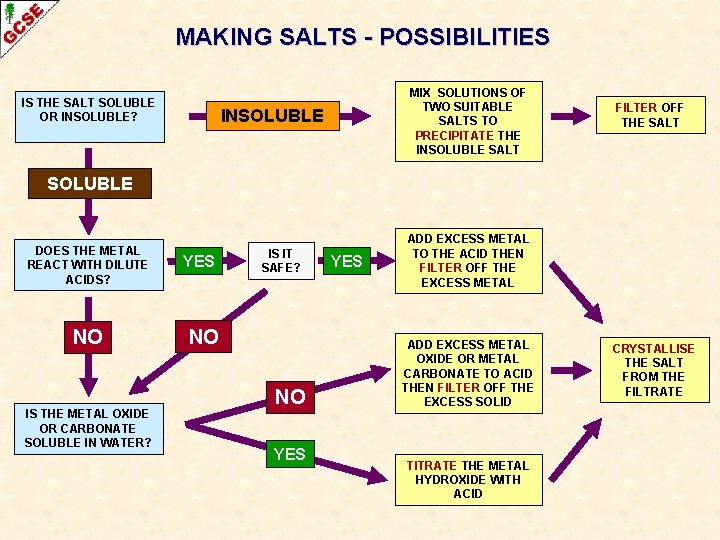
MAKING SALTS - POSSIBILITIES IS THE SALT SOLUBLE OR INSOLUBLE? MIX SOLUTIONS OF TWO SUITABLE SALTS TO PRECIPITATE THE INSOLUBLE SALT INSOLUBLE FILTER OFF THE SALT SOLUBLE DOES THE METAL REACT WITH DILUTE ACIDS? NO IS THE METAL OXIDE OR CARBONATE SOLUBLE IN WATER? YES IS IT SAFE? NO NO YES ADD EXCESS METAL TO THE ACID THEN FILTER OFF THE EXCESS METAL ADD EXCESS METAL OXIDE OR METAL CARBONATE TO ACID THEN FILTER OFF THE EXCESS SOLID TITRATE THE METAL HYDROXIDE WITH ACID CRYSTALLISE THE SALT FROM THE FILTRATE

PREPARATION OF SOLUBLE SALTS Salts can be made by NEUTRALISATION of acids; • • acidic solutions contain aqueous hydrogen ions H+(aq) sulphuric acid produces a sulphate hydrochloric acid produces a chloride nitric acid produces a nitrate

PREPARATION OF SOLUBLE SALTS Salts can be made by NEUTRALISATION of acids; • • acidic solutions contain aqueous hydrogen ions H+(aq) sulphuric acid produces a sulphate hydrochloric acid produces a chloride nitric acid produces a nitrate Other chemicals are formed, it depends on what is neutralising the acid. OXIDES HYDROXIDES CARBONATES METALS SALT + + WATER CARBON DIOXIDE + WATER HYDROGEN (not all metals are suitable; some are too reactive; others are not reactive enough)

PREPARATION OF SALTS BY NEUTRALISATION
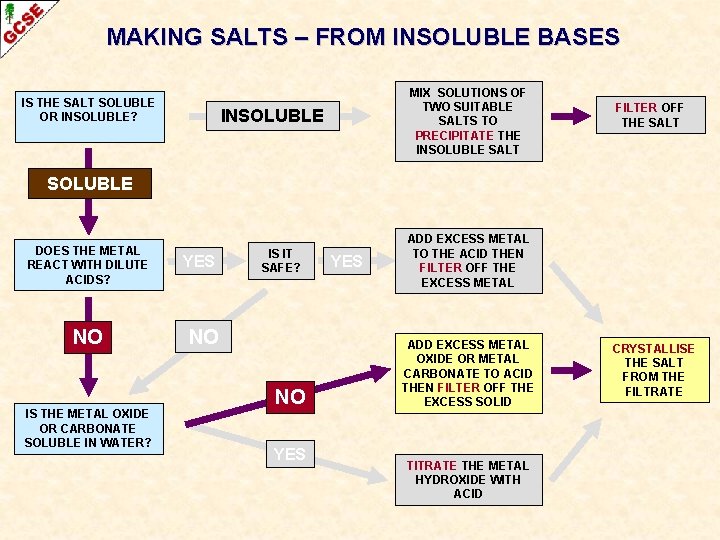
MAKING SALTS – FROM INSOLUBLE BASES IS THE SALT SOLUBLE OR INSOLUBLE? MIX SOLUTIONS OF TWO SUITABLE SALTS TO PRECIPITATE THE INSOLUBLE SALT INSOLUBLE FILTER OFF THE SALT SOLUBLE DOES THE METAL REACT WITH DILUTE ACIDS? NO IS THE METAL OXIDE OR CARBONATE SOLUBLE IN WATER? YES IS IT SAFE? NO NO YES ADD EXCESS METAL TO THE ACID THEN FILTER OFF THE EXCESS METAL ADD EXCESS METAL OXIDE OR METAL CARBONATE TO ACID THEN FILTER OFF THE EXCESS SOLID TITRATE THE METAL HYDROXIDE WITH ACID CRYSTALLISE THE SALT FROM THE FILTRATE

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 1 Place dilute acid in a beaker and warm it gently without letting it boil (reactions are faster at higher temperatures)

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 1 Place dilute acid in a beaker and warm it gently without letting it boil (reactions are faster at higher temperatures) 2. Remove the heat

