Acids Bases Edward Wen Ph D Learning Outcomes

Acids & Bases Edward Wen, Ph. D

Learning Outcomes • Properties of acids and bases and definitions • p. H scale and calculation of p. H • Completing and balancing Neutralization reactions • Titration calculations for neutralization reactions • Defining Weak vs. Strong electrolytes (using the concept of equilibrium) • Buffers – recognition of a buffer system, how a buffer works 2

Types of Ionic Compounds • Acids = form H+ ions in water solution • Bases = combine with H+ ions in water solution ü increases the OH- concentration Ø may either directly release OH- or pull H+ off H 2 O • Salts = Ionic compounds formed from Acid and Base. ü all strong electrolytes ü Cation: except H+ ü Anion: except OH 3

Properties of Acids • Sour taste • react with “active” Metals üi. e. Al, Zn, Fe, but NOT w/ Ag, Au Zn + 2 HCl ® Zn. Cl 2 + H 2 • react with Carbonates, Carbonates producing CO 2 ümarble, baking soda, limestone Ca. CO 3 + 2 HCl ® Ca. Cl 2 + CO 2 + H 2 O • change color of vegetable dyes üblue litmus turns red • react with Bases to form ionic salts 4

Most food contains acids • Citric acid: citrus fruits, tomato • Malic acid (HO 2 CCH 2 CHOHCO 2 H): green apple, tomato, grape • Ascorbic acid (aka Vitamin C) • Folic acid 5
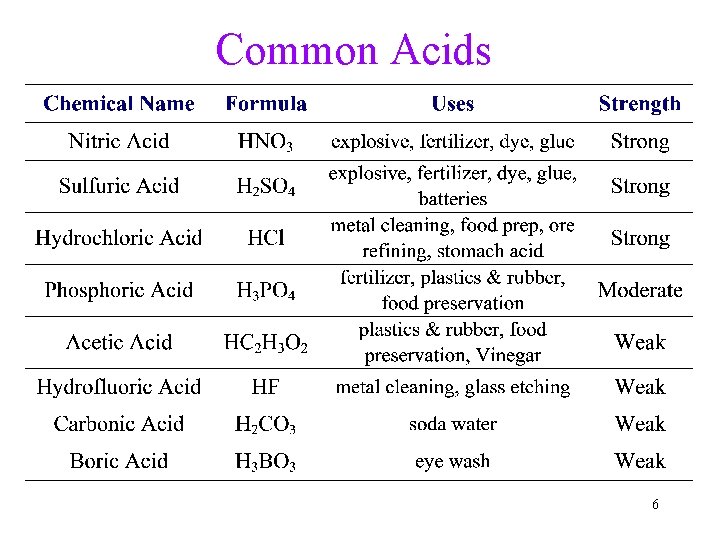
Common Acids 6

