Acids Bases AP Chemistry Acids Bases General Knowledge
Acids & Bases AP Chemistry

Acids & Bases (General Knowledge). Acids taste sour, are corrosive, and they turn blue indicators (vegetable colors, papers, etc. . ) red All these mean acid: H+, proton, H 3 O+ Bases (alkalies) feel soapy, taste bitter, and react with acids to produce salts "alkali'' is the arabic word for the ashes from burning certain plants. Potash (potassium carbonate) is a common form.

Acids & Bases (General Knowledge). A Salt is any ionic compound whose cation could come from a base and whose anion could come from an acid in an acid-base reaction An acid and a base react to form a salt & water, and maybe CO 2 if carbonates are involved. HCl + Na. OH Na. Cl + H 2 O HC 2 H 3 O 2 + KOH KC 2 H 3 O 2 + H 2 O
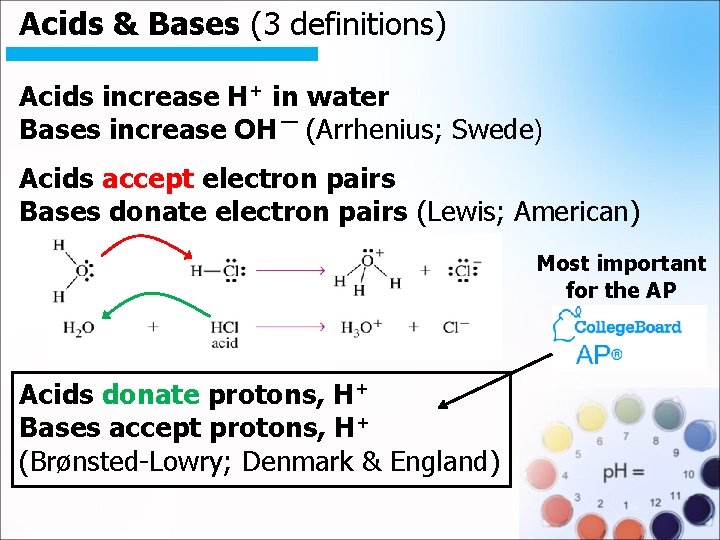
Acids & Bases (3 definitions). Acids increase H+ in water Bases increase OH ¯ (Arrhenius; Swede) Acids accept electron pairs Bases donate electron pairs (Lewis; American) Most important for the AP Acids donate protons, H+ Bases accept protons, H+ (Brønsted-Lowry; Denmark & England)
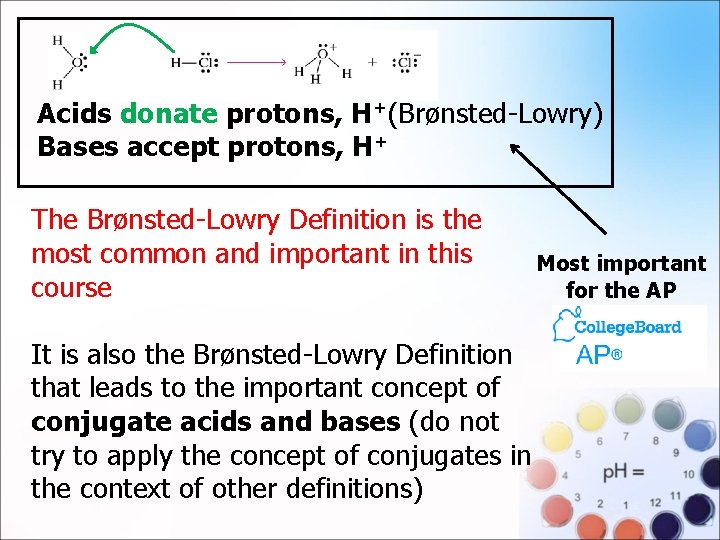
. Acids donate protons, H+(Brønsted-Lowry) Bases accept protons, H+ The Brønsted-Lowry Definition is the most common and important in this course It is also the Brønsted-Lowry Definition that leads to the important concept of conjugate acids and bases (do not try to apply the concept of conjugates in the context of other definitions) Most important for the AP
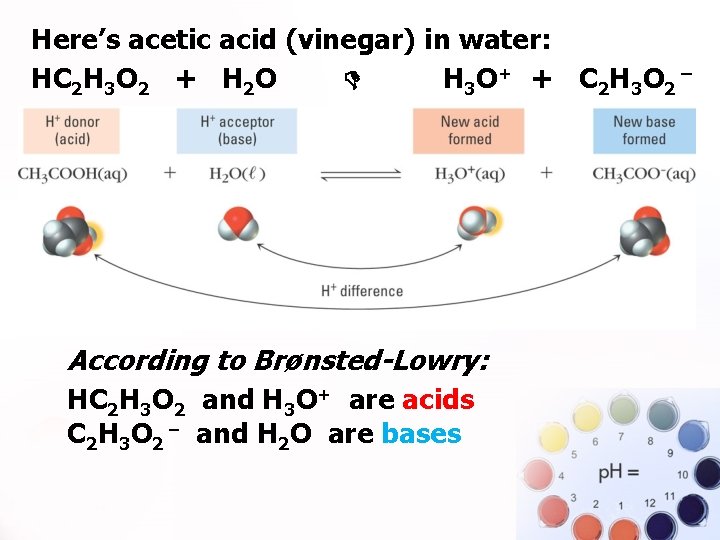
Here’s acetic acid (vinegar) in water: HC 2 H 3 O 2 + H 2 O H 3 O+ + C 2 H 3 O 2 ¯. According to Brønsted-Lowry: HC 2 H 3 O 2 and H 3 O+ are acids C 2 H 3 O 2 and H 2 O are bases

