Acids Bases I Introduction to Acids Bases A

Acids & Bases I. Introduction to Acids & Bases

A. Properties electrolytes sour taste bitter taste turn litmus red turn litmus blue react with metals to form H 2 gas slippery feel vinegar, milk, soda, apples, citrus fruits ammonia, lye, antacid, baking soda electrolytes Chem. ASAP
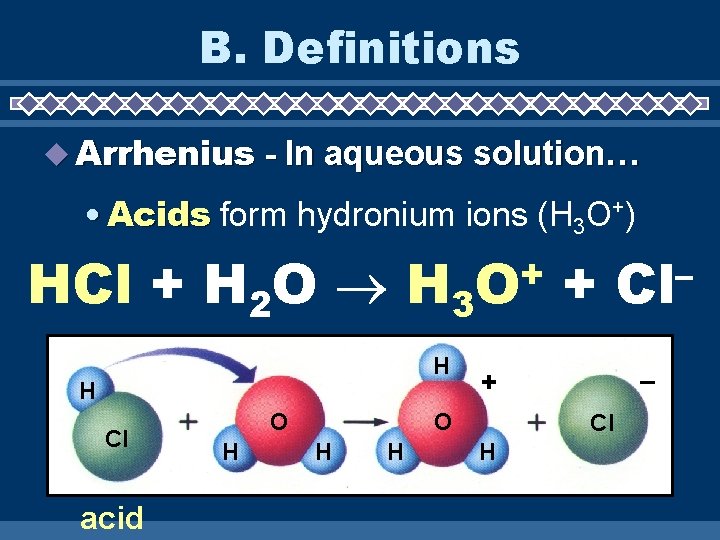
B. Definitions Arrhenius - In aqueous solution… • Acids form hydronium ions (H 3 O+) HCl + H 2 O H 3 H H Cl acid O H + O H – + O H + Cl H – Cl
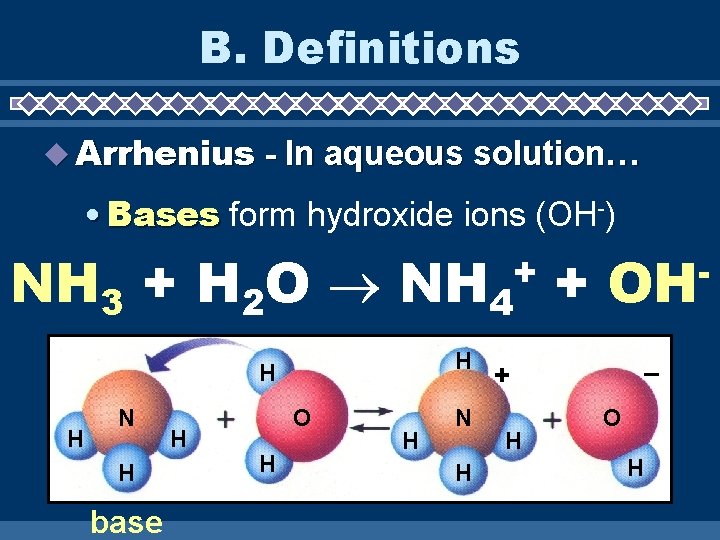
B. Definitions Arrhenius - In aqueous solution… • Bases form hydroxide ions (OH-) NH 3 + H 2 O NH 4 + + H H H N H base H O H H N H OH – + H O H
