ACIDS BASES Kenneth E Schnobrich ACIDS OPERATIONAL DEFINITION

ACIDS & BASES Kenneth E. Schnobrich

ACIDS - OPERATIONAL DEFINITION n n Have a sour taste (not recommended) React with bases to form water and a salt (Neutralizationn) React with some metals to form a salt and H 2 gas (see Table J) Reacts with indicators n n Litmus Blue -> Red Phenolphthalein - colorless

ACIDS - OPERATIONAL DEFINITION n n In aqueous solution they are good electrolytes (conduct current) Can be very corrosive and can cause severe burns

BASES - OPERATIONAL DEFINITION n n n Bases have a bitter taste (not recommended) They have a slippery feel (soapy) In aqueous solution they are good electrolytes (conduct electric current) React with acids to form a salt and water (neutralization) Cause indicators to change color n n Litmus Red -> Blue Phenolphthalein - pink

ACIDS - CONCEPTUAL DEFINITION (Arrhenius) n n ACIDS - are substances that will yield H+ as the only positive ion in an aqueous solution. HCl(aq) --> H+(aq) + Cl-(aq) Monoprotic acid n H 2 SO 4(aq) --> 2 H+(aq) + SO 4 -2(aq) Diprotic acid n H 3 PO 4(aq) --> 3 H+1(aq) + PO 4 -3(aq) Triprotic acid

BASES - CONCEPTUAL DEFINITION (Arrhenius) n BASES - are substances that will yield OH- as the only negative ion in an aqueous solution. n Na. OH(s) --> Na+(aq) + OH-(aq) n Ca(OH)2(s) --> Ca+2(aq) + 2 OH-1(aq) n Fe(OH)3(s) --> Fe+3(aq) + 3 OH-1(aq)

Acid/Base Reference Tables These are considered strong acids because they ionize almost completely in an aqueous solution. They yield a high concentration of H + or H 3 O+ ions in aqueous solutions. These are considered moderate to weak acids because they do not ionize completely in an aqueous solution. They yield a lower concentration of H + or H 3 O+ ions in aqueous solutions. These are considered strong bases because they ionize (dissociate) almost completely in an aqueous solution. They yield a high concentration of OH - ions in aqueous solutions. These are considered weak bases because they ionize (dissociate) to a small degree in an aqueous solution. They yield a low concentration of OH - ions in aqueous solutions.
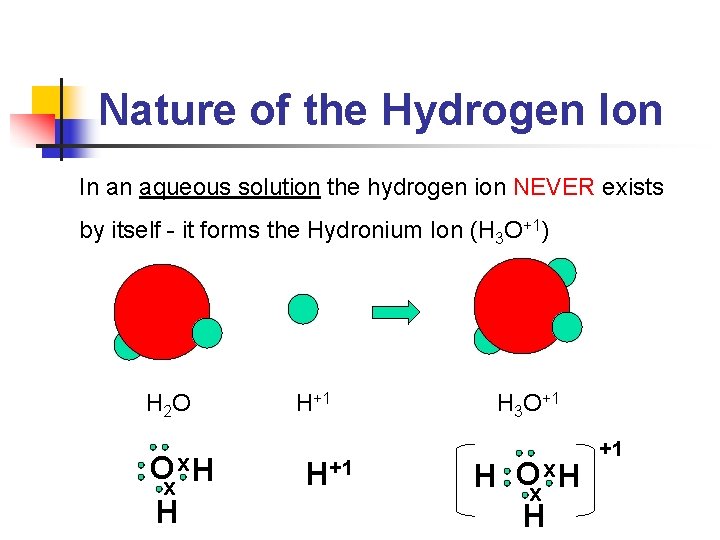
Nature of the Hydrogen Ion In an aqueous solution the hydrogen ion NEVER exists by itself - it forms the Hydronium Ion (H 3 O+1) H 2 O O x. H x H H+1 H 3 O+1 H Oxx. H H +1

