Acids Bases Arrhenius Acids Arrhenius acids produce H

Acids / Bases

Arrhenius Acids Arrhenius acids ▪ produce H+ ions in water. ▪ have a sour taste. ▪ turn blue litmus paper red. ▪ corrode some metals. © 2013 Pearson Education, Inc. Chapter 10, Section 1 Citrus fruits are sour because of the presence of acids.

Naming Acids ▪ Acids with H and a nonmetal are named with the prefix hydro− and end with −ic acid. HCl hydrochloric acid ▪ Acids with H and an oxygen-containing polyatomic ion are named by changing the end of the name of the polyatomic ion from −ate to −ic acid or −ite to −ous acid. Cl. O 3− chlorate HCl. O 3 chloric acid Cl. O 2− chlorite HCl. O 2 chlorous acid © 2013 Pearson Education, Inc. Chapter 10, Section 1
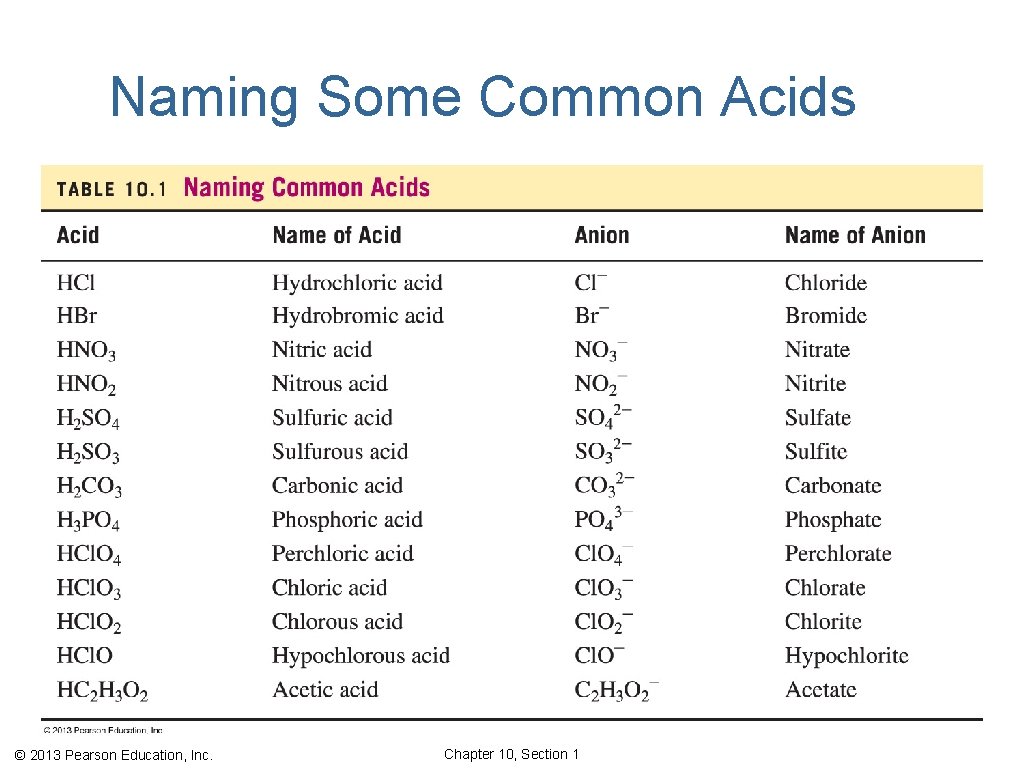
Naming Some Common Acids © 2013 Pearson Education, Inc. Chapter 10, Section 1

Learning Check Select the correct name for each of the following acids. 1. HBr A. bromic acid B. bromous acid C. hydrobromic acid 2. H 2 CO 3 A. carbonic acid B. hydrocarbonic acid C. carbonous acid 3. HBr. O 2 A. bromic acid B. hydrobromous acid C. bromous acid © 2013 Pearson Education, Inc. Chapter 10, Section 1

Solution 1. HBr C. hydrobromic acid The name of an acid with H and one nonmetal uses the prefix hydro− and ends with −ic acid. 2. H 2 CO 3 A. carbonic acid An acid with H and a polyatomic ion, bicarbonate, HCO 3− is named by changing the end of the ion’s name from −ate to −ic acid. 3. HBr. O 2 C. bromous acid This acid of bromite, (Br. O 2−) is bromous acid. © 2013 Pearson Education, Inc. Chapter 10, Section 1
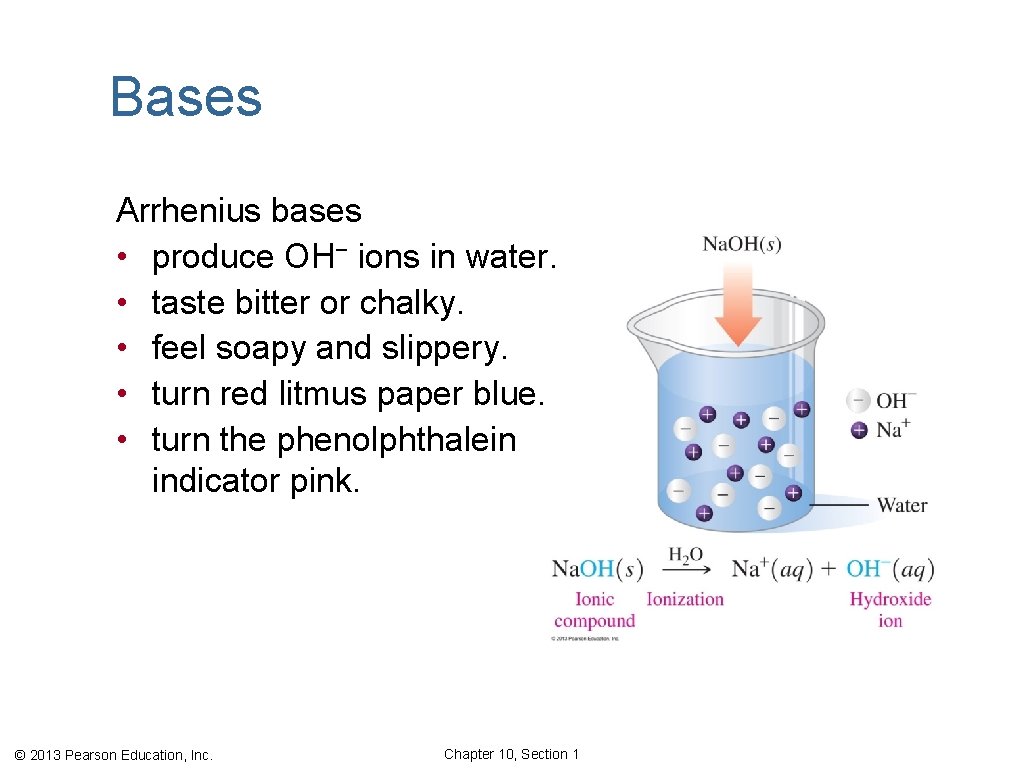
Bases Arrhenius bases • produce OH− ions in water. • taste bitter or chalky. • feel soapy and slippery. • turn red litmus paper blue. • turn the phenolphthalein indicator pink. © 2013 Pearson Education, Inc. Chapter 10, Section 1

