Acids Bases Properties of Acids Taste SOUR ex

Acids & Bases

Properties of Acids Taste SOUR (ex: lemons, vinegar) Corrosive to Metals Produces H+ ions Which is the same as H 3 O+ (hydronium ion) Proton (H+) Donor Example: HCl + H 2 O Cl- + H 3 O+

Strong Acid vs Weak Acid Strong Acid – an acid that completely dissociates to produce H+ ions in solution (lot of H+) HCl, HNO 3 Weak Acid – an acid that dissociates to a slight extent in aqueous solution (not that many H+) HC 2 H 3 O 2 (vinegar), lemon juice
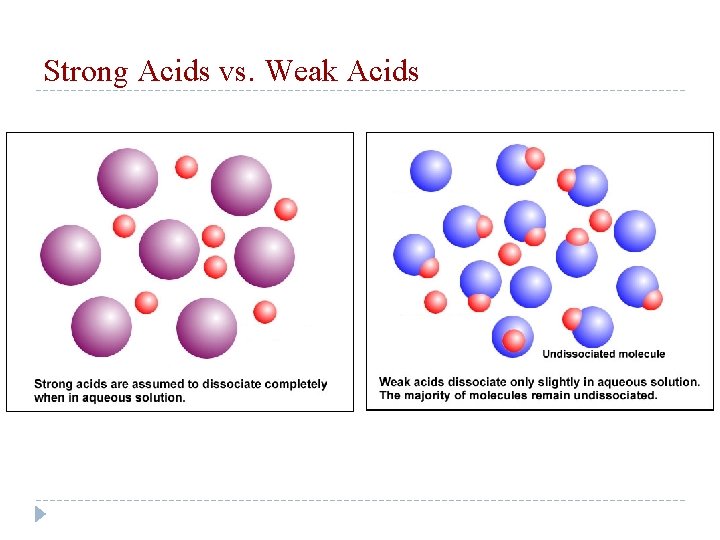
Strong Acids vs. Weak Acids

Indicators An indicator is a chemical that will change colors when placed in an acidic, basic or neutral environment Indicator Colors for Acids Litmus paper = red Phenolphthalein = clear Methyl orange = red

Properties of Bases Tastes Bitter (banana peel, dark chocolate, parsley) Feels slippery (soap) Produces OH- ions Proton (H+) Acceptor Example: NH 3 + H 2 O NH 4+ + OH -

Strong Bases v. Weak Bases Strong Base – a base that completely dissociates to produce many OH- ions in solution Na. OH, KOH Weak Base – a base that dissociates to a slight extent in aqueous solution (not that many OH-) NH 3 (ammonia) Strength & Weaknesses of A & B Video

Indicators Indicator Colors for Bases Litmus paper = blue Phenolphthalein = pink Methyl orange = yellow

Measuring the Amount of H+ and OH− Ions in a Solution p. H Scale- measures the _______ concentration of [H+] ions in a solution _____ p. OH Scale- measures the concentration of [ ____ OH− ] ions in a solution _____ With the p. H scale, we have another way to define acids and bases: Acids have a p. H _____7. 0 below Neutral Bases have a p. H _____7. 0 above = Neutral p. H ___7. 0 Acidic Basic

p. H of different household items Make predictions on whether a solution would be acidic, basic or neutral Go around the room, checking each cup with p. H paper (make sure used p. H paper goes in GARBAGE!) to save p. H paper, use HALF a side for each one. Approximate p. H colors: purple-ish red (p. H 2) red (p. H 4) brown (p. H 6) Stays the same (p. H 7) green (p. H 8) blue (p. H 10)

Examples of Common Acids citrus juices, ______, aspirin stomach acid, Pepsi, _____ battery acid, _______, ______ vinegar DNA

Examples of Common Bases Milk of magnesia, ______, ammonia drain cleaner, soap, blood, ______. antacid tablets, ______ baking soda
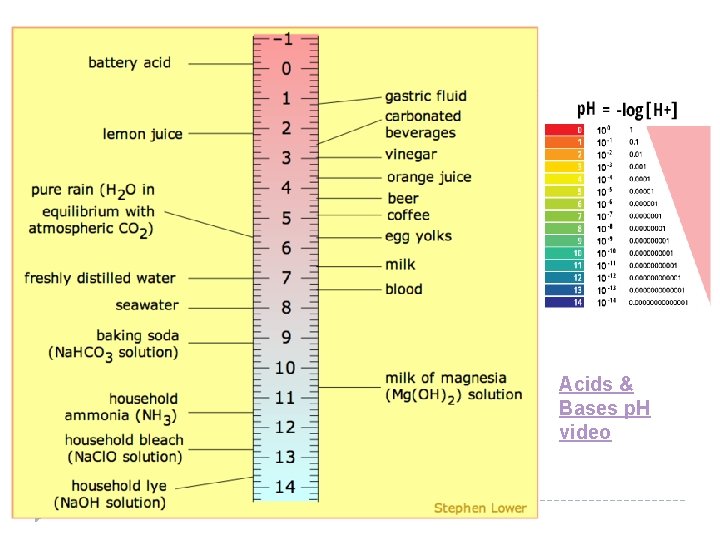
Acids & Bases p. H video

Exit Slip When using litmus paper, what color would it turn for an acid? If something has a p. H of 7, what is it considered?

