ACIDS BASES Arrhenius Theory 1 in aqueous solution

ACIDS & BASES

Arrhenius Theory 1. in aqueous solution + 2. Acid: produces H 3. Base: produces OH
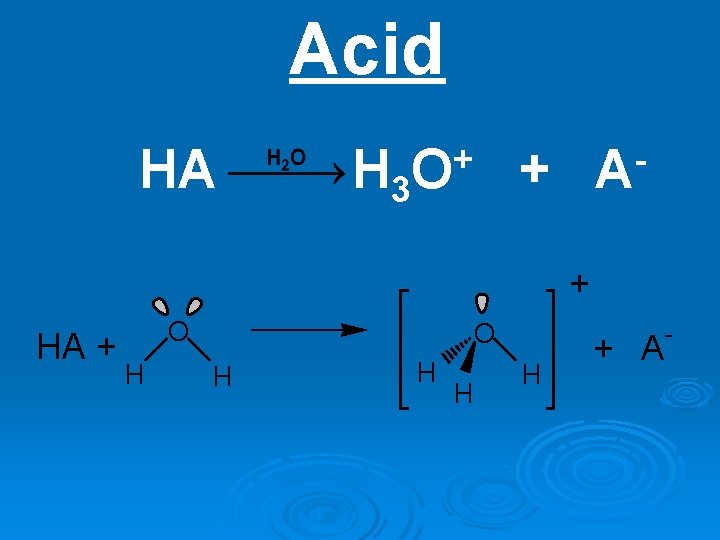
Acid HA H 3 + O A + + HA + O H H H H + A -
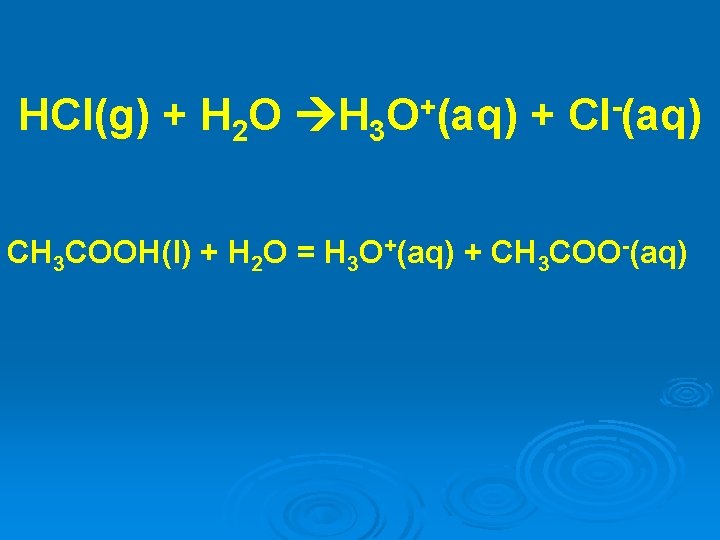
HCl(g) + H 2 O H 3 O+(aq) + Cl-(aq) CH 3 COOH(l) + H 2 O = H 3 O+(aq) + CH 3 COO-(aq)

careless, but often seen HCl + H + Cl CH 3 COOH H+ + CH 3 COO-
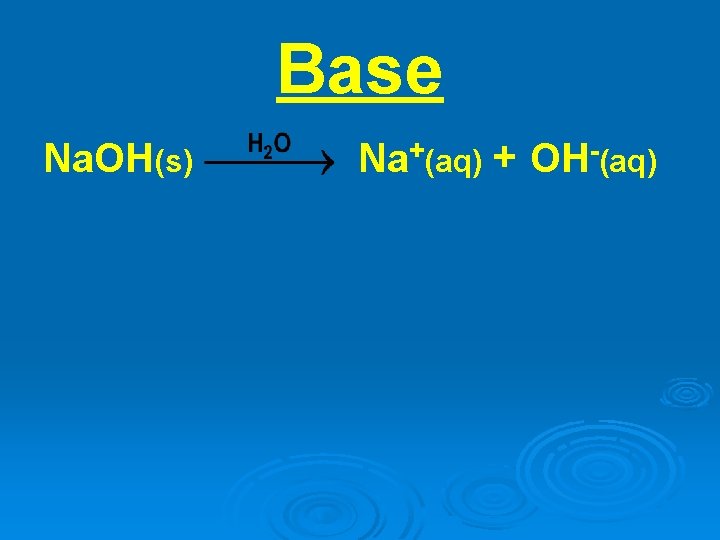
Base Na. OH(s) Na+(aq) + OH-(aq)

Arrhenius acid/base reaction acid + base H 2 O + a salt HA + MOH HOH + MA
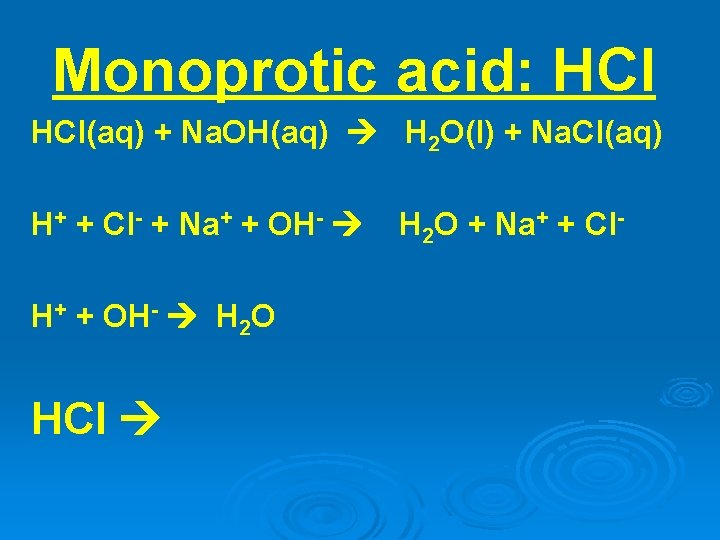
Monoprotic acid: HCl(aq) + Na. OH(aq) H 2 O(l) + Na. Cl(aq) H+ + Cl- + Na+ + OH- H 2 O HCl H 2 O + Na+ + Cl-
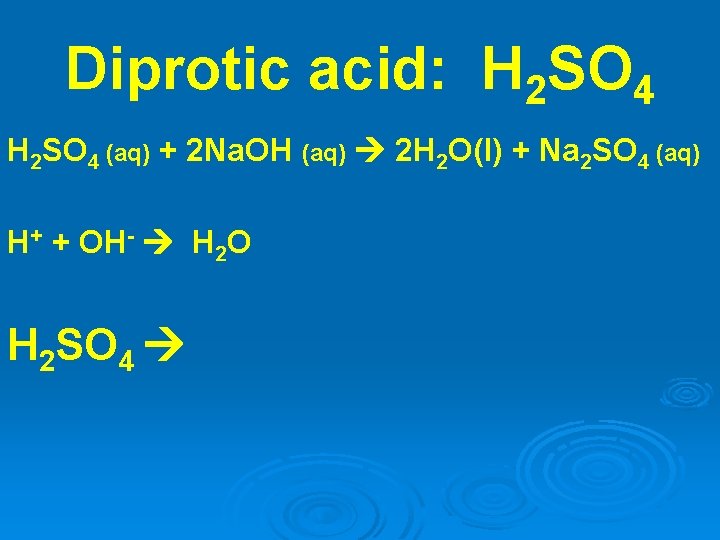
Diprotic acid: H 2 SO 4 (aq) + 2 Na. OH (aq) 2 H 2 O(l) + Na 2 SO 4 (aq) H+ + OH- H 2 O H 2 SO 4
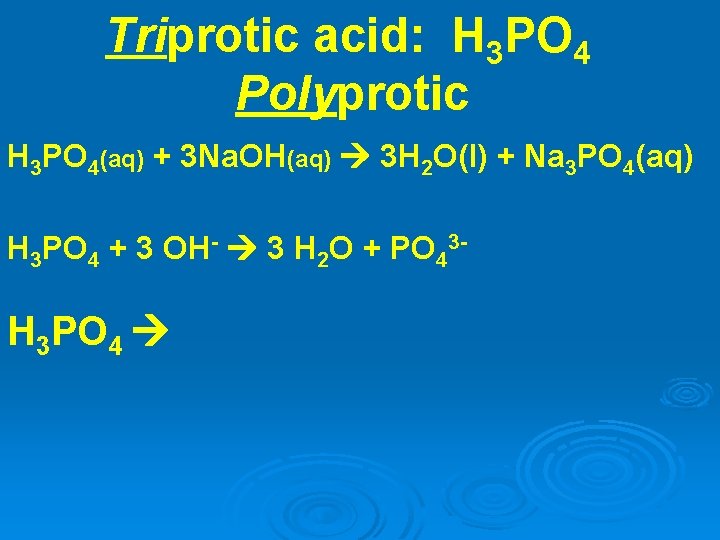
Triprotic acid: H 3 PO 4 Polyprotic H 3 PO 4(aq) + 3 Na. OH(aq) 3 H 2 O(l) + Na 3 PO 4(aq) H 3 PO 4 + 3 OH- 3 H 2 O + PO 43 - H 3 PO 4