Some Common Bases with OH− ions are named as the hydroxide of the metal in the formula. Na. OH KOH Ba(OH)2 Al(OH)3 Fe(OH)3 © 2013 Pearson Education, Inc. sodium hydroxide potassium hydroxide barium hydroxide aluminum hydroxide iron (III) hydroxide Chapter 10, Section 1

Learning Check Match the formulas with the names. 1. HNO 2 A. iodic acid 2. Ca(OH)2 B. sulfuric acid 3. H 2 SO 4 C. sodium hydroxide 4. HIO 3 D. nitrous acid 5. Na. OH E. calcium hydroxide © 2013 Pearson Education, Inc. Chapter 10, Section 1

Solution Match the formulas with the names. 1. HNO 2 D. nitrous acid 2. Ca(OH)2 E. calcium hydroxide 3. H 2 SO 4 B. sulfuric acid 4. HIO 3 A. iodic acid 5. Na. OH C. sodium hydroxide © 2013 Pearson Education, Inc. Chapter 10, Section 1
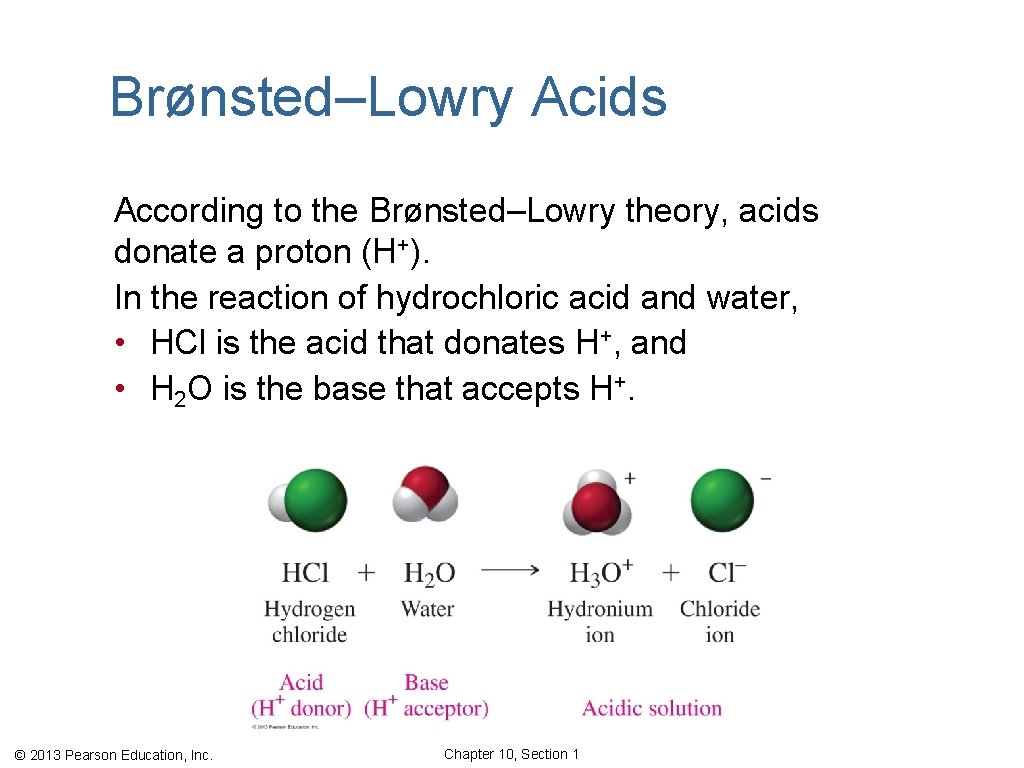
Brønsted–Lowry Acids According to the Brønsted–Lowry theory, acids donate a proton (H+). In the reaction of hydrochloric acid and water, • HCl is the acid that donates H+, and • H 2 O is the base that accepts H+. © 2013 Pearson Education, Inc. Chapter 10, Section 1
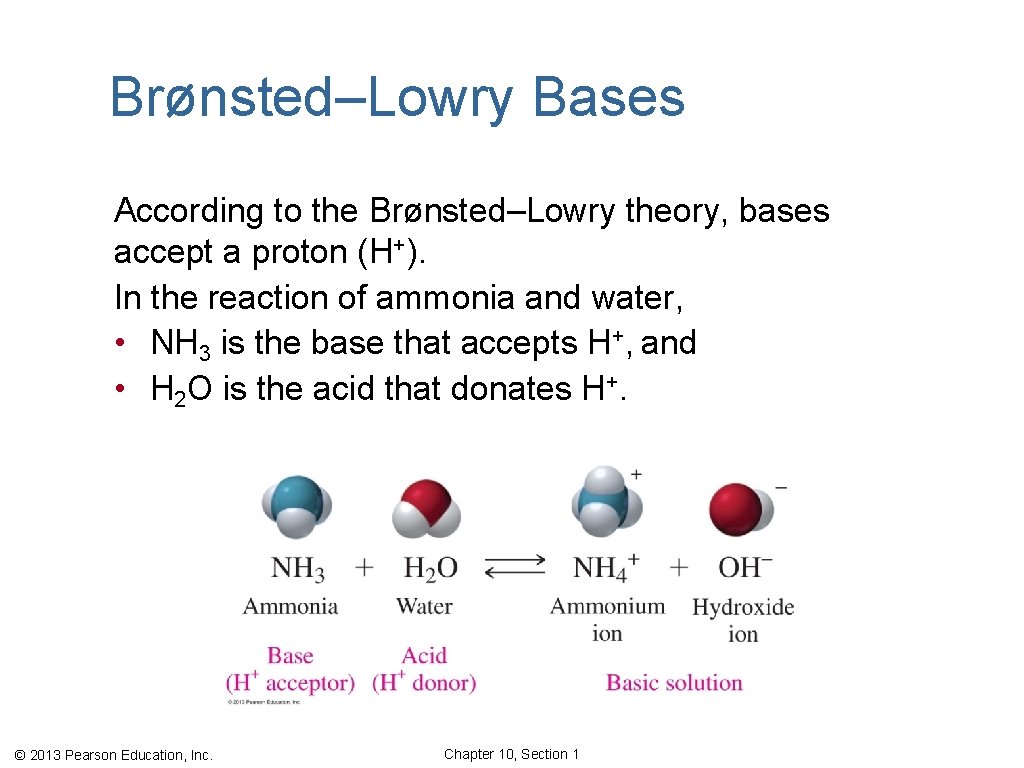
Brønsted–Lowry Bases According to the Brønsted–Lowry theory, bases accept a proton (H+). In the reaction of ammonia and water, • NH 3 is the base that accepts H+, and • H 2 O is the acid that donates H+. © 2013 Pearson Education, Inc. Chapter 10, Section 1