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 1 Place dilute acid in a beaker and warm it gently without letting it boil (reactions are faster at higher temperatures) 2. Remove the heat 3. Carefully and slowly add small amounts of the solid while stirring (most reactions of this type are exothermic i. e. they give off energy - the energy released would make the solution boil over)
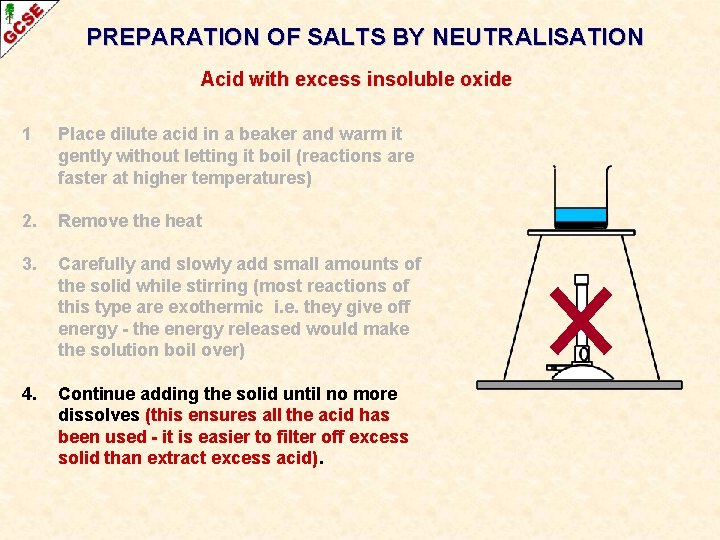
PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 1 Place dilute acid in a beaker and warm it gently without letting it boil (reactions are faster at higher temperatures) 2. Remove the heat 3. Carefully and slowly add small amounts of the solid while stirring (most reactions of this type are exothermic i. e. they give off energy - the energy released would make the solution boil over) 4. Continue adding the solid until no more dissolves (this ensures all the acid has been used - it is easier to filter off excess solid than extract excess acid).

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 5. Filter the solution into an evaporating dish to remove excess solid

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 5. Filter the solution into an evaporating dish to remove excess solid 6. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation.
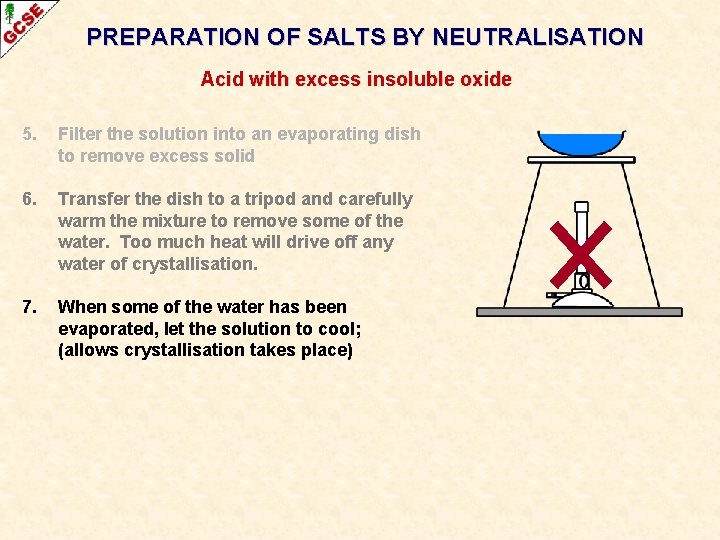
PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 5. Filter the solution into an evaporating dish to remove excess solid 6. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 7. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place)

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 5. Filter the solution into an evaporating dish to remove excess solid 6. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 7. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place) 8. When sufficient crystals have appeared, filter them off, wash with a little cold water and transfer them to a filter paper. Place another filter paper over the top and press gently to help remove some of the water.

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble oxide 5. Filter the solution into an evaporating dish to remove excess solid 6. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 7. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place) 8. When sufficient crystals have appeared, filter them off, wash with a little cold water and transfer them to a filter paper. Place another filter paper over the top and press gently to help remove some of the water. 9. Allow the crystals to dry naturally in air. (heat can cause the crystals to decompose and lose their water of crystallisation)

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate The method used is similar to that used with acids and metal oxides. The main differences is that it is not necessary to use heat and that carbon dioxide gas is given off so you have to make sure the solution doesn’t effervesce (fizz) too much.

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate The method used is similar to that used with acids and metal oxides. The main differences is that it is not necessary to use heat and that carbon dioxide gas is given off so you have to make sure the solution doesn’t effervesce (fizz) too much. 1 Place dilute acid in a beaker

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate The method used is similar to that used with acids and metal oxides. The main differences is that it is not necessary to use heat and that carbon dioxide gas is given off so you have to make sure the solution doesn’t effervesce (fizz) too much. 1 Place dilute acid in a beaker 2. Carefully and slowly add small amounts of the solid carbonate while stirring – make sure there is not too much effervescence.
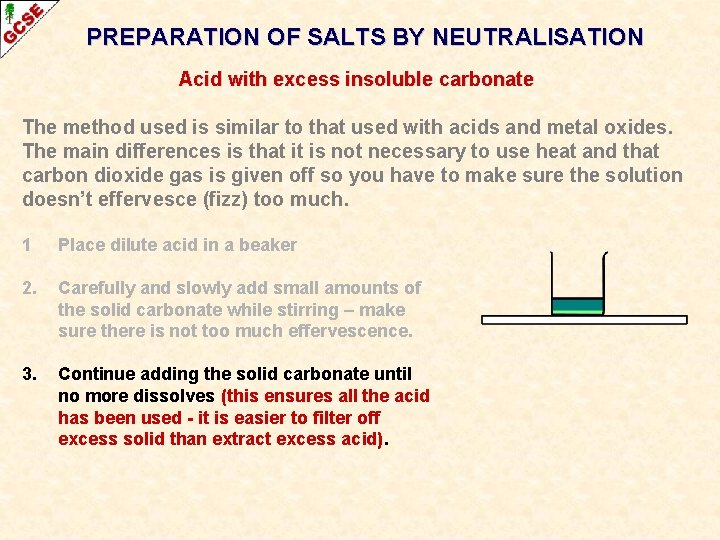
PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate The method used is similar to that used with acids and metal oxides. The main differences is that it is not necessary to use heat and that carbon dioxide gas is given off so you have to make sure the solution doesn’t effervesce (fizz) too much. 1 Place dilute acid in a beaker 2. Carefully and slowly add small amounts of the solid carbonate while stirring – make sure there is not too much effervescence. 3. Continue adding the solid carbonate until no more dissolves (this ensures all the acid has been used - it is easier to filter off excess solid than extract excess acid).

