Acids Bases I Properties of Acids Bases taste

Acids & Bases I. Properties of Acids & Bases -_____ taste ____ and produce a _______ sensation to the touch -______ acid and ____ acid, (also known as _______ __), are _____ responsible for the _______ of ______ and other ______, while ______ makes _______ taste ____, and in our body, the unpleasant ____ and _________ produced by our _____ is due to ______ in our ____

Acids & Bases I. Properties of Acids & Bases -because _____ and ________, (and all ______ are made of _______), in ______, they are often used as a _______ -____, ______, and ______ all use ______ to ______ themselves, while the human _______ is guarded against _______ by ______, the human ______ is guarded against _______ by ______ (produced by _____- _______ of the genus ______), and even human ____ is guarded by ____ in our _____

Acids & Bases I. Properties of Acids & Bases -_____ react with ______, especially the more ______, to produce ____ ___ -_____ react with _____ and _________ to produce _______

Acids & Bases I. Properties of Acids & Bases Write the balanced thermochemical equation for the reaction that occurs between: 1. Magnesium and Nitric acid 2. Aluminum and Sulfuric acid 3. Calcium carbonate and Hydrobromic acid 4. Potassium Hydrogen carbonate and Hydrochloric acid

Acids & Bases I. Properties of Acids & Bases 1. Hypothesis: 2. Prediction: 3. Gather Data: A. Safety: B. Procedure: Name_________
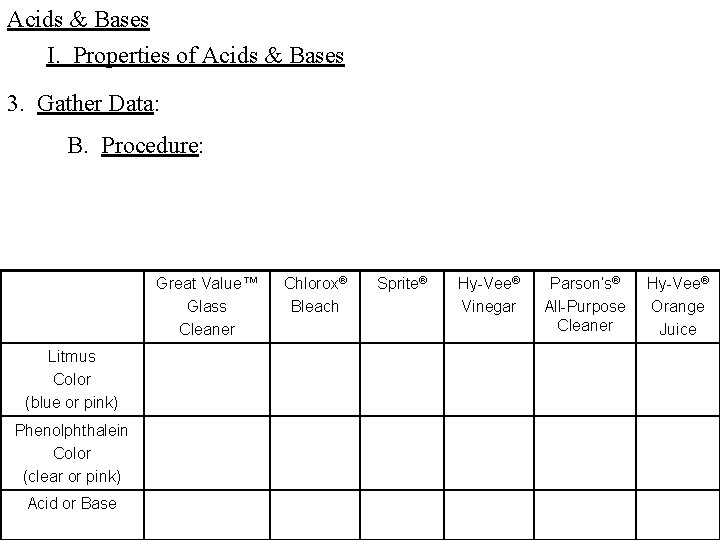
Acids & Bases I. Properties of Acids & Bases 3. Gather Data: B. Procedure: Great Value™ Glass Cleaner Litmus Color (blue or pink) Phenolphthalein Color (clear or pink) Acid or Base Chlorox® Bleach Sprite® Hy-Vee® Vinegar Parson’s® All-Purpose Cleaner Hy-Vee® Orange Juice
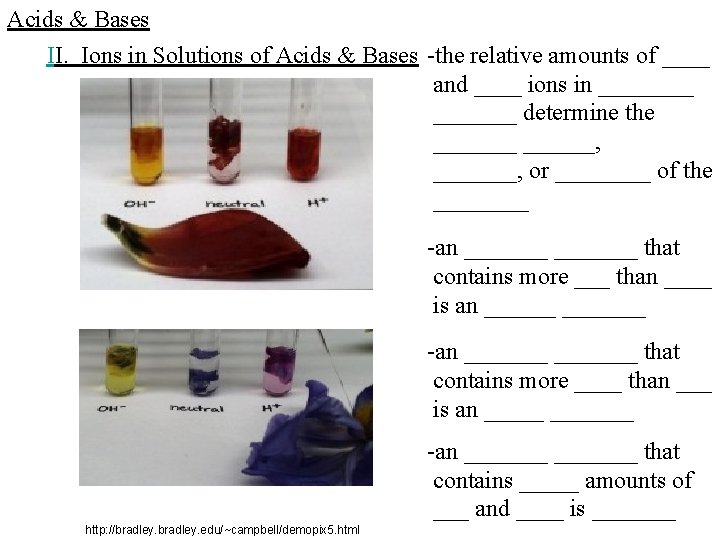
Acids & Bases II. Ions in Solutions of Acids & Bases -the relative amounts of ____ and ____ ions in _______ determine the _______, _______, or ____ of the ____ -an _______ that contains more ___ than ____ is an _______ -an _______ that contains more ____ than ___ is an _______ -an _______ that contains _____ amounts of ___ and ____ is _______ http: //bradley. edu/~campbell/demopix 5. html