Conjugate Acid–Base Pairs In any acid–base reaction, there are two conjugate acid–base pairs. • Each is related by the loss and gain of H+. • One occurs in the forward direction. • One occurs in the reverse direction. conjugate acid–base pair 1 conjugate acid–base pair 2 © 2013 Pearson Education, Inc. Chapter 10, Section 1
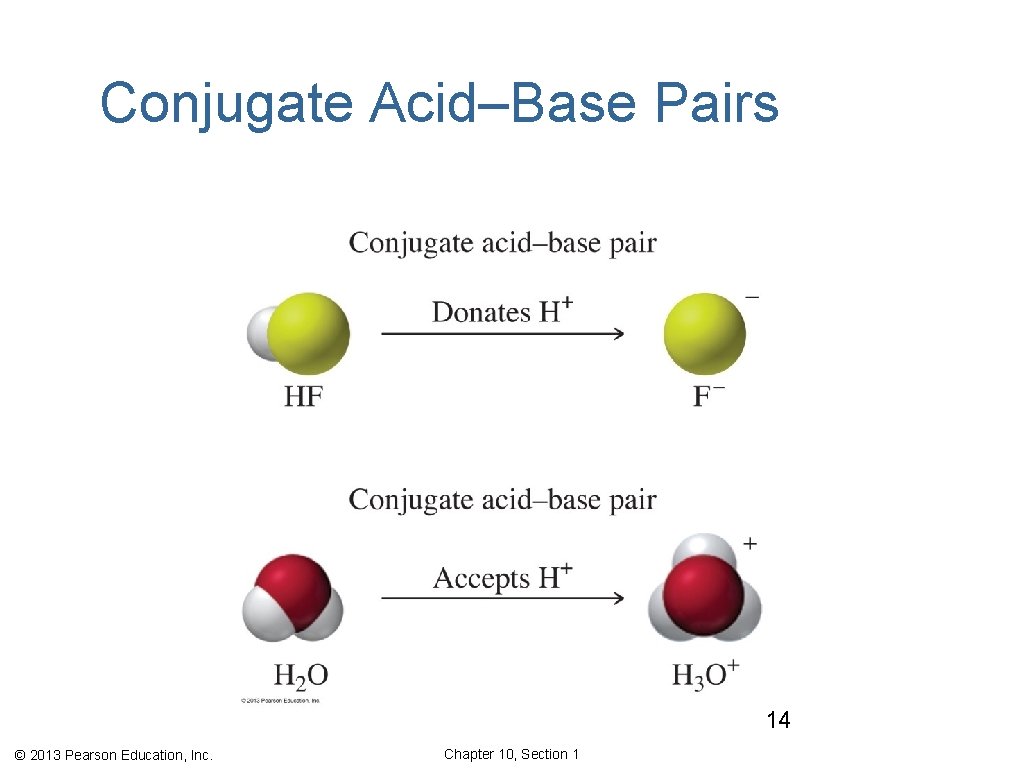
Conjugate Acid–Base Pairs 14 © 2013 Pearson Education, Inc. Chapter 10, Section 1