Arrhenius Theory ACIDS - substances whose water solutions (aqueous) contain Hydrogen ions (Hydronium ions) as the only positive ion. CHART K - contains a list of common Arrhenius acids Listed by strength (strongest ->weakest) BASES - substances whose water solutions (aqueous) contain Hydroxide ions as the only negative ion. CHART L - contains a list of common Arrhenius bases Listed by strength (strongest ->weakest)

ACID/BASE STRENGTH The Strength of an acid or base is determined by how much it ionizes in an aqueous solution. HCl, HNO 3, H 2 SO 4 - are considered strong because they ionize almost 100% in an aqueous solution KOH, Na. OH - are considered strong bases because they ionize (dissociate) almost 100% in a aqueous Solution Therefore, strong acids and bases are good electrolytes in an aqueous solution

BRONSTED THEORY The Bronsted-Lowry Theory is an expansion of the definition of acids and bases (not a replacement). ACIDS - substances that will donate a proton (all Arrhenius acids are also Bronsted acids) BASES - substances that will accept a proton (all Arrhenius bases are also Bronsted bases) HCl + H 2 O -> H 3 O+1 + Cl-1

REACTIONS Acids with Metals TABLE J of your Reference Tables for Chemistry is important in determining what metals will react with an acid - any metal above **H 2 on Table J will react with an acid to form H 2 gas and a salt. HCl(aq) + Mg(s) -> Mg. Cl 2(aq) + H 2(g)

Activity Series All of the metals listed above **H 2 on this activity series will react to replace hydrogen from an acid to produce H 2 as one of the products of the reaction. 2 HCl(aq) + Zn(s) -> Zn +2(aq) + 2 Cl-(aq) + H 2(g)
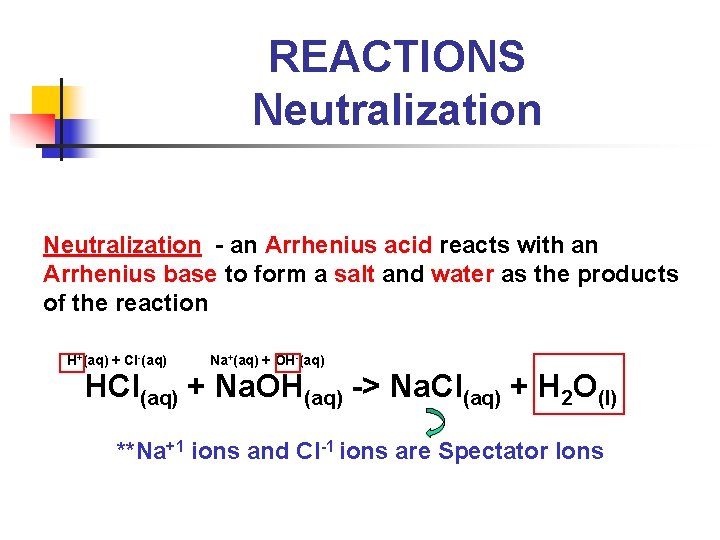
REACTIONS Neutralization - an Arrhenius acid reacts with an Arrhenius base to form a salt and water as the products of the reaction H+(aq) + Cl-(aq) Na+(aq) + OH-(aq) HCl(aq) + Na. OH(aq) -> Na. Cl(aq) + H 2 O(l) **Na+1 ions and Cl-1 ions are Spectator Ions
![p. H/Acid/Base - Strength p. H = -log of the [H+] in a solution. p. H/Acid/Base - Strength p. H = -log of the [H+] in a solution.](http://slidetodoc.com/presentation_image_h2/38cf60a7abf07b63746398ec1b5a10b9/image-15.jpg)
p. H/Acid/Base - Strength p. H = -log of the [H+] in a solution. The p. H scale describes the concentration of [H+] in an aqueous solution. The scale is based on neutral water where the [H+] = [OH-] concentration. HOH(l) -> H+(aq) + OH-(aq) **Important note: remember H+ = H 3 O+ in aqueous solution
![p. H SCALE Neutral 0 Becoming more Acidic [H]+>[OH]- 7 Becoming more Basic [H]+=[OH]*Remember p. H SCALE Neutral 0 Becoming more Acidic [H]+>[OH]- 7 Becoming more Basic [H]+=[OH]*Remember](http://slidetodoc.com/presentation_image_h2/38cf60a7abf07b63746398ec1b5a10b9/image-16.jpg)
p. H SCALE Neutral 0 Becoming more Acidic [H]+>[OH]- 7 Becoming more Basic [H]+=[OH]*Remember [ ] = concentration in mols/L 14 [H]+<[OH]-