Calculating p. H and p. OH
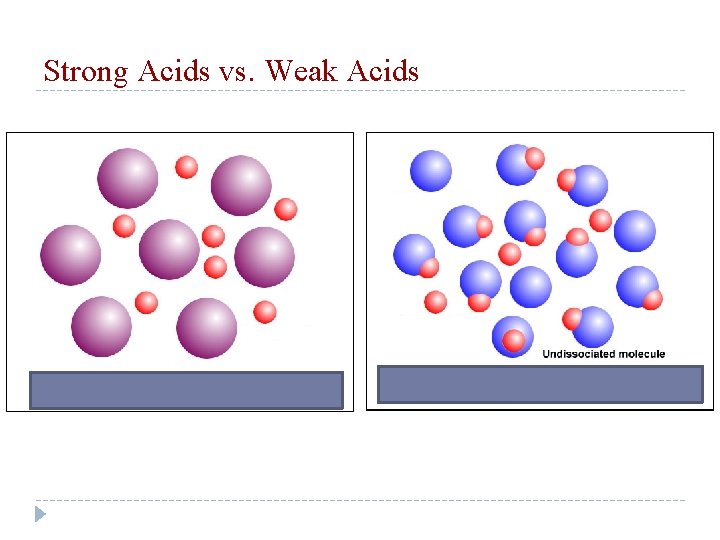
Strong Acids vs. Weak Acids

Strong Acids and Strong Bases Since they _______ disassociate… completely …so when HCl is placed in water… 100 molecules of HCl are dissolved in water, 100 H+ ions 100 Cl- ions _____and _____ions are produced. This comes into play for calculating the p. H for strong solutions!

Measuring the Amount of H+ and OH− Ions in a Solution p. H Scale- measures the _______ concentration of [H+] ions in a solution _____ p. OH Scale- measures the concentration of [ ____ OH− ] ions in a solution _____ Formulas p. H = − (log [H+]) [H+] = 10−p. H p. OH = −(log [OH−]) [OH−] = 10−p. OH p. H + p. OH = 14 With the p. H scale, we have another way to define acids and bases: below Acids have a p. H _____7. 0 above Bases have a p. H _____7. 0 = Neutral p. H ___7. 0
![[H+] = concentration of hydrogen ions Molarity ________ (M)! Before we try calculating p. [H+] = concentration of hydrogen ions Molarity ________ (M)! Before we try calculating p.](http://slidetodoc.com/presentation_image_h/024fb5626706e23a34d3b64a91c2ddf2/image-19.jpg)
[H+] = concentration of hydrogen ions Molarity ________ (M)! Before we try calculating p. H’s, watch video about background information on p. H

Example of Calculating p. H a) Calculate the p. H of a 0. 001 M HCl solution (so 0. 001 H+ ions) [H+] = 0. 001 M p. H = − (log 0. 001 M) p. H = 3 b) What is the p. OH of a solution with a p. H of 3? p. H + p. OH = 14 3+ p. OH = 14 p. OH = 11 c) What is the concentration of [OH−] ions in the solution? [OH−] = 10−p. OH [OH−] = 10− 11 M or 1 x 10− 11 M p. H = − (log [H+]) [H+] = 10−p. H p. OH = −(log [OH−]) [OH−] = 10−p. OH p. H + p. OH = 14

You Try: a) Calculate the p. OH of a Na. OH solution that has a p. H of 8. 50 p. H + p. OH = 14 8. 5 + p. OH = 14 p. OH = 5. 5 b) What is the [OH−] of this solution? [OH−] = 10−p. OH [OH−] = 10− 5. 5 M or 3. 16 x 10− 6 M c) What is the concentration of [H+] ions in the solution? [H+] = 10−p. H [H+] = 10− 8. 5 M or 3. 16 x 10− 9 M p. H = − (log [H+]) [H+] = 10−p. H p. OH = −(log [OH−]) [OH−] = 10−p. OH p. H + p. OH = 14

Exit Slip Which disassociates to more H+ ions: a strong acid or a weak acid? Calculate the p. H of. 005 M HCl solution Calculate the p. OH of a solution with a p. H of 6. p. H = − (log [H+]) [H+] = 10−p. H p. OH = −(log [OH−]) [OH−] = 10−p. OH p. H + p. OH = 14