Acids & Bases II. Ions in Solutions of Acids & Bases -_______ that are neither ______ or _____ are _______, but _____ is _______ because it is ____ and a ____ at the same time, containing _____ amounts of ___ and ____ -because of its _______, ___________: -but the ___ forms a _______ with a ______, producing the _____ http: //bradley. edu/~campbell/demopix 5. html

Acids & Bases III. The Arrhenius Model -according to _________, an of Acids and Bases ____ is a ____ that contains ____ and _______ to produce ____ in _______ -according to _____, ________ is an _____ -according to _____, a _____ is a ____ that contains a _____ and _____ to produce _____ in ________ -according to _____, _________ is a _____
Acids & Bases IV. The Brønsted –Lowry Model -according to ____-______, of Acids and Bases an _____ is a _____-______ and a _____ is a ____- ________ -the _____, _____ in _____ and _______ a ____ to a ________, which acts as a _____ by ____ the ____ -a ____ that has ____ a ___ now has the ability to ______ a ___, and so becomes an _____; the _____

Acids & Bases IV. The Brønsted –Lowry Model -according to ____-______, of Acids and Bases a _____ is the ______ produced when a ____ accepts a ____ ___ from an ____ -a _____ is the ______ that is left over from the ____ after it donates a ____ Playing Hacky Sack in Zurich, Switzerland http: //zurichdailyphoto. blogspot. com/2007/04/hacky-sack. html