Bronsted-Lowry Theory 1. aqueous & nonaqueous solutions 2. Acid: species donating a proton HCl H+ + Cl. H 2 SO 4 H+ + HSO 4 CH 3 COOH H+ + CH 3 COO-

Bronsted-Lowry Theory 3. Base: species accepting a proton OH- + H+ HOH H 2 O + H + H 3 O + NH 3 + H+ NH 4+
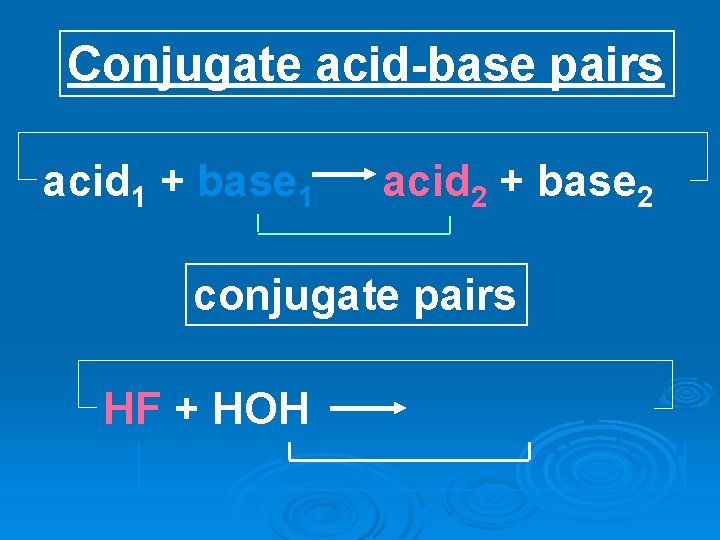
Conjugate acid-base pairs acid 1 + base 1 acid 2 + base 2 conjugate pairs HF + HOH
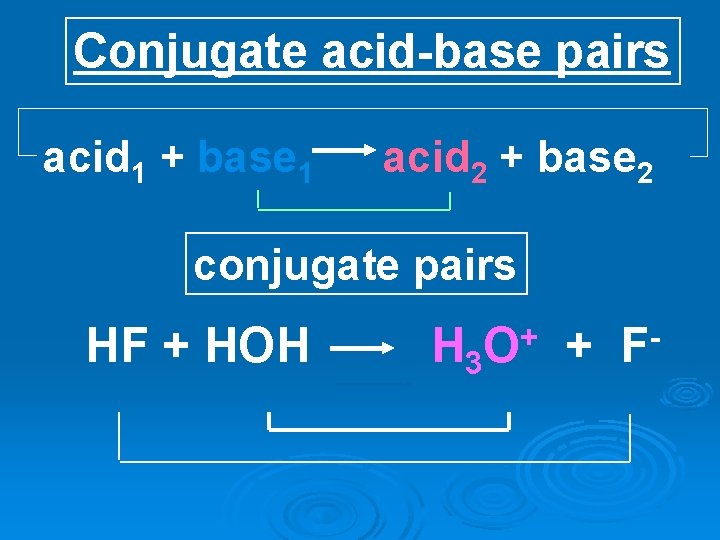
Conjugate acid-base pairs acid 1 + base 1 acid 2 + base 2 conjugate pairs HF + HOH H 3 + O + F

ALL Arrhenius reactions are Bronsted-Lowry reactions HCl + Na. OH H 2 O + Na. Cl

NOT all Bronsted reactions are Arrhenius reactions CH 3 COOH + NH 3 NH 4+ + CH 3 COO-
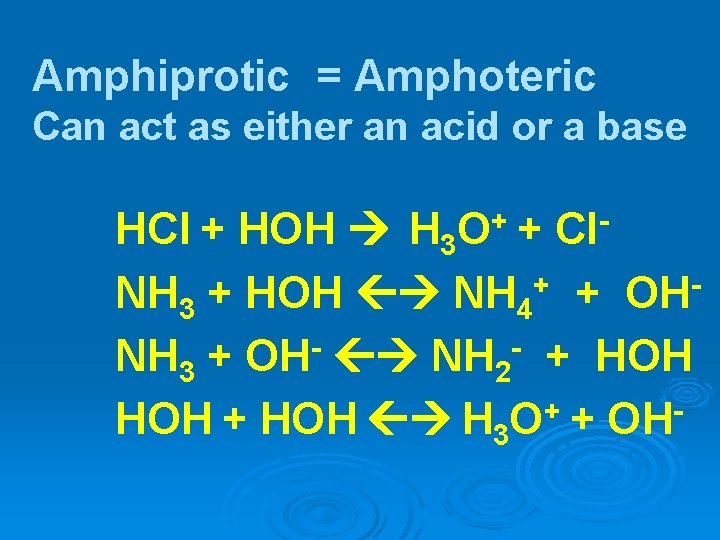
Amphiprotic = Amphoteric Can act as either an acid or a base + O Cl HCl + HOH H 3 + NH 3 + HOH NH 4+ + OHNH 3 + OH- NH 2 - + HOH H 3 O+ + OH-

ACID STRENGTH Relative ability of a compound to donate a proton Base strength is considered a result, not a cause

REVIEW Strong acid 100% dissociation Weak acid <100% dissociation Notice this is NOT related to concentration

Electronegativity is the most significant factor influencing the strength of acids & bases

HF > HCl > HBr > HI as acids in nonaqueous solvents, or as pure gases

Look at difference in electronegativities 2. 1 H - F 2. 1 H - Cl 2. 1 H - Br 2. 1 H - I 4. 0 3. 0 2. 8 2. 5

Most “ionic” is the most acidic Nonpolar Polar Ionic ED 0 ED 1. 7 ED 4. 0

However, as acids in aqueous solution HF < HCl = HBr = HI

2. 1 H - O 3. 5 competition! 2. 1 H - F 4. 0 2. 1 H - Cl 3. 0 2. 1 H - Br 2. 8 2. 1 H - I 2. 5

Is methane acidic as a gas or in aqueous solution? 2. 1 H - C 2. 5

The strength of oxy-acids are also dependent on electronegativity.

