Acids Bases Chemistry Ms Siddall Standard 5 a
Acids & Bases Chemistry. Ms. Siddall.
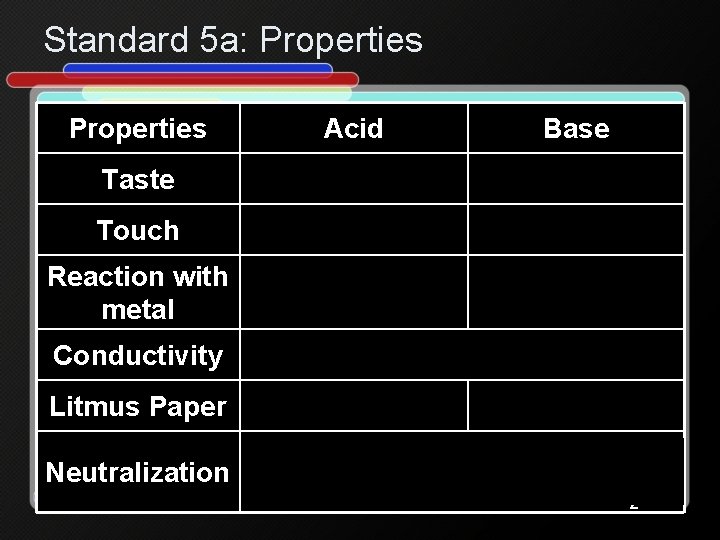
Standard 5 a: Properties Acid Base Taste Sour (lemon) Bitter (soap) Touch Like water Like soap Reaction with metal e. x. HCl + Mg HVigorous 2(g) + Mg. Cl 2 No reaction Conductivity Litmus Paper Neutralization Conducts electricity Blue red Red blue Acid + base salt + water e. x. HCl + Na. OH Na. Cl + H 2 O

Summary 1 1. Write the balanced equation for the reaction of aluminum with HCl to form hydrogen and aluminum chloride. 2. A substance turns red litmus blue and does not react with metal. What is it?

Standard 5 b: Brønsted-Lowry Definition • Acids donate hydrogen ions (Hydrogen ion = H+ = proton) e. x. HBr + H 2 O H 3 O+ + Br- • Bases accept hydrogen ions e. x. NH 3 + H 2 O NH 4+ + OH-

Summary 2 • Write the balanced equation for the reaction of HF (hydrofluoric acid) with water.

Strong acid ionization weak acid ionization
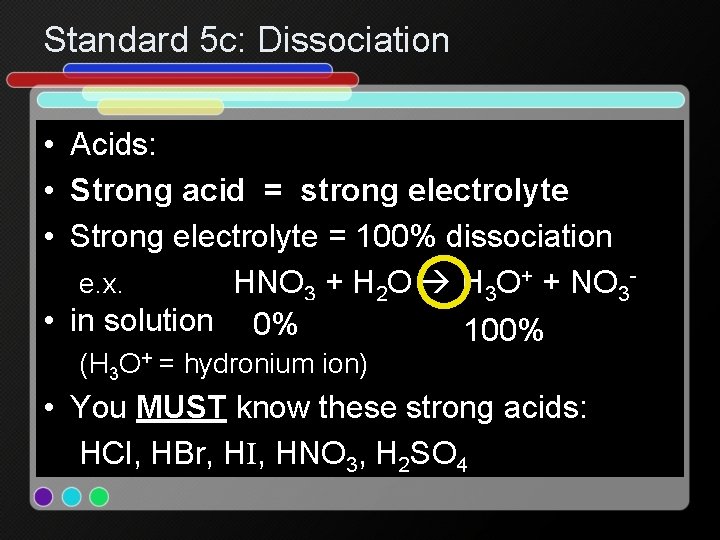
Standard 5 c: Dissociation • Acids: • Strong acid = strong electrolyte • Strong electrolyte = 100% dissociation e. x. HNO 3 + H 2 O H 3 O+ + NO 3 • in solution 0% 100% (H 3 O+ = hydronium ion) • You MUST know these strong acids: HCl, HBr, HI, HNO 3, H 2 SO 4
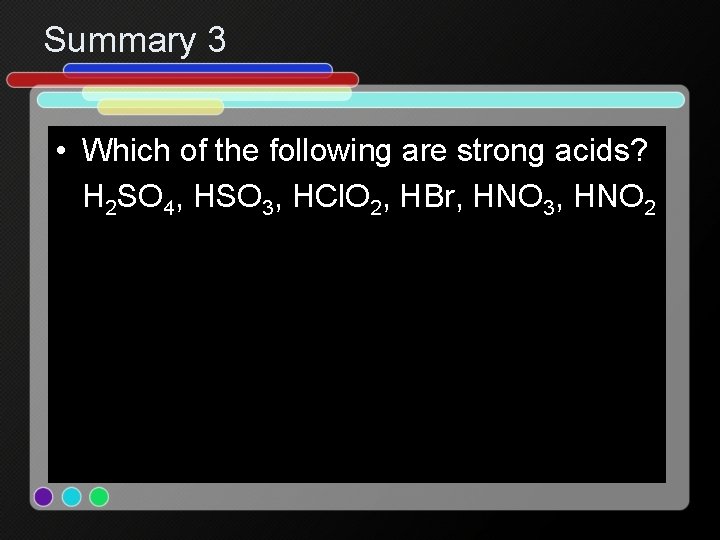
Summary 3 • Which of the following are strong acids? H 2 SO 4, HSO 3, HCl. O 2, HBr, HNO 3, HNO 2