Conjugate Acids and Bases, HF • The first conjugate acid–base pair is HF/F−. HF loses one H+ to form its conjugate base F−. • The other conjugate acid–base pair is H 2 O/H 3 O+. H 2 O acts as a base accepting one H+ to form its conjugate acid H 3 O+. © 2013 Pearson Education, Inc. Chapter 10, Section 1
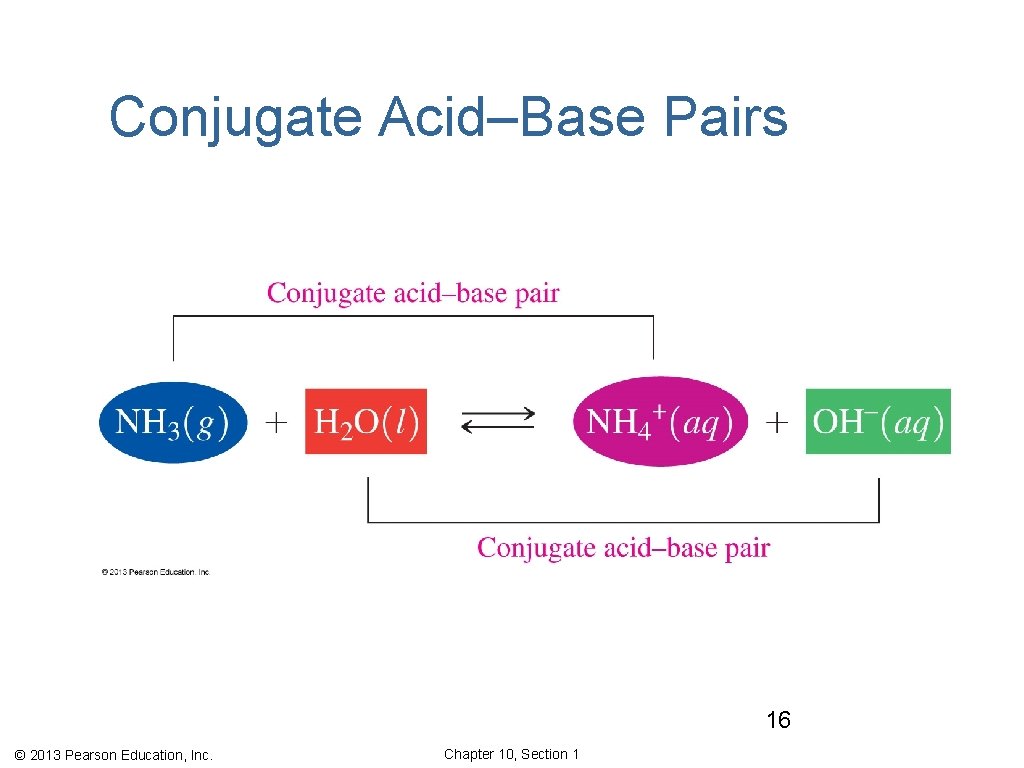
Conjugate Acid–Base Pairs 16 © 2013 Pearson Education, Inc. Chapter 10, Section 1
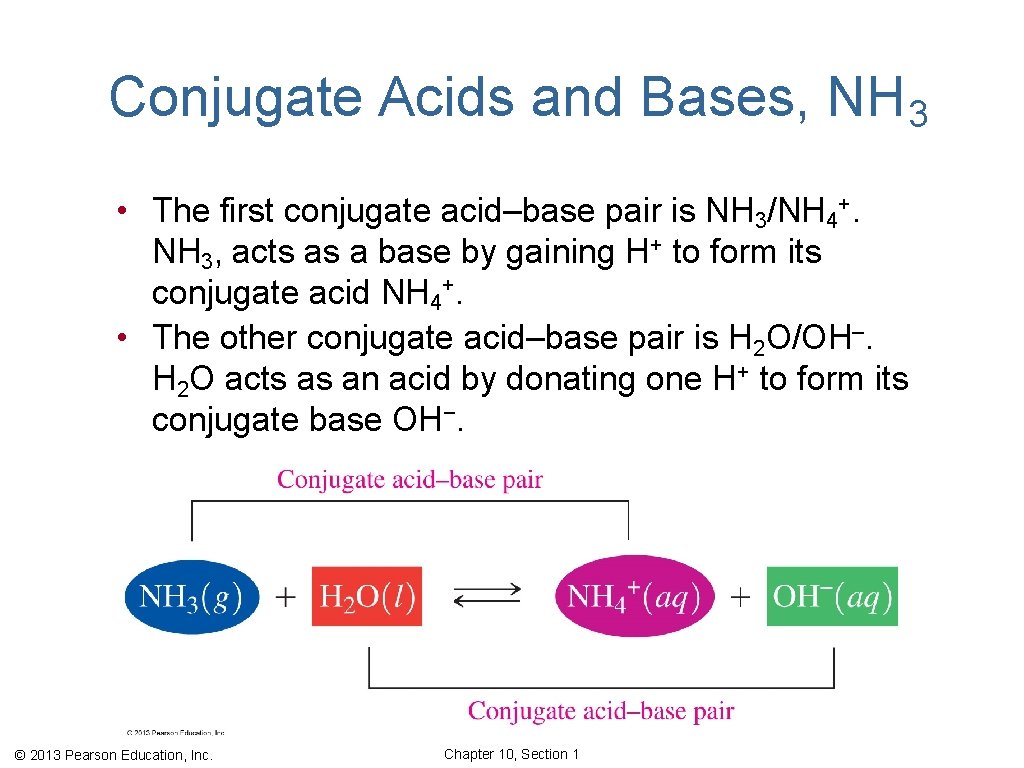
Conjugate Acids and Bases, NH 3 • The first conjugate acid–base pair is NH 3/NH 4+. NH 3, acts as a base by gaining H+ to form its conjugate acid NH 4+. • The other conjugate acid–base pair is H 2 O/OH–. H 2 O acts as an acid by donating one H+ to form its conjugate base OH−. © 2013 Pearson Education, Inc. Chapter 10, Section 1

Learning Check 1. Write the conjugate base of the following. A. HBr B. H 2 S C. H 2 CO 3 2. Write the conjugate acid of the following. A. NO 2− B. NH 3 C. OH− © 2013 Pearson Education, Inc. Chapter 10, Section 1

Solution 1. Write the conjugate base of the following. Remove H+ to write the conjugate base. A. HBr Br − B. H 2 S HS− C. H 2 CO 3 HCO 3− 2. Write the conjugate acid of the following. Add H+ to write the conjugate acid. A. NO 2− HNO 2 B. NH 3 NH 4+ C. OH− H 2 O © 2013 Pearson Education, Inc. Chapter 10, Section 1

Learning Check 1. The conjugate base of HCO 3− is A. CO 32−. B. HCO 3−. C. H 2 CO 3. 2. The conjugate acid of HCO 3− is A. CO 32−. B. HCO 3−. C. H 2 CO 3. 3. The conjugate base of H 2 O is A. OH−. B. H 2 O. C. H 3 O+. 4. The conjugate acid of H 2 O is A. OH−. B. H 2 O. C. H 3 O+. © 2013 Pearson Education, Inc. Chapter 10, Section 1

Solution 1. (A) The conjugate base of HCO 3− is, CO 32−. 2. (C) The conjugate acid of HCO 3− is H 2 CO 3. 3. (A) The conjugate base of H 2 O is OH−. 4. (C) The conjugate acid of H 2 O is H 3 O+. © 2013 Pearson Education, Inc. Chapter 10, Section 1

