Acids Bases Properties of Acids Taste sour Cause

Acids: Bases:
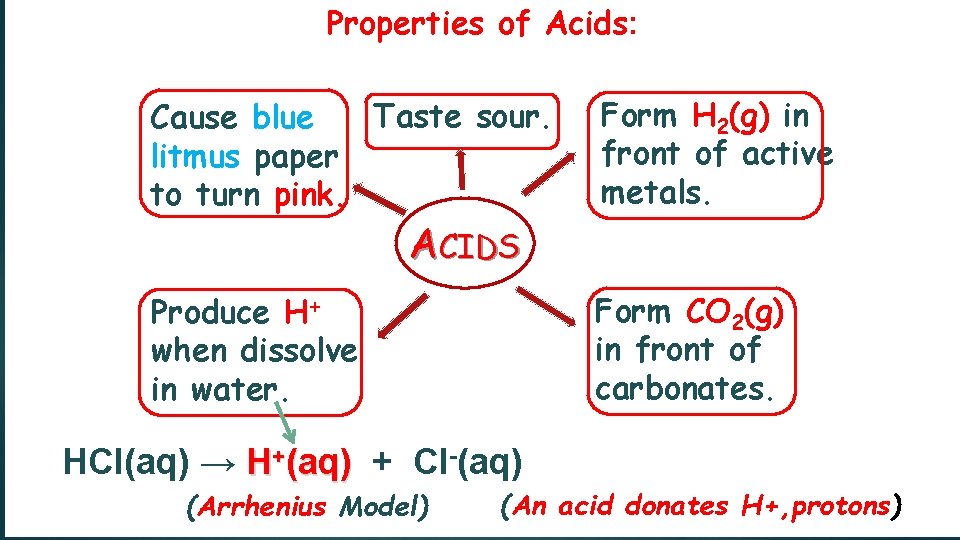
Properties of Acids: Taste sour. Cause blue litmus paper to turn pink. Form H 2(g) in front of active metals. Produce H+ when dissolve in water. Form CO 2(g) in front of carbonates. ACIDS HCl(aq) → H+(aq) + Cl-(aq) (Arrhenius Model) (An acid donates H+, protons)
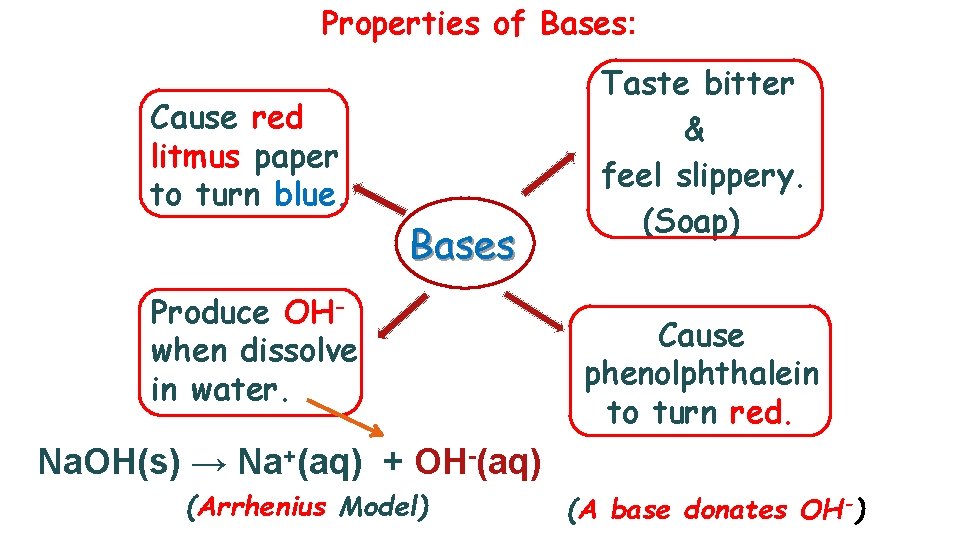
Properties of Bases: Cause red litmus paper to turn blue. Bases Produce OHwhen dissolve in water. Taste bitter & feel slippery. (Soap) Cause phenolphthalein to turn red. Na. OH(s) → Na+(aq) + OH-(aq) (Arrhenius Model) (A base donates OH-)
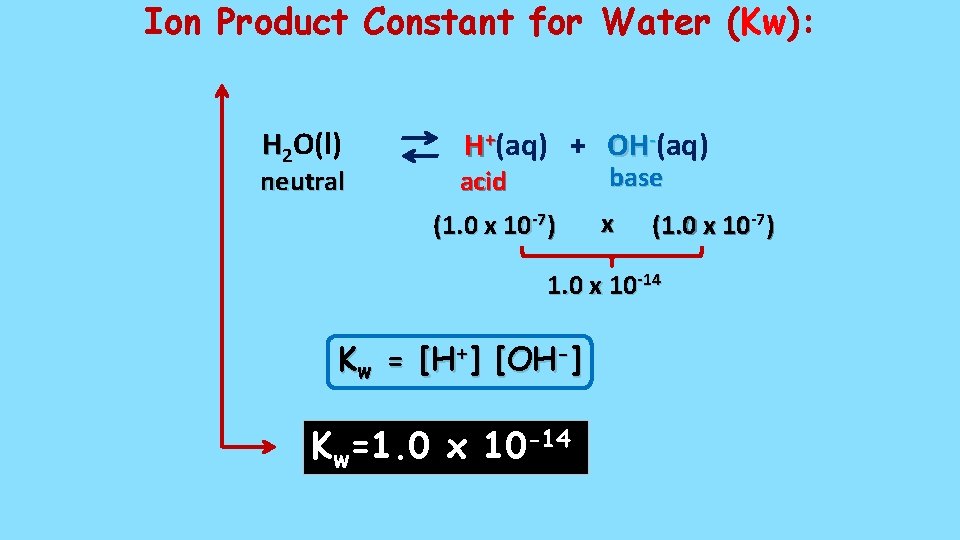
Ion Product Constant for Water (Kw): H 2 O(l) neutral H+(aq) + OH-(aq) base acid (1. 0 x 10 -7) x (1. 0 x 10 -7) 1. 0 x 10 -14 Kw = [H+] [OH-] Kw=1. 0 x 10 -14

Example: If the H+ ion concentration of an aqueous solution is 1 x 10 -5 M. What is the OH- ion concentration? Is the solution acid, basic, or neutral? [H+] =1 x 10 -5 M Kw = 1. 0 x 10 -14 [OH-] = ? Kw = [H+] [OH-] = 1. 0 x 10 -14 ÷ 1 x 10 -5 [OH-] = 1. 0 x 10 -14 -(-5) = 1. 0 x 10 -14 +5 [OH-] = 1 x 10 -9 M The solution is acidic (Makes sense? ) (Because [H+] > [OH-] )