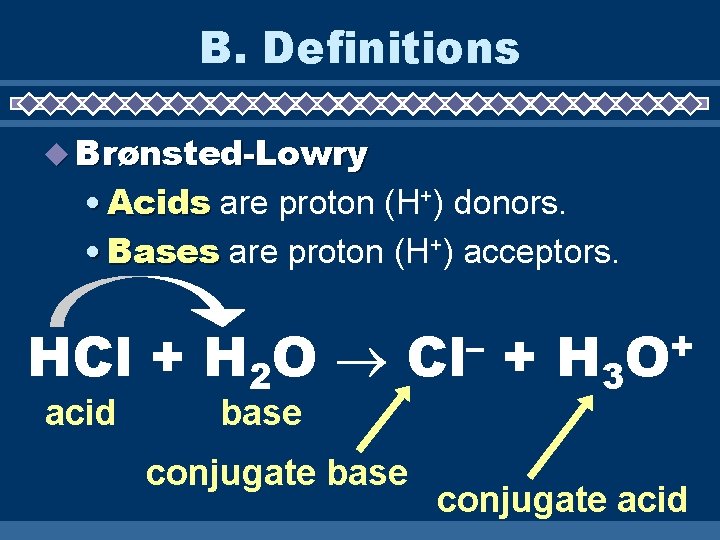
B. Definitions Brønsted-Lowry • Acids are proton (H+) donors. • Bases are proton (H+) acceptors. HCl + H 2 O acid – Cl base conjugate base + H 3 + O conjugate acid
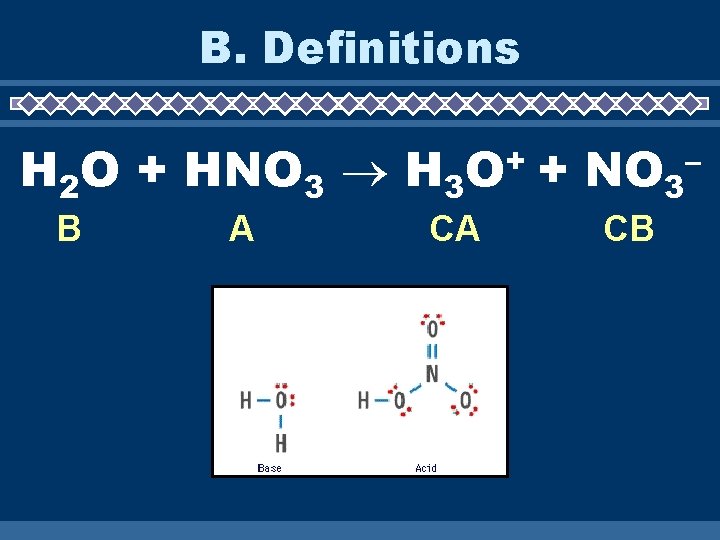
B. Definitions H 2 O + HNO 3 H 3 B A + O CA + NO 3 CB –
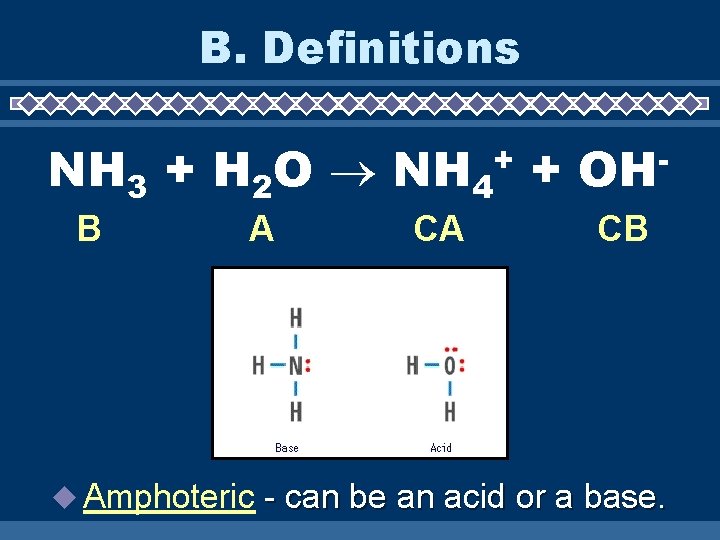
B. Definitions NH 3 + H 2 O NH 4 + + B A Amphoteric CA OH CB - can be an acid or a base.
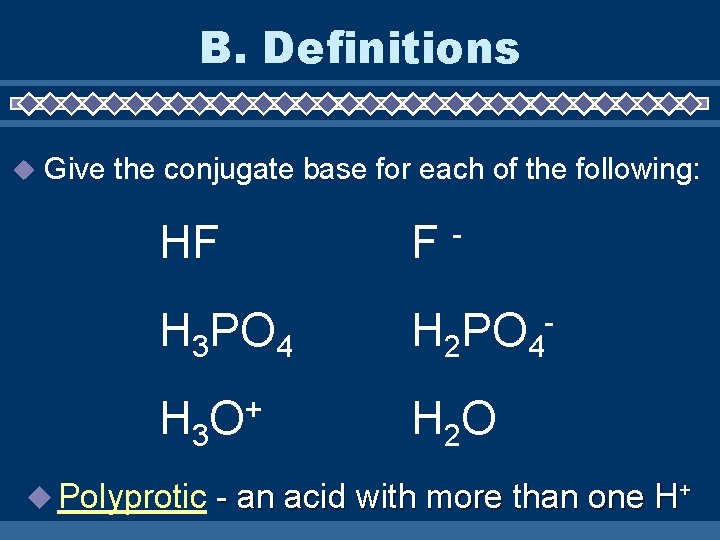
B. Definitions Give the conjugate base for each of the following: HF F H 3 PO 4 H 2 PO 4 - H 3 O + H 2 O Polyprotic - - an acid with more than one H+