Learning Check Identify the acid–base conjugate pairs. © 2013 Pearson Education, Inc. Chapter 10, Section 1
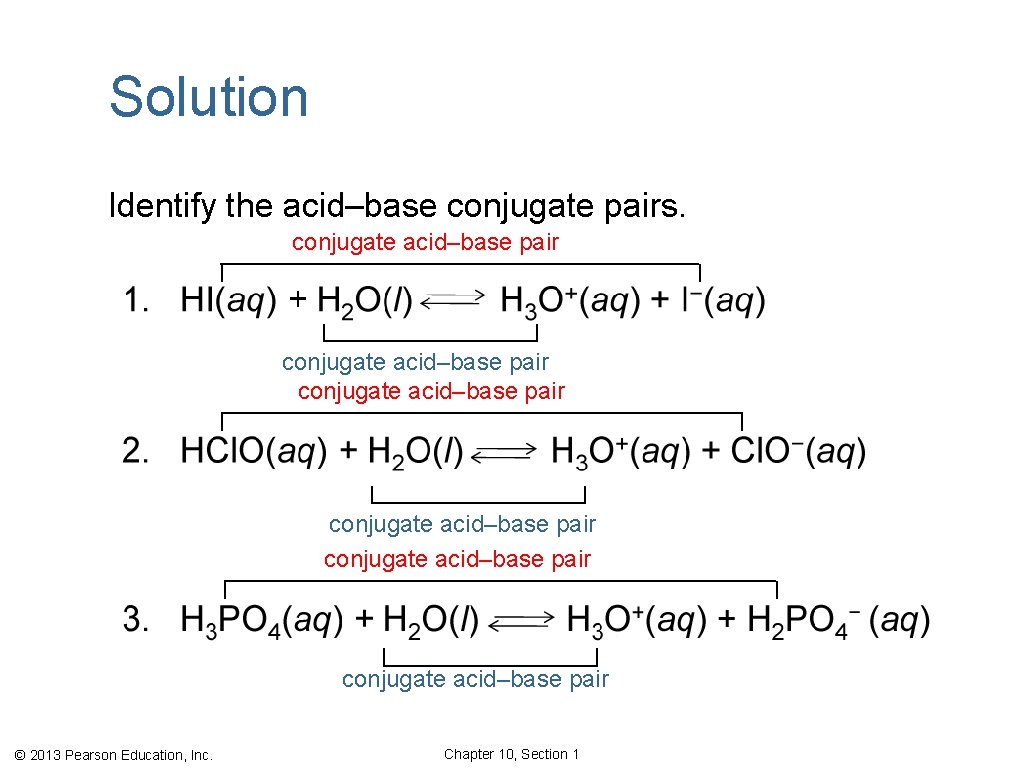
Solution Identify the acid–base conjugate pairs. conjugate acid–base pair conjugate acid–base pair © 2013 Pearson Education, Inc. Chapter 10, Section 1

Strengths of Acids • Strong acids completely ionize (100%) in aqueous solutions. We use a single arrow in the chemical equation. HCl(g) + H 2 O(l) H 3 O+(aq) + Cl−(aq) • Weak acids dissociate only slightly in water to form a solution of mostly molecules and a few ions. H 2 CO 3(aq) + H 2 O(l) © 2013 Pearson Education, Inc. H 3 O+(aq) + HCO 3−(aq) Chapter 10 Section 2
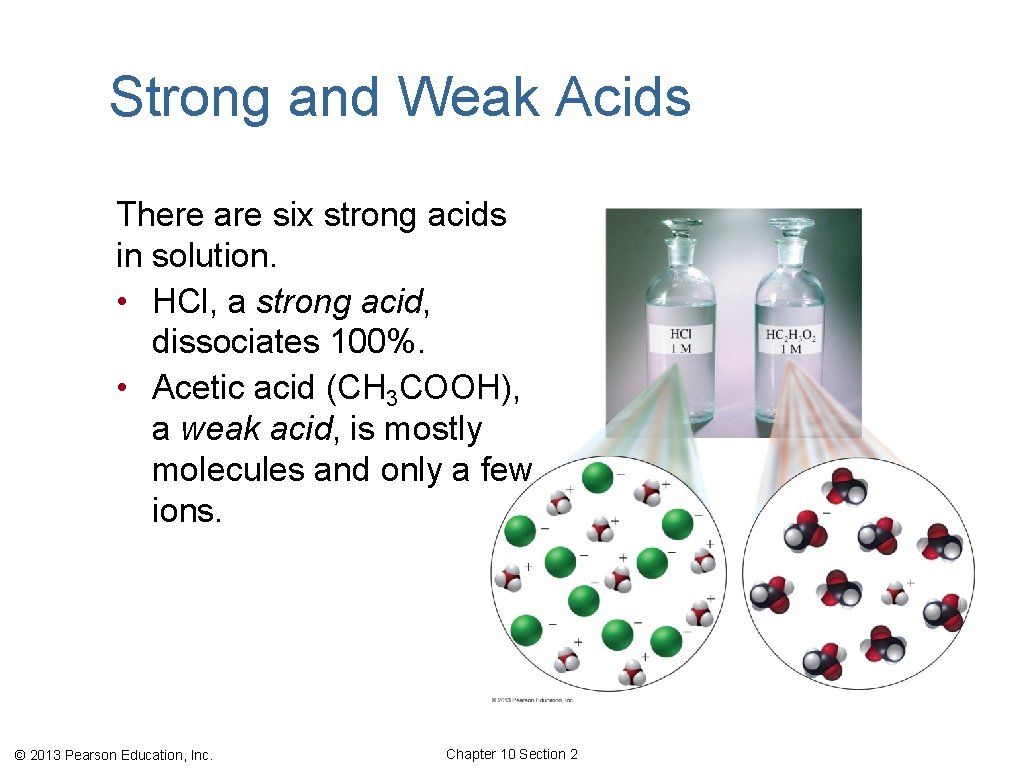
Strong and Weak Acids There are six strong acids in solution. • HCl, a strong acid, dissociates 100%. • Acetic acid (CH 3 COOH), a weak acid, is mostly molecules and only a few ions. © 2013 Pearson Education, Inc. Chapter 10 Section 2