Binary acids • (Hm. X): acid hydrogens attached to a nonmetal atom üHCl, HF, HBr, HI üH 2 S, H 2 Se 7
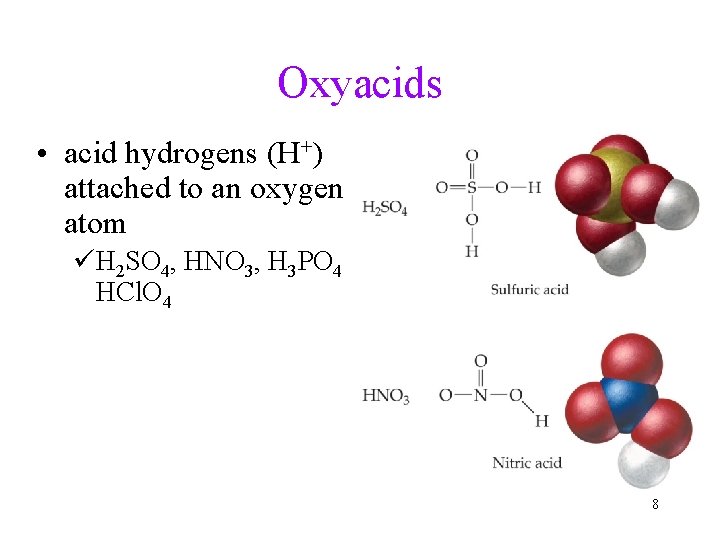
Oxyacids • acid hydrogens (H+) attached to an oxygen atom üH 2 SO 4, HNO 3, H 3 PO 4 HCl. O 4 8
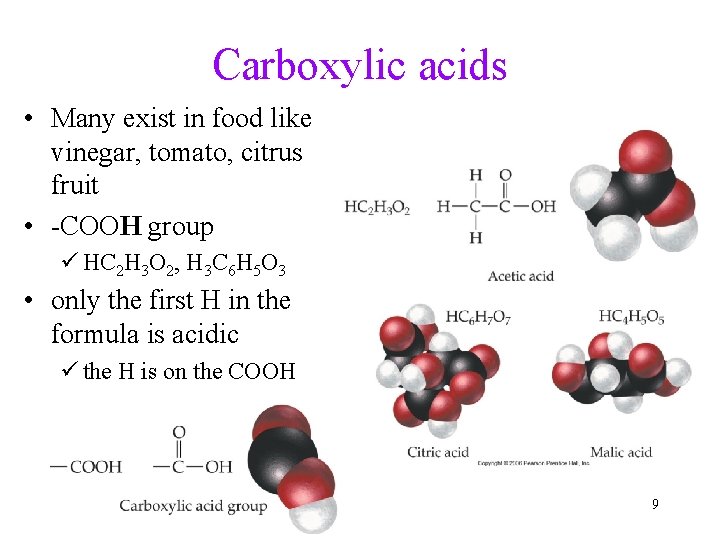
Carboxylic acids • Many exist in food like vinegar, tomato, citrus fruit • -COOH group ü HC 2 H 3 O 2, H 3 C 6 H 5 O 3 • only the first H in the formula is acidic ü the H is on the COOH 9

Properties of Bases • • also known as alkalis taste bitter solutions feel slippery change color of vegetable dyes ü different color than acid ü red litmus turns blue • react with acids to form ionic salts ü neutralization 10
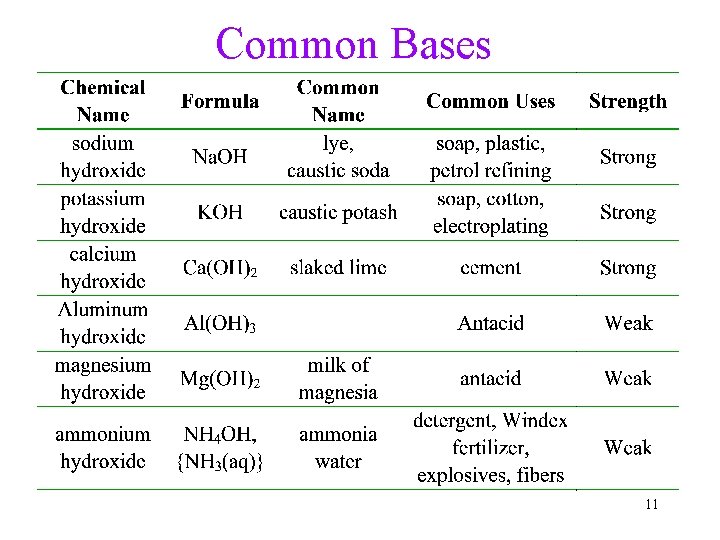
Common Bases 11

Structure of Bases • most ionic bases contain OH- ions ü Drano clog-remover: Na. OH, Ca(OH)2 • some contain CO 32 - ion: it produces OH- with water ü Baking soda: Ca. CO 3 ü Alka-Seltzer: Na. HCO 3 • molecular bases that react with H+ ü Windex: Ammonia (NH 3) 12

Acid-Base Reactions (Neutralization, Double Displacement Reaction) • H+ (from the acid) + OH- (from the base) H 2 O üit is often helpful to think of H 2 O as H-OH • Cation (from base) + Anion (from acid) Salt acid + base → salt + water HCl + Na. OH → Na. Cl + H 2 O 2 HNO 3 + Ba(OH)2 → Ba(NO 3)2 + 2 H 2 O H 2 SO 4 + 2 KOH → K 2 SO 4 + 2 H 2 O 13