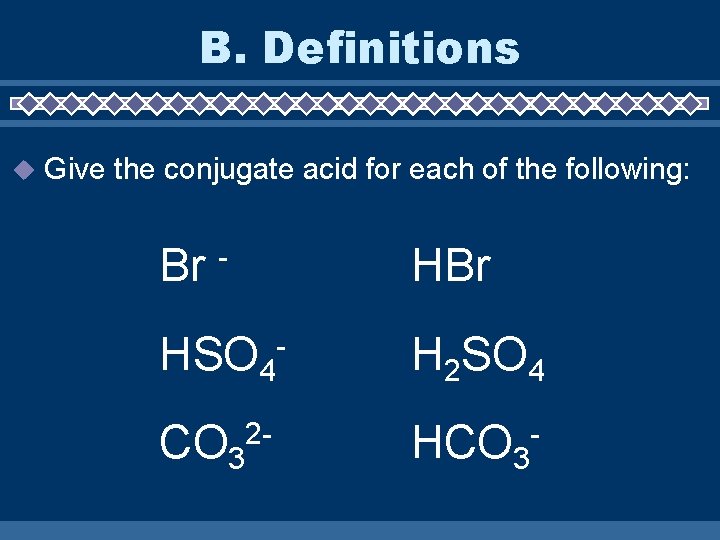
B. Definitions Give the conjugate acid for each of the following: Br HBr - HSO 4 CO 32 - - H 2 SO 4 HCO 3 -
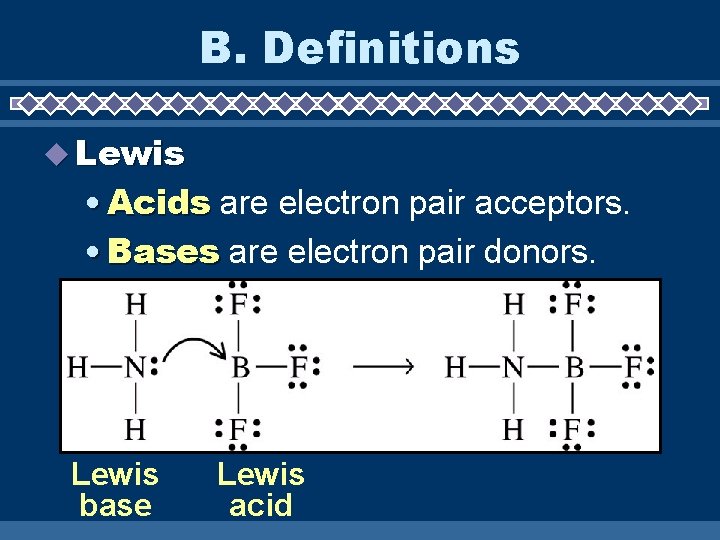
B. Definitions Lewis • Acids are electron pair acceptors. • Bases are electron pair donors. Lewis base Lewis acid
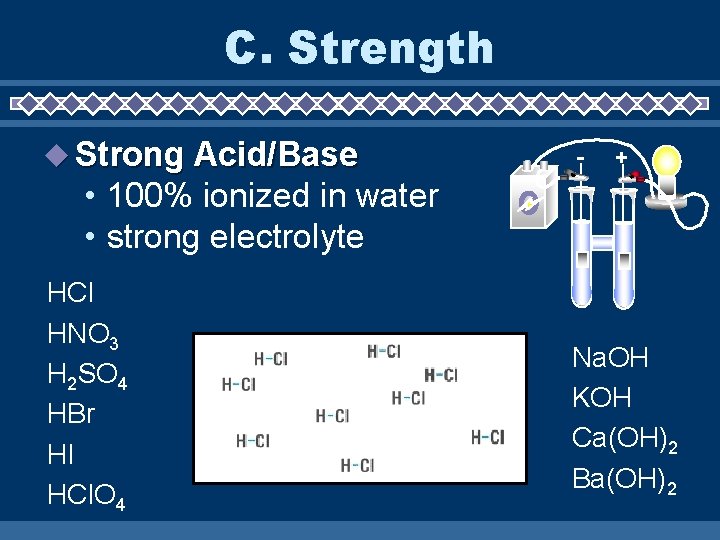
C. Strength Strong Acid/Base - + • 100% ionized in water • strong electrolyte HCl HNO 3 H 2 SO 4 HBr HI HCl. O 4 Na. OH KOH Ca(OH)2 Ba(OH)2
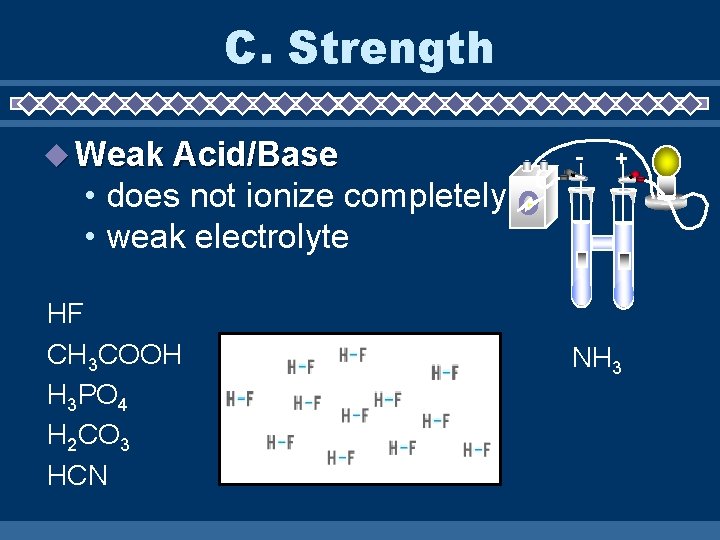
C. Strength Weak Acid/Base - + • does not ionize completely • weak electrolyte HF CH 3 COOH H 3 PO 4 H 2 CO 3 HCN NH 3
- Slides: 12