Acids Bases Properties and Indicators Properties electrolyte sour

Acids & Bases Properties and Indicators

Properties • • electrolyte sour taste sticky feel turn litmus red • • electrolyte bitter taste slippery feel turn litmus blue • react with bases to • react with acids to form water and a salt (ionic compound)

Acids Affect Indicators: Blue litmus paper turns red in contact with an acid.

Bases affect indicators: Red litmus paper turns blue in contact with a base. Phenolphthalein turns pink in a base.

Examples ACIDS: Most citrus fruits, tea, battery acid, vinegar, milk, soda, apples. BASES: Common household bases include baking soda, lye, ammonia, soap, and antacids. .
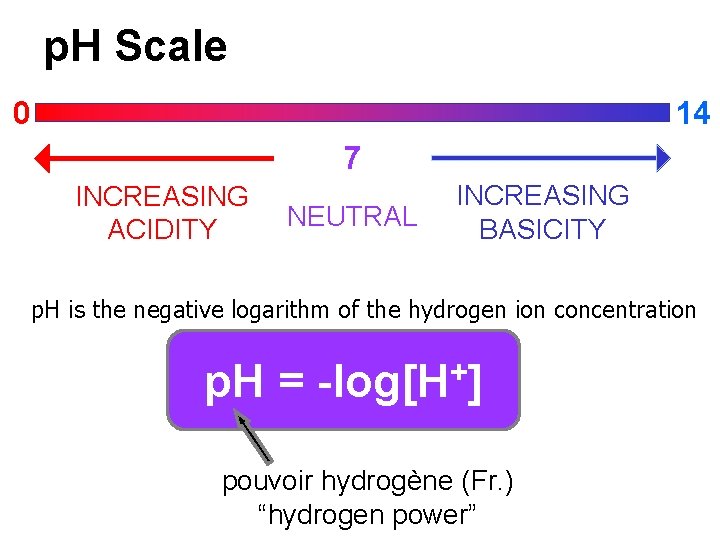
p. H Scale 14 0 7 INCREASING ACIDITY NEUTRAL INCREASING BASICITY p. H is the negative logarithm of the hydrogen ion concentration p. H = + -log[H ] pouvoir hydrogène (Fr. ) “hydrogen power”
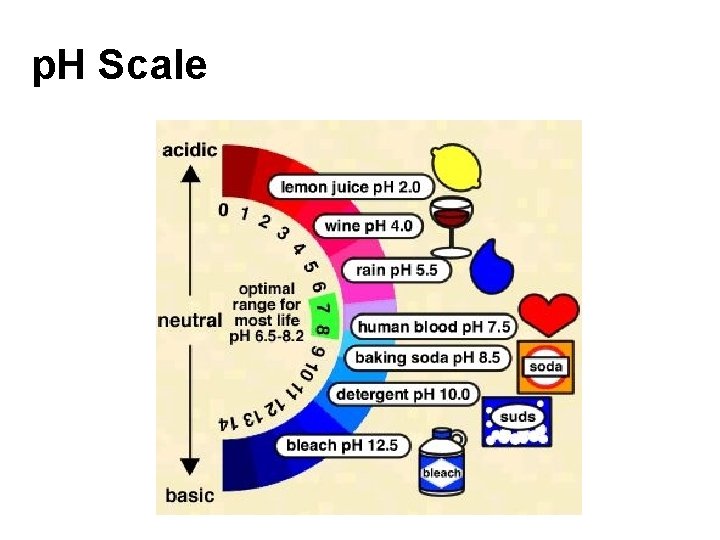
p. H Scale
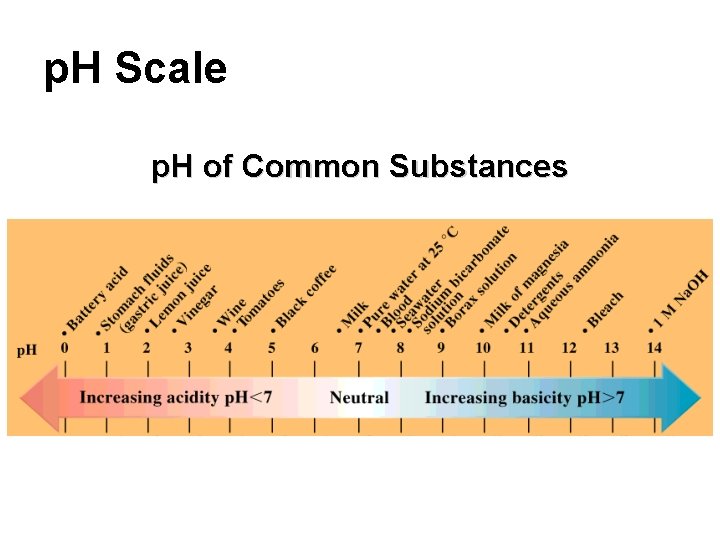
p. H Scale p. H of Common Substances

Titration standard solution • Titration – Analytical method in which a standard solution is used to determine the concentration of an unknown solution
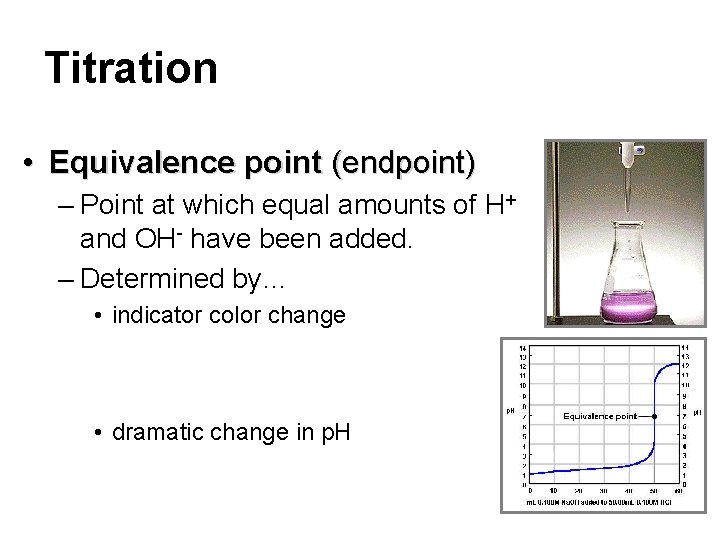
Titration • Equivalence point (endpoint) – Point at which equal amounts of H+ and OH- have been added. – Determined by… • indicator color change • dramatic change in p. H


Acids H+ ions- Hydrogen ions H 3 O+ ions- Hydronium ions Proton donor Bases OH- ions- hydroxide ions Electron donor



Molarity • Molarity= moles of a substance/liter water • Molarity= M • Ex. 18 M Sulfuric acid means that there are 18 moles of sulfuric acid per one liter of water • Molarity deals with liquid solutions
- Slides: 15