Acid Reactions. I. Reaction with Metals • Reaction with many metals: Al, Zn, Fe, Mg übut not all!! Not for Cu, Ag, etc. • Producing a Salt and hydrogen gas H 2 3 H 2 SO 4(aq) + 2 Al(s) → Al 2(SO 4)3(aq) + 3 H 2(g) 14

Acid Reactions. II Reaction with Metal Oxides • when acids react with metal oxides, they produce a salt and water 3 H 2 SO 4 + Al 2 O 3 → Al 2(SO 4)3 + 3 H 2 O • To remove rust from iron/steel, use vinegar or citric acid + Fe 2 O 3 → iron(III) salt + H 2 O 15

Acid Reactions. III Gas-evolving Reaction with Salts + metal carbonate, bicarbonate, sulfide, sulfite, and bisulfite Gas products 2 HNO 3 + Fe. CO 3 → Fe(NO 3)2 + CO 2 + H 2 O HCl + Na. HCO 3 → Na. Cl + CO 2 + H 2 O Zn. S + 2 HBr → Zn. Br 2 + H 2 S Ca. SO 3 + 2 HI → Ca. I 2 + SO 2 + H 2 O H 2 SO 4 + 2 NH 4 HSO 3 → (NH 4)2 SO 4 + 2 SO 2 + 2 H 2 O 16

Base Reactions • Neutralization of acids • Reaction with Nonmetal oxides, CO 2 2 Na. OH + CO 2 → Na 2 CO 3 + H 2 O • Strong bases will react with Al metal to form sodium aluminate and hydrogen gas Example: Dissolving recycled aluminum can with Na. OH solution 2 Na. OH + 2 Al + 6 H 2 O → 2 Na. Al(OH)4 + 3 H 2 17
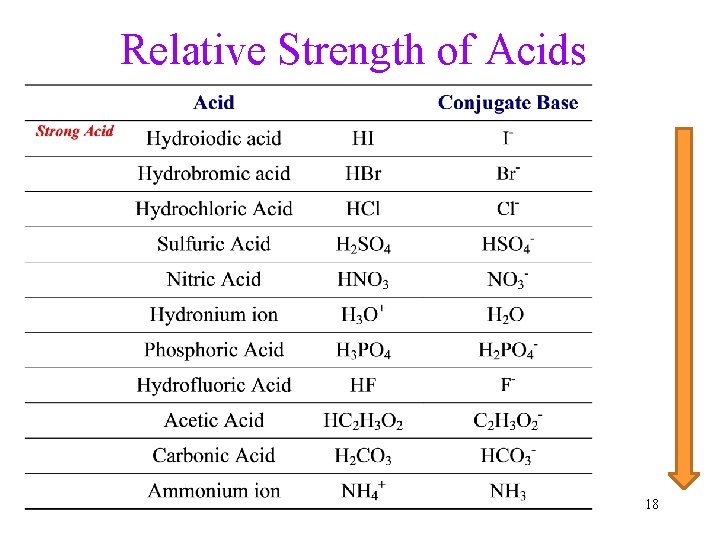
Relative Strength of Acids 18

Strong Acids • The stronger the acid, the more willing it is to donate H+ Stomach acid HCl ® H+ + Cl- ü use water as the standard base • Strong acids donate practically all their H+ ü 100% ionized in water • [H 3 O+] = [strong acid] ü [ ] = molarity 19

Strong Acids Examples: • Binary Acid: HCl, HBr, HI • Oxyacid: HNO 3, H 2 SO 4, HCl. O 4 • Example: HNO 3 = H+ + NO 3 H 2 SO 4 = 2 H+ + SO 42 - 20

Weak Acids • Weak acids donate a small fraction of their H+ Vinegar HC 2 H 3 O 2 Û H+ + C 2 H 3 O 2 - ümost of the weak acid molecules do not donate H+ to water • [H 3 O+] << [weak acid] 21