Stronger Acid, Weaker Conjugate Base Weaker acids have a varying degree of strength. The stronger the acid, the weaker the conjugate base. © 2013 Pearson Education, Inc. Chapter 10 Section 2
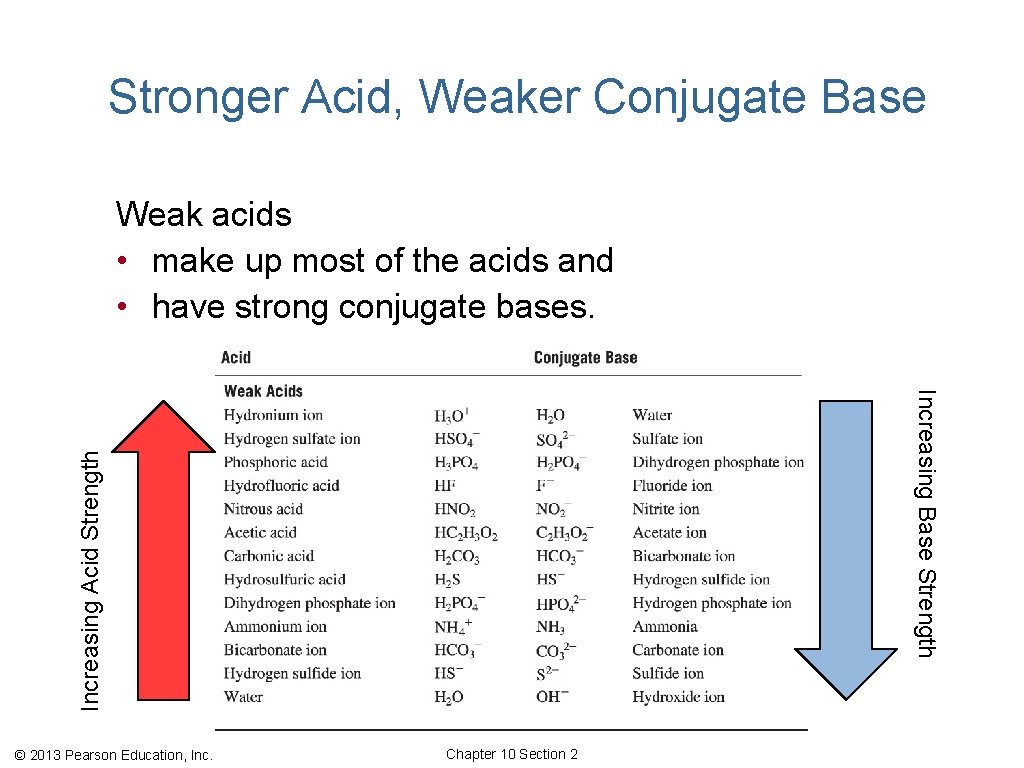
Stronger Acid, Weaker Conjugate Base Weak acids • make up most of the acids and • have strong conjugate bases. Increasing Acid Strength Increasing Base Strength © 2013 Pearson Education, Inc. Chapter 10 Section 2

H 2 CO 3, Weak Acid H 2 CO 3 is a diprotic acid. It partially dissociates in water, giving up one H+ at a time. H 2 CO 3(aq) + H 2 O(l) H 3 O+(aq) + HCO 3−(aq) Because HCO 3− is also a weak acid, a second dissociation can take place to produce another hydronium ion and the carbonate ion. HCO 3−(aq) + H 2 O(l) © 2013 Pearson Education, Inc. H 3 O+(aq) + CO 32−(aq) Chapter 10 Section 2

H 2 SO 4, Strong Acid H 2 SO 4 is also a diprotic acid. It completely dissociates in water. H 2 SO 4 (aq) + H 2 O(l) H 3 O+(aq) + HSO 4−(aq) Because HSO 4− is a weak acid, a second dissociation takes place to produce another hydronium ion and the sulfate ion. HSO 4−(aq) + H 2 O(l) © 2013 Pearson Education, Inc. H 3 O+(aq) + SO 42−(aq) Chapter 10 Section 2

Strong Bases Strong bases • are formed from metals of Groups 1 A(1) and 2 A(2). • include Li. OH, Na. OH, KOH, and Ca(OH)2. • dissociate completely in water. KOH(s) © 2013 Pearson Education, Inc. K+(aq) + OH−(aq) Chapter 10 Section 2

Weak Bases Weak bases • are most other bases. • dissociate only slightly in water. • form only a few ions in water. NH 3(g) + H 2 O(l) © 2013 Pearson Education, Inc. NH 4+(aq) + OH−(aq) Chapter 10 Section 2

Learning Check Identify each of the following as a strong or weak acid or base. A. HBr B. HNO 2 C. Na. OH D. H 2 SO 4 E. Cu(OH)2 © 2013 Pearson Education, Inc. Chapter 10 Section 2

Solution Identify each of the following as a strong or weak acid or base. A. HBr strong acid B. HNO 2 weak acid C. Na. OH strong base D. H 2 SO 4 strong acid E. Cu(OH)2 weak base © 2013 Pearson Education, Inc. Chapter 10 Section 2

Acid Dissociation Constant, Ka • In a weak acid, the rate of the dissociation of the acid is equal to the rate of the association. HA + H 2 O H 3 O + + A – • The equilibrium expression is indicated below. © 2013 Pearson Education, Inc. Chapter 10 Section 2

