ACIDS BASES Properties of Acids Taste sour Contain

ACIDS & BASES

Properties of Acids �Taste sour �Contain H+ ion �p. H less than 7 �React with bases to form a salt and water �React with some metals to produce hydrogen gas

Properties of Acids �Turn litmus paper red �Phenolphthalein is colorless in the presence of an acid �Bromothymol blue is yellow in the presence of an acid �Found in citrus fruits in the form of citric acid

Properties of Acids �Found in soured milk and in sore muscles in the form of lactic acid �Found in vitamin C in the form of ascorbic acid �Found in carbonated beverages in the form of carbonic acid…that’s also what you exhale

Properties of Bases �Taste bitter �Contain OH- ion �p. H greater than 7 �React with acids to form a salt and water �React with organic material

Properties of Bases �Feel slippery because they immediately begin to dissolve the outer layer of skin tissue �Turn litmus paper blue �Phenolphthalein is fuchsia in the presence of a base �Bromothymol blue is blue in the presence of a base

Properties of Bases �Found in drain cleaners usually in the form of sodium hydroxide �Found in ammonia-based cleaners like Windex �Lye (Na. OH) is used to make soaps

Classifying Acids and Bases �Svante Arrhenius—Swedish guy who put forth his definitions of acids and bases in 1884 at the age of 25 �Worked with our buddy van’t Hoff �Received 1903 Nobel Chemistry Prize for electrolytic dissociation discoveries

Arrhenius Acids �are substances that will dissociate in water to yield hydrogen ion (H+)

Arrhenius Bases �are substances that will dissociate in water to yield hydroxide ion (OH-)

Classifying Acids and Bases �Johannes Brønsted (Danish) and Thomas Lowry (English) came up with a new way to classify acids and bases and their conjugates (pairs that have features in common but are opposites) in the 1920’s �never received a Nobel for furthering these acid/base concepts, and Arrhenius never accepted them!

Brønsted-Lowry Acid �A reactant that donates a proton in a chemical reaction �The proton is actually the hydrogen ion…since a hydrogen atom has 1 proton and 1 electron and the ion with a 1+ charge indicates that it has lost an electron
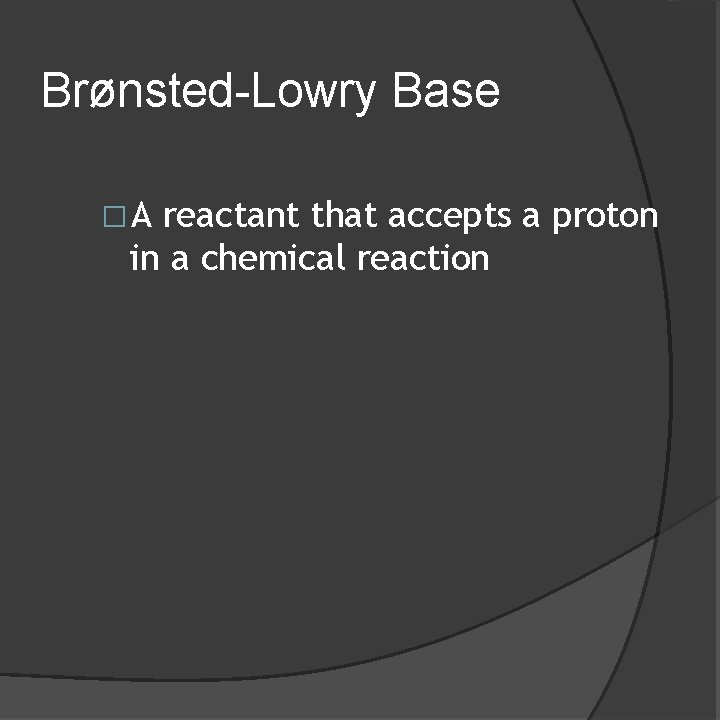
Brønsted-Lowry Base �A reactant that accepts a proton in a chemical reaction

Brønsted-Lowry Conjugate Acid �A product that �is formed when a base accepts a proton �in the reverse reaction, will donate a proton

Brønsted-Lowry Conjugate Base �A product that �is formed when an acid donates a proton (what’s left after the donation occurs) �in the reverse reaction, will accept a proton

Classifying Acids and Bases �Around the same time that Brønsted and Lowry were devising their acid/base scheme, our buddy Gilbert Lewis (yep, the same guy who did the dots) came up with yet another method of classifying them…it’s a broader method than Arrhenius, Brønsted, or Lowry ever postulated