Oxy-acids and bases have the same fundamental structure
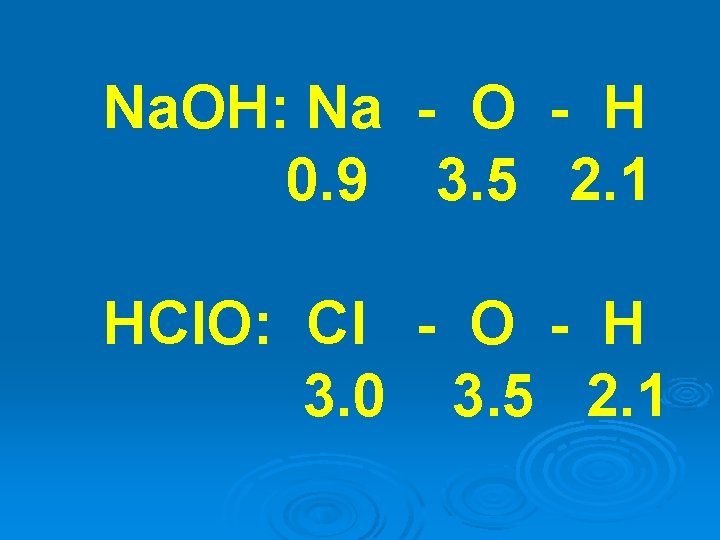
Na. OH: Na - O - H 0. 9 3. 5 2. 1 HCl. O: Cl - O - H 3. 0 3. 5 2. 1

In water, the more “ionic” bond dissociates, forming the acid or base
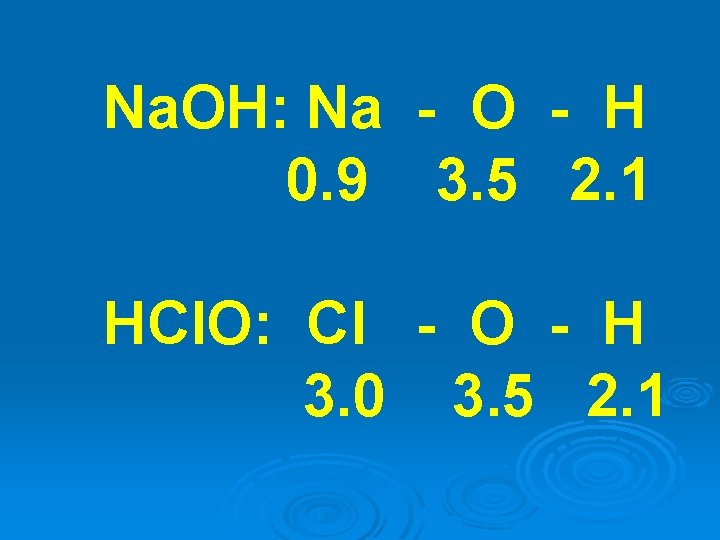
Na. OH: Na - O - H 0. 9 3. 5 2. 1 HCl. O: Cl - O - H 3. 0 3. 5 2. 1

Are alcohols acids or bases? C-O-H 2. 5 3. 5 2. 1

Acids in homologous series are of different strength
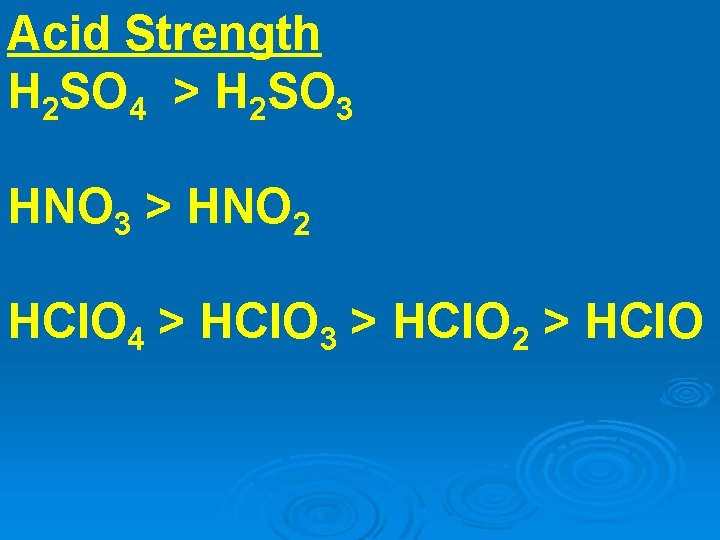
Acid Strength H 2 SO 4 > H 2 SO 3 HNO 3 > HNO 2 HCl. O 4 > HCl. O 3 > HCl. O 2 > HCl. O
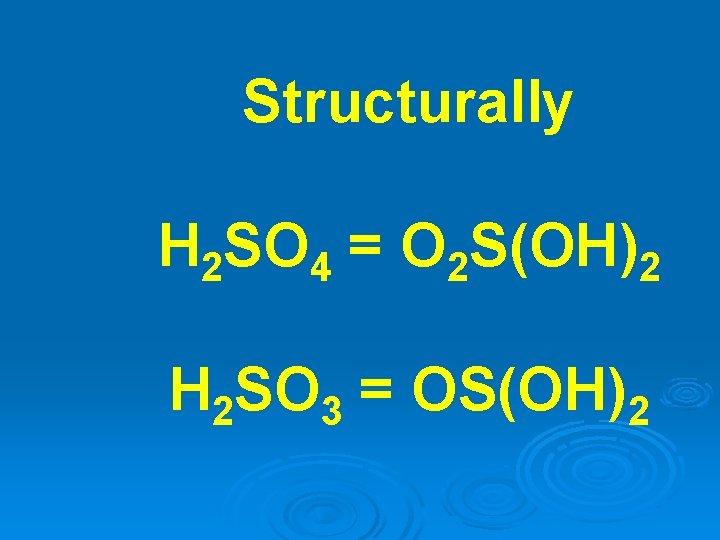
Structurally H 2 SO 4 = O 2 S(OH)2 H 2 SO 3 = OS(OH)2

Need to examine formal charge of central atom.
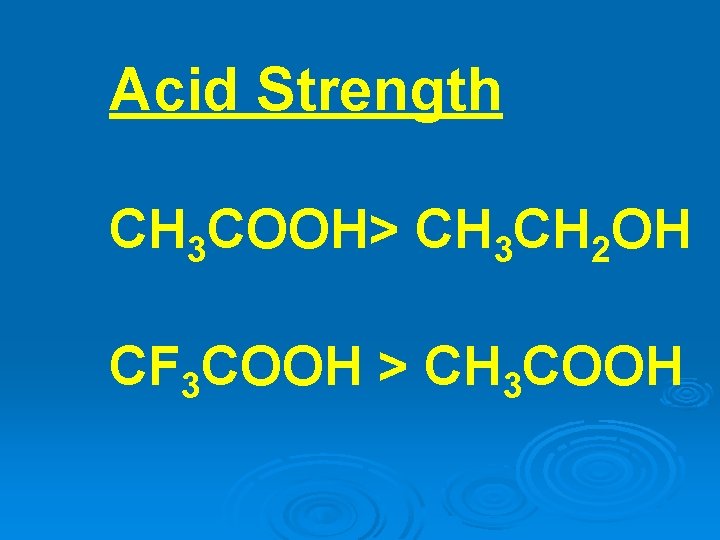
Acid Strength CH 3 COOH> CH 3 CH 2 OH CF 3 COOH > CH 3 COOH

