Acids Bases I Introduction to Acids Bases Properties

Acids & Bases I. Introduction to Acids & Bases

Properties u BASES u ACIDS u OH- anions u H+ or H 3 O+ cations u Bitter taste u Sour taste u React with metals to u Slippery feel form H 2 gas u p. H > 7 u p. H < 7 u Ex: ammonia, antacid, baking soda u Ex: vinegar, soda, apples, citrus fruits

p. H indicators Indicators are dyes that will change color in the presence of an acid or base. u Some indicators only work in a specific range of p. H u Examples • Phenolphthalein • Bromothymol blue • Methyl Red u

p. H indicators u Some dyes are natural, like radish skin or red cabbage u Litmus paper, p. H paper, or p. H meters can also be used to determine p. H values
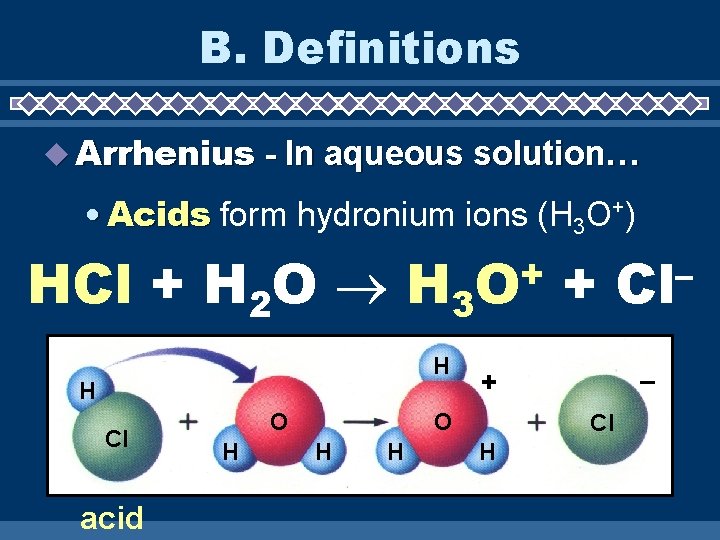