Lewis Acid �A reactant that accepts an electron pair
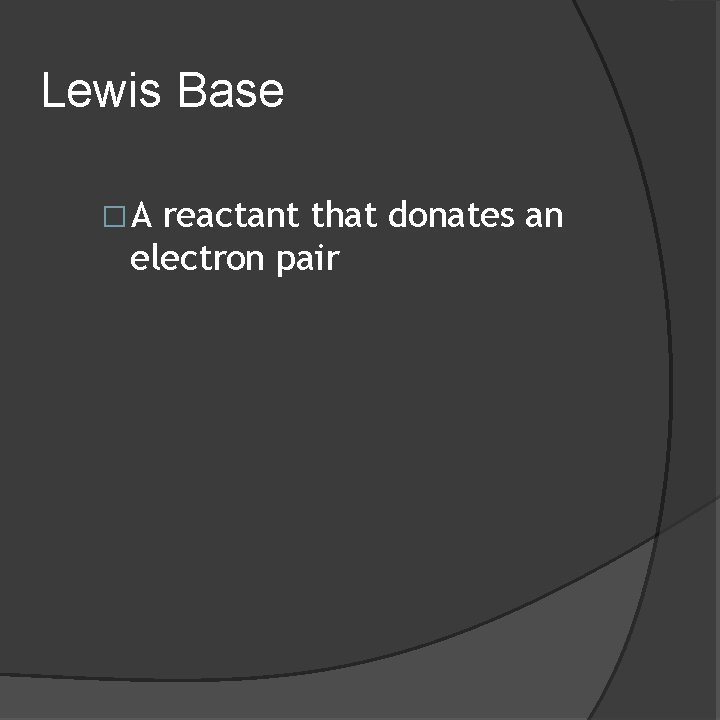
Lewis Base �A reactant that donates an electron pair
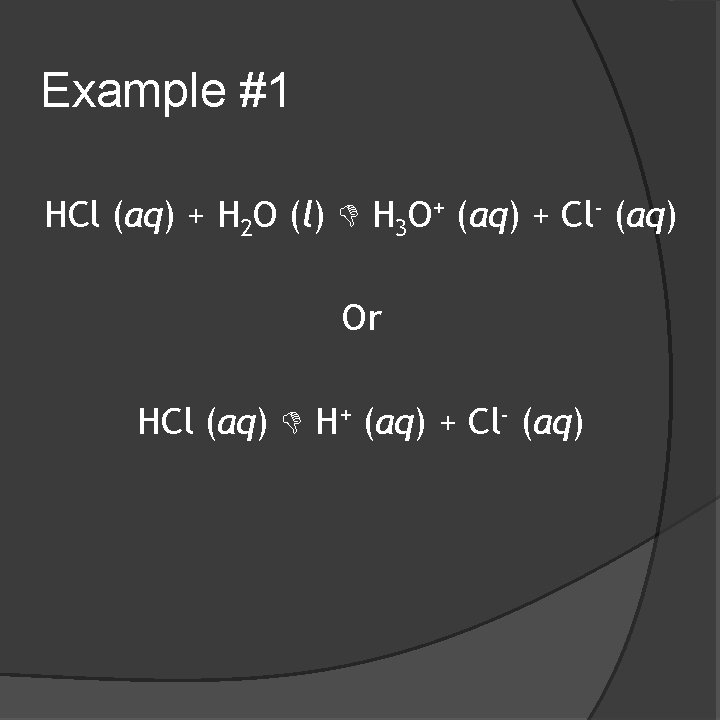
Example #1 HCl (aq) + H 2 O (l) H 3 O+ (aq) + Cl- (aq) Or HCl (aq) H+ (aq) + Cl- (aq)