Need to examine inductive effect of neighboring atoms.

p. H p. K Ka , K b , Kw
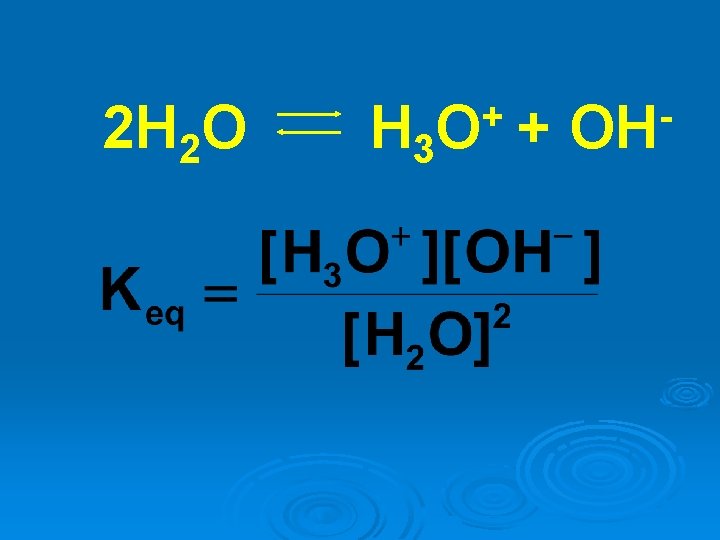
2 H 2 O H 3 + OH
![Keq [H 2 2 O] Kw = [H 3 + O ][OH ] where Keq [H 2 2 O] Kw = [H 3 + O ][OH ] where](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-41.jpg)
Keq [H 2 2 O] Kw = [H 3 + O ][OH ] where o -14 Kw (25 C ) = 1 x 10
![in a neutral solution [H 3 O+ ] = [OH-] 1 x 10 -14 in a neutral solution [H 3 O+ ] = [OH-] 1 x 10 -14](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-42.jpg)
in a neutral solution [H 3 O+ ] = [OH-] 1 x 10 -14 = [H 3 O+ ]2 = [OH-]2 [H 3 + O ]= [OH ] =1 x -7 10
![p. X = -log X p. K = -log K + O] p. H p. X = -log X p. K = -log K + O] p. H](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-43.jpg)
p. X = -log X p. K = -log K + O] p. H = -log [H 3 p. OH = -log [OH ]
![leveling effect of + H 2 O limits [H 3 O ] & [OH leveling effect of + H 2 O limits [H 3 O ] & [OH](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-44.jpg)
leveling effect of + H 2 O limits [H 3 O ] & [OH ] to that controlled by H 2 O
![upper limit [H 3 lower limit [H 3 + O ]=1 ]= -14 1 upper limit [H 3 lower limit [H 3 + O ]=1 ]= -14 1](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-45.jpg)
upper limit [H 3 lower limit [H 3 + O ]=1 ]= -14 1 x 10
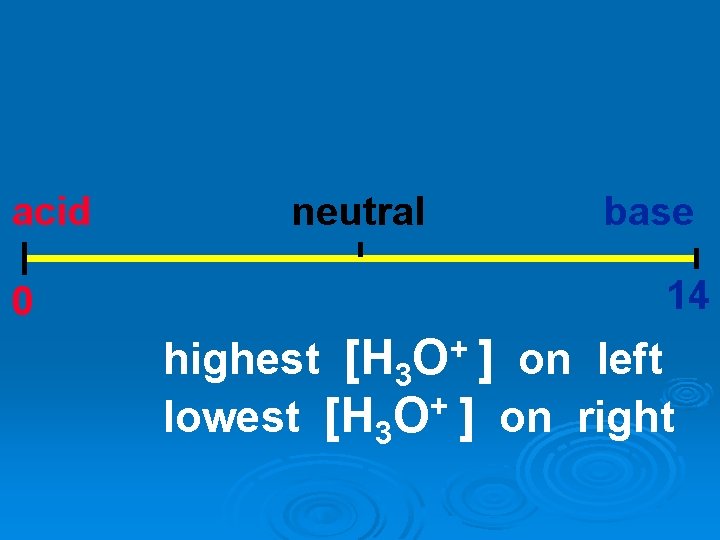
p. H scale acid 0 neutral 7 base 14 + O highest [H 3 ] on left lowest [H 3 O+ ] on right
![[H 3 + O ] and [OH ] must be considered together [H 3 + O ] and [OH ] must be considered together](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-47.jpg)
[H 3 + O ] and [OH ] must be considered together
![Kw = [H 3 + O ][OH ] -log Kw = -log {[H 3 Kw = [H 3 + O ][OH ] -log Kw = -log {[H 3](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-48.jpg)
Kw = [H 3 + O ][OH ] -log Kw = -log {[H 3 + O ][OH ]} -log Kw = {-log [H 3 O+ ]} + {-log[OH-]}

p. Kw = p. H + p. OH but Kw = 1 x -14 10 14 = p. H + p. OH
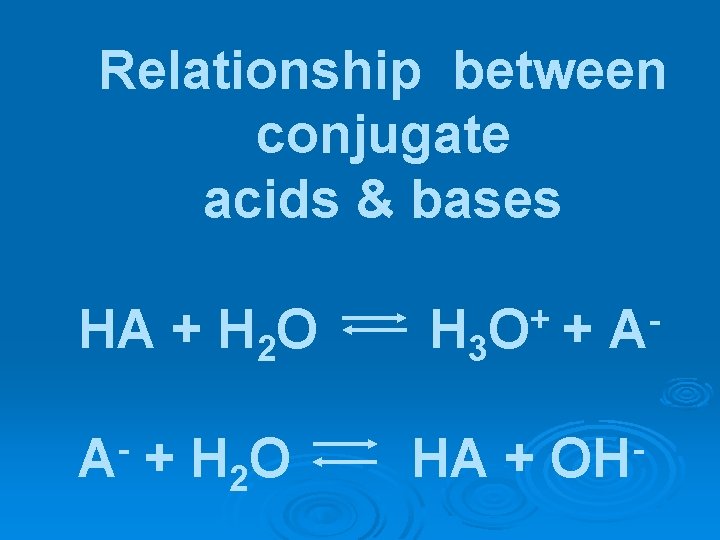
Relationship between conjugate acids & bases HA + H 2 O H 3 + O HA + + A OH
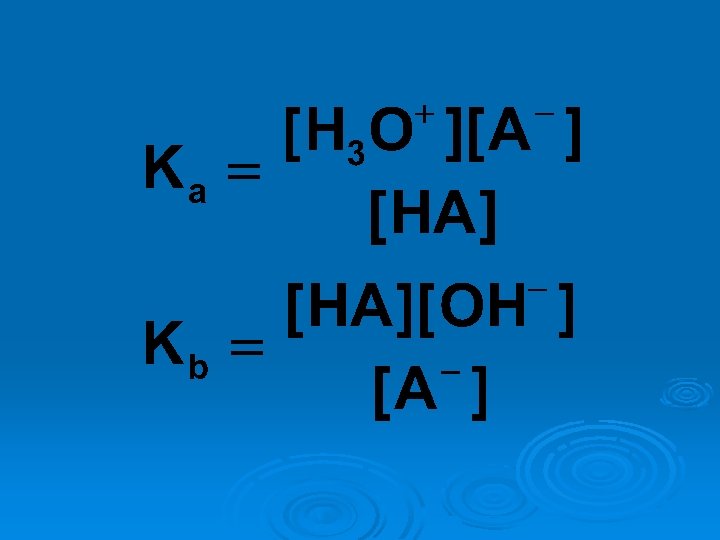