Weak Acids Examples: • Binary Acid: HF, H 2 Se • Oxyacid: HNO 2, H 2 SO 3, H 3 PO 4, HCl. O • Most carboxylic acids, such as acetic acid 22

Strong Bases • The stronger the base, the more willing it is to accept H+ Drano. TM ü use water as the standard acid Na. OH ® Na+ + OH- • Strong bases: practically all molecules are dissociated into OH– or accept H+ ü 1 mol Na. OH = 1 mol OHü 1 mol Ca(OH)2 = 2 mol OH- 23

Weak Bases • Definition: a small fraction of molecules accept H+ Windex. TM NH 3 + H 2 O Û NH 4+ + OH- ümost of the weak base molecules do not take H+ from water • [HO–] << [weak base] 24

Autoionization of Water • Water: extremely Weak electrolyte • about 2 out of every 1 billion water molecules form Ions: Autoionization H 2 O Û H+ + OH– • ALL aqueous solutions contain both H+ and OH– üthe concentration of H+ and OH– are equal in water ü@ 25°C: [H+] = [OH–] = 10 -7 M 25
![Ion Product of Water • [H+] x [OH–] = constant: Ion Product of water, Ion Product of Water • [H+] x [OH–] = constant: Ion Product of water,](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-26.jpg)
Ion Product of Water • [H+] x [OH–] = constant: Ion Product of water, Kw • At 25°C, [H+] x [OH–] = _____= Kw • as [H+] increases, [OH–] must decrease so the product stays constant 26
![Acidic and Basic Solutions • Neutral solutions have equal [H+] and [OH–] ü[H+] = Acidic and Basic Solutions • Neutral solutions have equal [H+] and [OH–] ü[H+] =](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-27.jpg)
Acidic and Basic Solutions • Neutral solutions have equal [H+] and [OH–] ü[H+] = [OH–] = 1 x 10 -7 M • Acidic solutions : [H+] > [OH–] ü[H+] > 1 x 10 -7 M [OH–] < 1 x 10 -7 M • Basic solutions: [OH–] > [H+] ü[H+] < 1 x 10 -7 M [OH–] > 1 x 10 -7 M 27
![Practice - Determine the [H+] concentration and whether the solution is acidic, basic or Practice - Determine the [H+] concentration and whether the solution is acidic, basic or](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-28.jpg)
Practice - Determine the [H+] concentration and whether the solution is acidic, basic or neutral for the following • All [H+] compared to 1 x 10 -7 M • [OH–] = 3. 50 x 10 -8 M • [Na. OH] = 0. 000250 M • [HCl] = 0. 50 M 2. 86 x 10 -7 M; acidic 4. 00 x 10 -11 M; basic 0. 50 M; acidic 28
![Acidic/Basic: [H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 Acidic/Basic: [H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-29.jpg)
Acidic/Basic: [H+] vs. [OH-] [H+] 100 10 -1 + H OH- Acid 10 -3 10 -5 + H OH- [OH-]10 -14 10 -13 10 -11 10 -9 10 -7 10 -9 Base 10 -11 H+ + H OH OH 10 -7 10 -5 10 -3 10 -14 H+ OH 10 -1 100 even though it may look like it, neither H+ of OH- will ever be 0 the sizes of the H+ and OH- are not to scale because the divisions are powers of 10 rather than units 29

p. H • The measure of the acidity/basicity of a solution • p. H = ______ , • exponent on 10 with a positive sign ü p. Hwater = -log[10 -7] = 7 ü need to know the [H+] concentration to find p. H • p. H = 7 : Neutral; p. H < 7 : Acidic; p. H > 7 : Basic • [H+] = _____ 30

p. H scale • p. H↓, Acidity↑ • p. H↑, basicity↑ ü 1 p. H unit corresponds to a factor of 10 difference in acidity ü • normal range 0 to 14 üp. H 0 is [H+] = 1 M, p. H 14 is [OH–] = 1 M 31

p. H measurement p. H can be measured by p. H meter: • The change in [H+] affects the voltage of a standard cell 32