Acids & Bases IV. The Brønsted –Lowry Model of Acids and Bases Label the acid, base, conjugate acid, and conjugate base in the following: 1 NH 3 (aq) + 1 H 2 O (l) 1 NH 4+(aq) + 1 OH- (aq) 1 HBr (aq) + 1 H 2 O (l) 1 H 3 O+(aq) + 1 Br- (aq) 1 CO 32 - (aq) + 1 H 2 O (l) 1 HSO 4 - (aq) + 1 H 2 O (l) 1 HCO 3 -(aq) + 1 OH- (aq) 1 H 3 O+(aq) + 1 SO 42 - (aq)
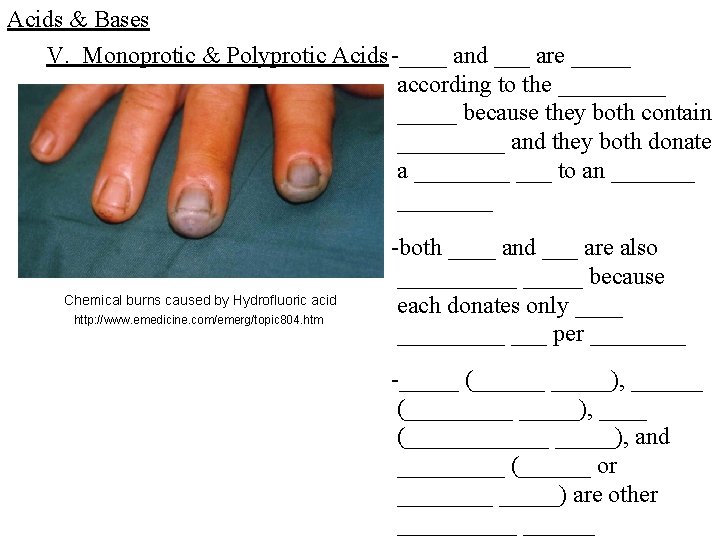
Acids & Bases V. Monoprotic & Polyprotic Acids -____ and ___ are _____ according to the _____ because they both contain _____ and they both donate a ____ ___ to an ________ Chemical burns caused by Hydrofluoric acid http: //www. emedicine. com/emerg/topic 804. htm -both ____ and ___ are also _____ because each donates only _________ per ____ -_____ (______), ______ (_____), ____ (______), and _____ (______ or _____) are other ______
Acids & Bases V. Monoprotic & Polyprotic Acids -_____ that donate _____ than _________ are called _____ -______(_______) and ______(_____) are both _______, while ______ (_____) and ______ (______) are both ______
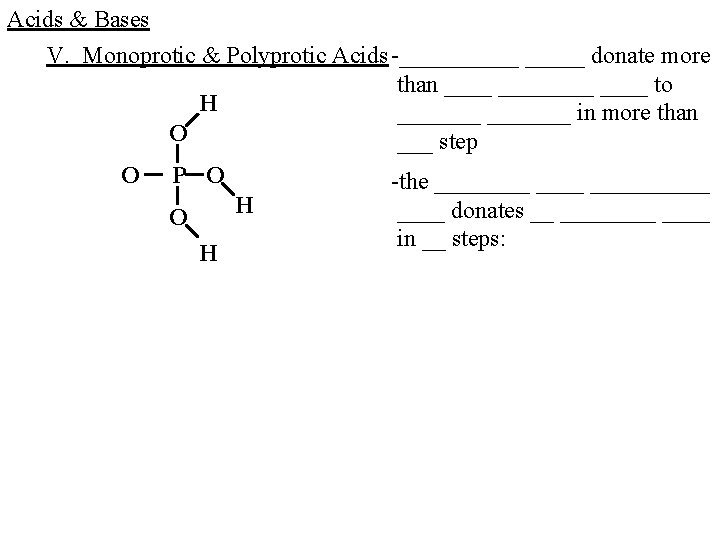
Acids & Bases V. Monoprotic & Polyprotic Acids -_____ donate more than ________ to H _______ in more than O ___ step O P O -the __________ H ____ donates __ ____ O in __ steps: H

Acids & Bases V. Monoprotic & Polyprotic Acids Write the steps for complete ionization of the following polyprotic acids: H 2 Se (aq) H 3 As. O 4 (aq) H 2 SO 3 (aq)
Acids & Bases VI. Acid & Base Anhydrides -_____ are _______ that form _____ and _____ when added to ______ -______ of ______ elements produce _____ in _______, while ______ of ____ elements produce _____ in ________

Acids & Bases VII. Strength of Acids -______ are _____ that _________ Hydrofluoric acid is considered a weak acid because it doesn’t ionize completely; it is expected to be a strong acid, because it is so corrosive, but its corrosiveness is due to the chemical reactivity of its anion, the fluoride ion. The strongest acids, the carborane superacids, are a million times stronger than concentrated sulfuric acid, but not at all corrosive because the anion to the acid is so unreactive. -____ (______), ______ (_____), _____ (______), ____ (______), ______ (_____), and ___ (______) are all ______ -______ are _____ that ______ only ____ -____ (______), ______ (_____), ___ (______), ______ (_____), and _____ (______) are _____

Acids & Bases VII. Strength of Acids -according to _______, the ____ on the _______ side of the _______ produces a ____ on the ____ side of the _______ by _______ a ____ ___ -if the ____ is a ______, then its _____ is _____, that is, if the ____ readily _______ a ____ ___, then its ____ is not very good at ____a ____ ___, or holding on to one, while a ____, which is not very good at _______ a ____ ___ has a ________