![Ka x Kb = [H 3 O+ ][OH-] = Kw Ka x Kb = [H 3 O+ ][OH-] = Kw](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-53.jpg)
Ka x Kb = [H 3 O+ ][OH-] = Kw
![Ka . K = [H b 3 b + O = Kw ][OH ] Ka . K = [H b 3 b + O = Kw ][OH ]](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-54.jpg)
Ka . K = [H b 3 b + O = Kw ][OH ] = Kw
![SUMMARY + p. H = -log [H 3 O ] p. OH = -log SUMMARY + p. H = -log [H 3 O ] p. OH = -log](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-55.jpg)
SUMMARY + p. H = -log [H 3 O ] p. OH = -log [OH ] + -14 [H 3 O ][OH ] = 1 x 10 p. H + p. OH = 14. Ka Kb = Kw

Applications of Acid-Base Concepts

for weak acids & bases, refer to Appendix H for Ka & Appendix I for Kb values in Kotz & Treichel

1. What is the p. H of a solution that is 0. 025 M KOH?

2. What is the p. H of a 0. 20 M acetic acid solution?

3. 100 m. L of 0. 10 M CH 3 COOH are mixed with 20. 0 m. L of 0. 10 M Na. OH. What is the p. H of the solution?
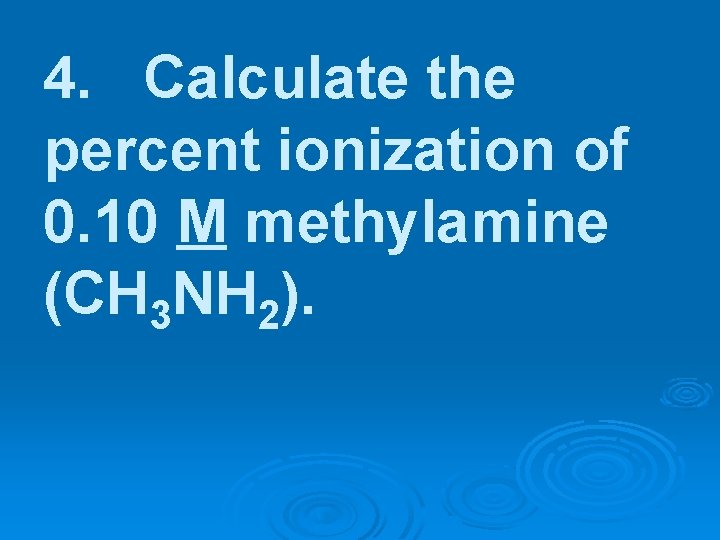
4. Calculate the percent ionization of 0. 10 M methylamine (CH 3 NH 2).

p. H of Salts & Oxides

What effect does the addition of a salt to water have upon the p. H of the water?

H 2 O equilibrium is the prime factor in the behavior of solutions.

p. H of a salt solution is dependent upon the strength of the salt as an electrolyte.
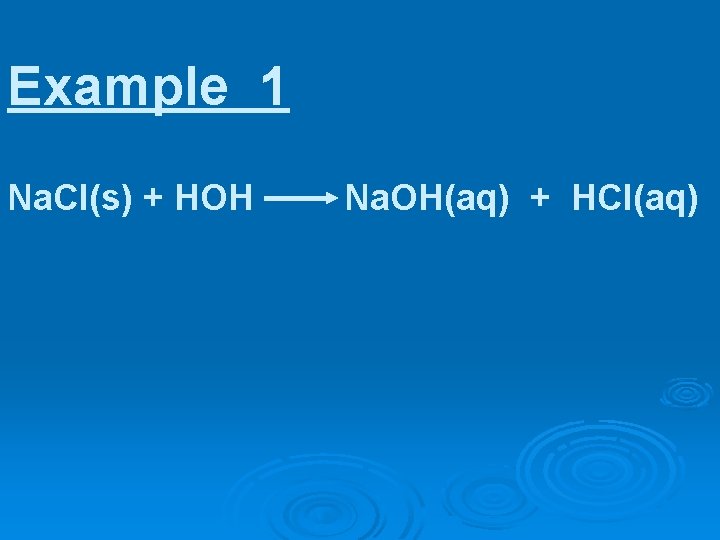
Example 1 Na. Cl(s) + HOH Na. OH(aq) + HCl(aq)

Example 1 Na. Cl(s) + HOH Na. OH (aq) + HCl(aq) strong base strong acid Na+ + OH- + H+ + Cl- Na+ + HOH + Cl-

thus, Na. Cl in water has NO effect on p. H

Example 2 Na. CN(s) + HOH Na. OH(aq) + HCN(aq)
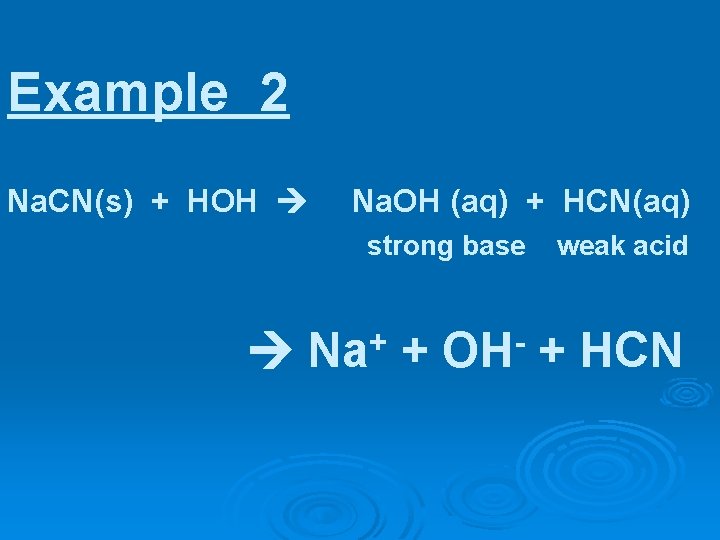
Example 2 Na. CN(s) + HOH Na. OH (aq) + HCN(aq) strong base weak acid Na+ + OH- + HCN