Ka, Formic Acid • Formic acid, HCHO 2, the acid found in bee and ant stings, is a weak acid. • The equilibrium expression is indicated below © 2013 Pearson Education, Inc. Chapter 10 Section 2
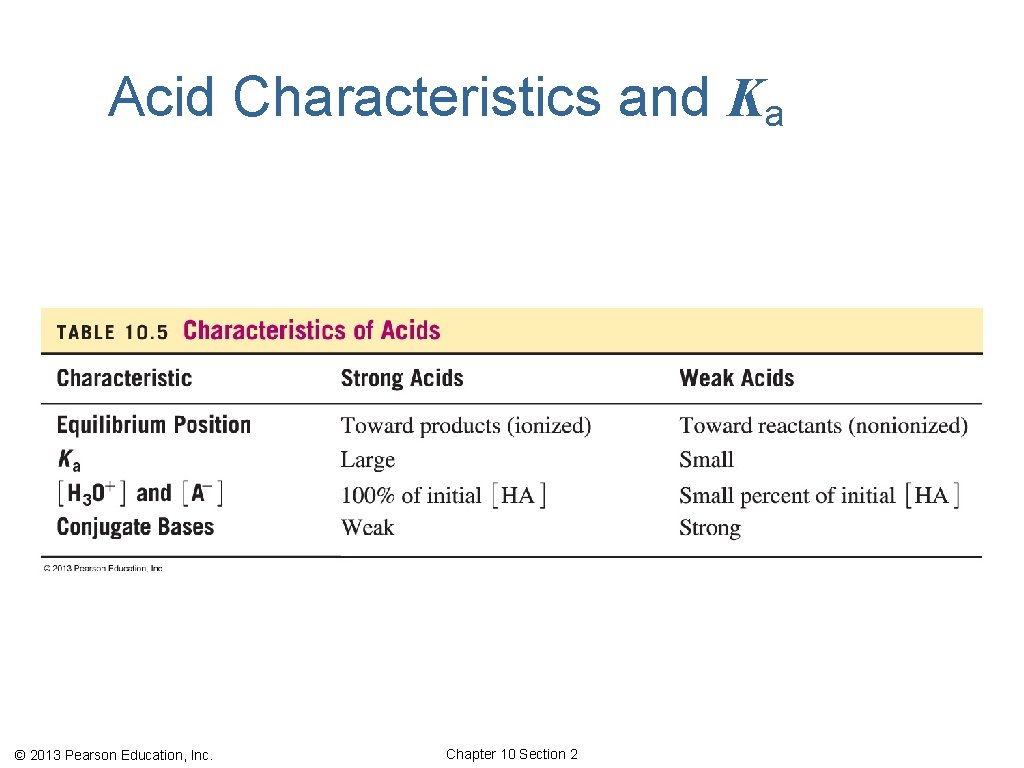
Acid Characteristics and Ka © 2013 Pearson Education, Inc. Chapter 10 Section 2
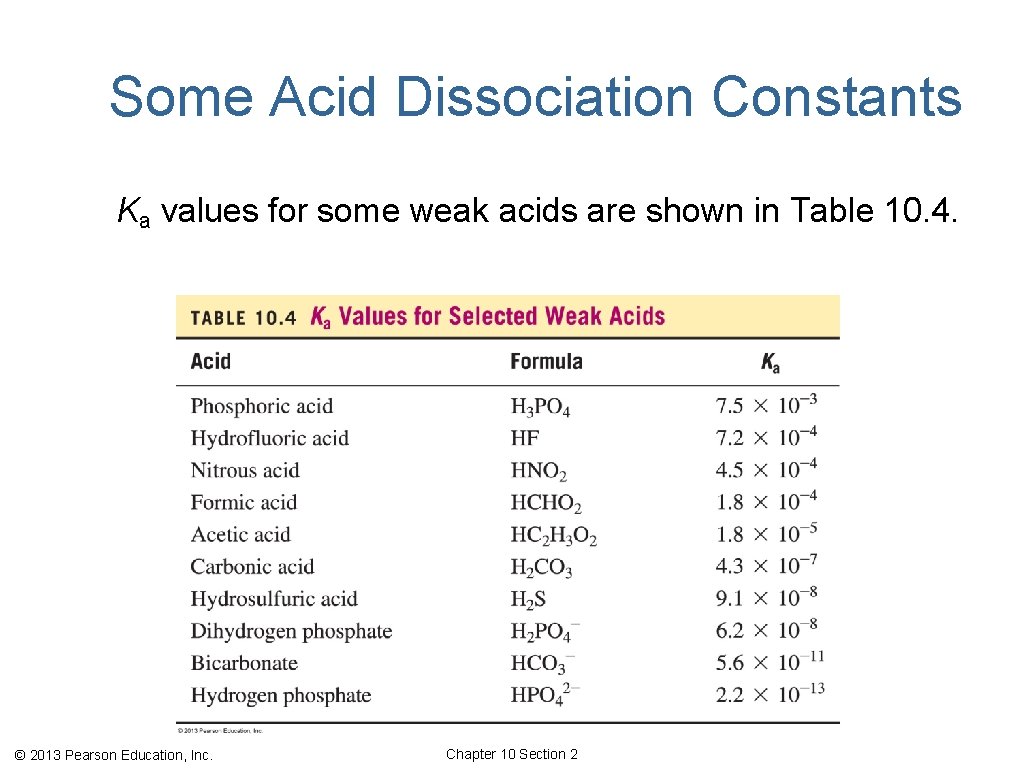
Some Acid Dissociation Constants Ka values for some weak acids are shown in Table 10. 4. © 2013 Pearson Education, Inc. Chapter 10 Section 2

Writing Ka for a Weak Acid Write the Ka for H 2 S. 1. Write the equation for the dissociation of H 2 S. 2. Set up the Ka expression. © 2013 Pearson Education, Inc. Chapter 10 Section 2

Learning Check Write the Ka for hypochlorous acid, HCl. O. © 2013 Pearson Education, Inc. Chapter 10 Section 2

Solution Write the Ka for hypochlorous acid, HCl. O. 1. Write the equation for the dissociation of the weak acid, HCl. O. 2. Set up the Ka expression. © 2013 Pearson Education, Inc. Chapter 10 Section 2

Ionization of Water In the ionization of water, • H+ is transferred from one H 2 O molecule to another. • one water molecule acts as an acid, while another acts as a base. © 2013 Pearson Education, Inc. Chapter 10 Section 3

