Acids Bases p Hused to measure how acidic

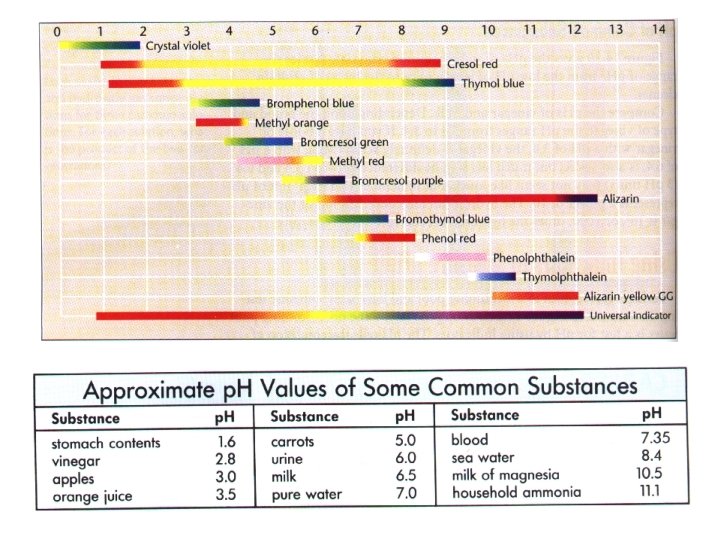
Acids & Bases p. H-used to measure how acidic or basic a substance is. Determining p. H: 1)p. H Meter-device used to determine p. H of a solution using a voltmeter +2 electrodes. . : V p. H 2)Indicators-dyes used to determine p. H of a substance. Turns different colors at various p. H’s Ex/phenolphthalein litmus universal clear pink p. H>7. 6 red blue p. H>7 red purple p. H 1 -14

Some natural dyes such as Cabbage Juice can be used as p. H indicators. How would you calibrate these colors so if you used Cabbage Juice as an indicator, you would know the p. H?

Even in ancient civilizations such as Teotihuacan in 1 -200 A. D. , Mesoamericans used natural pigments from the local cactus plant to produce 2 different colored dyes to paint the Pyramid of the Sun, Pyramid of the Moon, and other structures at the site. See: http: //studentweb. tulane. edu/~dhixson/teo. html http: //archaeology. la. asu. edu/teo/ http: //www. teotihuacan. com/

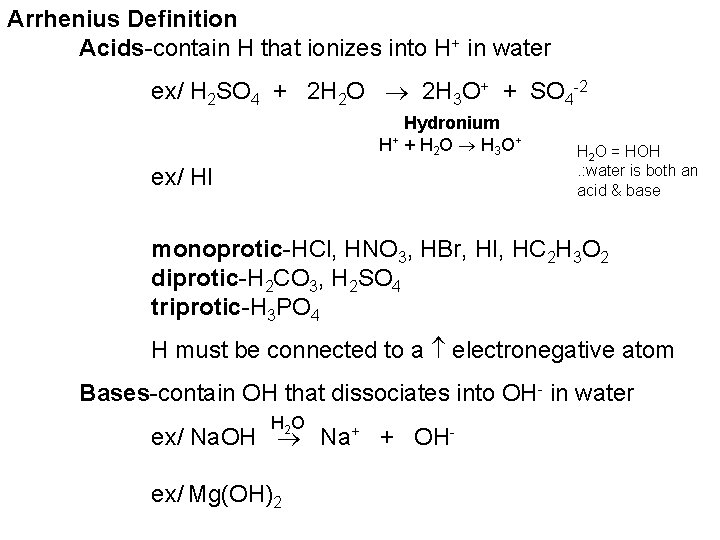
Arrhenius Definition Acids-contain H that ionizes into H+ in water ex/ H 2 SO 4 + 2 H 2 O 2 H 3 O+ + SO 4 -2 Hydronium H+ + H 2 O H 3 O + ex/ HI H 2 O = HOH. : water is both an acid & base monoprotic-HCl, HNO 3, HBr, HI, HC 2 H 3 O 2 diprotic-H 2 CO 3, H 2 SO 4 triprotic-H 3 PO 4 H must be connected to a electronegative atom Bases-contain OH that dissociates into OH- in water H 2 O ex/ Na. OH Na+ + OHex/ Mg(OH)2
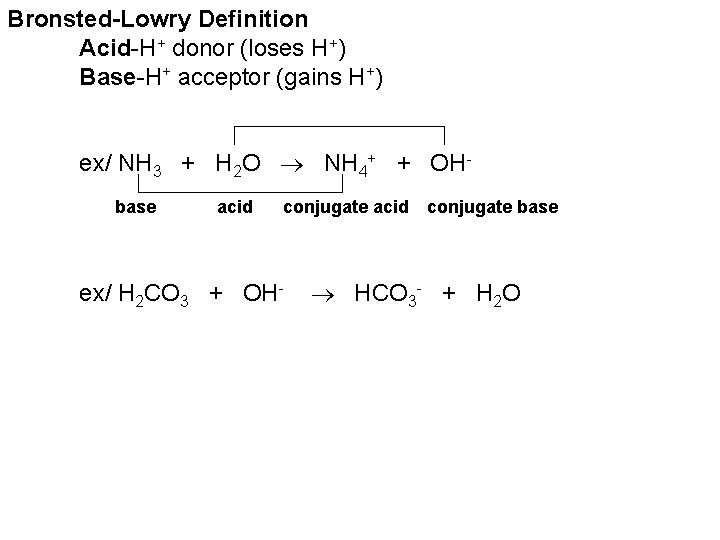
Bronsted-Lowry Definition Acid-H+ donor (loses H+) Base-H+ acceptor (gains H+) ex/ NH 3 + H 2 O NH 4+ + OHbase acid conjugate acid ex/ H 2 CO 3 + OH- conjugate base HCO 3 - + H 2 O

Strong Acids & Bases-Fully separate(ionize) into ions in water. : Good conductors electricity in water! Ex/ HCl + H 2 O H 3 O+ + Cl. Na. OH Na+ + OHWeak Acids & Bases-Not fully separate(dissociate) into ions in water. : poor conductors electricity in water! Ex/ HC 2 H 3 O 2 + H 2 O H 3 O+ + C 2 H 3 O 2 NH 4 OH NH 4+ + OH-

ex/ Normal CO 2 + H 2 O H 2 CO 3 Acid Rain p. H=5 -6 Abnormal NO 2 + H 2 O HNO 3 p. H=3 -4 Abnormal SO 2 + H 2 O H 2 SO 4 p. H=3 -4 Strong Acid or Base Weak Acid or Base

Uses of Acids & Bases Neutralization-Reaction involving an Acid & Base reacting together to produce a Salt & H 2 O p. H=7 Ex/ upset stomach? HCl + Mg(OH)2 Mg. Cl 2 + H 2 O HCl + Ca(OH)2 Ca. Cl 2 + H 2 O No Acid/Base Properties Ph=7 Buffers-Compounds used to keep p. H set at a value. Weak acid + its salt Ex/ lakes, pools, spas, blood H 2 CO 3 + Na 2 CO 3 p. H=7. 6

Titrations-Scientists use to determine concentration of unknown acidic/basic solution. Use Buret -Uses: neutralization reaction balanced equation standardized solution unknown solution p. H=7 equivalence point Macid vacid = Mbase vbase p. H=7 If 1: 1 in balanced equation Ex/ Concentrated stomach acid? HCl + Na. OH Na. Cl + H 2 O 32 m. L ? 25. 3 m. L 1. 0 mol/L
- Slides: 11