CN is the anion of the weak acid HCN CN- + HOH HCN + OH-

Na. CN(s) + HOH Na+ + OH- + HCN strong base weak acid

thus, Na. CN in water produces a/n ? ? solution

thus, Na. CN in water produces a BASIC solution

5. What is the p. H of a 0. 010 M sodium cyanide solution?

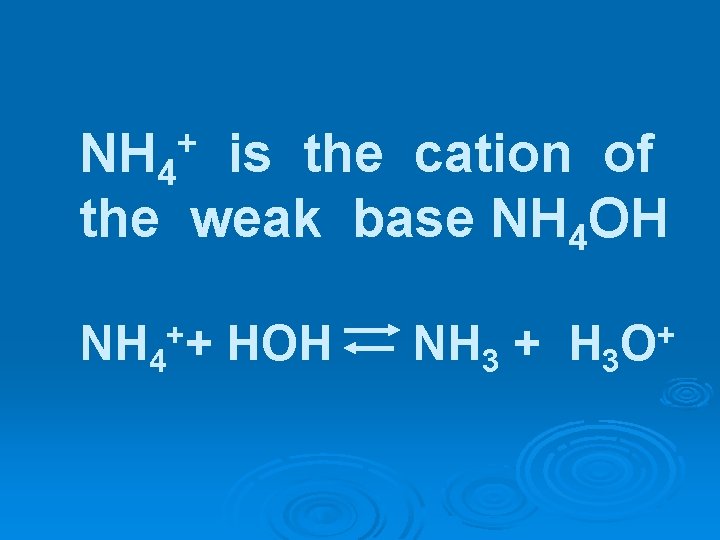
Example 3 NH 4 Cl(s) + HOH NH 4 OH (aq) + HCl(aq)
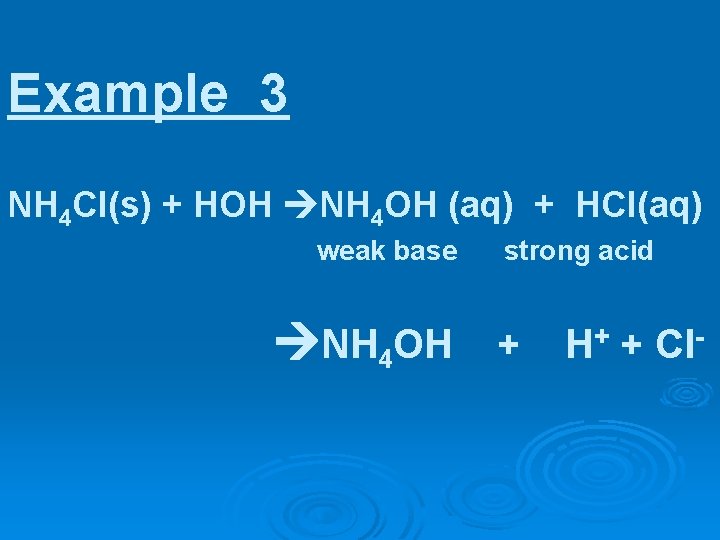
Example 3 NH 4 Cl(s) + HOH NH 4 OH (aq) + HCl(aq) weak base strong acid NH 4 OH + H+ + Cl-
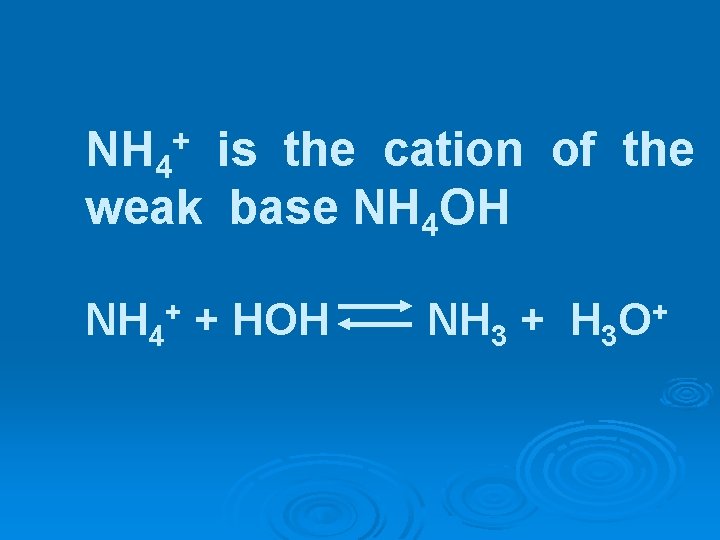
NH 4 is the cation of the weak base NH 4 OH + NH 4+ + HOH NH 3 + H 3 O+
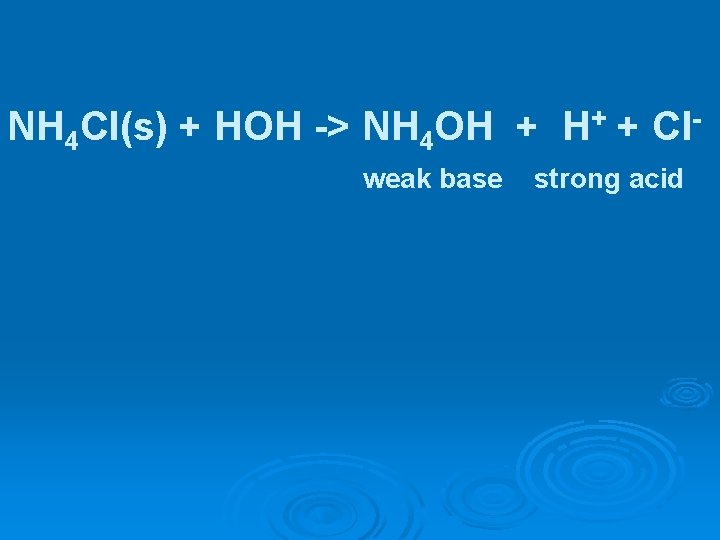
NH 4 Cl(s) + HOH -> NH 4 OH + H+ + Clweak base strong acid

thus, NH 4 Cl in water produces a/n ? ? solution

thus, NH 4 Cl in water produces an ACID solution

6. What is p. H of a 0. 10 M ammonium chloride solution?
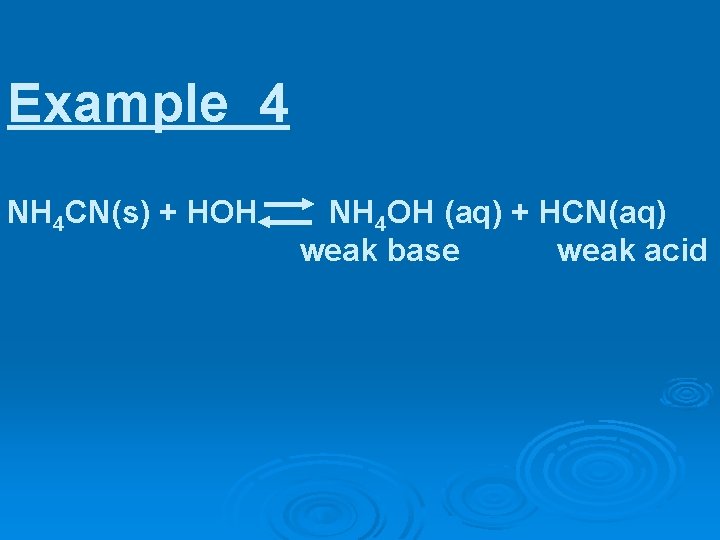
Example 4 NH 4 CN(s) + HOH NH 4 OH (aq) + HCN(aq) weak base weak acid
NH 4 is the cation of the weak base NH 4 OH + NH 4 ++ HOH NH 3 + O