Standard 5 c: continued • Weak acids = weak electrolytes • Weak electrolyte = Partial dissociation (= equilibrium) e. x. HCN + H 2 O H 3 O+ + CN • In solution ~97% ~3% • All other acids are weak acids • Examples: acetic acid HC 2 H 3 O 2, carbonic acid H 2 CO 3, HCN, H 3 PO 4

Summary 4 • At equilibrium does a weak acid solution contain mostly products or mostly reactants? • At equilibrium does a strong acid solution contain mostly products or mostly reactants?

Standard 5 c: continued Conjugate acids & bases: e. x. HF + H 2 O H 3 O+ + F • acid A B CA – Donates hydrogen ion • Conjugate base – Different from acid by a proton • Base – Accepts hydrogen ion • Conjugate acid – Different from base by a proton CB

Summary 5 • Label the acid (A), base (B), conjugate acid (ca), and conjugate base (cb) in the following reaction: HNO 3 + H 2 O H 3 O+ + NO 3 -
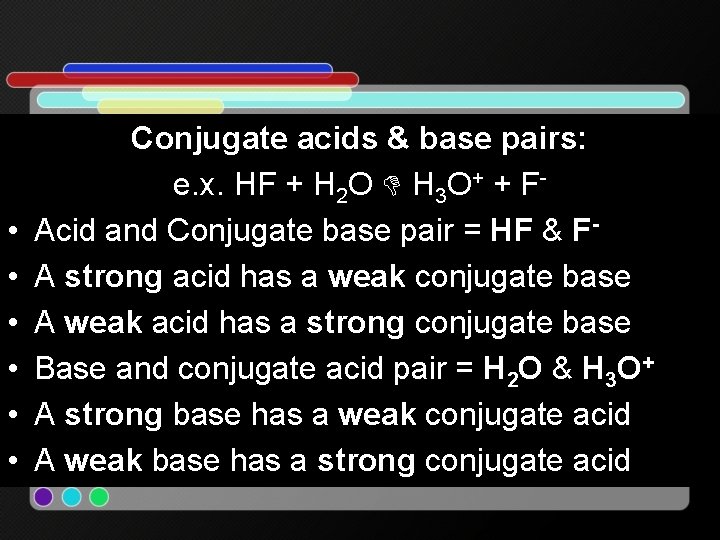
• • • Conjugate acids & base pairs: e. x. HF + H 2 O H 3 O+ + FAcid and Conjugate base pair = HF & FA strong acid has a weak conjugate base A weak acid has a strong conjugate base Base and conjugate acid pair = H 2 O & H 3 O+ A strong base has a weak conjugate acid A weak base has a strong conjugate acid