Acids & Bases VII. Strength of Acids -the _____ for ______ is: -the __________ for the _____ of ______ is: -the ______ of ______ is considered to be _______, and so can be _____ with ___ to give the _________, or ___

Acids & Bases VII. Strength of Acids Write ionization equations and acid ionization constant expressions for the following acids: HCl. O 2 (aq) HNO 2 (aq) HIO (aq)


Acids & Bases VIII. Strength of Bases -the ________ for _____ is: -the __________ for the _____ of ______ is:

Acids & Bases VIII. Strength of Bases Write ionization equations and base ionization constant expressions for the following bases: C 3 H 7 NH 2 (aq) CO 32 -(aq) HSO 3 - (aq)
Acids & Bases IX. Ion Product Constant for Water -the ________ for ______ is: -the ________ can be simplified to: -the __________ is:

Acids & Bases IX. Ion Product Constant for Water -since _____ is a ______, its _______ is equal to its _______, which is _______ at a ___________ and ____, so the _________ can be simplified by _____ both sides by ______, creating the _______ ___ _____, ___ -at _____, the ____- _____ of _____ produces ____ = _____ and a ______ = _____, so ___ = _______

Acids & Bases IX. Ion Product Constant for Water What is the concentration of the hydroxide ion in an aqueous solution with a hydrogen ion concentration of 1 x 10 -5 M? Is the solution acidic, basic, or neutral? What is the concentration of the hydrogen ion in an aqueous solution with a hydroxide ion concentration of 1 x 10 -3 M? Is the solution acidic, basic, or neutral?

Acids & Bases X. p. H (pondus hydrogenii) -in 1909, ________ developed the ___ (_____) _____ as a way of conveniently expressing the ______ of _____ and _____ What is the p. H of water? What is the p. H of an aqueous solution in which the [H+] = 1. 0 x 10 -2 M?

Acids & Bases X. p. H (pondus hydrogenii) Name________ 1. Hypothesis: What is the relationship between the concentration of an acid and its p. H? 2. Prediction: 3. Gather Data: A. Safety: The acids used in this lab are corrosive and cause irritation and damage to the skin, eyes, and mucous membranes. Avoid contact. Use caution. Goggles mandatory. B. Procedure: Serial Dilution 1. Using a pipette, place 10 drops of 1. 0 M HCl in test well 1 A of a microplate.

Acids & Bases X. p. H (pondus hydrogenii) 3. Gather Data: B. Procedure: 2. Rinse the pipette, and transfer 1 drop of acid solution from test well 1 A to test well 2 A, and add 9 drops of distilled water. 3. Repeat Step 2, transferring 1 drop of acid solution from test well 2 A to test well 3 A and adding 9 drops of distilled water. 4. Repeat Step 2 six more times, each time transferring 1 drop of acid solution from the previous test well and adding 9 drops of distilled water.

Acids & Bases X. p. H (pondus hydrogenii) 3. Gather Data: B. Procedure: 5. Test the p. H in each well with p. H indicator paper. Record. 6. Test the p. H in each well with 1 drop of universal indicator solution. Record. Test Well Number [H+] Concentration p. H paper p. H Universal Indicator A 1 A 2 A 3 A 4 A 5 A 6 B 1 B 2

Acids & Bases X. p. H (pondus hydrogenii) 4. Analyze Data: A. What is [H+] in test well A 4? ________. B. What is [H+] in test well A 6? ________. C. What is theoretical p. H in test well A 2? ________. D. What is theoretical p. H in test well B 2? ________. E. The color of p. H paper in test well A 1 was _______, indicating an approximate p. H of _______. F. In test well A 2 universal indicator was ____ in color. 5. Draw Conclusions: A. Each step in the serial dilution ______ the concentration of the hydrogen ion by a factor of _______ and _____ the p. H by a factor of _______

Acids & Bases XI. p. OH (potential Hydroxide) -the _______, or ____, of a _______ can be expressed by the ____, or _________: What is the p. OH of water? What is the p. H of an aqueous solution in which the [OH-] = 1. 0 x 10 -3 M? What is the p. OH of an aqueous solution in which the [OH-] = 1. 00 x 10 -6 M?