p. H of Common Substances Substance p. H 1. 0 M HCl 0. 0 0. 1 M HCl 1. 0 stomach acid 1. 0 to 3. 0 lemons 2. 2 to 2. 4 soft drinks 2. 0 to 4. 0 plums 2. 8 to 3. 0 apples 2. 9 to 3. 3 cherries 3. 2 to 4. 0 unpolluted rainwater 5. 6 human blood 7. 3 to 7. 4 egg whites 7. 6 to 8. 0 milk of magnesia (sat’d Mg(OH)2) 10. 5 household ammonia 10. 5 to 11. 5 1. 0 M Na. OH 14 33
![Example - Calculate the p. H of the following solutions • [H+] = 5. Example - Calculate the p. H of the following solutions • [H+] = 5.](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-34.jpg)
Example - Calculate the p. H of the following solutions • [H+] = 5. 2 x 10 -5 M • 0. 0020 M HCl • 0. 010 M Na. OH 4. 3 2. 7 12. 0 34
![Example - Calculate the concentration of [H+] for a solution with p. H 3. Example - Calculate the concentration of [H+] for a solution with p. H 3.](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-35.jpg)
Example - Calculate the concentration of [H+] for a solution with p. H 3. 7 [H+] = 10 -p. H 0. 0002 M 35
![p. H in everyday life Stomach acid Vinegar p. H 0 1 [H+] 100 p. H in everyday life Stomach acid Vinegar p. H 0 1 [H+] 100](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-36.jpg)
p. H in everyday life Stomach acid Vinegar p. H 0 1 [H+] 100 10 -1 + H OH- Acid 3 10 -3 Pure water Windex 5 7 9 10 -5 10 -7 10 -9 + H OH- [OH-]10 -14 10 -13 10 -11 10 -9 + H Base 11 13 10 -11 14 10 -13 10 -14 H+ H+ OH OH OH 10 -7 Drano 10 -5 10 -3 10 -1 100 36

Practice: - Write and Balance equation for the reaction of aqueous nitric acid with aqueous calcium hydroxide 1. Write the formulas of the reactants 2. Determine the ions present for each reactant 3. Exchange the ions, H+ combines with OH- to make H 2 O(l). Tip: write water as HOH 4. Write the formulas of the products 5. Balance the Equation (may be quickly balanced by matching the numbers of H and OH to make HOH) 6. Determine the solubility of the salt 7. Write an (s) after the insoluble products and a (aq) after the soluble products. Water exists as liquid, so (l) for water. 37
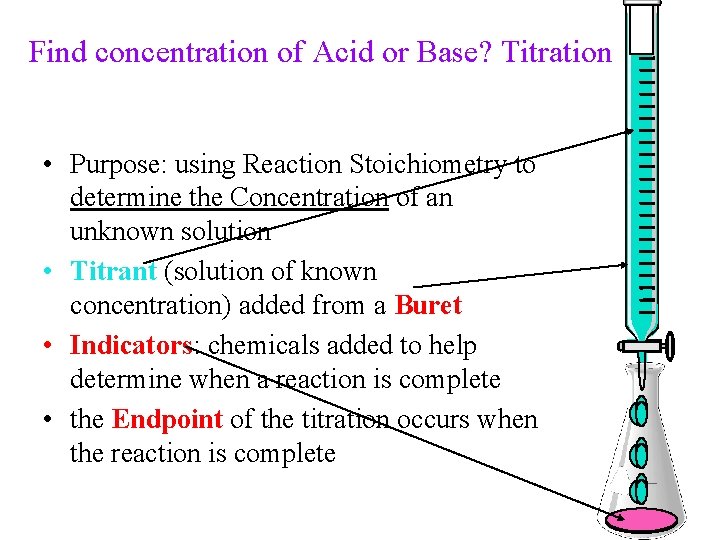
Find concentration of Acid or Base? Titration • Purpose: using Reaction Stoichiometry to determine the Concentration of an unknown solution • Titrant (solution of known concentration) added from a Buret • Indicators: chemicals added to help determine when a reaction is complete • the Endpoint of the titration occurs when the reaction is complete 38