Acid & Bases Intro to Calculations Answers 1) concentration 2) Concentration of H+ (or Hydronium) Ions 3) Concentration of H+ (or Hydronium) ions 1. a. –log(. 005) 2. 3 b. –log(2) -. 301 2. a. 5, A b. 12, B c. 7, N d. 10, A 3. a. 10 -1. 04 0. 0912 b. 1013. 1 1. 26 x 10 -13 4. a. –log(. 4) 0. 398 b. –log(4) -0. 602 *Calculating out a negative p. H value does happen when concentrations are above 1. 0 M (so for a very concentrated acid)

Recognizing Acids & Bases Neutralization

Recognizing Acids & Bases hydrogen Acids start with the element: ________ Bases generally have the polyatomic ion: hydroxide _________ Another common base: NH 3 ammonia! Hydroxide: OHHydrogen: H+
![Conjugate Acid-Base Pairs Acid substance formed when a _______ base gains a [H+] Conjugate Conjugate Acid-Base Pairs Acid substance formed when a _______ base gains a [H+] Conjugate](http://slidetodoc.com/presentation_image_h/024fb5626706e23a34d3b64a91c2ddf2/image-26.jpg)
Conjugate Acid-Base Pairs Acid substance formed when a _______ base gains a [H+] Conjugate ______ion. Base substance formed when an ______ acid loses a [H+] Conjugate ______ion. Practice Problems: Label the acid & base on the left side of the reaction and the conjugate acid & conjugate base on the right side. a) HCl + H 2 O H 3 O+ + Cl− acid base C. A. C. B. ______ b) NH 3 + H 2 O NH 4+ + OH− ______ base acid C. A. C. B.

Neutralization Reactions Strong acid solutions contain: H+ ions Strong base solutions contians: OH- ions Neutralization reaction: an acid-base reaction that has initial concentrations and volume of reactants equal. Equal H+ and OH- NEUTRAL solution (p. H = 7)
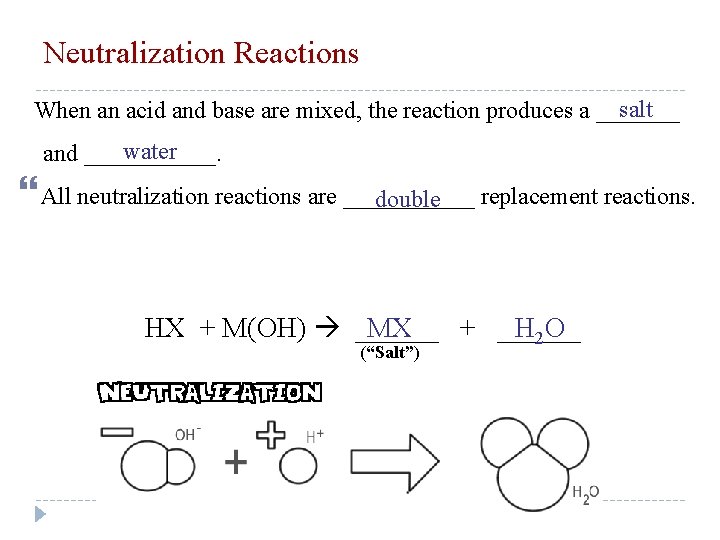
Neutralization Reactions salt When an acid and base are mixed, the reaction produces a _______ water and ______. All neutralization reactions are ______ replacement reactions. double HX + M(OH) ______ MX + ______ H 2 O (“Salt”)

Neutralization (or Acid-Base Reaction) Hydrochloric acid reacts with Sodium hydroxide… Na. Cl H 2 O (l) HCl (aq) + Na. OH (aq) _____ + _____ Salt Neutralization Video • an ionic compound produced from a neutralization reaction • composed of equal amount of cations & anions • thus neutral • can be aqueous or depending on solubility may be a precipitate