Look at this reversible reaction for ammonia: . NH 3 + H 2 O NH 4+ + According to Brønsted-Lowry: NH 3 and OH are bases. H 2 O and NH 4+ are acids OH ¯
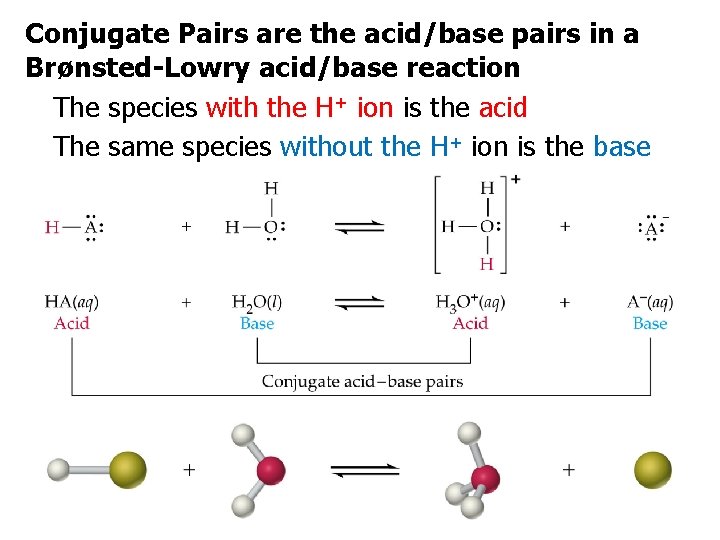
Conjugate Pairs are the acid/base pairs in a Brønsted-Lowry acid/base reaction The species with the H+ ion is the acid The same species without the H+ ion is the base.
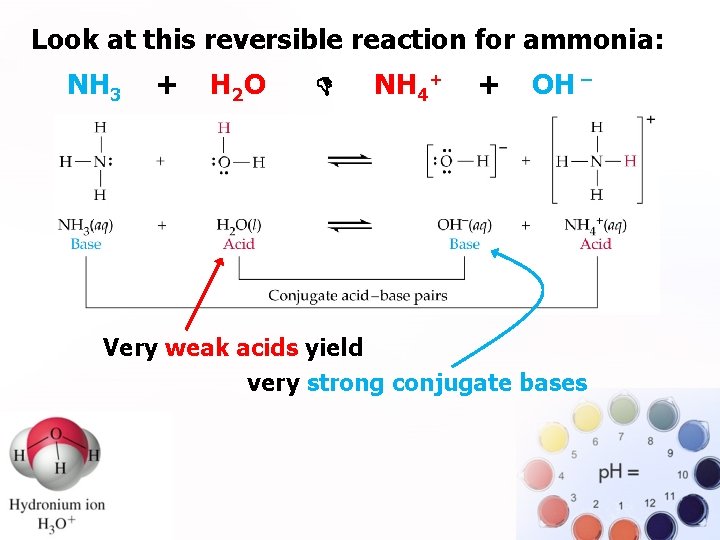
Look at this reversible reaction for ammonia: . NH 3 + H 2 O NH 4+ + OH ¯ Very weak acids yield very strong conjugate bases

. Notice that moderately weak acids or bases yield moderately strong conjugates… and vice versa.