Titration: Color change w/ Indicator 39

Titration Start: The base solution as titrant in the buret. Titrating: As the Base is added to the Acid, H+ + OH– HOH. But still excess Acid present so the color does not change. Endpoint: just enough Base to neutralize all the acid. The indicator changes color. 40

Calculations in Titration • At the Endpoint of the titration, acid base neutralization reaction is complete. The mole ratio between acid and base in the reaction mixture is the same as in the balanced equation. • mole of titrant = Molarity titrant x Volume titrant (L) • mole of the other reactant mole of titrant x mole ratio in the equation (review stoichiometry: mole-tomole). • Finally, the molarity of other reactant = mole reactant divided by volume of the reactant (L)

Example: Acid-Base Titration The titration of 10. 00 m. L of HCl solution of unknown concentration requires 12. 51 m. L of 0. 100 M Ba(OH)2 solution to reach the endpoint. What is the concentration of the unknown HCl solution? First, write balanced equation: 2 mole HCl = 1 mole Ba(OH)2

Example: The titration of 10. 00 m. L of HCl solution of unknown concentration requires 12. 51 m. L of 0. 100 M Ba(OH)2 solution to reach the end point. What is the concentration of the unknown HCl solution? 1. 25 x 10 -3 mol Ba(OH)2 Information Given: 10. 00 m. L HCl 12. 51 m. L Ba(OH)2 Find: M HCl SM: m. L Ba(OH)2 → mol HCl; m. L HCl → L HCl & mol M 2. 50 x 10 -3 mol HCl 0. 250 M HCl 43
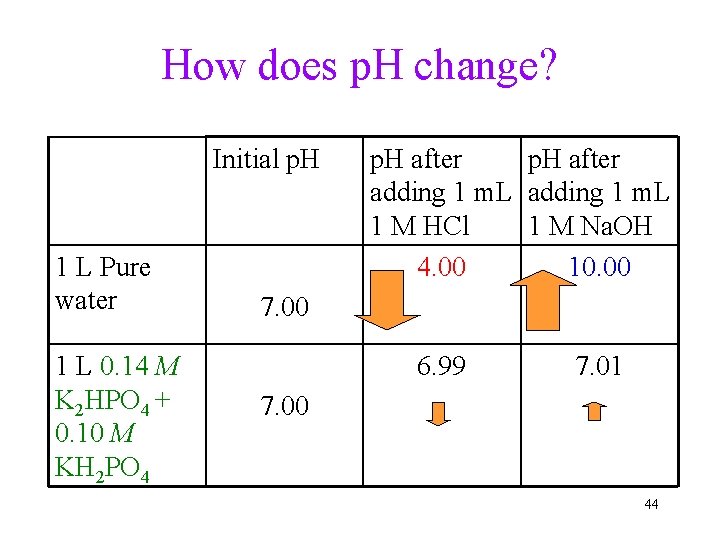
How does p. H change? Initial p. H 1 L Pure water 1 L 0. 14 M K 2 HPO 4 + 0. 10 M KH 2 PO 4 p. H after adding 1 m. L 1 M HCl 1 M Na. OH 4. 00 10. 00 7. 00 6. 99 7. 01 7. 00 44