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate 4. Filter the solution into an evaporating dish to remove excess solid

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate 4. Filter the solution into an evaporating dish to remove excess solid 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation.
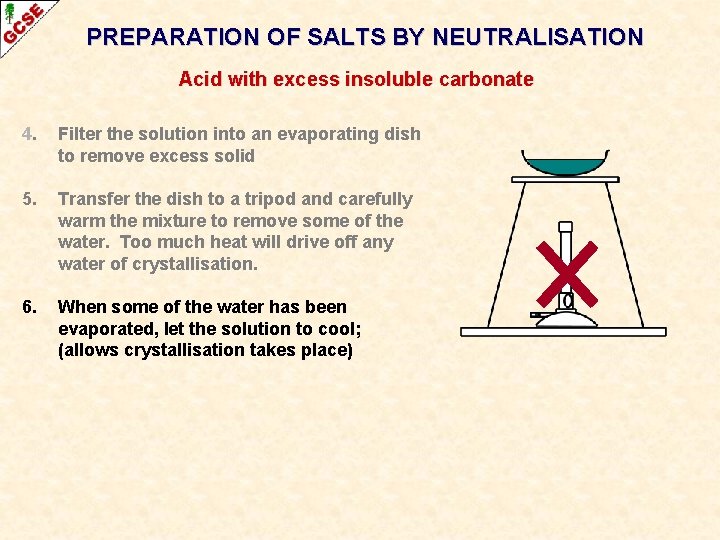
PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate 4. Filter the solution into an evaporating dish to remove excess solid 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 6. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place)

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate 4. Filter the solution into an evaporating dish to remove excess solid 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 6. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place) 7. When sufficient crystals have appeared, filter them off, wash with a little cold water and transfer them to a filter paper. Place another filter paper over the top and press gently to help remove some of the water.

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess insoluble carbonate 4. Filter the solution into an evaporating dish to remove excess solid 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 6. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place) 7. When sufficient crystals have appeared, filter them off, wash with a little cold water and transfer them to a filter paper. Place another filter paper over the top and press gently to help remove some of the water. 8. Allow the crystals to dry naturally in air. (heat can cause the crystals to decompose and lose their water of crystallisation)
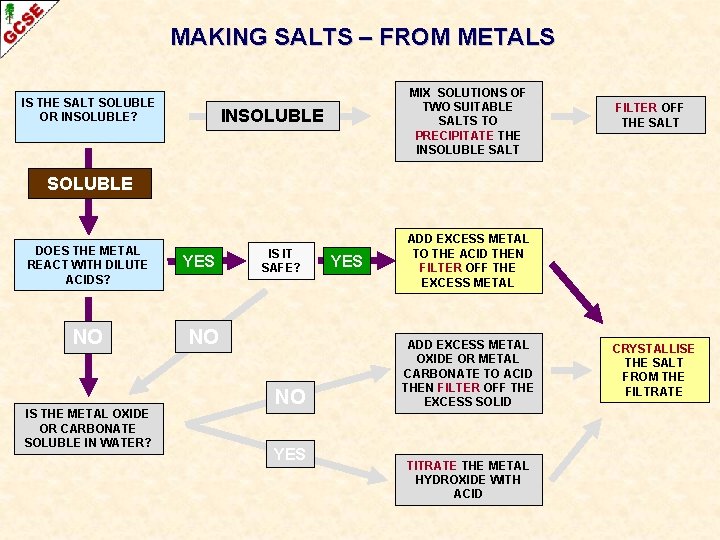
MAKING SALTS – FROM METALS IS THE SALT SOLUBLE OR INSOLUBLE? MIX SOLUTIONS OF TWO SUITABLE SALTS TO PRECIPITATE THE INSOLUBLE SALT INSOLUBLE FILTER OFF THE SALT SOLUBLE DOES THE METAL REACT WITH DILUTE ACIDS? NO IS THE METAL OXIDE OR CARBONATE SOLUBLE IN WATER? YES IS IT SAFE? NO NO YES ADD EXCESS METAL TO THE ACID THEN FILTER OFF THE EXCESS METAL ADD EXCESS METAL OXIDE OR METAL CARBONATE TO ACID THEN FILTER OFF THE EXCESS SOLID TITRATE THE METAL HYDROXIDE WITH ACID CRYSTALLISE THE SALT FROM THE FILTRATE

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal The method used is similar to that used with acids and metal oxides or metal carbonates. However, because hydrogen is produced during the experiment, Bunsen burners must not be used while the chemical reaction is taking place. Not all metals are suitable: some are too reactive some don’t react K, Na, Ca Cu