A few conjugates (identify bases as strong or weak). Acid HC 2 H 3 O 2 NH 4+ H 3 O+ H 2 O and and Base C 2 H 3 O 2 ¯ NH 3 H 2 O OH ¯ (strong or weak base? ) Water acts as either an acid or a base.

Water Auto-ionization. Water naturally ionizes into H+ and OH ¯ ions: H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH ¯(aq) In pure water, the reverse reaction is favored. At equilibrium: [H 3 O+] = 1. 0 10 -7 moles/L [OH ¯] = 1. 0 10 -7 moles/L From an equilibrium perspective: Kw = [H 3 O+] [OH ¯] = 1. 0 x 10 -14 Kw = Ka Kb = 1. 0 x 10 -14 This is true for water or aqueous solutions

Where p. H comes in: . We state the concentration of H+ ions in water as its "p. H" which is based on the exponent on the 10. * For pure water: [H+] = 1. 0 x 10 -7 moles/L & p. H = -log[H+] * So in pure water the p. H = 7. 0 (it’s the exponent) Logs are the exponents on the 10. . . Example: 102. 3 = 199. 5 As a Log function: log(199. 5) = 2. 3 * Technically p. H depends on the chemical activity a of the substance, just as the equilibrium expression did (see your equilibrium notes). The chemical activity a is still a = γ where γ is a unit-less activity coefficient and the units of concentration/standard concentration (molarity/molarity) cancel out. Therefore p. H is unit-less. So far in the course, the equilibrium K’s and p. H depend on chemical activities and are unit-less.

If we increased the H+ ion in pure water from 0. 0000001 to 0. 001 (from 10 -7 to 10 -3), the p. H is now 3, a stronger acid. p. H = -log(0. 001 mols/L) = -(-3) = 3. But the product is still the same: (1. 0 x 10 -3) (1. 0 x 10 -11) = 1. 0 x 10 -14 So the p. H and p. OH always add up to 14 Kw = [H 3 O+] [OH ¯] = 1. 0 x 10 -14 Kw = Ka Kb = 1. 0 x 10 -14 p. H from 1 to 7 p. H 7 = neutral p. H from 7 to 14 = acid = base
![Important: . Increasing p. H means decreasing [H+], which means fewer H+ ions and Important: . Increasing p. H means decreasing [H+], which means fewer H+ ions and](http://slidetodoc.com/presentation_image_h2/3e226f972e953012702b44a768a05d53/image-15.jpg)
Important: . Increasing p. H means decreasing [H+], which means fewer H+ ions and the solution is less acidic (or, it’s basic) Decreasing p. H means the [H+] is increasing, which means that there are more H+ ions and the solution is more acidic
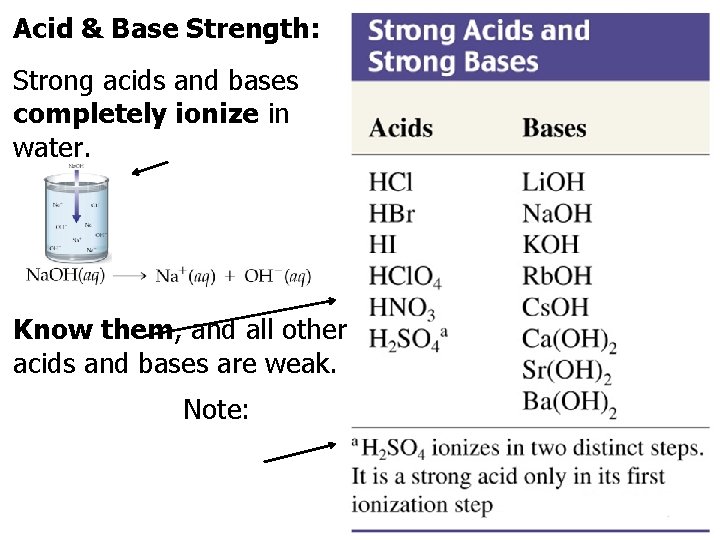
Acid & Base Strength: . Strong acids and bases completely ionize in water. Know them, and all other acids and bases are weak. Note:

Finding the p. H of STRONG Acids & Bases: . For strong acids & bases, it’s pretty easy: Strong acids & bases dissociate* completely, so the final concentration of H+ or OH¯ ions will be the same as the initial concentration of the strong acid or base. Example: In a 0. 040 -molar solution of HCl H+ + Cl¯ 0. 040 M + 0. 040 M So [HCl] is the same as [H+] = 0. 040 M p. H = -log [H+] = -log (0. 040) = 1. 4 * Technically the term dissociate is best applied to ionic substances. The more general term ionize is often used in the context of molecular substances. Within the scope of this course, however, the two terms may be used interchangeably.

Finding the p. H of STRONG Acids & Bases: . To get the p. H of a strong base: 1. Get the p. OH first 2. Subtract the p. OH from 14 to get the p. H Calculate the p. H of a 0. 050 M solution of KOH. p. OH = - log (0. 050) p. OH = 1. 30 So the p. H = 14. 00 - 1. 30 = 12. 70 Can you go backwards? Calculate the Molarity of a HBr solution with a p. H of 2. 45:

Finding the p. H of STRONG Acids & Bases: . Can you go backwards? Calculate the Molarity of a HBr solution with a p. H of 2. 45: p. H = -log[x] ; or -2. 45 = log[x] Calculator Tip: It’s the inverse of the “log” function Enter the 2. 45, make it negative, press the "10 x" key. so [HBr] = x = 3. 5 x 10 -3

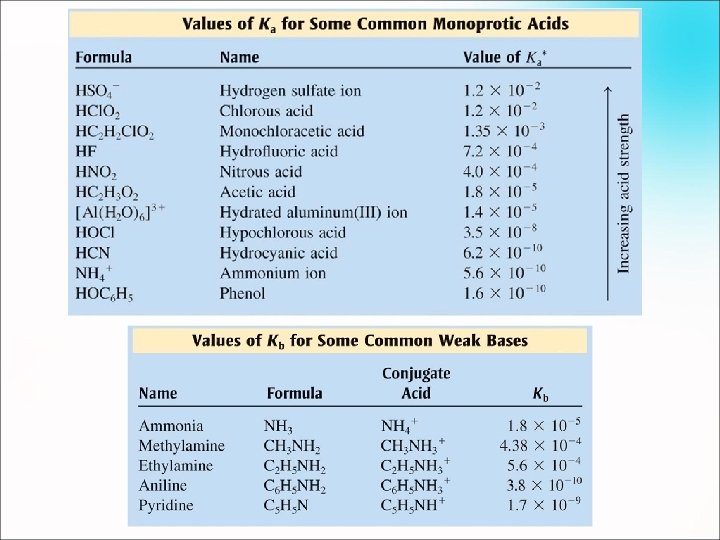
Finding the p. H of WEAK Acids & Bases: When a weak acid is placed in water, a small fraction of its molecules will ionize into hydrogen ions (H+) and conjugate base ions (B ¯). Most of the acid molecules will remain in solution as undissociated aqueous particles. Ka and Kb are measures of the strengths of weak acids and bases
![Here’s a generic weak acid equilibrium: HA H+ + A ¯ [A ¯] = Here’s a generic weak acid equilibrium: HA H+ + A ¯ [A ¯] =](http://slidetodoc.com/presentation_image_h2/3e226f972e953012702b44a768a05d53/image-21.jpg)
Here’s a generic weak acid equilibrium: HA H+ + A ¯ [A ¯] = molarity of conjugate base ions [HA] = molarity of undissociated acid molecules Here’s a generic weak base equilibrium: B + H 2 O HB+ + OH ¯ [HB+] = protonated base ions (M) [OH ¯] = molarity of hydroxide ions [B] = unprotonated base molecules The greater the value of K , the greater the extent of the dissociation and the greater the strength of the weak acid or base.

