Acids Bases I Introduction to Acids Bases A

Acids & Bases I. Introduction to Acids & Bases

A. Properties electrolytes sour taste electrolytes turn blue litmus red react with metals to form H 2 gas vinegar, milk, soda, apples, citrus fruits bitter taste turn red litmus blue slippery feel ammonia, lye, antacid, baking soda Chem. ASAP
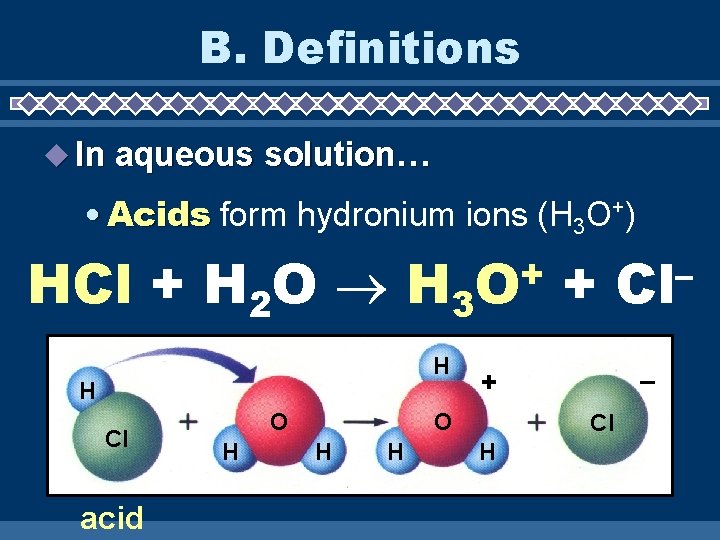
B. Definitions In aqueous solution… • Acids form hydronium ions (H 3 O+) HCl + H 2 O H 3 H H Cl acid O H + O H – + O H + Cl H – Cl
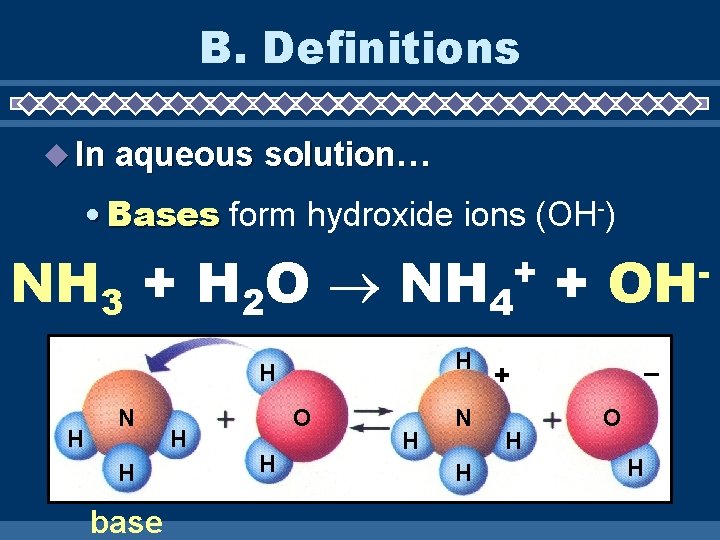
B. Definitions In aqueous solution… • Bases form hydroxide ions (OH-) NH 3 + H 2 O NH 4 + + H H H N H base H O H H N H OH – + H O H
C. Strength Strong Acid/Base - + • 100% ionized in water • strong electrolyte HCl HNO 3 H 2 SO 4 HBr HI HCl. O 4 Na. OH KOH Ca(OH)2 Ba(OH)2
C. Strength Weak Acid/Base - + • does not ionize completely • weak electrolyte HF CH 3 COOH H 3 PO 4 H 2 CO 3 HCN NH 3