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal The method used is similar to that used with acids and metal oxides or metal carbonates. However, because hydrogen is produced during the experiment, Bunsen burners must not be used while the chemical reaction is taking place. Not all metals are suitable: 1 Place dilute acid in a beaker some are too reactive some don’t react K, Na, Ca Cu

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal The method used is similar to that used with acids and metal oxides or metal carbonates. However, because hydrogen is produced during the experiment, Bunsen burners must not be used while the chemical reaction is taking place. Not all metals are suitable: some are too reactive some don’t react 1 Place dilute acid in a beaker 2. Carefully and slowly add small amounts of the metal while stirring – make sure there is not too much effervescence. K, Na, Ca Cu

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal The method used is similar to that used with acids and metal oxides or metal carbonates. However, because hydrogen is produced during the experiment, Bunsen burners must not be used while the chemical reaction is taking place. Not all metals are suitable: some are too reactive some don’t react 1 Place dilute acid in a beaker 2. Carefully and slowly add small amounts of the metal while stirring – make sure there is not too much effervescence. 3. Continue adding the metal until no more dissolves (this ensures all the acid has been used - it is easier to filter off excess metal than extract excess acid). K, Na, Ca Cu

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal 4. Filter the solution into an evaporating dish to remove excess metal

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal 4. Filter the solution into an evaporating dish to remove excess metal 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation.

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal 4. Filter the solution into an evaporating dish to remove excess metal 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 6. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place)

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal 4. Filter the solution into an evaporating dish to remove excess metal 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 6. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place) 7. When sufficient crystals have appeared, filter them off, wash with a little cold water and transfer them to a filter paper. Place another filter paper over the top and press gently to help remove some of the water.

PREPARATION OF SALTS BY NEUTRALISATION Acid with excess metal 4. Filter the solution into an evaporating dish to remove excess metal 5. Transfer the dish to a tripod and carefully warm the mixture to remove some of the water. Too much heat will drive off any water of crystallisation. 6. When some of the water has been evaporated, let the solution to cool; (allows crystallisation takes place) 7. When sufficient crystals have appeared, filter them off, wash with a little cold water and transfer them to a filter paper. Place another filter paper over the top and press gently to help remove some of the water. 8. Allow the crystals to dry naturally in air. (heat can cause the crystals to decompose and lose their water of crystallisation)
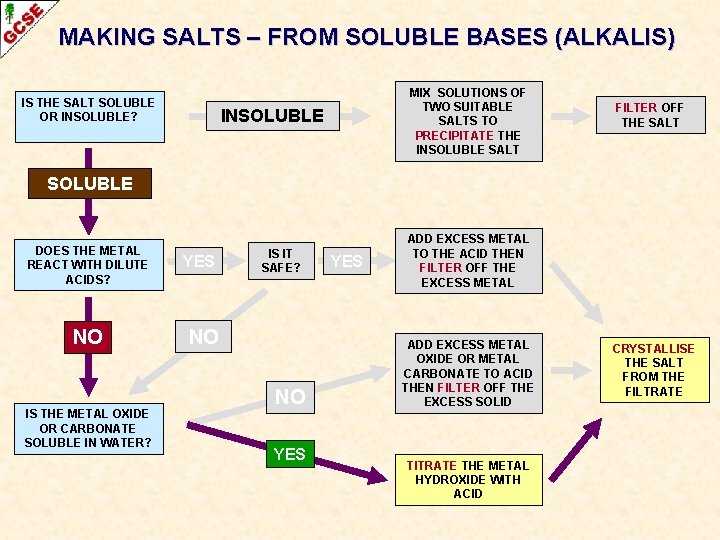
MAKING SALTS – FROM SOLUBLE BASES (ALKALIS) IS THE SALT SOLUBLE OR INSOLUBLE? MIX SOLUTIONS OF TWO SUITABLE SALTS TO PRECIPITATE THE INSOLUBLE SALT INSOLUBLE FILTER OFF THE SALT SOLUBLE DOES THE METAL REACT WITH DILUTE ACIDS? NO IS THE METAL OXIDE OR CARBONATE SOLUBLE IN WATER? YES IS IT SAFE? NO NO YES ADD EXCESS METAL TO THE ACID THEN FILTER OFF THE EXCESS METAL ADD EXCESS METAL OXIDE OR METAL CARBONATE TO ACID THEN FILTER OFF THE EXCESS SOLID TITRATE THE METAL HYDROXIDE WITH ACID CRYSTALLISE THE SALT FROM THE FILTRATE

ALKALIS

ALKALIS Definition ALKALIS ARE SOLUBLE BASES Their solutions contain hydroxide ions, OH¯(aq)

ALKALIS Definition ALKALIS ARE SOLUBLE BASES Their solutions contain hydroxide ions, OH¯(aq) STRONG ALKALIS sodium hydroxide (Na. OH) potassium hydroxide (KOH) WEAK ALKALIS ammonia solution (ammonium hydroxide) (NH 4 OH)

ALKALIS Definition ALKALIS ARE SOLUBLE BASES Their solutions contain hydroxide ions, OH¯(aq) STRONG ALKALIS sodium hydroxide (Na. OH) potassium hydroxide (KOH) WEAK ALKALIS ammonia solution (ammonium hydroxide) (NH 4 OH) Making salts using alkalis cannot be done by the methods used for insoluble bases because you cannot tell when all the acid has been neutralised – the excess alkali is soluble.