Summary 6 HF + H 2 O H 3 O+ + F 1. Identify the acid and conjugate base. Label each as ‘weak’ or ‘strong’ 2. Identify the base and conjugate acid. Label each as ‘weak’ or ‘strong’
![Standard 5 d: p. H scale Definition: p. H = -log[H 3 O+] • Standard 5 d: p. H scale Definition: p. H = -log[H 3 O+] •](http://slidetodoc.com/presentation_image/b79c2fcf59a8a42a8c7a6a2a57aefd26/image-15.jpg)
Standard 5 d: p. H scale Definition: p. H = -log[H 3 O+] • p. H measures the concentration (amount) of H 3 O+ ions in solution

Summary 7 1. Write the equation for HCl reacting with water to make a hydronium ion and a chloride ion 2. Is the concentration of hydronium ions in this solution high or low?
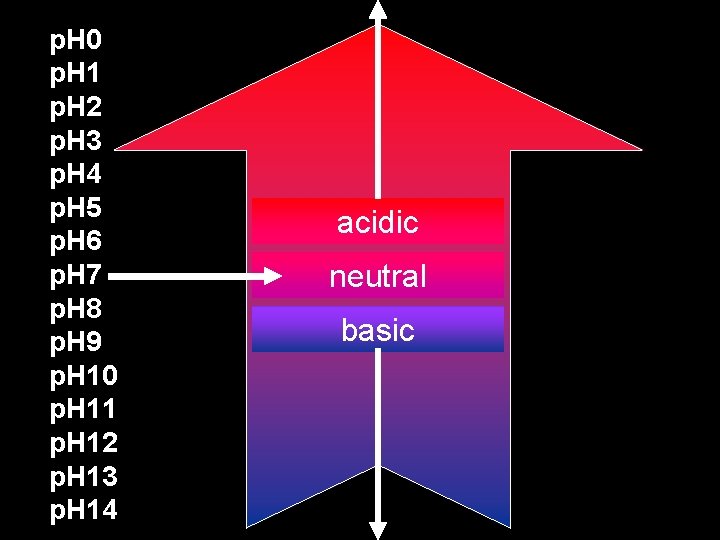
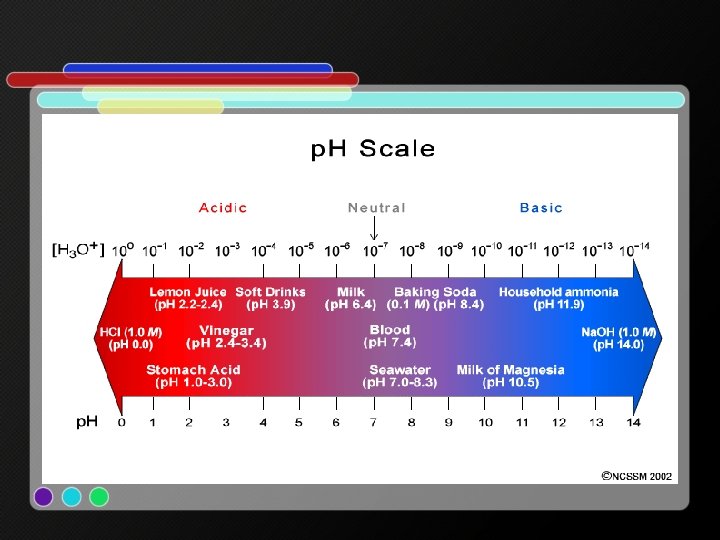
p. H 0 p. H 1 p. H 2 p. H 3 p. H 4 p. H 5 p. H 6 p. H 7 p. H 8 p. H 9 p. H 10 p. H 11 p. H 12 p. H 13 p. H 14 acidic neutral basic
Summary 8 Provide an example of: 1. An acidic compound 2. A basic compound 3. A neutral compound Add a minimum of 3 acids and 3 bases to your p. H scale
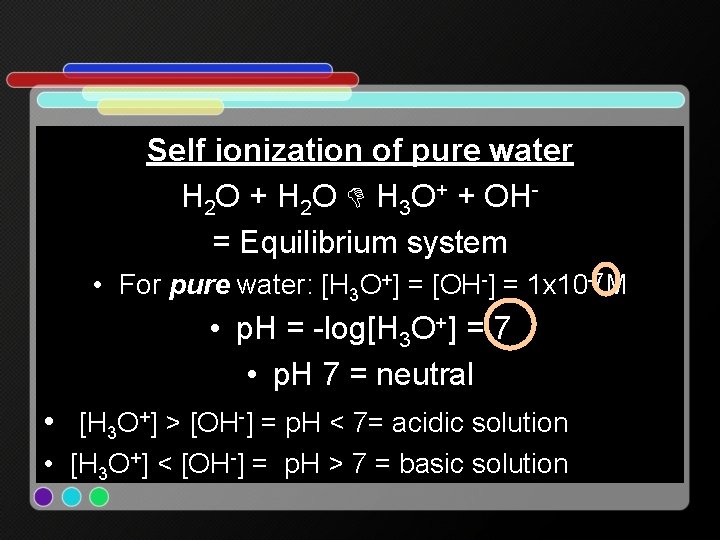
Self ionization of pure water H 2 O + H 2 O H 3 O+ + OH= Equilibrium system • For pure water: [H 3 O+] = [OH-] = 1 x 10 -7 M • p. H = -log[H 3 O+] = 7 • p. H 7 = neutral • [H 3 O+] > [OH-] = p. H < 7= acidic solution • [H 3 O+] < [OH-] = p. H > 7 = basic solution
Summary 9 Identify the following solutions as acidic, basic, or neutral: 1. p. H 3 2. p. H 5 3. p. H 10 4. p. H 7