Example #1 �HCl is an acid �It dissociates to yield H 3 O+ (hydronium ion), which is really water with an extra H+. (Arrhenius) �It donates a proton (H+) to water in the first reaction written. (Brønsted-Lowry)
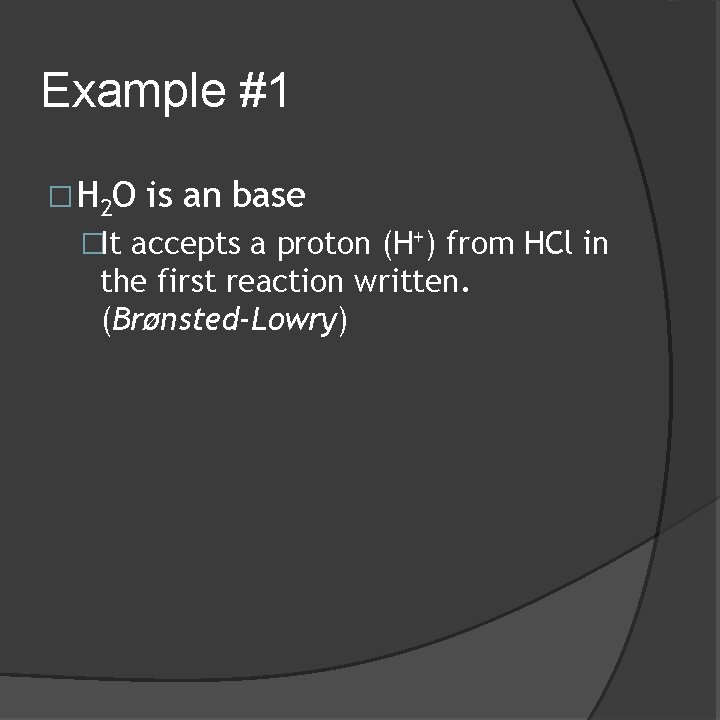
Example #1 � H 2 O is an base �It accepts a proton (H+) from HCl in the first reaction written. (Brønsted-Lowry)
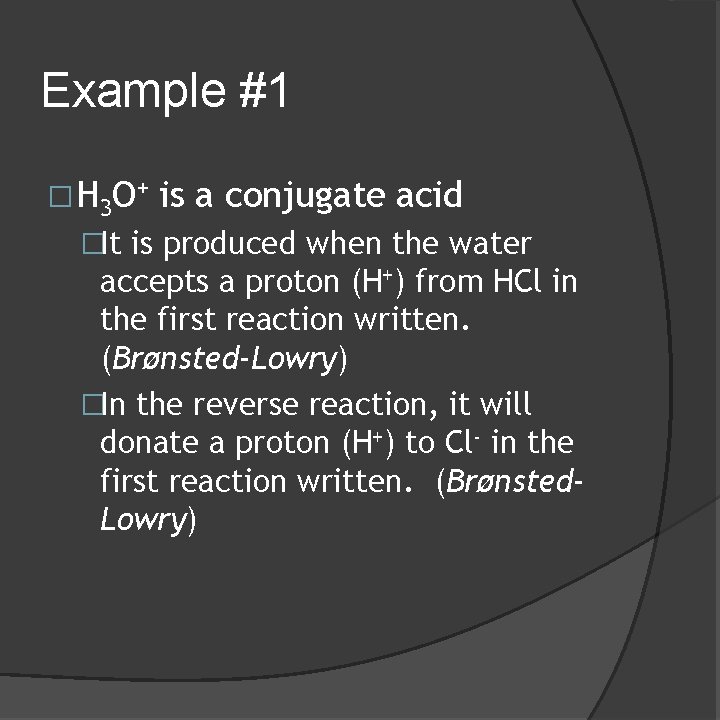
Example #1 � H 3 O + is a conjugate acid �It is produced when the water accepts a proton (H+) from HCl in the first reaction written. (Brønsted-Lowry) �In the reverse reaction, it will donate a proton (H+) to Cl- in the first reaction written. (Brønsted. Lowry)