B. Definitions u Arrhenius - In aqueous solution… • Acids form hydronium ions (H 3 O+) HCl + H 2 O H 3 H H Cl acid O H + O H – + O H + Cl H – Cl
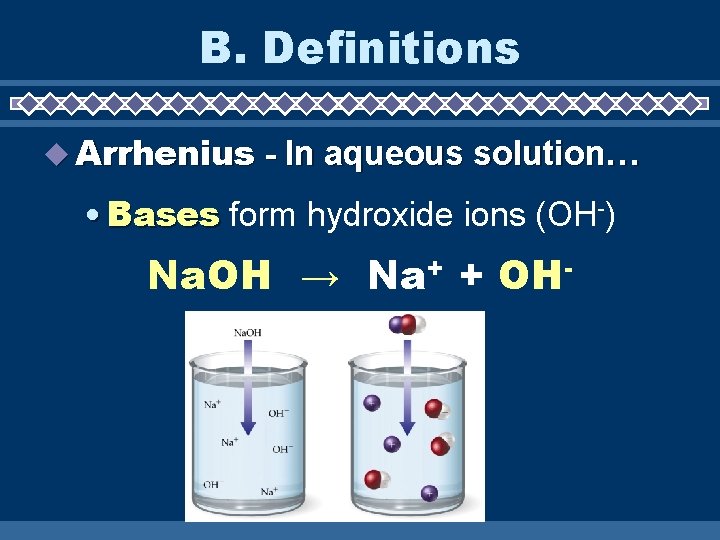
B. Definitions u Arrhenius - In aqueous solution… • Bases form hydroxide ions (OH-) Na. OH → Na+ + OH-
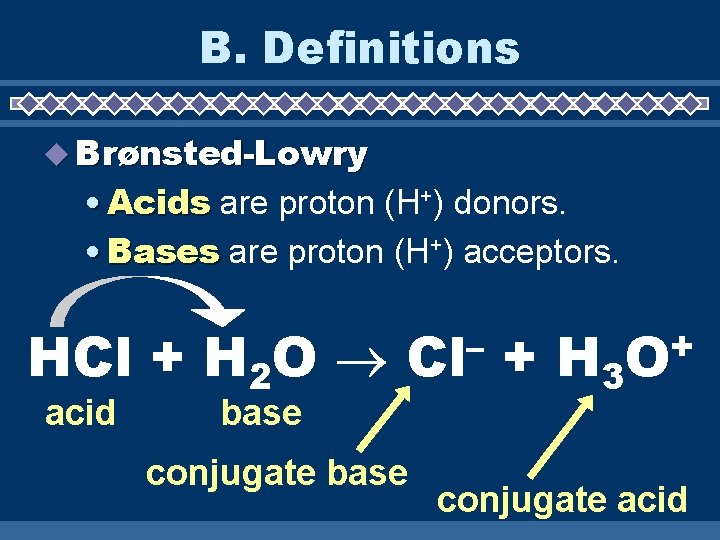
B. Definitions u Brønsted-Lowry • Acids are proton (H+) donors. • Bases are proton (H+) acceptors. HCl + H 2 O acid – Cl base conjugate base + H 3 + O conjugate acid
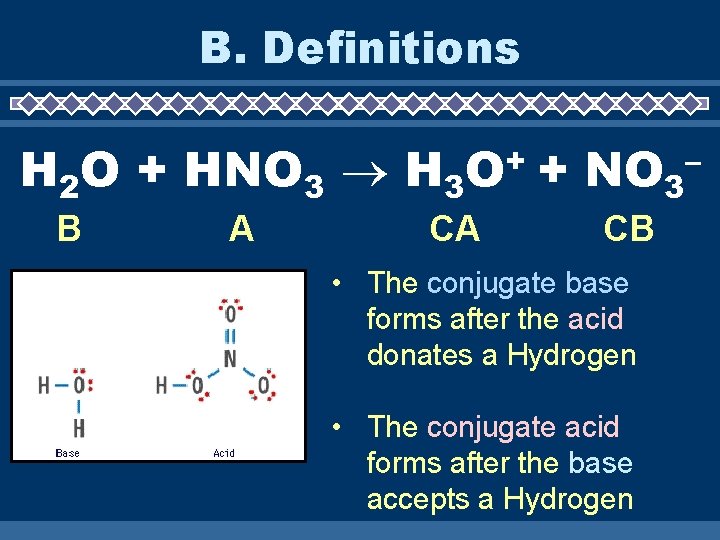
B. Definitions H 2 O + HNO 3 H 3 B A + O CA + NO 3 CB • The conjugate base forms after the acid donates a Hydrogen • The conjugate acid forms after the base accepts a Hydrogen –
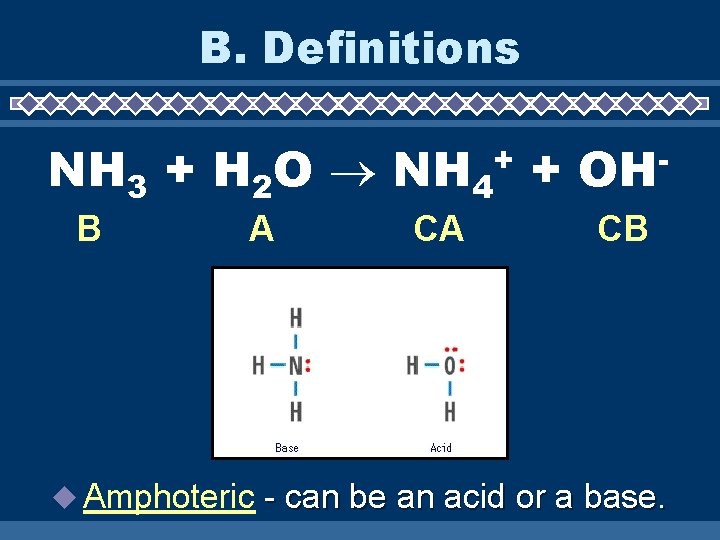