Acids ‘produce’ H 3 O+ in solution e. x. HCl + H 2 O H 3 O+ + Cl- • p. H measurement: [H 3 O+] = 0. 1 M = 1 x 10 -1 M p. H = -log [H 3 O+] = -log [1 x 10 -1] p. H = 1 • High [H 3 O+] = ‘Low’ p. H reading = acidic solution

Summary 10 1. p. H measures the concentration of __________ ions in solution. 2. A solution with p. H = 2: a) has a high / low concentration of these ions b) is acidic / basic
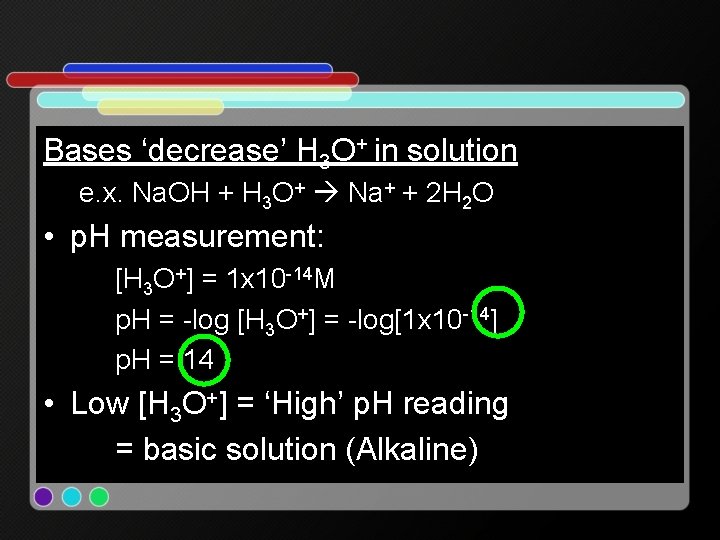
Bases ‘decrease’ H 3 O+ in solution e. x. Na. OH + H 3 O+ Na+ + 2 H 2 O • p. H measurement: [H 3 O+] = 1 x 10 -14 M p. H = -log [H 3 O+] = -log[1 x 10 -14] p. H = 14 • Low [H 3 O+] = ‘High’ p. H reading = basic solution (Alkaline)
Summary 11 1. What is the hydronium ion concentration for a solution with p. H = 12? 2. Is the solution acidic or basic?
![5 f: calculate p. H = -log[H 3 O+] Example: [H 3 O+] = 5 f: calculate p. H = -log[H 3 O+] Example: [H 3 O+] =](http://slidetodoc.com/presentation_image/b79c2fcf59a8a42a8c7a6a2a57aefd26/image-26.jpg)
5 f: calculate p. H = -log[H 3 O+] Example: [H 3 O+] = 4. 5 x 10 -5 M • [H 3 O+] > 1 x 10 -5 M • p. H should be between 4 - 5 • p. H = -log[4. 5 x 10 -5] = 4. 35
Summary 13 • Estimate then calculate the p. H for a solution with: • [H 3 O+] = 7 x 10 -2 M • [H 3 O+] = 2. 2 x 10 -9 M
![Relationship between [OH-] and [H 3 O+] p. OH = -log[OH-] p. OH + Relationship between [OH-] and [H 3 O+] p. OH = -log[OH-] p. OH +](http://slidetodoc.com/presentation_image/b79c2fcf59a8a42a8c7a6a2a57aefd26/image-28.jpg)
Relationship between [OH-] and [H 3 O+] p. OH = -log[OH-] p. OH + p. H = 14 Example: • [OH-] = 1 x 10 -2 M • p. OH = -log[1 x 10 -2] = 2 • p. H = 14 - 2 = 12
![Summary 14 Complete the following table: [H 3 O+] [OH-] p. H p. OH Summary 14 Complete the following table: [H 3 O+] [OH-] p. H p. OH](http://slidetodoc.com/presentation_image/b79c2fcf59a8a42a8c7a6a2a57aefd26/image-29.jpg)
Summary 14 Complete the following table: [H 3 O+] [OH-] p. H p. OH 10 -5 M 1