Example #1 �Cl- is a conjugate base �It is produced when the HCl donates a proton (H+) to water in the first reaction written. (Brønsted-Lowry) �In the reverse reaction, it will accept a proton (H+) from H 3 O+ in the first reaction written. (Brønsted-Lowry)
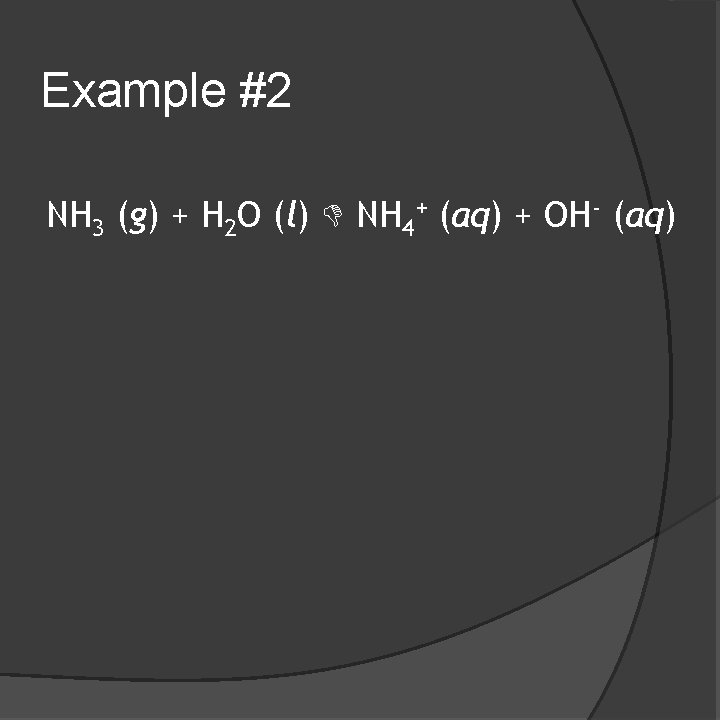
Example #2 NH 3 (g) + H 2 O (l) NH 4+ (aq) + OH- (aq)
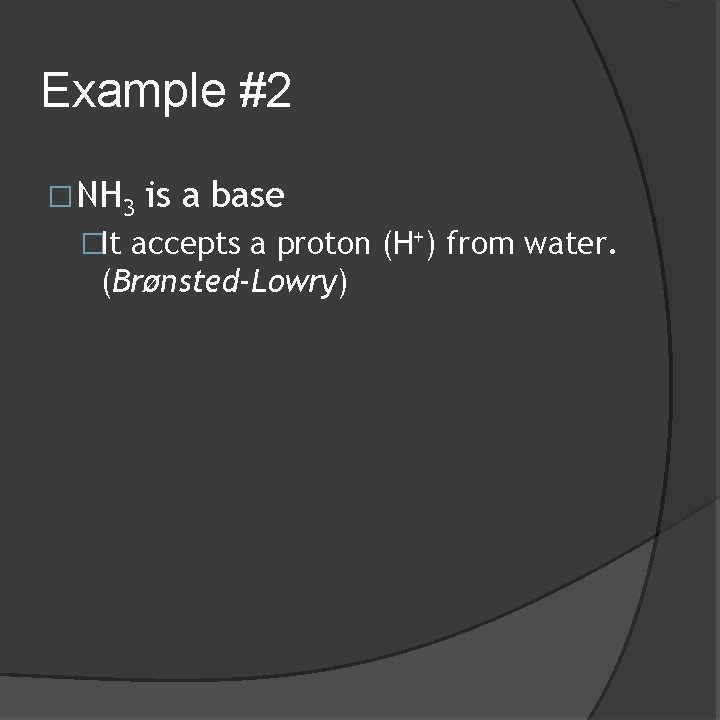
Example #2 �NH 3 is a base �It accepts a proton (H+) from water. (Brønsted-Lowry)

Example #2 � H 2 O is an acid �It donates a proton (H+) to ammonia. (Brønsted-Lowry)

Example #2 �NH 4+ is a conjugate acid �It is produced when the ammonia accepts a proton (H+) from water. (Brønsted-Lowry) �In the reverse reaction, it will donate a proton (H+) to OH-. (Brønsted-Lowry)

Example #2 �OH- is a conjugate base �It is produced when the water donates a proton (H+) to ammonia. (Brønsted-Lowry) �In the reverse reaction, it will accept a proton (H+) from NH 4+. (Brønsted-Lowry)