![[OH-] Solution 10 -3 M > 10 -11 M 10 -12 M < 10 [OH-] Solution 10 -3 M > 10 -11 M 10 -12 M < 10](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-6.jpg)
[OH-] Solution 10 -3 M > 10 -11 M 10 -12 M < 10 -2 M 10 -4 M > 10 -10 M 10 -7 M = 10 -7 M acidic basic acidic neutral Kw (10 -14) = [H+] x p. H: Conclusion: If [H+] > [OH-] The solution is acidic. If [H+] < [OH-] The solution is basic. If [H+] = [OH-] The solution is neutral.
The p. H Scale:
![p. H and p. OH: p. H: Is the negative logarithm of the [H+]. p. H and p. OH: p. H: Is the negative logarithm of the [H+].](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-8.jpg)
p. H and p. OH: p. H: Is the negative logarithm of the [H+]. p. H = - log [H+] Example: [H+] =1 x 10 -6 M (log XP = p log X) p. H= - log [H+] = - log 10 -6 = -(-6) log 10 = - (-6) = 6 p. H= 6 1
![Kw (10 -14) = [H+] [OH-] 10 -3 M > 10 -11 M 10 Kw (10 -14) = [H+] [OH-] 10 -3 M > 10 -11 M 10](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-9.jpg)
Kw (10 -14) = [H+] [OH-] 10 -3 M > 10 -11 M 10 -12 M < 10 -2 M 10 -4 M > 10 -10 M 10 -7 M = 10 -7 M Solution acidic basic acidic neutral p. H: 3 12 4 7 Conclusion: If p. H < 7 The solution is acidic. If p. H > 7 If p. H = 7 The solution is basic. The solution is neutral.