CN is the anion of the weak acid HCN CN + HOH HCN + OH

thus, NH 4 CN in water produces a/n ? ? solution

The p. H of a solution formed from the cation of a weak base and the anion of a weak acid is dependent on the relative strength of the weak acid and weak base.
![-10 10 Ka(HCN) = 6. 2 x [Text: Table 5. 1] Appendix H A-23 -10 10 Ka(HCN) = 6. 2 x [Text: Table 5. 1] Appendix H A-23](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-88.jpg)
-10 10 Ka(HCN) = 6. 2 x [Text: Table 5. 1] Appendix H A-23 -5 Kb(NH 4 OH) = 1. 8 x 10 [Text: Table 5. 3] Appendix I A-25

thus, NH 4 CN in water produces a/n ? ? solution

thus, NH 4 CN in water produces a BASIC solution, because the weak base is stronger (ionizes more) than the weak acid

Acidity of Oxides
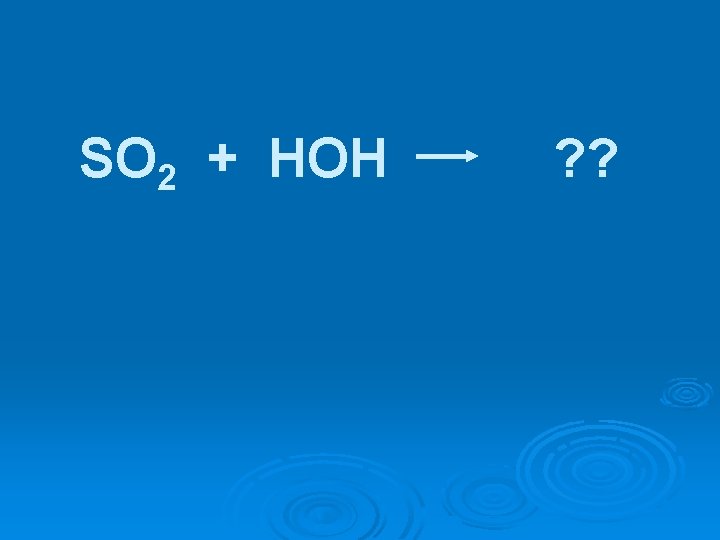
SO 2 + HOH ? ?
![SO 2 + HOH H 2 SO 3 [O 2] H 2 SO 4 SO 2 + HOH H 2 SO 3 [O 2] H 2 SO 4](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-93.jpg)
SO 2 + HOH H 2 SO 3 [O 2] H 2 SO 4
![SO 2 + HOH H 2 SO 3 [O 2] H 2 SO 4 SO 2 + HOH H 2 SO 3 [O 2] H 2 SO 4](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-94.jpg)
SO 2 + HOH H 2 SO 3 [O 2] H 2 SO 4 Covalent oxides are acidic & are referred to as acid anhydrides
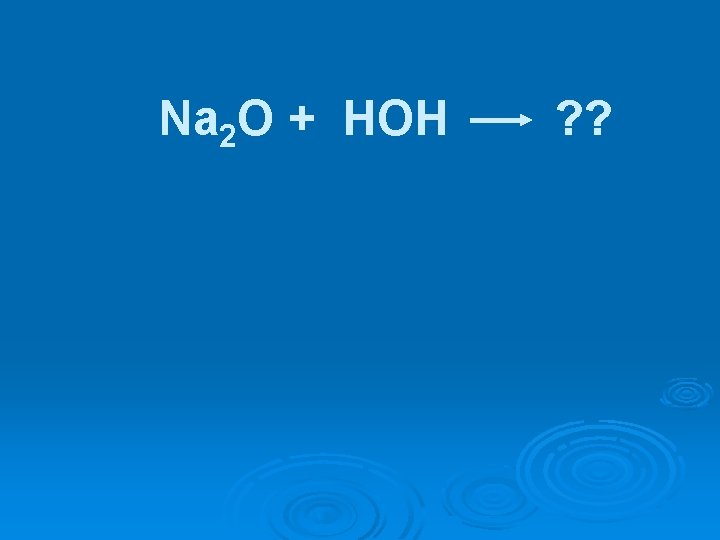
Na 2 O + HOH ? ?
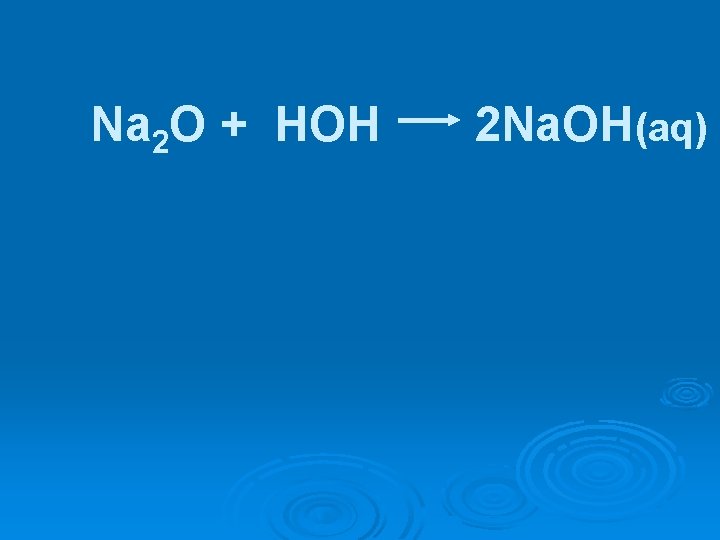
Na 2 O + HOH 2 Na. OH(aq)

Na 2 O + HOH 2 Na. OH(aq) Ionic oxides are basic & are referred to as basic anhydrides

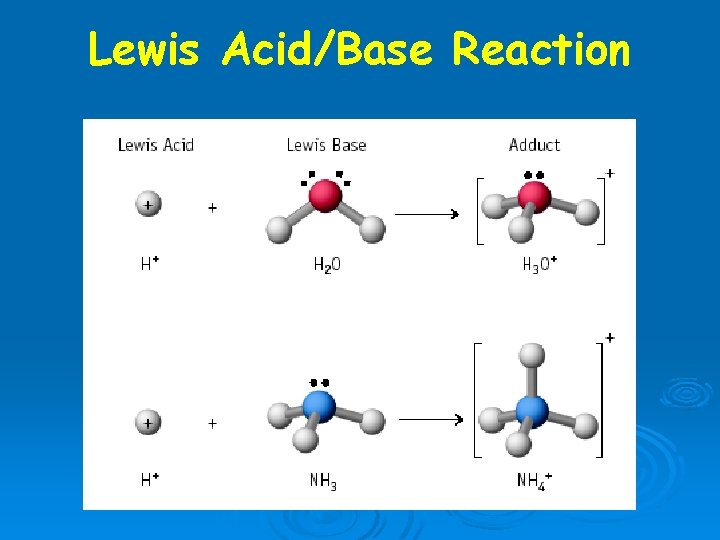
Lewis Acid-Base Theory

Acid substance capable of accepting an e- pair

Lewis acid must have an empty valence level orbital + H i. e. has an empty 1 s orbital which can accept an e pair

+ H Thus, is an acid under all three theories Arrhenius Bronsted-Lowry Lewis

Lewis Acid-Base Theory Acid: substance capable of accepting an e pair Base substance capable of donating an e pair

Examples of Lewis bases OH F , NH 3 , all have unbonded pairs of e available for donation

Elements of Group 13 (3 A) form compounds that make excellent Lewis acids

another “typical” Lewis acid-base reaction:

Reaction of a Lewis Acid and Lewis Base Ø New bond formed using electron pair from the Lewis base. Ø Coordinate covalent bond Ø Notice geometry change on reaction.

