ACIDS BASES and SALTS Slide numbering system Slide

ACIDS, BASES and SALTS

Slide numbering system Slide title: A, B, C… - Points on a slide: 1, 2, 3, - How to find it on your note handout: A 1, A 2, B 1, B 2…

A. What you need to know? A 1. What is a/an acid, base, salt? A 2. How to name them A 3. Acid, base, salt properties A 4. Identify at least 3 products that are acids, bases, and salts used in daily life

B. Common Acids Name Formula Organic(= Carbon-containing) acids acetic acid (vinegar) HC 2 H 3 O 2 citric acid HC 6 H 7 O 2 ascorbic acid HC 6 H 7 O 6 lactic acid HC 3 H 5 O 3 acetylsalicylic acid HC 9 H 7 O 4 Inorganic (=no carbon) acids sulfuric acid H 2 SO 4 carbonic acid H 2 CO 3 phosphoric acid H 3 PO 4 nitric acid HNO 3 hydrochloric acid HCl hydrobromic acid HBr hydrofluoric acid HF

C. What is an acid? C 1. An acid is a compound that releases hydrogen ions (H+) when added to water.
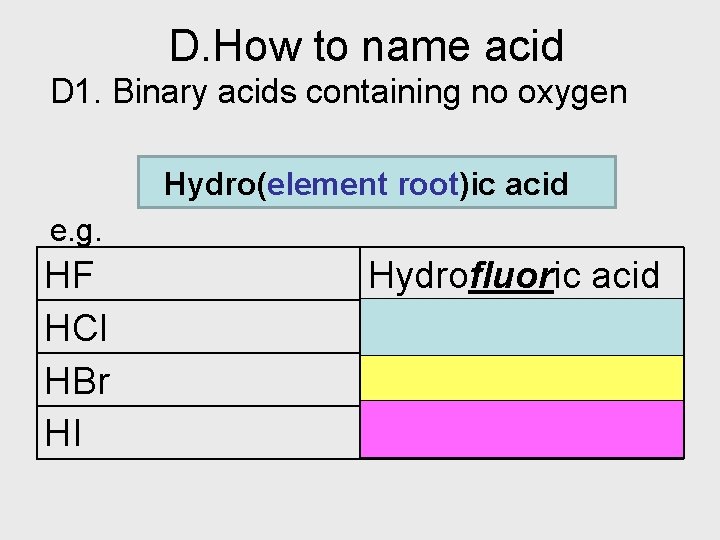
D. How to name acid D 1. Binary acids containing no oxygen Hydro(element root)ic acid e. g. HF HCl HBr HI Hydrofluoric acid Hydrochloric acid Hydrobromic acid Hydroiodic acid

How to name acid (cont. ) D 2. Oxoacids (oxygen-containing acids): (Non-ate part)ic acid Oxoacids HCl. O 3 HNO 3 H 2 CO 3 HBr. O 3 HIO 3 HC 2 H 3 O 2 Non-ate part Chlor. Nitr. Carbon. Brom. Iod. Acet- Names Chloric acid Nitric acid Carbonic acid Bromic acid Iodic acid Acetic acid

How to name acid (cont. ) D 3. Oxoacids that are slight exceptions to the naming rules of: (Non-ate part) + ic acid Oxoacids Non-ate part Names H 2 SO 4 Sulfuric acid H 3 PO 4 Phosphoric acid

Try again
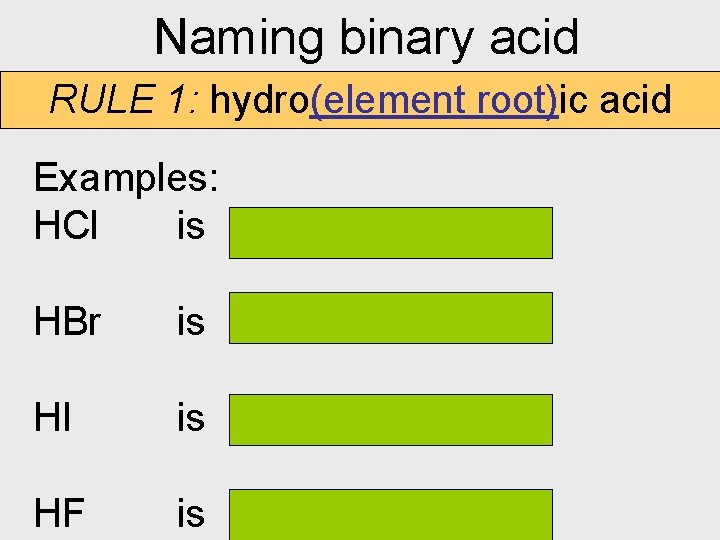
Naming binary acid RULE 1: hydro(element root)ic acid Examples: HCl is hydrochloric acid HBr is hydrobromic acid HI is hydroiodic HF is hydrofluoric acid
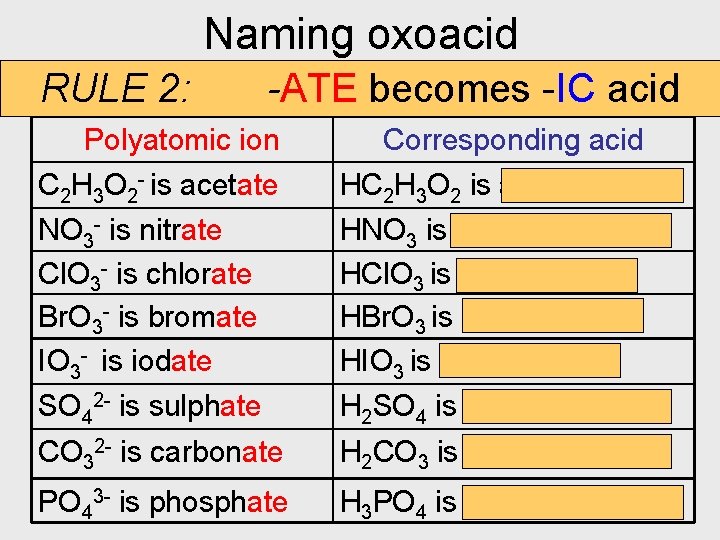
Naming oxoacid RULE 2: -ATE becomes -IC acid Polyatomic ion Corresponding acid C 2 H 3 O 2 - is acetate HC 2 H 3 O 2 is acetic acid NO 3 - is nitrate Cl. O 3 - is chlorate Br. O 3 - is bromate IO 3 - is iodate HNO 3 is nitric acid HCl. O 3 is chloric acid HBr. O 3 is bromic acid HIO 3 is iodic acid SO 42 - is sulphate H 2 SO 4 is sulphuric acid CO 32 - is carbonate H 2 CO 3 is carbonic acid PO 43 - is phosphate H 3 PO 4 is phosphoric acid