Buffers • Definition: solutions that resist changing p. H when small amounts of acid or base are added • The mixture of 0. 14 M K 2 HPO 4 + 0. 10 M KH 2 PO 4 solution has much smaller p. H change when strong acid or base is added, thus is called Buffer. • Ingredient: mixing together a weak acid and its conjugate base ü or weak base and it conjugate acid Online demo: https: //www. youtube. com/watch? v=P-R-Cqvb 5 yo • Human body fluid as buffer: H 2 CO 3/HCO 3 - 45
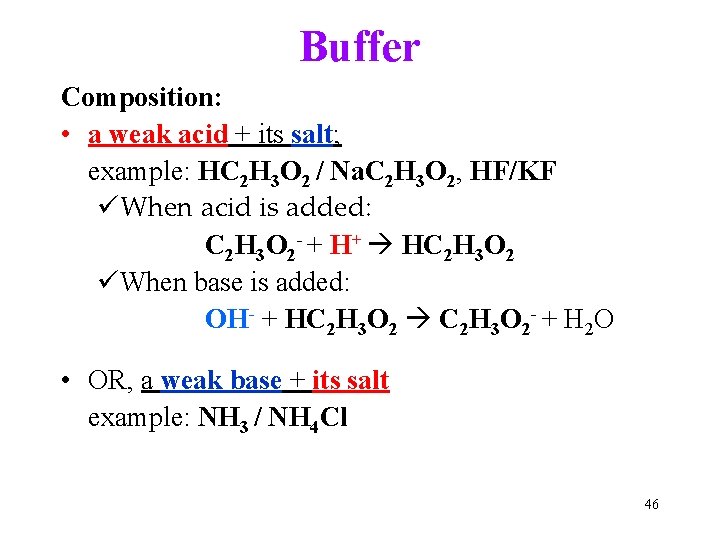
Buffer Composition: • a weak acid + its salt; example: HC 2 H 3 O 2 / Na. C 2 H 3 O 2, HF/KF üWhen acid is added: C 2 H 3 O 2 - + H+ HC 2 H 3 O 2 üWhen base is added: OH- + HC 2 H 3 O 2 C 2 H 3 O 2 - + H 2 O • OR, a weak base + its salt example: NH 3 / NH 4 Cl 46
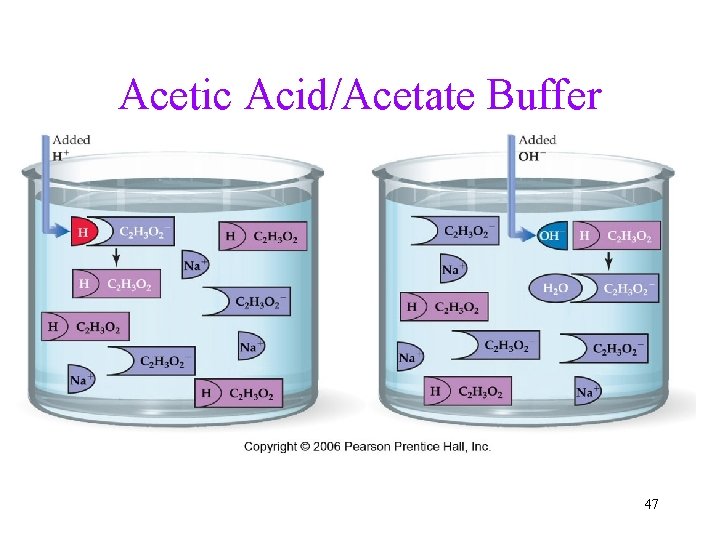
Acetic Acid/Acetate Buffer 47

Treasure Hunt: Which of the following pairs of compounds can combine into a Buffer? a) b) c) d) e) f) g) h) NH 3 /NH 4 Cl HC 2 H 3 O 2/Na. C 2 H 3 O 2 KNO 3/HNO 3 Na 2 SO 4/H 2 SO 4 KOH/H 2 O NH 3 /NH 4 F NH 3 /NH 4 NO 3 KNO 2/HNO 2 a, b, f, h Hint: Which is strong acid? Which is strong base? 48
![Practice - Determine the [H+] concentration and whether the solution is acidic, basic or Practice - Determine the [H+] concentration and whether the solution is acidic, basic or](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-49.jpg)
Practice - Determine the [H+] concentration and whether the solution is acidic, basic or neutral for the following • [OH–] = 3. 50 x 10 -8 M [H+] -14 1 x 10 -7 M = = 2. 86 x 10 3. 50 x 10 -8 [H+] >[OH-], therefore acidic • Na. OH = 0. 000250 M [H+] -14 1 x 10 = = 4. 00 x 10 -11 M [H+] < [OH-], therefore basic 0. 000250 • [HCl] = 0. 50 M [H+] > 1. 0 x 10 -7 M therefore acidic