ALKALIS Definition ALKALIS ARE SOLUBLE BASES Their solutions contain hydroxide ions, OH¯(aq) STRONG ALKALIS sodium hydroxide (Na. OH) potassium hydroxide (KOH) WEAK ALKALIS ammonia solution (ammonium hydroxide) (NH 4 OH) Making salts using alkalis cannot be done by the methods used for insoluble bases because you cannot tell when all the acid has been neutralised – the excess alkali is soluble. The salts are made by titration using an indicator ACID + ALKALINE HYDROXIDE ——> NEUTRAL SALT + WATER SOLUTION

ALKALIS Definition ALKALIS ARE SOLUBLE BASES Their solutions contain hydroxide ions, OH¯(aq) STRONG ALKALIS sodium hydroxide (Na. OH) potassium hydroxide (KOH) WEAK ALKALIS ammonia solution (ammonium hydroxide) (NH 4 OH) Making salts using alkalis cannot be done by the methods used for insoluble bases because you cannot tell when all the acid has been neutralised – the excess alkali is soluble. The salts are made by titration using an indicator ACID + ALKALINE HYDROXIDE ——> NEUTRAL SALT + WATER SOLUTION The indicator tells you when the acid has been neutralised

AMMONIUM SALTS Ammonia is very soluble in water giving an alkaline solution which can be neutralised by acids to produce AMMONIUM SALTS e. g. AMMONIA + HYDROCHLORIC —> AMMONIUM SOLUTION ACID CHLORIDE

AMMONIUM SALTS Ammonia is very soluble in water giving an alkaline solution which can be neutralised by acids to produce AMMONIUM SALTS e. g. AMMONIA + HYDROCHLORIC —> AMMONIUM SOLUTION ACID CHLORIDE If ammonium hydroxide is specified you can write the equation AMMONIUM + HYDROCHLORIC —> AMMONIUM + WATER HYDROXIDE ACID CHLORIDE Ammonium salts make very important fertilisers

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION This method is limited to the soluble salts of ammonia or alkali metals e. g. ammonium chloride NH 4 Cl sodium chloride Na. Cl potassium sulphate K 2 SO 4 sodium nitrate Na. NO 3 Method 1 2 3 4 5 6 React an acid with an alkali (hydroxides of Group I metals and ammonia). The method involves TITRATION. Place a measured quantity of acid in a conical flask. Add three or four drops of a suitable indicator. Place the alkali in a burette. Note the level of the liquid. Add the alkali to the acid until the indicator just changes colour Note the new level of liquid. Repeat experiment with exactly the same volumes but no indicator. Evaporate a small amount of the water in an evaporating basin and let the solution cool to allow crystals to form.

MAKING SALTS BY TITRATION Example word equations: sodium hydroxide + hydrochloric acid —> sodium chloride + water sodium hydroxide + sulphuric acid —> sodium sulphate potassium hydroxide + nitric acid —> potassium nitrate + water ammonium hydroxide + nitric acid —> ammonium nitrate + water

MAKING SALTS BY TITRATION Example word equations: sodium hydroxide + hydrochloric acid —> sodium chloride + water sodium hydroxide + sulphuric acid —> sodium sulphate potassium hydroxide + nitric acid —> potassium nitrate + water ammonium hydroxide + nitric acid —> ammonium nitrate + water Now complete these: - + water

MAKING SALTS BY TITRATION Example word equations: sodium hydroxide + hydrochloric acid —> sodium chloride + water sodium hydroxide + sulphuric acid —> sodium sulphate potassium hydroxide + nitric acid —> potassium nitrate + water ammonium hydroxide + nitric acid —> ammonium nitrate + water Now complete these: potassium hydroxide + hydrochloric acid —> potassium chloride + water

MAKING SALTS BY TITRATION Example word equations: sodium hydroxide + hydrochloric acid —> sodium chloride + water sodium hydroxide + sulphuric acid —> sodium sulphate potassium hydroxide + nitric acid —> potassium nitrate + water ammonium hydroxide + nitric acid —> ammonium nitrate + water Now complete these: potassium hydroxide + hydrochloric acid —> potassium chloride + water sodium hydroxide + nitric acid —> sodium nitrate + water

MAKING SALTS BY TITRATION Example word equations: sodium hydroxide + hydrochloric acid —> sodium chloride + water sodium hydroxide + sulphuric acid —> sodium sulphate potassium hydroxide + nitric acid —> potassium nitrate + water ammonium hydroxide + nitric acid —> ammonium nitrate + water Now complete these: potassium hydroxide + hydrochloric acid —> potassium chloride + water sodium hydroxide + nitric acid —> sodium nitrate + water ammonium hydroxide + hydrochloric acid —> ammonium chloride + water

MAKING SALTS BY TITRATION Example word equations: sodium hydroxide + hydrochloric acid —> sodium chloride + water sodium hydroxide + sulphuric acid —> sodium sulphate potassium hydroxide + nitric acid —> potassium nitrate + water ammonium hydroxide + nitric acid —> ammonium nitrate + water Now complete these: potassium hydroxide + hydrochloric acid —> potassium chloride + water sodium hydroxide + nitric acid —> sodium nitrate + water ammonium hydroxide + hydrochloric acid —> ammonium chloride + water

NEUTRALISATION - IONIC EQUATION In all alkali – acid reactions, the same ionic reaction takes place…

NEUTRALISATION - IONIC EQUATION In all alkali – acid reactions, the same ionic reaction takes place… sodium hydroxide + dil. nitric acid Na. OH(aq) + HNO 3 (aq) —> —> sodium nitrate + water Na. NO 3 (aq) + H 2 O (l)