5 e: acid/base definitions Arrhenius: • acids are hydrogen containing compounds that ionize to yield H+ ions in aqueous solution. e. x. HCl(aq) H+(aq) + Cl-(aq) • Bases ionize to yield OH- ions in aqueous solutions. e. x. Na. OH(aq) Na+(aq) + OH-(aq) • Problems: NH 3 is not a base according to Arrhenius
Summary 15 • Explain why NH 3 is not a base according to the Arrhenius definition of a base.
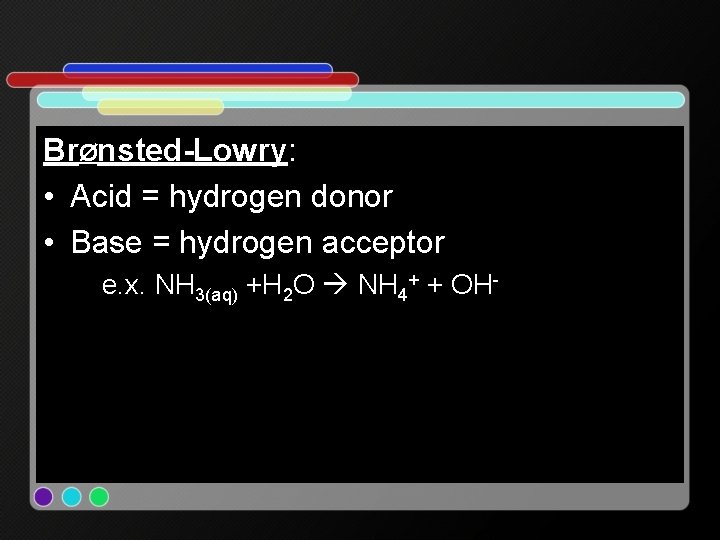
BrØnsted-Lowry: • Acid = hydrogen donor • Base = hydrogen acceptor e. x. NH 3(aq) +H 2 O NH 4+ + OH-

