Acids Bases Properties electrolytes sour taste bitter taste

Acids & Bases

Properties ö electrolytes ö sour taste ö bitter taste ö turn litmus red ö turn litmus blue ö react with metals ö slippery feel to form H 2 gas ö vinegar, milk, soda, apples, citrus fruits ö Alkaline ö ammonia, lye, antacid, baking soda Chem. ASAP
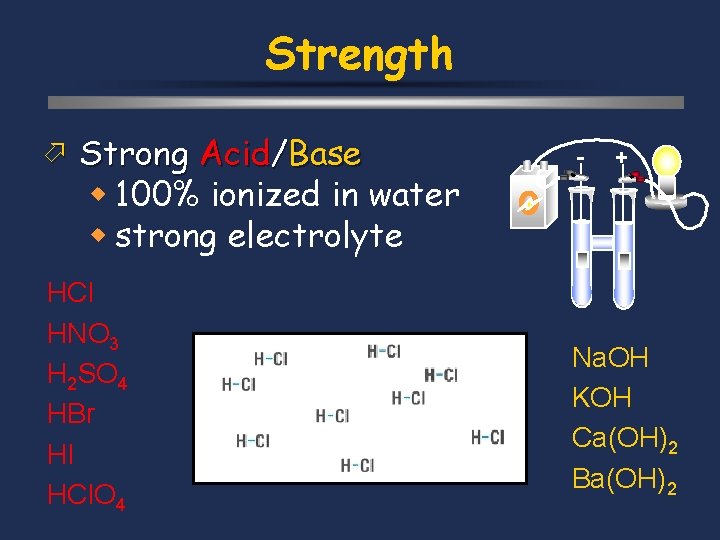
Strength ö Strong Acid/Base w 100% ionized in water w strong electrolyte HCl HNO 3 H 2 SO 4 HBr HI HCl. O 4 - + Na. OH KOH Ca(OH)2 Ba(OH)2
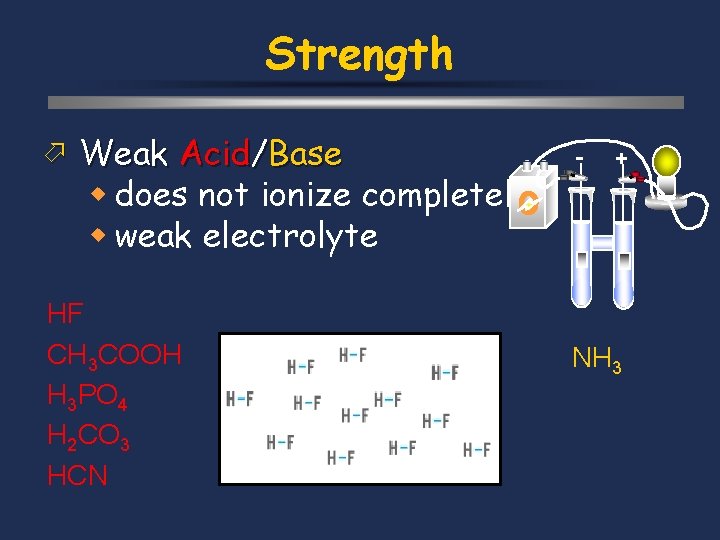
Strength ö Weak Acid/Base w does not ionize completely w weak electrolyte HF CH 3 COOH H 3 PO 4 H 2 CO 3 HCN - + NH 3

p. H Scale p. H of Common Substances

Practice 0 to __ 14 The range of the p. H scale is __ 7 0 to __ acids range in p. H from __ 7 to __ 14 Bases range in p. H from __ neutral A p. H of 7 is considered ______ Weak acid Vinegar is a _____ Weak base Baking soda is a ______ Strong base Bleach is a ______ Strong acid Stomach juice is a _____
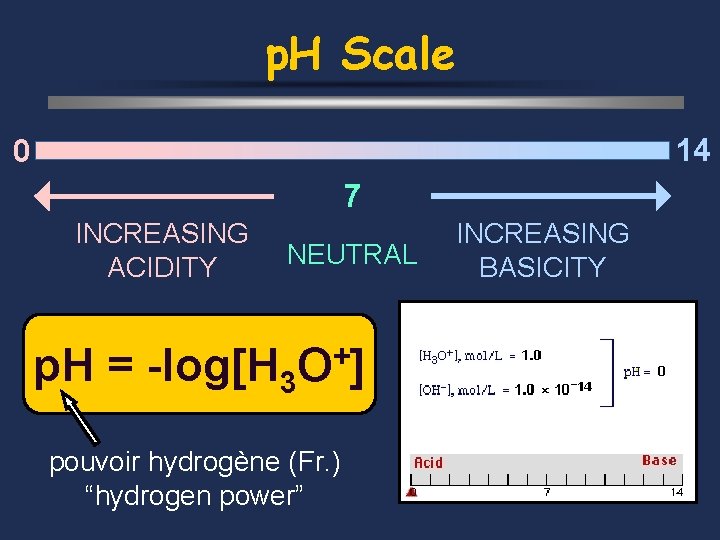
p. H Scale 14 0 7 INCREASING ACIDITY NEUTRAL p. H = -log[H 3 + O] pouvoir hydrogène (Fr. ) “hydrogen power” INCREASING BASICITY
![p. H Scale p. H = -log[H 3 p. OH = + O] -log[OH p. H Scale p. H = -log[H 3 p. OH = + O] -log[OH](http://slidetodoc.com/presentation_image_h2/380da33dcc4580218fb5999c0523eb03/image-8.jpg)
p. H Scale p. H = -log[H 3 p. OH = + O] -log[OH ] p. H + p. OH = 14

Indicators pg. 495 A dye that turns colors depending on the presence of an acid or a base- colors change as the p. H of the solution changes Litmus paper p. H paper Universal Indicator

Neutralization ö Chemical reaction between an acid and a base. ö Products are a salt (ionic compound) and water.

Neutralization ACID + BASE SALT + WATER HCl + Na. OH Na. Cl + H 2 O strong neutral HC 2 H 3 O 2 + Na. OH Na. C 2 H 3 O 2 + H 2 O weak strong basic w Salts can be neutral, acidic, or basic. w Neutralization does not mean p. H = 7.

Self Ionization of Water H 2 O + H 2 O H 3 O+ + OH- ö Amphoteric ~ can be an acid or a base. ö Water is amphoteric
- Slides: 12