NEUTRALISATION - IONIC EQUATION In all alkali – acid reactions, the same ionic reaction takes place… sodium hydroxide + dil. nitric acid Na. OH(aq) + HNO 3 (aq) —> —> sodium nitrate + water Na. NO 3 (aq) + H 2 O (l) Na+(aq) + OH¯(aq) + H+(aq) + NO 3¯(aq) —> Na+(aq) + NO 3¯(aq) + H 2 O(l)

NEUTRALISATION - IONIC EQUATION In all alkali – acid reactions, the same ionic reaction takes place… sodium hydroxide + dil. nitric acid Na. OH(aq) + HNO 3 (aq) —> —> sodium nitrate + water Na. NO 3 (aq) + H 2 O (l) Na+(aq) + OH¯(aq) + H+(aq) + NO 3¯(aq) —> Na+(aq) + NO 3¯(aq) + H 2 O(l) cancel ions H+(aq) + from acid OH¯(aq) —> from alkali H 2 O(l)
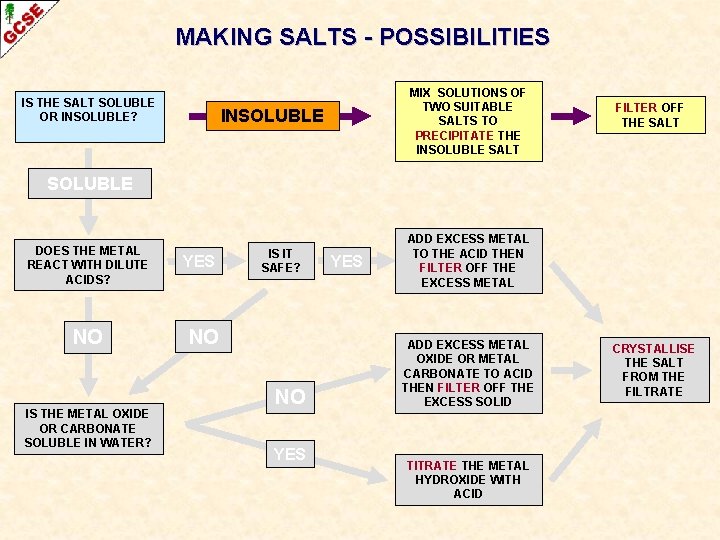
MAKING SALTS - POSSIBILITIES IS THE SALT SOLUBLE OR INSOLUBLE? MIX SOLUTIONS OF TWO SUITABLE SALTS TO PRECIPITATE THE INSOLUBLE SALT INSOLUBLE FILTER OFF THE SALT SOLUBLE DOES THE METAL REACT WITH DILUTE ACIDS? NO IS THE METAL OXIDE OR CARBONATE SOLUBLE IN WATER? YES IS IT SAFE? NO NO YES ADD EXCESS METAL TO THE ACID THEN FILTER OFF THE EXCESS METAL ADD EXCESS METAL OXIDE OR METAL CARBONATE TO ACID THEN FILTER OFF THE EXCESS SOLID TITRATE THE METAL HYDROXIDE WITH ACID CRYSTALLISE THE SALT FROM THE FILTRATE

PREPARATION OF INSOLUBLE SALTS

PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed.

PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. EXAMPLES silver chloride barium sulphate lead iodide silver nitrate and sodium chloride barium chloride and sodium sulphate lead nitrate and potassium iodide

PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride silver nitrate sodium chloride. plus

PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 1 silver nitrate sodium chloride. Prepare a solution of sodium chloride. plus

PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 2 silver nitrate sodium chloride. plus The sodium chloride lattice breaks up and dissolves in the water.
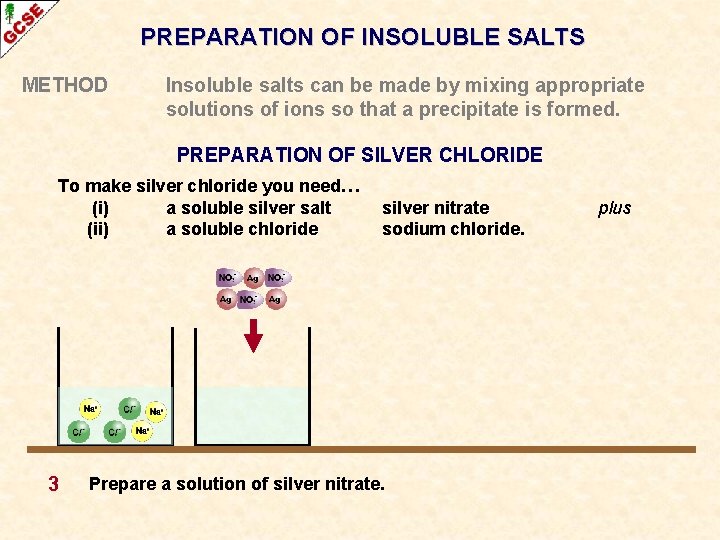
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 3 silver nitrate sodium chloride. Prepare a solution of silver nitrate. plus
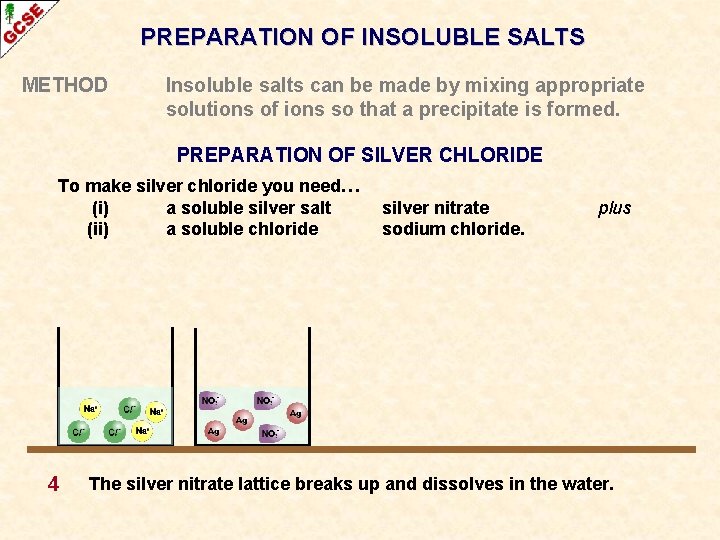
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 4 silver nitrate sodium chloride. plus The silver nitrate lattice breaks up and dissolves in the water.
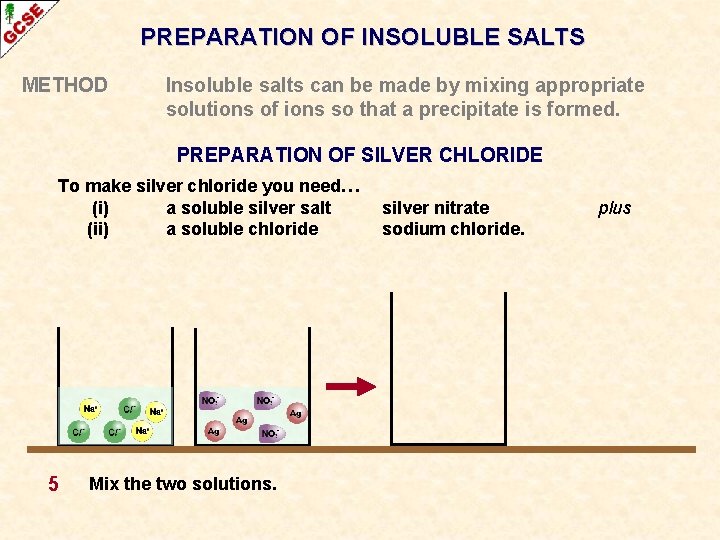
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 5 Mix the two solutions. silver nitrate sodium chloride. plus
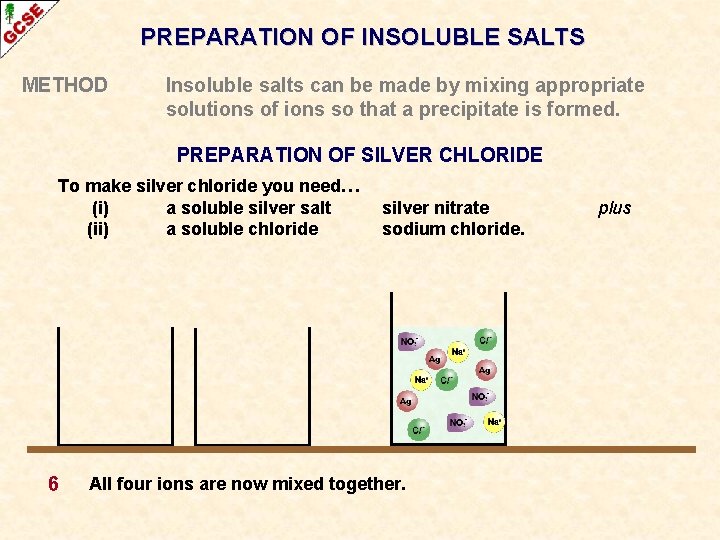
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 6 silver nitrate sodium chloride. All four ions are now mixed together. plus
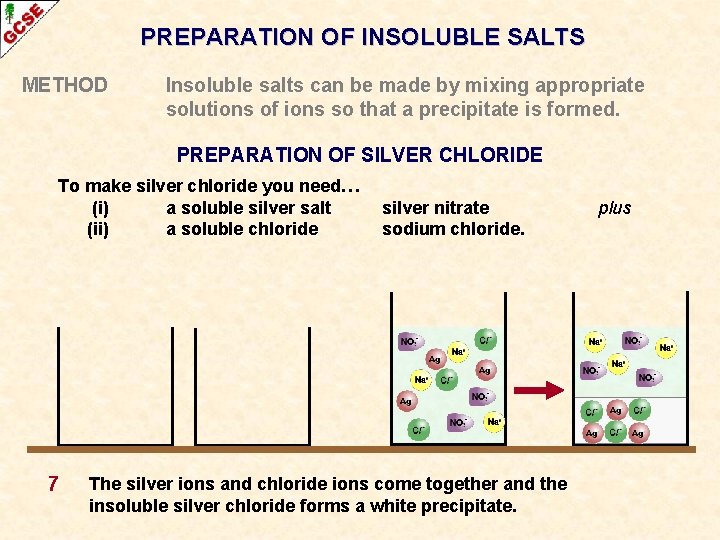
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 7 silver nitrate sodium chloride. The silver ions and chloride ions come together and the insoluble silver chloride forms a white precipitate. plus
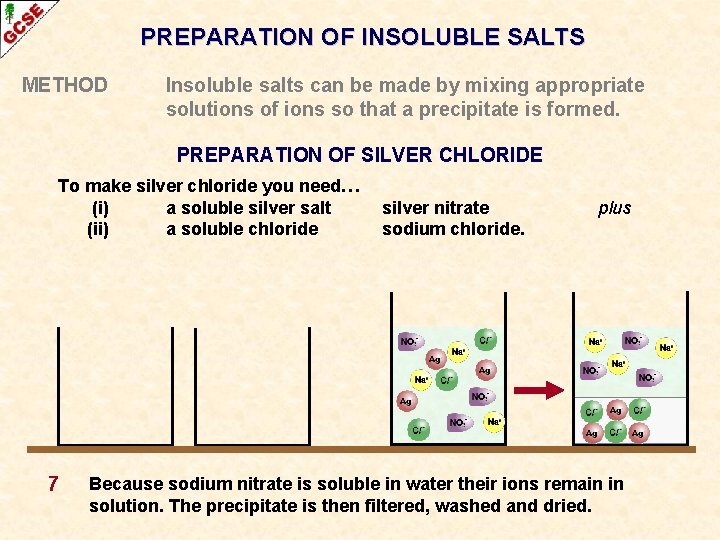
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride 7 silver nitrate sodium chloride. plus Because sodium nitrate is soluble in water their ions remain in solution. The precipitate is then filtered, washed and dried.
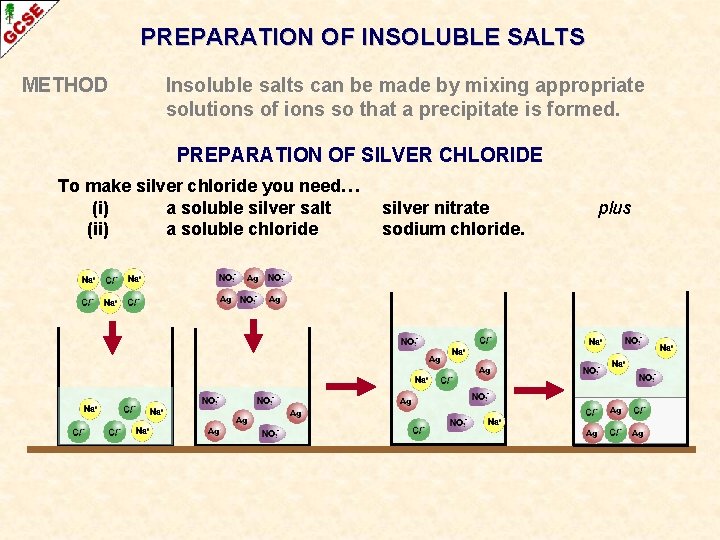
PREPARATION OF INSOLUBLE SALTS METHOD Insoluble salts can be made by mixing appropriate solutions of ions so that a precipitate is formed. PREPARATION OF SILVER CHLORIDE To make silver chloride you need… (i) a soluble silver salt (ii) a soluble chloride silver nitrate sodium chloride. plus