Finding the p. H of WEAK Acids (and Bases). Remember ICE Initial Change Equilibrium You will also need to know the Ka (given or easily looked up) and the Molarity for a weak acid, then you can find the p. H.
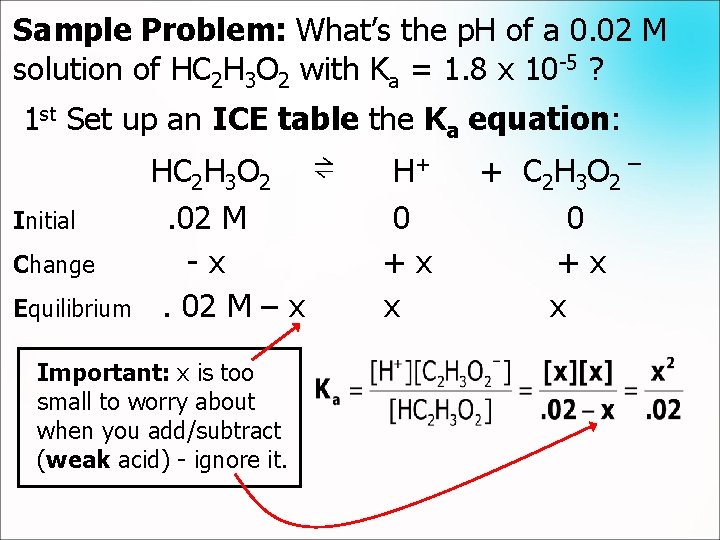
Sample Problem: What’s the p. H of a 0. 02 M solution of HC 2 H 3 O 2 with Ka = 1. 8 x 10 -5 ? 1 st Set up an ICE table the Ka equation: Initial Change Equilibrium HC 2 H 3 O 2. 02 M -x. 02 M – x Important: x is too small to worry about when you add/subtract (weak acid) - ignore it. ⇌ H+ 0 +x x + C 2 H 3 O 2 ¯ 0 +x x

Sample Problem: What’s the p. H of a 0. 02 M solution of HC 2 H 3 O 2 with Ka = 1. 8 x 10 -5 ? Now just solve: Important: Notice that x is less than 5% of 0. 02, so we have confirmed our early assumption was good. Now that we know [H+], we can calculate the p. H: p. H = -log [H+] = -log (6. 0 x 10 -4) = 3. 2

For a weak base, do everything the same except you’ll end up with the p. OH. Remember to subtract it from 14 to get the p. H. The p. H should be between 7 and 14. Also, remember that: So you can quickly find the Kb for the conjugate base if you know the Ka for the weak acid.

Polyprotic Acids with more than one H+ to give. Examples: H 2 SO 4 and H 3 PO 4 Polyprotic acids ionize in successive steps. For phosphoric acid, the steps and their respective Ka values are: H 3 PO 4(aq) H+ (aq) + H 2 PO 4¯ (aq) H 2 PO 4 ¯(aq) H+(aq) + HPO 42 ¯ (aq) H+(aq) + PO 43 ¯(aq) Ka 1 = 7. 5 x l 0 -5 Ka 2 = 6. 2 x 10 -8 Ka 3 = 4. 8 x l 0 -13
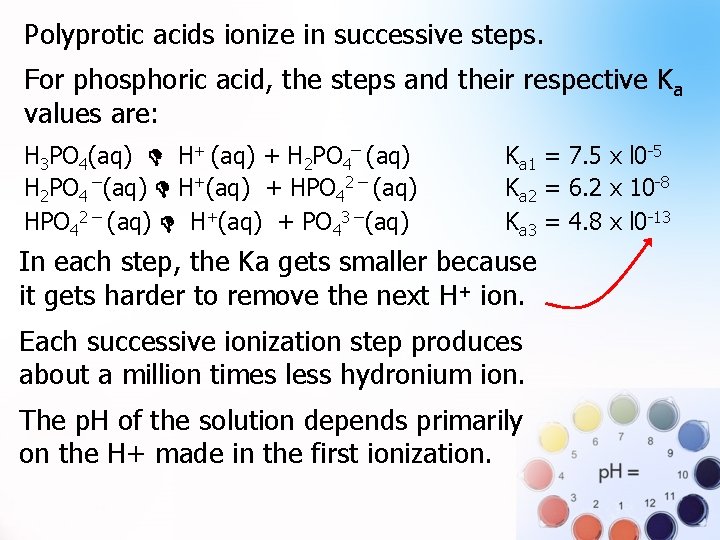
Polyprotic acids ionize in successive steps. . For phosphoric acid, the steps and their respective Ka values are: H 3 PO 4(aq) H+ (aq) + H 2 PO 4¯ (aq) H 2 PO 4 ¯(aq) H+(aq) + HPO 42 ¯ (aq) H+(aq) + PO 43 ¯(aq) Ka 1 = 7. 5 x l 0 -5 Ka 2 = 6. 2 x 10 -8 Ka 3 = 4. 8 x l 0 -13 In each step, the Ka gets smaller because it gets harder to remove the next H+ ion. Each successive ionization step produces about a million times less hydronium ion. The p. H of the solution depends primarily on the H+ made in the first ionization.