Example - Calculate the p. H of the following strong acid or base solutions • 0. 0020 M HCl as strong acid, so [H+] = 0. 0020 M p. H = - log (2. 0 x 10 -3) = 2. 7 • 0. 010 M Na. OH as strong base, so [OH-] = 0. 010 M [H+] = 1 x 10 -14= 1 x 10 -12 M 1 x 10 -2 p. H = - log (1. 0 x 10 -12) = 12 50
![Example - Calculate the concentration of [H+] for a solution with p. H 3. Example - Calculate the concentration of [H+] for a solution with p. H 3.](http://slidetodoc.com/presentation_image_h2/8d9e8016bfb09d4b4e09d01c3a7c656a/image-51.jpg)
Example - Calculate the concentration of [H+] for a solution with p. H 3. 7 [H+] = 10 -p. H [H+] = 10 -3. 7 = 2 x 10 -4 M = 0. 0002 M 51

Example: Acid-Base Titration The titration of 10. 00 m. L of HCl solution of unknown concentration requires 12. 51 m. L of 0. 100 M Ba(OH)2 solution to reach the endpoint. What is the concentration of the unknown HCl solution? First, write balanced equation: 2 HCl(aq) + Ba(OH)2(aq) → Ba. Cl 2 (aq) + 2 H 2 O(l) 2 mole HCl = 1 mole Ba(OH)2

Example: The titration of 10. 00 m. L of HCl solution of unknown concentration requires 12. 54 m. L of 0. 100 M Ba(OH)2 solution to reach the end point. What is the concentration of the unknown HCl solution? Given: 10. 00 m. L HCl 12. 54 m. L 0. 100 M Ba(OH)2 Find: M HCl 0. 100 M Ba(OH)2 0. 100 mol Ba(OH)2 1 L sol’n 53
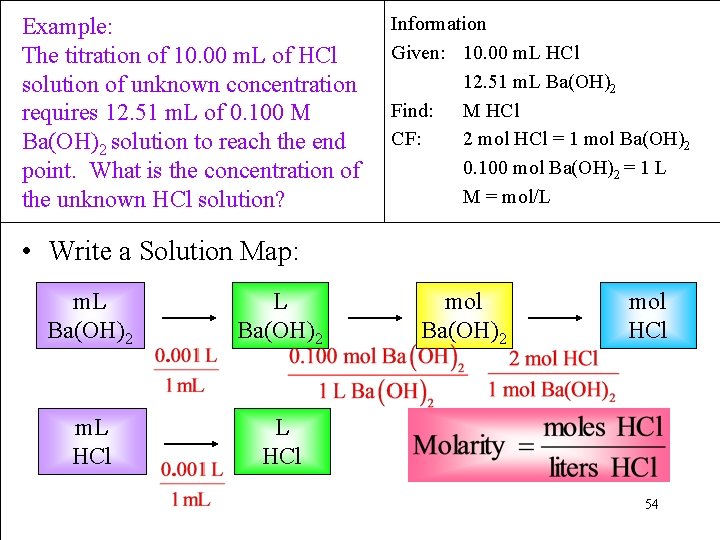
Example: The titration of 10. 00 m. L of HCl solution of unknown concentration requires 12. 51 m. L of 0. 100 M Ba(OH)2 solution to reach the end point. What is the concentration of the unknown HCl solution? Information Given: 10. 00 m. L HCl 12. 51 m. L Ba(OH)2 Find: M HCl CF: 2 mol HCl = 1 mol Ba(OH)2 0. 100 mol Ba(OH)2 = 1 L M = mol/L • Write a Solution Map: m. L Ba(OH)2 m. L HCl mol Ba(OH)2 mol HCl 54

Example: The titration of 10. 00 m. L of HCl solution of unknown concentration requires 12. 51 m. L of 0. 100 M Ba(OH)2 solution to reach the end point. What is the concentration of the unknown HCl solution? 1. 25 x 10 -3 mol Ba(OH)2 Information Given: 10. 00 m. L HCl 12. 51 m. L Ba(OH)2 Find: M HCl SM: m. L Ba(OH)2 → mol HCl; m. L HCl → L HCl & mol M 2. 50 x 10 -3 mol HCl 0. 250 M HCl 55
- Slides: 55