Water—our special friend �Did you notice that it behaved as a base in the first example and as an acid in the second example? n. A substance that can behave as either an acid or a base is called, amphoteric or amphiprotic.
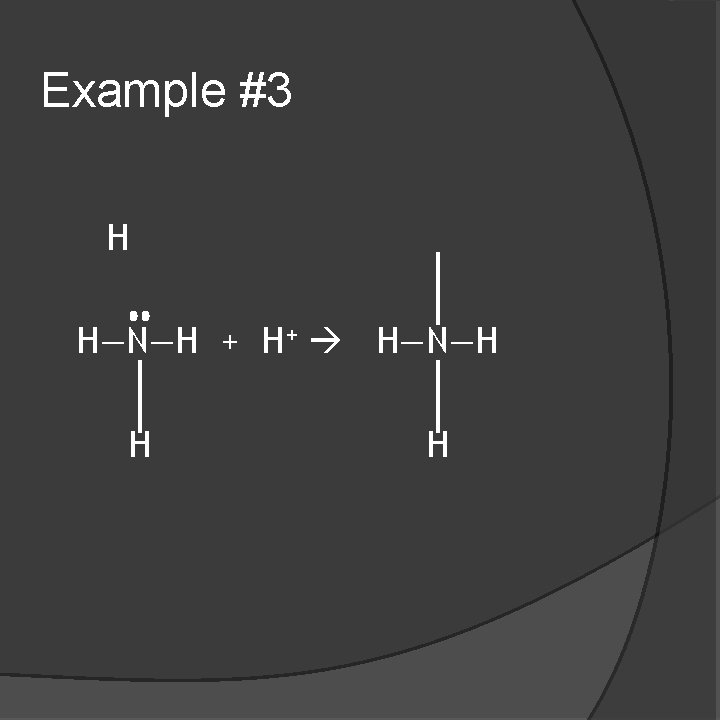
Example #3 H H—N—H + H+ H—N—H H H
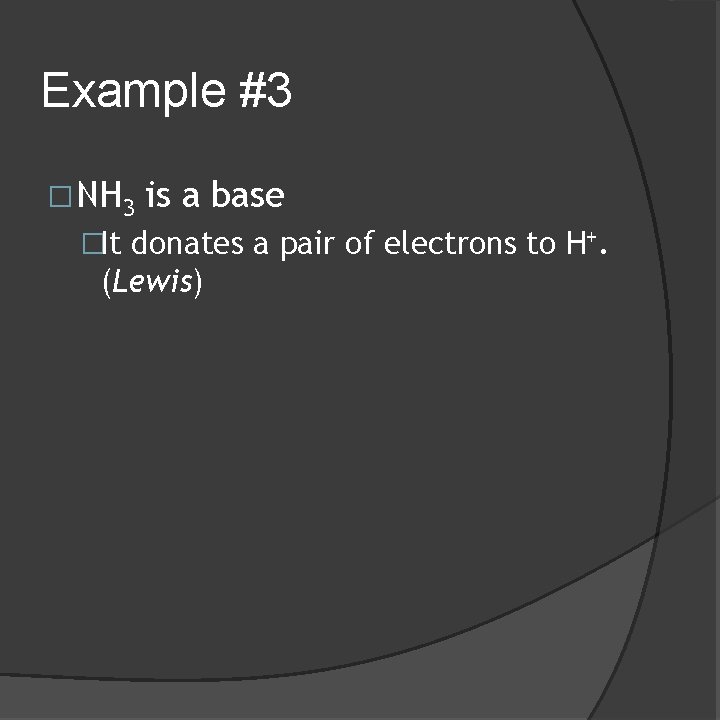
Example #3 �NH 3 is a base �It donates a pair of electrons to H+. (Lewis)

Example #3 � H+ is an acid �It accepts a pair of electrons from NH 3. (Lewis)

Autoionization of Water �Every acid and base will dissociate in water…even water (since it’s amphoteric)! 2 H 2 O (l) H 3 O+ (aq) + OH- (aq) or H 2 O (l) H+ (aq) + OH- (aq)

Autoionization of Water �Usually, the 2 nd reaction is the one we will use since H 3 O+ is just water with an extra H+. �Write the K expression for the 2 nd reaction, keeping in mind that we only include gaseous and aqueous phases.
![Autoionization of Water �K = [H+][OH-] �note that water is not included because it Autoionization of Water �K = [H+][OH-] �note that water is not included because it](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-35.jpg)
Autoionization of Water �K = [H+][OH-] �note that water is not included because it is a liquid n This expression is known as the Kw, or equilibrium constant for water, expression n K w = [H+][OH-]

Autoionization of Water �The value of Kw is 1 x 10 -14 M 2 at 25°C. �This is a small K value. n If the temperature changes, so does the value of Kw

Autoionization of Water �Make an equilibrium chart for the dissociation, or autoionization, of water.
![Autoionization of Water [H 2 O] Initial Change Eq. -- + [H ] [OH Autoionization of Water [H 2 O] Initial Change Eq. -- + [H ] [OH](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-38.jpg)
Autoionization of Water [H 2 O] Initial Change Eq. -- + [H ] [OH ] 0 0
![Autoionization of Water [H 2 O] + [H ] [OH ] Initial -- 0 Autoionization of Water [H 2 O] + [H ] [OH ] Initial -- 0](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-39.jpg)
Autoionization of Water [H 2 O] + [H ] [OH ] Initial -- 0 0 Change -- +x +x Eq.
![Autoionization of Water [H 2 O] + [H ] [OH ] Initial -- 0 Autoionization of Water [H 2 O] + [H ] [OH ] Initial -- 0](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-40.jpg)
Autoionization of Water [H 2 O] + [H ] [OH ] Initial -- 0 0 Change -- +x +x Eq. -- x x