Ion Product of Water, Kw The ion product constant, Kw, for water at 25 °C • is the product of the concentrations of the hydronium and hydroxide ions. • is obtained from the concentrations in pure water. © 2013 Pearson Education, Inc. Chapter 10 Section 3
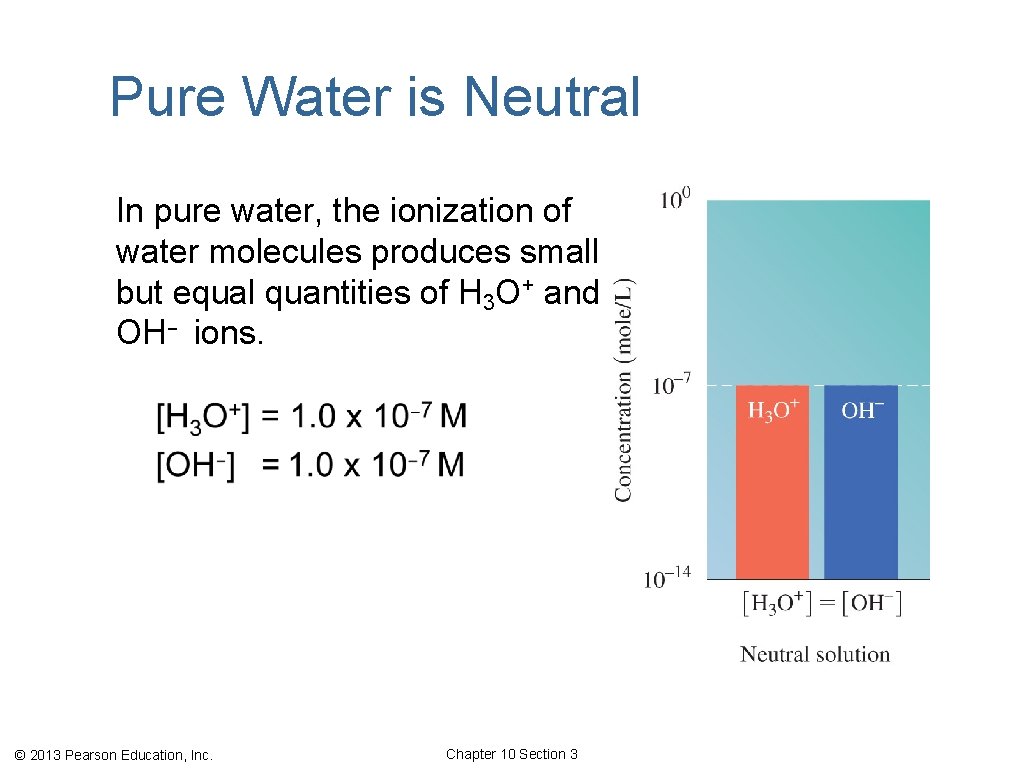
Pure Water is Neutral In pure water, the ionization of water molecules produces small but equal quantities of H 3 O+ and OH− ions. © 2013 Pearson Education, Inc. Chapter 10 Section 3
![Acidic Solutions Adding an acid to pure water • increases the [H 3 O+], Acidic Solutions Adding an acid to pure water • increases the [H 3 O+],](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-44.jpg)
Acidic Solutions Adding an acid to pure water • increases the [H 3 O+], • decreases the [OH−], and • causes the [H 3 O+] to exceed 1. 0 x 10− 7 M. © 2013 Pearson Education, Inc. Chapter 10 Section 3
![Basic Solutions Adding a base to pure water, • increases the [OH−], • decreases Basic Solutions Adding a base to pure water, • increases the [OH−], • decreases](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-45.jpg)
Basic Solutions Adding a base to pure water, • increases the [OH−], • decreases the [H 3 O+], and • causes the [OH−] to exceed 1. 0 x 10− 7 M. © 2013 Pearson Education, Inc. Chapter 10 Section 3
![Comparison of [H 3 © 2013 Pearson Education, Inc. + O] Chapter 10 Section Comparison of [H 3 © 2013 Pearson Education, Inc. + O] Chapter 10 Section](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-46.jpg)
Comparison of [H 3 © 2013 Pearson Education, Inc. + O] Chapter 10 Section 3 and − [OH ]
![[H 3 + O] and − [OH ] in Solutions In neutral, acidic, or [H 3 + O] and − [OH ] in Solutions In neutral, acidic, or](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-47.jpg)
[H 3 + O] and − [OH ] in Solutions In neutral, acidic, or basic solutions, the Kw at 25 °C is always 1. 0 x 10− 14. © 2013 Pearson Education, Inc. Chapter 10 Section 3

Guide to Calculating [H 3 © 2013 Pearson Education, Inc. Chapter 10 Section 3 + O] and − [OH ]
![Calculating [H 3 + O] What is the [H 3 O+] of a solution Calculating [H 3 + O] What is the [H 3 O+] of a solution](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-49.jpg)
Calculating [H 3 + O] What is the [H 3 O+] of a solution if [OH−] is 5. 0 x 10− 8 M? Step 1 Write the Kw for water. Step 2 Solve the Kw for the unknown [H 3 O+]. Step 3 Substitute the known [OH−] into the equation and calculate. © 2013 Pearson Education, Inc. Chapter 10 Section 3

p. H Scale The p. H of a solution • is used to indicate the acidity of a solution. • has values that range from 0 to 14 representing the H 3 O+ concentration of a solution. • is acidic when the values are less than 7. • is neutral with a p. H of 7. • is basic when the values are greater than 7. © 2013 Pearson Education, Inc. Chapter 10, Section 4
![Calculating p. H is the negative logarithm (base 10) of the [H 3 O+]. Calculating p. H is the negative logarithm (base 10) of the [H 3 O+].](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-51.jpg)
Calculating p. H is the negative logarithm (base 10) of the [H 3 O+]. For a solution with [H 3 O+] = 1 x 10− 4 M: © 2013 Pearson Education, Inc. Chapter 10, Section 4
![Learning Check The [H 3 O+] of tomato juice is 2 x 10− 4 Learning Check The [H 3 O+] of tomato juice is 2 x 10− 4](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-52.jpg)
Learning Check The [H 3 O+] of tomato juice is 2 x 10− 4 M. What is the p. H of the solution? A. 4. 0 B. 3. 7 C. 10. 3 © 2013 Pearson Education, Inc. Chapter 10, Section 4
![Learning Check What is the p. H of coffee if the [H 3 O+] Learning Check What is the p. H of coffee if the [H 3 O+]](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-53.jpg)
Learning Check What is the p. H of coffee if the [H 3 O+] is 1 x 10− 5 M? A. p. H = 9. 0 B. p. H = 7. 0 C. p. H = 5. 0 © 2013 Pearson Education, Inc. Chapter 10, Section 4
![Learning Check The [OH−] of a solution is 1. 0 x 10− 3 M. Learning Check The [OH−] of a solution is 1. 0 x 10− 3 M.](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-54.jpg)
Learning Check The [OH−] of a solution is 1. 0 x 10− 3 M. What is the p. H? A. 3. 00 B. 11. 00 C. – 11. 00 © 2013 Pearson Education, Inc. Chapter 10, Section 4
![[H 3 + O ], © 2013 Pearson Education, Inc. [OH ], and p. [H 3 + O ], © 2013 Pearson Education, Inc. [OH ], and p.](http://slidetodoc.com/presentation_image_h2/7f7e743b8cd2395a097df0f777bd2a98/image-55.jpg)
[H 3 + O ], © 2013 Pearson Education, Inc. [OH ], and p. H Values Chapter 10, Section 4

Buffers A buffer solution • maintains p. H by neutralizing added acid or base. • in the body, absorbs H 3 O+ or OH− from foods and cellular processes to maintain p. H. © 2013 Pearson Education, Inc. Chapter 10, Section 6

Buffers (continued) When an acid or base is added • to water, the p. H changes drastically. • to a buffer solution, the p. H does not change very much; p. H is maintained. © 2013 Pearson Education, Inc. Chapter 10, Section 6
- Slides: 57