Amphoteric Substances that can act as either acids or bases H 2 PO 4 ¯ can act as an acid, giving up a proton to become HPO 42 ¯ It can also act as a base, accepting a proton to become H 3 PO 4 H 2 O can act as an acid, giving up a proton to become OH ¯ It can also act as a base, accepting a proton to become H 3 O+

Other Acid-Base Formation Reactions. These do not involve clear Bronsted-Lowry type conjugates or patterns – just know them Nonmetal oxides are acid anhydrides: CO 2 + H 2 O H 2 CO 3 (Carbonic acid) SO 3 + H 2 O H+ + HSO 4 ¯ (Sulfurous acid) Descriptive Chem Tip! The non metal will have the same Oxidation# in the product! Metals oxides are basic anhydrides: Ca. O + H 2 O Ca(OH)2 Na 2 O +H 2 O 2 Na+ + 2 OH ¯

Salt Solutions (Neutral, Acidic, & Basic). Salts composed of a strong acid’s conjugate and strong base’s conjugate will be neutral (Na. Cl).

Salts composed of a strong acid’s conjugate and strong base’s conjugate will be neutral (Na. Cl). . Cl ¯ is the conjugate of HCl (strong acid) Na+ is the conjugate of Na. OH (strong base) Neither ion will react with water HCl 2 O ¯ Na+ + H 2 O Cl ¯ Na+ + H 2 O Two ways to show an ion in water There is no H+ or OH ¯ in the solution!

Salt Solutions (Neutral, Acidic, & Basic). Salts composed of a strong acid’s conjugate and strong base’s conjugate will be neutral (Na. Cl). Salts composed of a weak base’s conjugate and a strong acid’s conjugate will be acidic (NH 4 Cl).

Salts composed of a weak base’s conjugate and a strong acid’s conjugate will be acidic (NH 4 Cl). . NH 4+ is the conjugate of NH 3 (weak base) Cl ¯ is the conjugate of HCl (strong acid) Only NH 4+ is strong enough to will react with water NH 4+ H 2 O NH 3 + H+ Cl ¯ + H 2 O There is now a little H+ in the solution! Note: NH 4+ will act like an acid. The NH 3 produced will only take up the H+ again – it will not act as a base to form OH ¯ with water. The solution is acidic.

Salt Solutions (Neutral, Acidic, & Basic). Salts composed of a strong acid’s conjugate and strong base’s conjugate will be neutral (Na. Cl). Salts composed of a weak base’s conjugate and a strong acid’s conjugate will be acidic (NH 4 Cl). Salts composed of a weak acid’s conjugate and a strong base’s conjugate will be basic (Na. C 2 H 3 O 2).
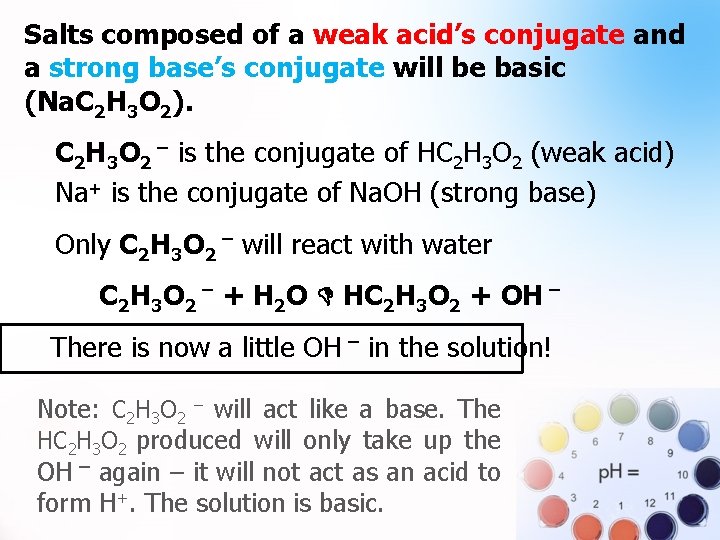
Salts composed of a weak acid’s conjugate and a strong base’s conjugate will be basic (Na. C 2 H 3 O 2). . C 2 H 3 O 2 ¯ is the conjugate of HC 2 H 3 O 2 (weak acid) Na+ is the conjugate of Na. OH (strong base) Only C 2 H 3 O 2 ¯ will react with water C 2 H 3 O 2 ¯ + H 2 O HC 2 H 3 O 2 + OH ¯ There is now a little OH ¯ in the solution! Note: C 2 H 3 O 2 ¯ will act like a base. The HC 2 H 3 O 2 produced will only take up the OH ¯ again – it will not act as an acid to form H+. The solution is basic.