Acids & Bases II. p. H
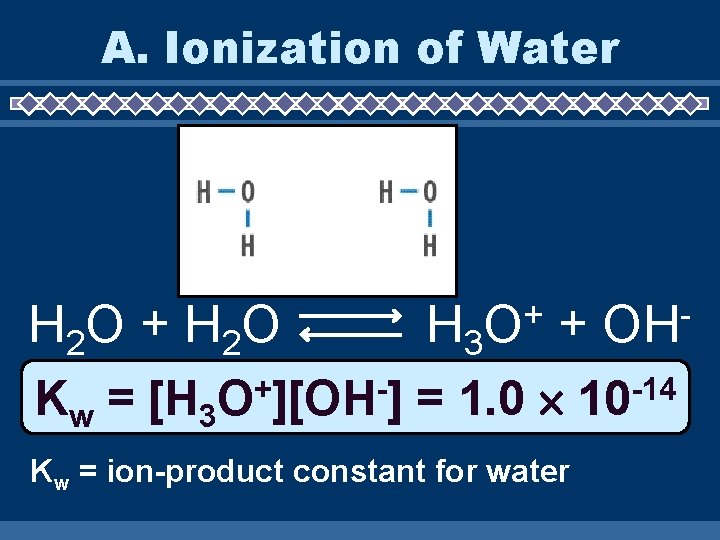
A. Ionization of Water H 2 O + H 2 O H 3 + OH Kw = [H 3 O+][OH-] = 1. 0 10 -14 Kw = ion-product constant for water

A. Ionization of Water Find the hydroxide ion concentration of 3. 0 10 -2 M HCl. [H 3 O+][OH-] = 1. 0 10 -14 [3. 0 10 -2][OH-] = 1. 0 10 -14 [OH-] = 3. 3 10 -13 M Acidic or basic? Acidic
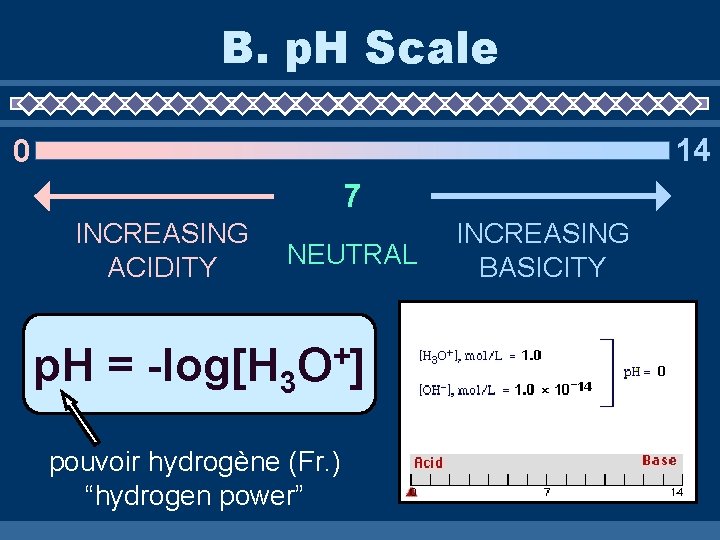
B. p. H Scale 14 0 7 INCREASING ACIDITY NEUTRAL p. H = -log[H 3 + O] pouvoir hydrogène (Fr. ) “hydrogen power” INCREASING BASICITY

B. p. H Scale p. H of Common Substances
![B. p. H Scale p. H = -log[H 3 p. OH = + O] B. p. H Scale p. H = -log[H 3 p. OH = + O]](http://slidetodoc.com/presentation_image_h/2131bc8b3133701be36e480cf389e009/image-12.jpg)
B. p. H Scale p. H = -log[H 3 p. OH = + O] -log[OH ] p. H + p. OH = 14

B. p. H Scale What is the p. H of 0. 050 M HNO 3? p. H = -log[H 3 O+] p. H = -log[0. 050] p. H = 1. 3 Acidic or basic? Acidic

Example p. H (p. OH) calculations A. If you had a sol’n with the concentration of 0. 025 M [H 3 O+], what is the sol’ns p. H? What is the sol’ns p. OH? 1. Given: [H 3 O+] = 0. 025 M Unknown: p. H = ? ? 2. p. H = -log [H 3 O+] = -log[0. 025 M] = 1. 6 3. p. OH = 14 – p. H = 14 - 1. 6 = 12. 4

Example p. H (p. OH) calculations B. What is the p. H of 0. 070 M HNO 3? Is it acidic or basic? 1. p. H = -log[H 3 O+] 2. p. H = -log[0. 070] = 1. 15 3. It is acidic because 1. 15 7.