B. Definitions NH 3 + H 2 O NH 4 + + B A u Amphoteric CA OH CB - can be an acid or a base.
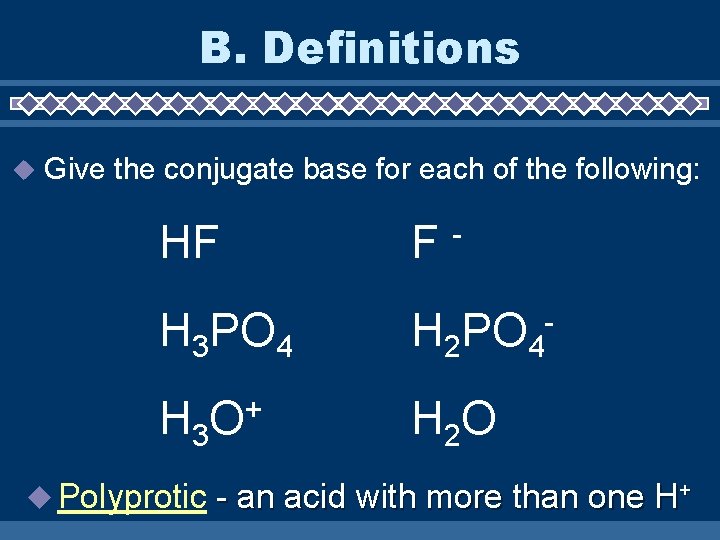
B. Definitions u Give the conjugate base for each of the following: HF F H 3 PO 4 H 2 PO 4 - H 3 O + H 2 O u Polyprotic - - an acid with more than one H+
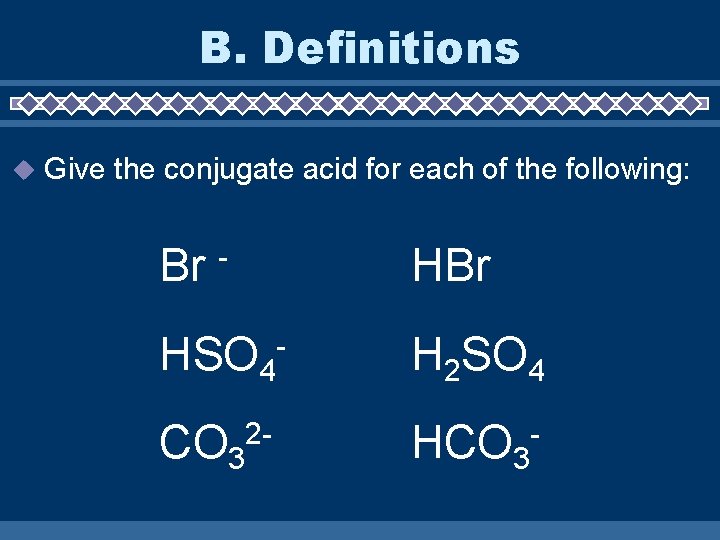
B. Definitions u Give the conjugate acid for each of the following: Br HBr - HSO 4 CO 32 - - H 2 SO 4 HCO 3 -
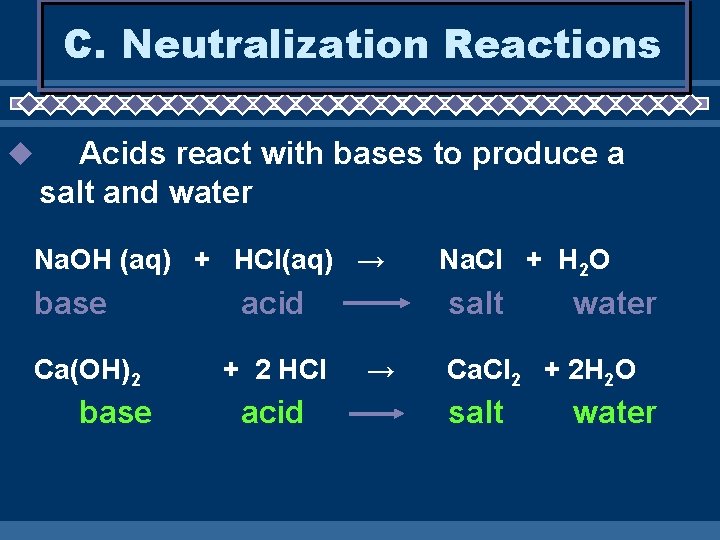
C. Neutralization Reactions u Acids react with bases to produce a salt and water Na. OH (aq) + HCl(aq) → base Ca(OH)2 base acid + 2 HCl acid Na. Cl + H 2 O salt → water Ca. Cl 2 + 2 H 2 O salt water
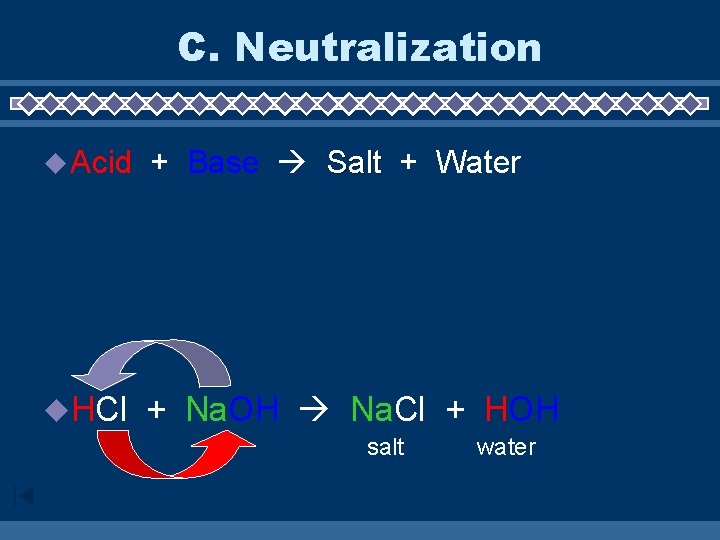
C. Neutralization u Acid + Base Salt + Water u. HCl + Na. OH Na. Cl + HOH salt water