Lewis Acids & Bases Formation of hydronium ion is also an excellent example. • Electron pair of the new O-H bond originates on the Lewis base.
Lewis Acid/Base Reaction
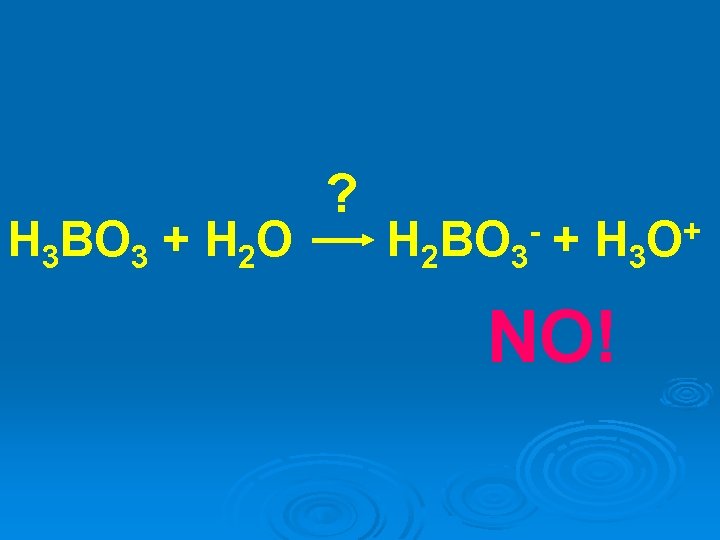
H 3 BO 3 + H 2 O ? H 2 BO 3 + H 3 - NO! + O
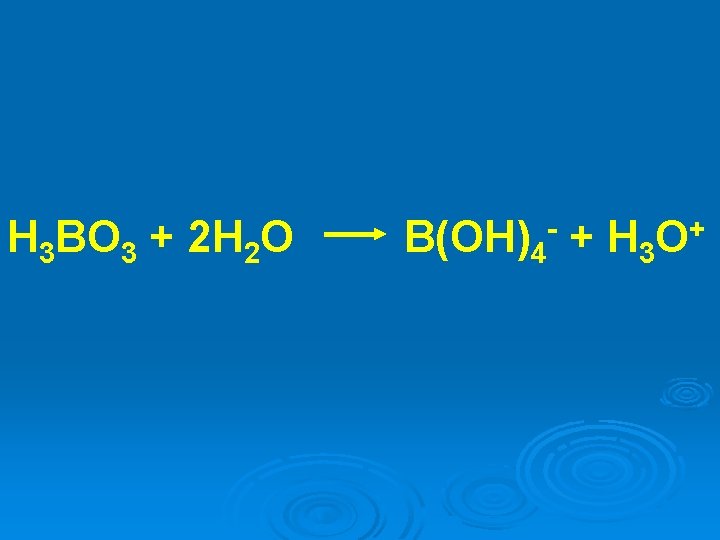
H 3 BO 3 + 2 H 2 O B(OH)4 + H 3 - + O

is Al(OH)3 an acid or base?

Amphoterism of Al(OH)3

Lewis Acids & Bases This explains AMPHOTERIC nature of some metal hydroxides. Al(OH)3(s) + 3 H+ Al 3+ + 3 H 2 O Here Al(OH)3 is a Brønsted base. Al(OH)3(s) + OH- Al(OH)4 Here Al(OH)3 is a Lewis acid.

Transition metal ions also very good Lewis Acids
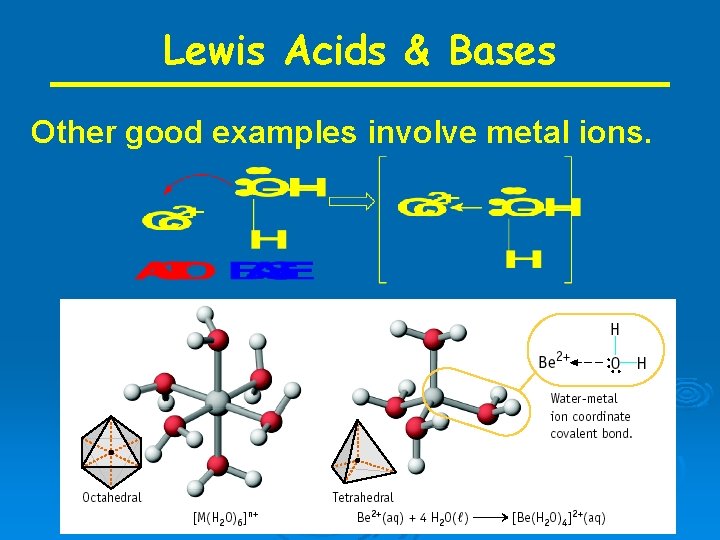
Lewis Acids & Bases Other good examples involve metal ions.
![2+ Zn . . + HOH. . => [Ar] 0 4 s ? 10 2+ Zn . . + HOH. . => [Ar] 0 4 s ? 10](http://slidetodoc.com/presentation_image_h/931ec707ea856d5f19f6a0493c20d83c/image-116.jpg)
2+ Zn . . + HOH. . => [Ar] 0 4 s ? 10 3 d 0 4 p Zn(H 2 O)4 2+
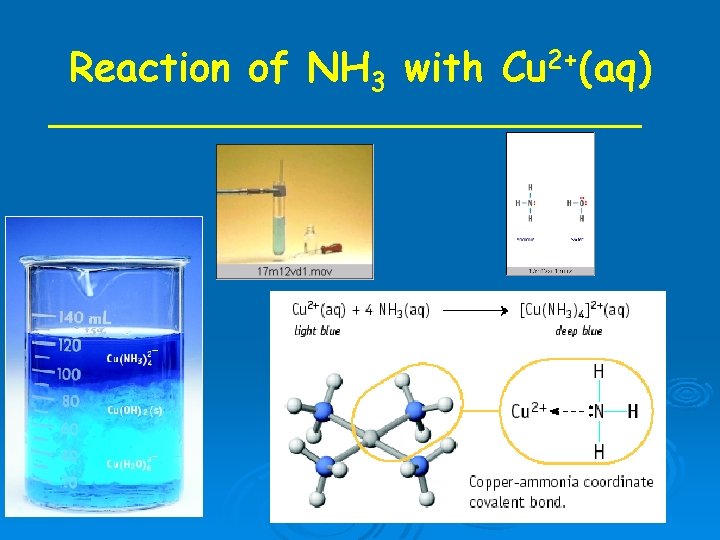
Reaction of NH 3 with Cu 2+(aq)

Formation of complex ions is a Lewis acid-base reaction

Lewis Acid-Base Interactions in Biology Ø The heme group in hemoglobin can interact with O 2 and CO. Ø The Fe ion in hemoglobin is a Lewis acid Ø O 2 and CO can act as Heme group Lewis bases

Inclusiveness of the Acid/Base Definitions Lewis Bronstead Arrhenius
- Slides: 120