Acids & Bases XI. p. OH (potential Hydroxide) What is the p. H and the p. OH of an aqueous solution with a hydroxide ion concentration of 1. 0 x 10 -6 M? Is the solution acidic, basic, or neutral? What is the p. H and the p. OH of an aqueous solution with a hydroxide ion concentration of 6. 5 x 10 -4 M? Is the solution acidic, basic, or neutral? What is the p. H and the p. OH of an aqueous solution with a hydrogen ion concentration of 3. 6 x 10 -9 M? Is the solution acidic, basic, or neutral?

Acids & Bases XI. p. OH (potential Hydroxide) What is the hydrogen ion concentration and the hydroxide ion concentration of a person’s blood that has a p. H of 7. 40 at 298 K? What is the hydrogen ion concentration and the hydroxide ion concentration of an aqueous solution that has a p. H of 2. 37 at 298 K? What is the hydrogen ion concentration and the hydroxide ion concentration of a person’s blood that has a p. H of 11. 05 at 298 K?

Acids & Bases XII. Calculating p. H of Strong Acids and Strong Bases What is the p. H of a 1. 0 M HI solution? What is the p. H of a 0. 050 M HNO 3 solution? What is the p. OH of a 2. 4 x 10 -5 M Mg(OH)2 solution?

Acids & Bases XIII. Calculating p. H of Weak Acids and Weak Bases What is Ka of a 0. 100 M formic (methanoic) acid solution with p. H 2. 38?

Acids & Bases XIII. Calculating p. H of Weak Acids and Weak Bases What is Ka of a 0. 220 M arsenic acid solution with p. H 1. 50?

Acids & Bases XIII. Calculating p. H of Weak Acids and Weak Bases What is Ka of a 0. 0400 M chlorous acid solution with p. H 1. 80?

Acids & Bases XIII. Calculating p. H of Weak Acids and Weak Bases What is Ka of a 1. 000 M propanoic acid solution with p. H 2. 43?

Acids & Bases XIV. Neutralization -when an ____ and a _____, the resulting _______ has _____ that are characteristic of _______ an ____ or a ____ -a _______ is a _________ in which an ____ and a ____ react in ________ to produce a ____ and _____ -a ____ is an _________ made up of the ______ from the ____ and the _____ from the ____ -the _____ for a _______ is:

Acids & Bases XIV. Neutralization Write the complete balanced thermochemical equations for the following neutralization reactions and name the products: Nitric acid + Cesium hydroxide Hydrobromic acid + Calcium hydroxide Sulfuric acid + Potassium hydroxide
Acids & Bases XV. Titration -a _______is a method for determining an ____________ of a solution by _______ a ______ of a _______ solution of ____________ -to find the ___________ of a ______ of ____ solution, you can ______ the solution with a ______ of an ____ solution of ____________

Acids & Bases XV. Titration What is the molarity of a Cs. OH solution if 30. 0 m. L of the solution is neutralized by 26. 4 m. L of 0. 250 M HBr solution? What is the molarity of a nitric acid solution if 20. 00 m. L of the solution is neutralized by 43. 33 m. L of 0. 1000 M KOH solution?

Acids & Bases XV. Titration -during the course of a _______, a known _______ of a _______ of known ______ is added until the _____ is reached, at which the _______ of ____ equals the _______ of _____ -corresponding closely with the _____ is the _____, at which the ____ changes _____
Acids & Bases XVI. Buffered Solutions -_______ are _____ that _____ changes in ___ -a ______ is a _______ of a _____ and its _____
Acids & Bases XVI. Buffered Solutions -a ______ can also be a _______ of a _____ and its _____

Acids & Bases XVI. Buffered Solutions What is the p. H of buffer system made by mixing equal amounts of H 2 PO 4 - and HPO 42 -?
- Slides: 48