Acids u Acids can be recognized because the start with H u Examples • HCl • H 2 SO 4 • HI

Acids & Bases II. p. H
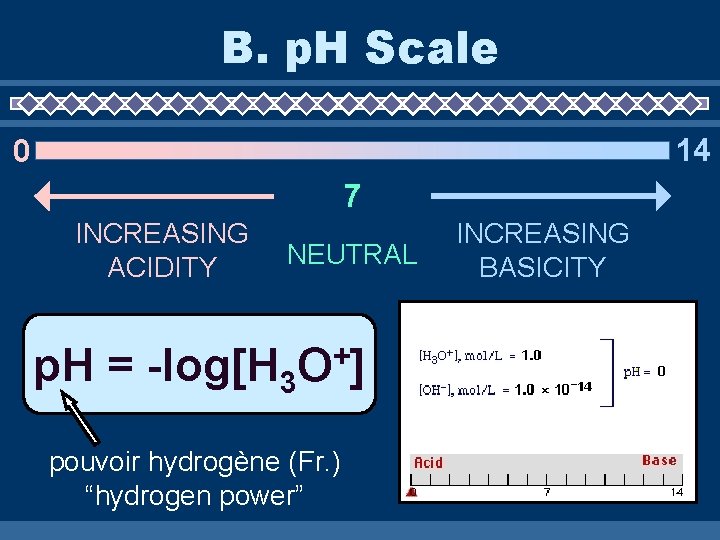
B. p. H Scale 14 0 7 INCREASING ACIDITY NEUTRAL p. H = -log[H 3 + O] pouvoir hydrogène (Fr. ) “hydrogen power” INCREASING BASICITY

B. p. H Scale p. H of Common Substances
![B. p. H Scale + O] p. H = -log[H 3 p. OH = B. p. H Scale + O] p. H = -log[H 3 p. OH =](http://slidetodoc.com/presentation_image_h/2bab306be7d3a0dbcbf088d3c8657631/image-18.jpg)
B. p. H Scale + O] p. H = -log[H 3 p. OH = -log[OH-] p. H + p. OH = 14 [H 3 O+] = 10(-p. H) (-p. OH) [OH ] = 10

B. p. H Scale u What is the p. H of 0. 050 M HNO 3? p. H = -log[H 3 O+] p. H = -log[0. 050] p. H = 1. 3 Acidic or basic? Acidic

B. p. H Scale u What is the molarity of HBr in a solution that has a p. OH of 9. 6? p. H + p. OH = 14 p. H = -log[H 3 O+] 4. 4 = -log[H 3 O+] p. H + 9. 6 = 14 -4. 4 = log[H 3 O+] p. H = 4. 4 Inverse log (–p. H) = Molarity Acidic [H 3 O+] = 4. 0 10 -5 M HBr
- Slides: 20