Example p. H (p. OH) calculations C. What is the p. H of a 0. 100 M HCl sol’n? 1. Given: Identity Concentration: [0. 100 M] Unknown: p. H = ? ? 2. [H 3 O+] = 0. 100 M p. H = -log[0. 100 M] = 1. 00

Example p. H (p. OH) calculations D. What is the p. H of a 1. 5 M sol’n of KOH? (this is a base) 1. Given: [1. 5 M] Unknown: p. H = ? ? 2. p. OH = - log [OH-] p. OH = - log [1. 5] p. OH = -0. 18 3. p. H + p. OH = 14 14 – p. OH = p. H 14 - (-0. 18) = 14. 18 p. H = 14. 18 Check : Answer does indicate that KOH forms a sol’n p. H 7, which is basic.

Acids & Bases III. Titration

A. Neutralization Chemical reaction between an acid and a base. Products are a salt (ionic compound) and water.

A. Neutralization ACID + BASE SALT + WATER HCl + Na. OH Na. Cl + H 2 O strong neutral HC 2 H 3 O 2 + Na. OH Na. C 2 H 3 O 2 + H 2 O weak strong basic • Salts can be neutral, acidic, or basic. • Neutralization does not mean p. H = 7.

B. Titration standard solution • Analytical method in which a standard solution is used to determine the concentration of an unknown solution
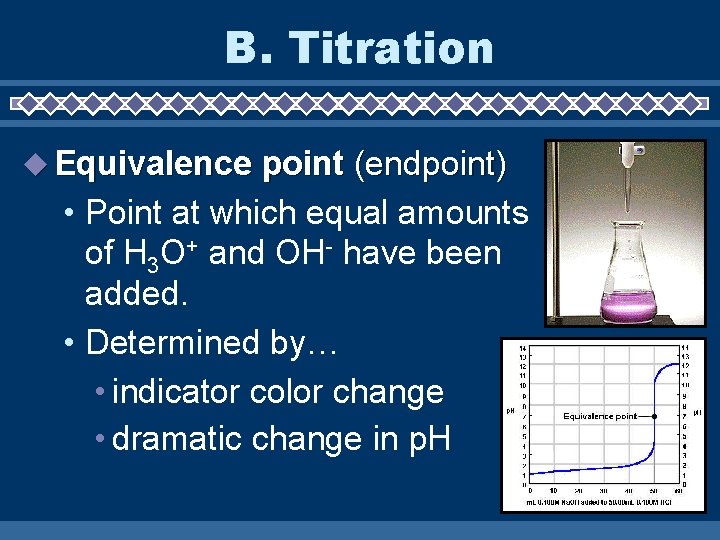
B. Titration Equivalence point (endpoint) • Point at which equal amounts of H 3 O+ and OH- have been added. • Determined by… • indicator color change • dramatic change in p. H

B. Titration + O moles H 3 = moles M V n = M V n M: Molarity V: volume n: # of H+ ions in the acid or OH- ions in the base OH

B. Titration 42. 5 m. L of 1. 3 M KOH are required to neutralize 50. 0 m. L of H 2 SO 4. Find the molarity of H 2 SO 4. H 3 O + M=? V = 50. 0 m. L n=2 OHM = 1. 3 M V = 42. 5 m. L n=1 MV# = MV# M(50. 0 m. L)(2) =(1. 3 M)(42. 5 m. L)(1) M = 0. 55 M H 2 SO 4
- Slides: 24