Salt Solutions (Neutral, Acidic, & Basic). Salts composed of a strong acid’s conjugate and strong base’s conjugate will be neutral (Na. Cl). Salts composed of a weak base’s conjugate and a strong acid’s conjugate will be acidic (NH 4 Cl). Salts composed of a weak acid’s conjugate and a strong base’s conjugate will be basic (Na. C 2 H 3 O 2). Overall: conjugates of strong acids/bases are neutral and irrelevant.

Salt Solutions (Neutral, Acidic, & Basic). Salts composed of a strong acid’s conjugate and strong base’s conjugate will be neutral (Na. Cl). Salts composed of a weak base’s conjugate and a strong acid’s conjugate will be acidic (NH 4 Cl). Salts composed of a weak acid’s conjugate and a strong base’s conjugate will be basic (Na. C 2 H 3 O 2). The p. H of salts composed of a weak acid’s conjugate and a weak base’s conjugate will depend on Ka and Kb.

AP Multiple Choice Question: 1999 #62 (31% got this correct). HC 2 H 3 O 2(aq) + CN–(aq) HCN(aq) + C 2 H 3 O 2–(aq) The reaction represented above has an equilibrium constant equal to 3. 7 104. Which of the following can be concluded from this information? (a) CN–(aq) is a stronger base than C 2 H 3 O 2–(aq) (b) HCN(aq) is a stronger acid than HC 2 H 3 O 2 (aq) (c) The conjugate base of CN–(aq) is C 2 H 3 O 2–(aq) (d) The equilibrium constant will increase with an increase in temperature (e) The p. H of a solution containing equimolar amounts of CN–(aq) and HC 2 H 3 O 2(aq) is 7. 0
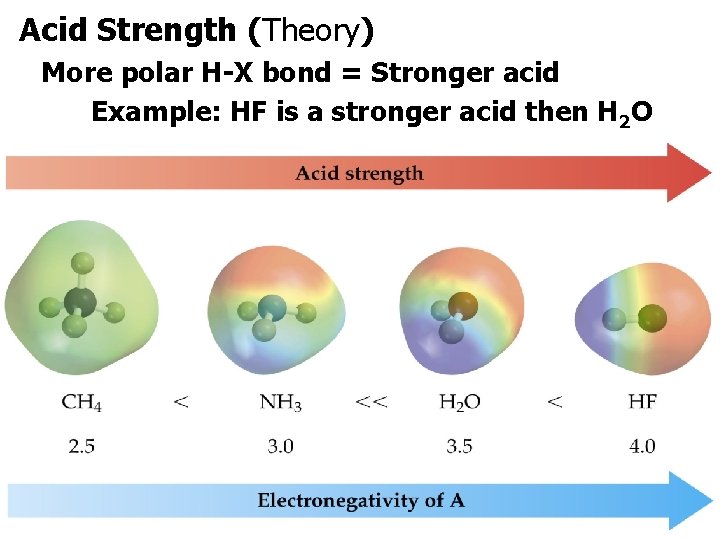
Acid Strength (Theory) More polar H-X bond = Stronger acid Example: HF is a stronger acid then H 2 O.
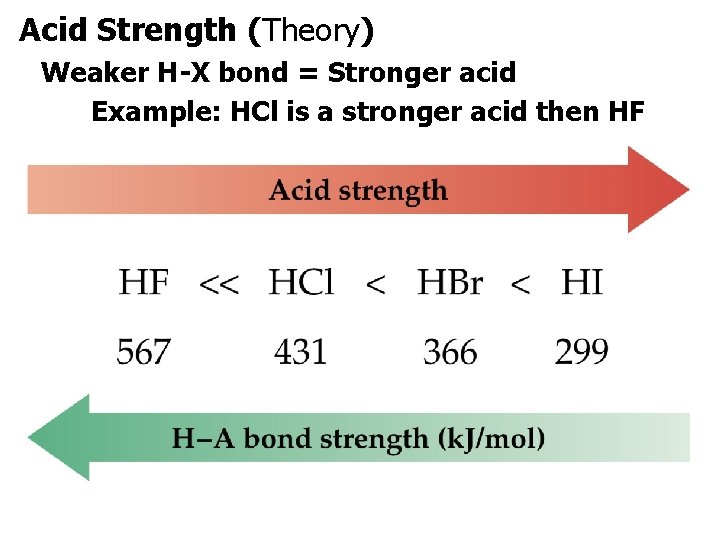
Acid Strength (Theory) Weaker H-X bond = Stronger acid Example: HCl is a stronger acid then HF.
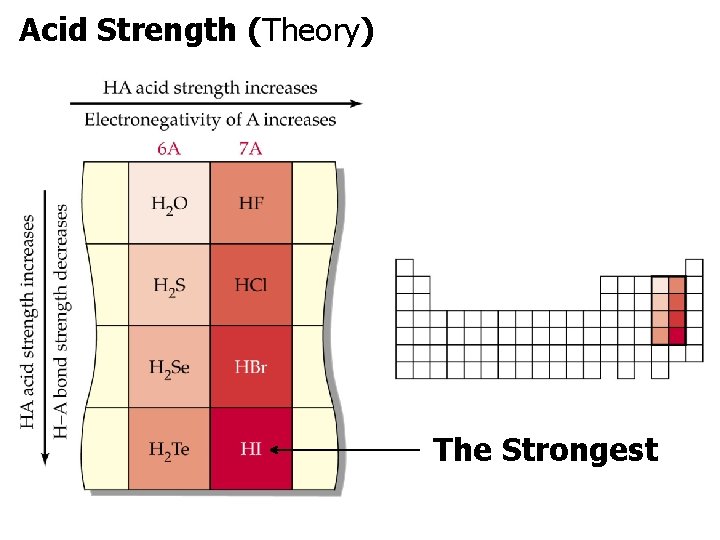
Acid Strength (Theory). The Strongest