Autoionization of Water �Determine the equlibrium concentrations of both the hydrogen ion and the hydroxide ion by plugging into the Kw expression. 1 x 10 -14 M 2 = [x][x]

Autoionization of Water 1 x 10 -14 M 2 = x 2 1 x 10 -7 M = x [H+] = 1 x 10 -7 M [OH-] = 1 x 10 -7 M

Autoionization of Water �Because the H+ and OHconcentrations are equal, the solution is neutral. �If [H+] > [OH-], then the solution is an acid. �If [H+] < [OH-], then the solution is a base.

Autoionization of Water �Since every acid or base dissociation we will entertain occurs in water, then the Kw expression is applicable to any of these dissociations.
![Autoionization of Water �Thus, if you know the [H+] concentration of a solution, you Autoionization of Water �Thus, if you know the [H+] concentration of a solution, you](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-45.jpg)
Autoionization of Water �Thus, if you know the [H+] concentration of a solution, you can determine the [OH-] concentration. �And, if you know the [OH-] concentration of a solution, you can determine the [H+] concentration.

p. H �Represents the “power of hydrogen” �Calculated by taking the opposite of the logarithm of the hydrogen ion concentration… p. H = -log [H+]
![p. H �Calculate the p. H of water at 25°C knowing that the [H+] p. H �Calculate the p. H of water at 25°C knowing that the [H+]](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-47.jpg)
p. H �Calculate the p. H of water at 25°C knowing that the [H+] is 1 x 10 -7 M. p. H = -log [1 x 10 -7] p. H = 7

p. H �If you already know the p. H of a solution, then you can find the [H+] using: [H+] = 10 -p. H So, [H+] = 10 -7 [H+] = 1 x 10 -7 M

p. OH �Represents the “power of hydroxide” �Calculated by taking the opposite of the logarithm of the hydroxide ion concentration… p. OH = -log [OH-]
![p. OH �Calculate the p. OH of water at 25°C knowing that the [OH-] p. OH �Calculate the p. OH of water at 25°C knowing that the [OH-]](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-50.jpg)
p. OH �Calculate the p. OH of water at 25°C knowing that the [OH-] is 1 x 10 -7 M. p. OH = -log [1 x 10 -7] p. OH = 7

Ooh, ah! �So, the sum of p. H and p. OH for all aqueous solutions will be 14. p. H + p. OH = 14
![Example #4—show all work �Find the p. H, p. OH, and [OH-] and state Example #4—show all work �Find the p. H, p. OH, and [OH-] and state](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-52.jpg)
Example #4—show all work �Find the p. H, p. OH, and [OH-] and state whether the solution is acidic, neutral or basic if the hydrogen ion concentration is 3. 48 x 10 -4 M. p. H = 3. 46 p. OH = 10. 54 [OH-] = 2. 87 x 10 -11 M acidic
![Example #5—show all work �Find the p. OH, [H+], and [OH-] and state whether Example #5—show all work �Find the p. OH, [H+], and [OH-] and state whether](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-53.jpg)
Example #5—show all work �Find the p. OH, [H+], and [OH-] and state whether the solution is acidic, neutral or basic if the p. H is 9. 84. p. OH = 4. 16 [H+] = 1. 45 x 10 -10 M [OH-] = 6. 91 x 10 -5 M basic
![Example #6—show all work �Find the p. H, [H+], and [OH-] and state whether Example #6—show all work �Find the p. H, [H+], and [OH-] and state whether](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-54.jpg)
Example #6—show all work �Find the p. H, [H+], and [OH-] and state whether the solution is acidic, neutral or basic if the p. OH is 12. 7. p. H = 1. 30 [H+] = 5. 01 x 10 -2 M [OH-] = 1. 99 x 10 -13 M acidic
![Example #7—show all work �Find the p. H, p. OH, and [H+], and state Example #7—show all work �Find the p. H, p. OH, and [H+], and state](http://slidetodoc.com/presentation_image_h2/3ccc9dcaa3d968048567e49be30c738b/image-55.jpg)
Example #7—show all work �Find the p. H, p. OH, and [H+], and state whether the solution is acidic, neutral or basic if the [OH-] is 5. 26 x 10 -2 M. p. H = 12. 7 p. OH = 1. 28 [H+] = 1. 90 x 10 -13 M basic
- Slides: 55