Lewis: • An acid accepts a pair of electrons (accepts a negative charge) • Acid = proton donor = electron acceptor • A base donates a pair of electrons • Base = proton acceptor = electron donor o e. x. HCl + H 2 O H 3 O+ + Cl-
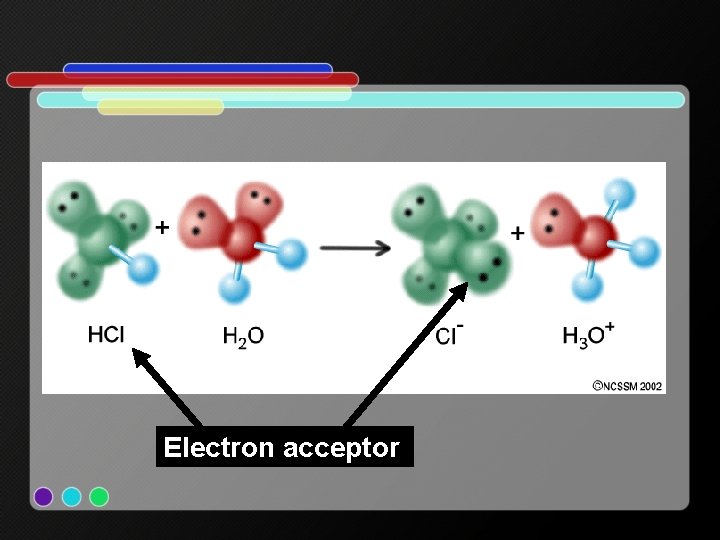
Electron acceptor
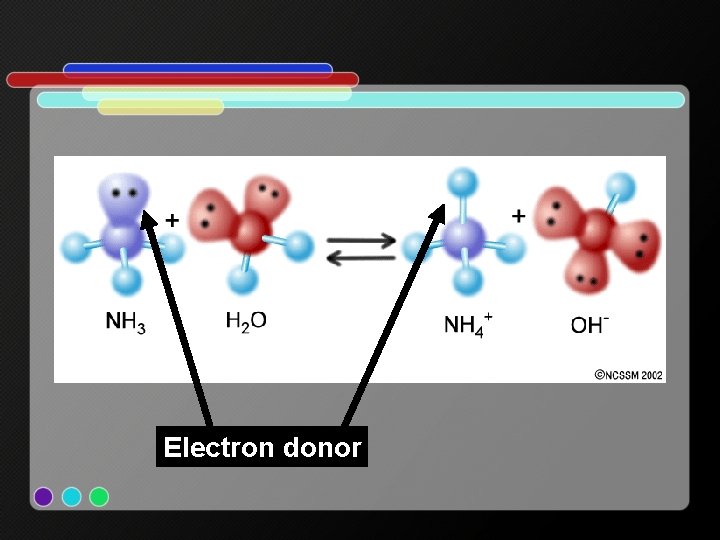
Electron donor

Summary 16 • Explain the difference between the Bronsted-Lowry definition and the Lewis definition of acids and bases
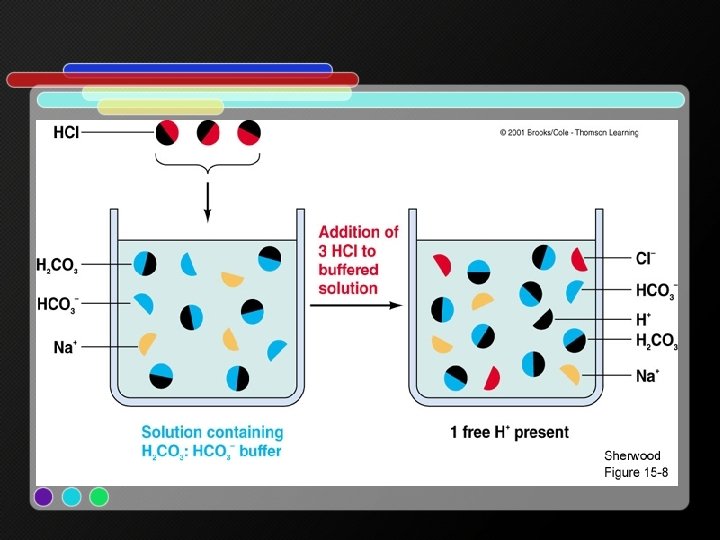
5 g: buffers Buffer = a solution whose p. H does not change (much) when acid or base are added. • A buffer is a solution made from an acid and its conjugate base

• Example: human blood must have a p. H between 7. 35 – 7. 45 (or we will die) • There are 2 buffer systems in human blood: H 2 PO 4 -/HPO 42 - & H 2 CO 3/HCO 3 B- + H 2 PO 4 - HPO 42 - + HB B- + H 2 CO 3 HCO 3 - + HB HB = acid (H+ donor) B- = base (H+ acceptor)
Summary 17 • Write a general equation showing the buffering ability of HSO 4 - and its conjugate base. (use B- as the base that reacts with HSO 4 -)
- Slides: 40