Alka-Seltzer Activity Each group needs: 1 small (150 or 250 m. L) beaker, 1 alka-seltzer tablet, 1 p. H paper 2) The Indicator is in the front table, add your 10 to 15 drops and then return it to the front! Vinegar will be at the lab stations. 3) When you add 1) 3 dropper fulls of vinegar!! *When finished, you may pick up a study guide for the quiz on Wednesday! (will have class time tomorrow to work on the study guide).
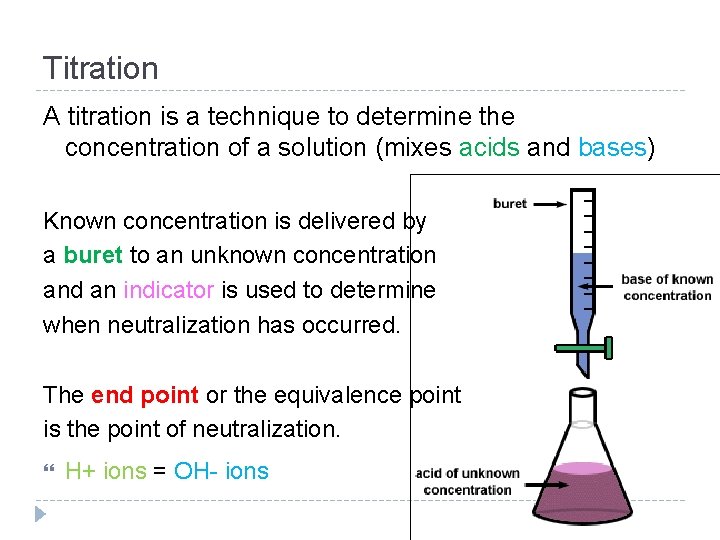
Titration A titration is a technique to determine the concentration of a solution (mixes acids and bases) Known concentration is delivered by a buret to an unknown concentration and an indicator is used to determine when neutralization has occurred. The end point or the equivalence point is the point of neutralization. H+ ions = OH- ions

Determining the Concentration of an Acid (or Base) by Titration Macid. Vacid = Mbase. Vbase A 25 m. L solution of HNO 3 is neutralized by 18 m. L of 1. 0 M Na. OH standard solution using phenolphthalein as an indicator. What is the concentration of the HNO 3 solution? ( Macid ) x ( 25 m. L ) = ( 1. 0 M ) x (18 m. L ) Macid = 0. 72 M

Determining the Concentration of an Acid (or Base) by Titration You Try How many m. L of 2. 0 M KOH will it take to neutralize 55 m. L of a 0. 76 M HCl standard solution? A 0. 05 L solution of HNO 3 is neutralized by 0. 025 L of 1. 5 M Na. OH standard solution using phenolphthalein as an indicator. What is the concentration of the HNO 3 solution?

Exit Slip How many m. L of 1. 5 M KOH will it take to neutralize 150 m. L of a 0. 75 M HCl standard solution? What is the p. H of a solution has a 1. 0 M concentration of H+ ions? If you could be a strong acid, weak acid, strong base, weak base, giraffe, kangaroo or the moon, which would you be and why?

Exit Slip Label the acid, base, conjugate acid and conjugate base HF + H 2 O H 3 O+ + F−

Answer Key Practice II 2. False – there is a small amount of H+, p. H measures concentration of H ions. If you have a high p. H there is a small, if you have a low p. H there are more H+ ions 4. A, N, A, B, A 5)100 m. L 6) 0. 14 M 7) 0. 125 M 8) p. H = 10. 37, p. OH = 3. 63, [OH-] = 10 -3. 63 0. 000234 M 9) 25 m. L 10) Water and a salt

* = indicates that it completely breaks apart into the ions (the arrows both ways, indicate that it does not) Semi-answer Key to Acids 4. Hydrofluoric acid, hydrobromic acid, hydroiodic acid, hydrosulfuric acid 1) Phosphoric acid 3) Carbonic acid 5) Nitrous acid H 2 SO 3 2) Phosphorous acid 4) Hydronitric acid 6) H 3 P 7) HNO 3 1) Bariumn Hydroxide 3) Iron (II) Hydroxide 4) Na. OH 5) Ca(OH)2 7) A, B, CA, CB 1) Calcium Hydroxide (B) 5) Sulfuric Acid (A) 8)

Acids & Bases 2

Strong Acids and Strong Bases Since they _______ disassociate… completely …so when HCl is placed in water… 100 molecules of HCl are dissolved in water, 100 H+ ions 100 Cl- ions _____and _____ions are produced. This comes into play for calculating the p. H for strong solutions!

Find the p. H of the following solution… A solution containing 1 mole of hydrochloric acid in 12 L of water p. H measures ________________. concentration of H+ ions M = mol/L M = 1 mol / 12 L M = _____ 0. 0833 M HCl _____ 0. 0833 M H+ p. H = -log( 0. 0833 )

Find the p. H of the following solution… A solution containing 5 moles of HCl in 10 L of water

Find the p. H of the following solution… A solution containing 20 grams of HCl in 10 L of water
Exit Slip Calculate the p. H of a solution containing 50 grams of HCl in 2. 5 L of water.

Measuring the Amount of H+ and OH− Ions in a Solution p. H Scale- measures the _______ concentration of [H+] ions in a solution _____ p. OH Scale- measures the concentration of [ ____ OH− ] ions in a solution _____ Formulas p. H = − (log [H+]) [H+] = 10−p. H p. OH = −(log [OH−]) [OH−] = 10−p. OH p. H + p. OH = 14 With the p. H scale, we have another way to define acids and bases: below Acids have a p. H _____7. 0 above Bases have a p. H _____7. 0 = Neutral p. H ___7. 0
- Slides: 44