p. H and Indicators • Indicators tend to be weak organic acids – HIn(aq) H+(aq) + In-(aq) Color 1 Color 2 • A change in color indicates a change in the p. H of the solution by shifting the equilibrium above to decrease or increase the amounts of HIn or In- in solution. If you started with a p. H of 2. 0 and used bromcresol green as your indicator the solution would appear yellow. If you now added a base like Na. OH, as the p. H increases to something between 3. 8 and 5. 4, the solution would appear green in color (a combination of yellow and blue).

Titration using an Indicator • Acid-Base titrations are very common in chemistry labs. What is a titration? • A titration is a procedure in which a base is added to an acid (or vice versa), a neutralization reaction occurs; an indicator is used to determine the point at which you have equivalent quantities of H+(H 3 O+) and OH-. • A commonly used acid-base indicator is Phenolphthalein. • Using Phenolphthalein, you are looking for a faint pink color to indicate the so-called “end point” or “equivalence point”. At the “end point” even a trace amount of base will cause a drastic change in color because of the equilibrium shift. HIn (aq) H+(aq) + In-(aq) Colorless in acid solution Deep pink in base solution Phenolphthalein

Titration In the burette you would place a known quantity of base like Na. OH (unknown molarity) – deliver it slowly using the stopcock You would add base until a very faint pink color appears in the flask – the equivalence point. In the Erlenmeyer flask you would have an acid solution with a known volume and molarity. You would also have 4 -5 drops of Phenolphthalein indicator (it would appear colorless in the acid solution)
![Titration Calculation • Since we assume at the equivalence point that the [H+] = Titration Calculation • Since we assume at the equivalence point that the [H+] =](http://slidetodoc.com/presentation_image_h2/38cf60a7abf07b63746398ec1b5a10b9/image-20.jpg)
Titration Calculation • Since we assume at the equivalence point that the [H+] = [OH-] we can say #mols H+ = #mols OH-. • M(V) = # mols • at the equivalence point Ma. Va = Mb. Vb (see Table T) • If we had 20 m. L of a 1. 0 M acid and it took 40 m. L of the base, what is the molarity of the base? • (1. 0 M)(20 m. L) = (X)(40 m. L) • X = (1. 0 M)(20 m. L)/40 m. L = 0. 5 M for the base
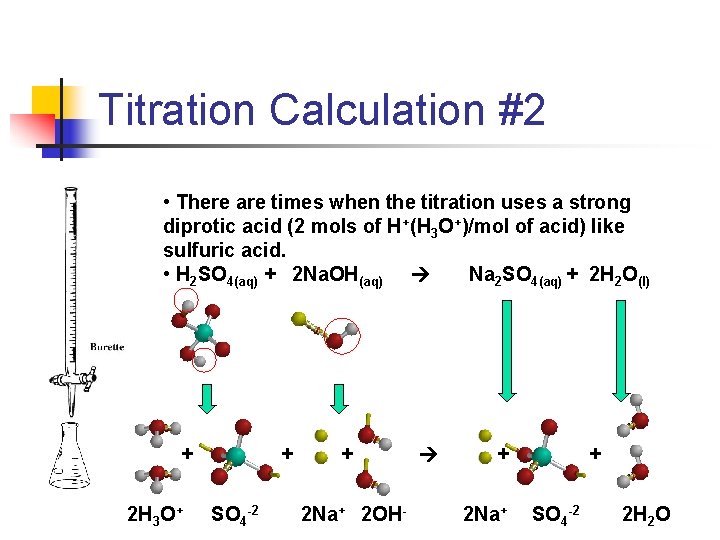
Titration Calculation #2 • There are times when the titration uses a strong diprotic acid (2 mols of H+(H 3 O+)/mol of acid) like sulfuric acid. • H 2 SO 4(aq) + 2 Na. OH(aq) Na 2 SO 4(aq) + 2 H 2 O(l) + 2 H 3 O+ + SO 4 -2 + 2 Na+ 2 OH- + 2 Na+ + SO 4 -2 2 H 2 O