SALTS - QUESTIONS

SALTS - QUESTIONS 1. Work out what you get if the following chemicals are mixed. a) b) c) d) e) f) g) zinc and sulphuric acid copper carbonate and nitric acid sodium hydroxide and hydrochloric acid lead oxide and nitric acid magnesium and hydrochloric acid ammonium hydroxide and sulphuric acid magnesium carbonate and sulphuric acid

SALTS - QUESTIONS 1. Work out what you get if the following chemicals are mixed. a) b) c) d) e) f) g) zinc and sulphuric acid copper carbonate and nitric acid sodium hydroxide and hydrochloric acid lead oxide and nitric acid magnesium and hydrochloric acid ammonium hydroxide and sulphuric acid magnesium carbonate and sulphuric acid zinc sulphate + hydrogen copper nitrate + carbon dioxide + water sodium chloride + water lead nitrate + water magnesium chloride + hydrogen ammonium sulphate + water magnesium + carbon + water sulphate dioxide

SALTS - QUESTIONS 1. Work out what you get if the following chemicals are mixed. a) b) c) d) e) f) g) zinc and sulphuric acid copper carbonate and nitric acid sodium hydroxide and hydrochloric acid lead oxide and nitric acid magnesium and hydrochloric acid ammonium hydroxide and sulphuric acid magnesium carbonate and sulphuric acid zinc sulphate + hydrogen copper nitrate + carbon dioxide + water sodium chloride + water lead nitrate + water magnesium chloride + hydrogen ammonium sulphate + water magnesium + carbon + water sulphate dioxide 2. Answer the following. . . Which ion is found in (i) acidic solutions; H+(aq) (ii) alkaline solutions? What is formed when you mix these two ions together? What name do we give to this type of reaction? When making salts from metals, oxides and carbonates, how do you know when all the acid is used up? When making salts from soluble Group I hydroxides, what do you use to check when all the acid is used up?

SALTS - QUESTIONS 1. Work out what you get if the following chemicals are mixed. a) b) c) d) e) f) g) zinc and sulphuric acid copper carbonate and nitric acid sodium hydroxide and hydrochloric acid lead oxide and nitric acid magnesium and hydrochloric acid ammonium hydroxide and sulphuric acid magnesium carbonate and sulphuric acid zinc sulphate + hydrogen copper nitrate + carbon dioxide + water sodium chloride + water lead nitrate + water magnesium chloride + hydrogen ammonium sulphate + water magnesium + carbon + water sulphate dioxide 2. Answer the following. . . Which ion is found in (i) acidic solutions; H+(aq) (ii) alkaline solutions? OH¯(aq) What is formed when you mix these two ions together? WATER What name do we give to this type of reaction? NEUTRALISATION When making salts from metals, oxides and carbonates, how do you know when all the acid is used up? WHEN THE SOLID NO LONGER DISSOLVES When making salts from soluble Group I hydroxides, what do you use to check when all the acid is used up? AN INDICATOR

ACIDS, BASES & SALTS THE END © JONATHAN HOPTON AND KNOCKHARDY PUBLISHING
- Slides: 116