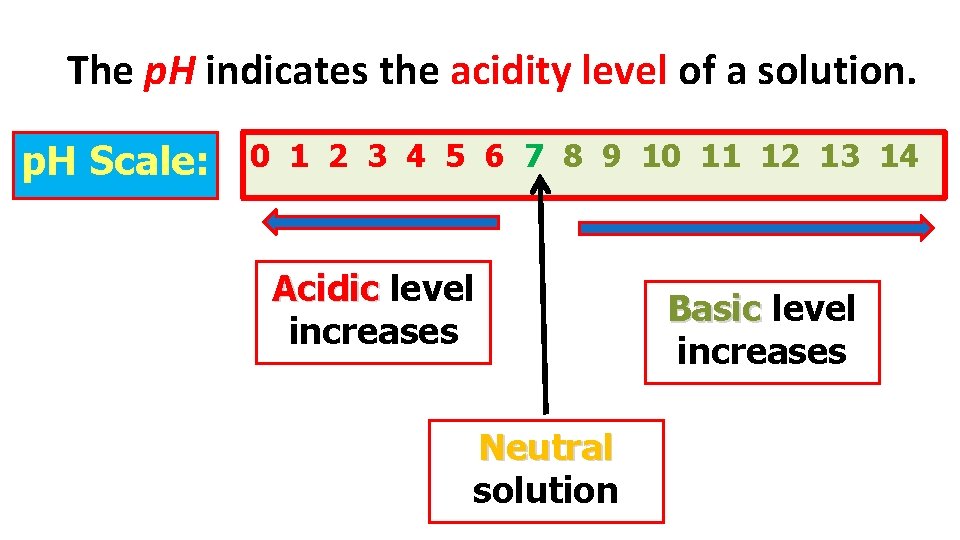
The p. H indicates the acidity level of a solution. p. H Scale: 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Acidic level increases Neutral solution Basic level increases
![p. H and p. OH: Is the negative logarithm of the [OH]. p. OH p. H and p. OH: Is the negative logarithm of the [OH]. p. OH](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-11.jpg)
p. H and p. OH: Is the negative logarithm of the [OH]. p. OH = - log [OH-] p. H + p. OH = 14
![Example: Calculate the p. H and p. OH of a solution in which [H+] Example: Calculate the p. H and p. OH of a solution in which [H+]](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-12.jpg)
Example: Calculate the p. H and p. OH of a solution in which [H+] = 1 x 10 -4 M. [H+] =1 x 10 -4 M p. H = ? p. OH = ? p. H= - log [H+] = -log 10 -4 = - (-4) = 4 p. H + p. OH = 14 4 + p. OH = 14 - 4 p. OH = 10 (acidic)
![[H+] = 10 -4 M p. H= 4 [H+] = 10 -p. H If [H+] = 10 -4 M p. H= 4 [H+] = 10 -p. H If](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-13.jpg)
[H+] = 10 -4 M p. H= 4 [H+] = 10 -p. H If p. H= 8 [H+] = 10 -8 M p. OH = 6 [OH-] = 10 -6 M [OH-] = 10 -p. OH
![Complete the following table: [OH-] [H+] -4 M -8 M -7 M 0 1 Complete the following table: [OH-] [H+] -4 M -8 M -7 M 0 1](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-14.jpg)
Complete the following table: [OH-] [H+] -4 M -8 M -7 M 0 1 x 1 0 1 x 1 -13 0 1 x 1 -10 0 1 x 1 -6 0 1 x 1 M 4 10 8 6 M 7 7 -1 M 13 1 x 10 1 M p. OH -7 0 1 x 1 M p. H acid base neutral √ √
![SUMMARY: Kw=[H+] [OH-]=1 x 10 -14 p. H=- log [H+] p. OH=- log [OH-] SUMMARY: Kw=[H+] [OH-]=1 x 10 -14 p. H=- log [H+] p. OH=- log [OH-]](http://slidetodoc.com/presentation_image_h2/ac0c64ad3c33d63e6c1ba5705a5d5c54/image-15.jpg)
SUMMARY: Kw=[H+] [OH-]=1 x 10 -14 p. H=- log [H+] p. OH=- log [OH-] p. H + p. OH =14 [H+] = 10 -p. H [OH-] = 10 -p. OH
- Slides: 15