Titration Calculation #2 • On the previous slide you notice it takes 2 mols of Na. OH to neutralize the 2 mols of H+ (H 3 O+) using the diprotic acid, sulfuric acid (H 2 SO 4). • If we have 20. 0 m. L of 1. 0 M sulfuric acid in the flask and it takes 40 m. L of the base, Na. OH, what is the molarity of the base? • M a V a = M b. V b • 2. 0 M(20. 0 m. L) = X(40 m. L) • X = 2. 0 M(20. 0 m. L)/40 m. L) = 1. 0 MNa. OH • Notice the adjusted molarity of the acid to 2. 0 M because it is a diprotic acid. Sulfuric acid will yield 2 mols of hydrogen ions (hydronium ions) per mol of acid ionized.

Kw for Water • The Kw for H 2 O = 1 x 10 -14 at 25°C. This means that the [H+] and the [OH-] equals 1 x 10 -7. • This means that in an aqueous solution the [H+][OH-] = 1 x 10 -14. • In a 0. 001 M HCl solution the concentration of [H+] = 1 x 10 -3 and the [OH-] = 1 x 10 -11 because the product of the two concentrations must always equal 1 x 10 -14 in an aqueous solution. • The p. H of this solution would be 3. • p. H = -log [H+] = -(log 1 x 10 -3) = -(-3) = 3 • At p. H of 3 it would be considered a fairly strong acid solution HCl H 2 O H 3 O+ Cl-

Hydrolysis of a Salt • Hydrolysis of a salt is the reverse of neutralization. In neutralization, an acid and a base react to form a salt and water. • Salts can be formed by reactions of 1. strong acid and a strong base 2. strong acid and a weak base 3. strong base and weak acid 4. Weak acid and weak base • In cases 1, 2, and 3 it is easy to predict the outcome of hydrolysis of the resulting salt. In case 4 you would have to know the values for the strengths of the acid and base (information you do not have).

Hydrolysis Case #1 Na. Cl(s) + HOH(l) = Na+(aq) + OH-(aq) + H+(aq) + Cl-(aq) Na. Cl is the salt of a strong acid and a strong base so when it dissolves in water there will be an equal number of H + and OH- ions in solution. The aqueous solution would have a p. H of 7.

Hydrolysis Case #2 NH 4 NO 3(s) + HOH = NH 4+(aq) + OH-(aq) + H+(aq) + NO 3 -(aq) Since NH 4 NO 3 is the salt of a weak base, NH 4 OH and a strong acid, HNO 3 the salt will yield a higher concentration of H+ in the aqueous solution than the OH- ions. The solution of NH 4 NO 3 with have an acidic p. H.

Hydrolysis Case #3 K 2 CO 3(s) + 2 HOH(l) = 2 K+(aq) + 2 OH-(aq) + 2 H+(aq) + CO 3 -2(aq) Since K 2 CO 3 is the salt of a strong base, KOH and a weak acid, H 2 CO 3 the salt will yield a higher concentration of OHin the aqueous solution than the H+ ions. The solution of K 2 CO 3 with have an basic p. H.
- Slides: 27