Acid Strength (Theory) Oxoacids: hydrogen, oxygen and one other element (H-O-E) Higher electronegativity on E = Stronger Acid.
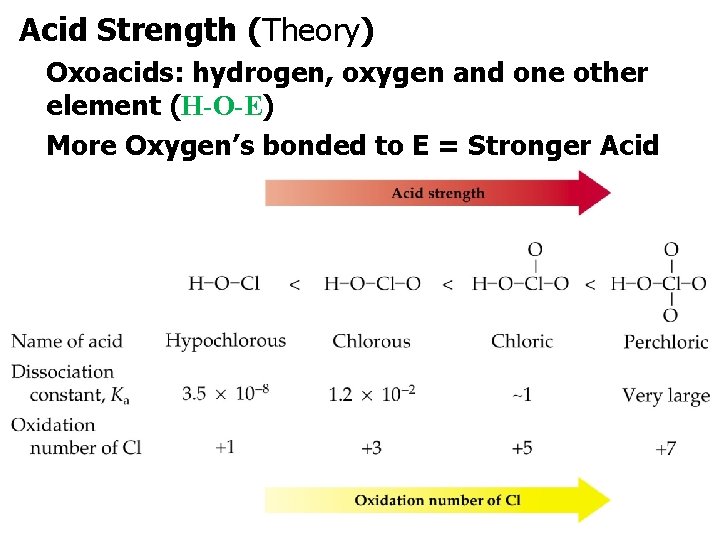
Acid Strength (Theory) Oxoacids: hydrogen, oxygen and one other element (H-O-E) More Oxygen’s bonded to E = Stronger Acid.

AP Multiple Choice Question: 1994 #61 (21% got this correct). A 1–molar solution of which of the following salts has the highest p. H? (A) Na. NO 3 (B) Na 2 CO 3 (D) Na. HSO 4 (E) Na 2 SO 4 (C) NH 4 Cl
- Slides: 45