E. Common Acid Properties • E 1. Sour • E 2. Corrosive • E 3. Water-soluble • E 4. Good electricity conductors

F. Acids in everyday life F 1. Phosphoric acid (H 3 PO 4) F 2. Citric acid Also, ascorbic acid (Vitamin C)

F. Acids in everyday life F 4. Cheese (lactic acid) F 3. Aspirin (acetylsalicylic acid) F 5. Sour milk (lactic acid)

F 6. Sorbic Acid: a preservative
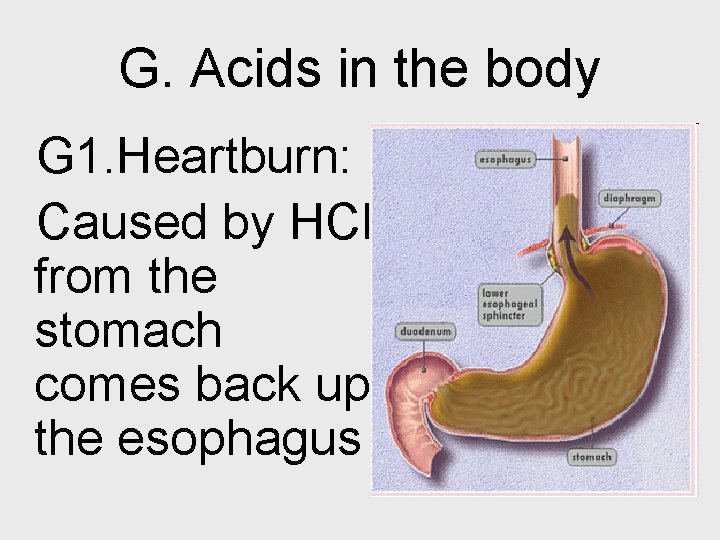
G. Acids in the body G 1. Heartburn: Caused by HCl from the stomach comes back up the esophagus
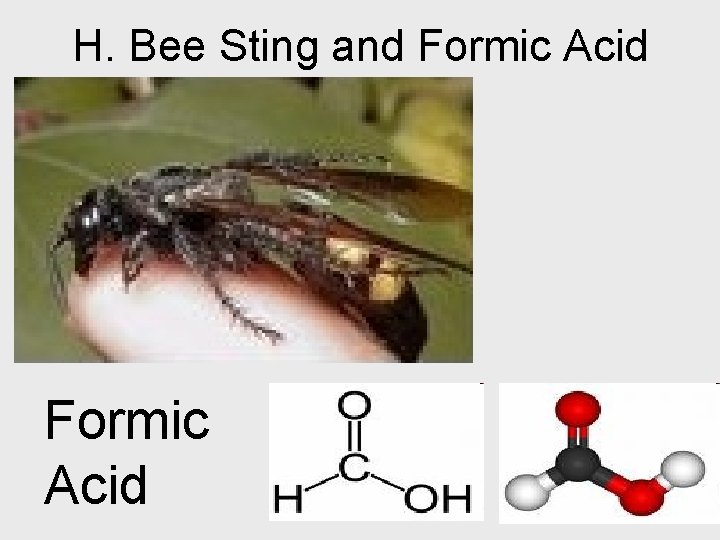
H. Bee Sting and Formic Acid

BASES

I. Common Bases Name Sodium hydroxide Potassium hydroxide Aluminum hydroxide Ammonium hydroxide Sodium bicarbonate Formula Na. OH KOH Al(OH)3 NH 4 OH Na. HCO 3

J. What is a base? J 1. A base is a compound which releases hydroxide ions (OH ) when added to water.
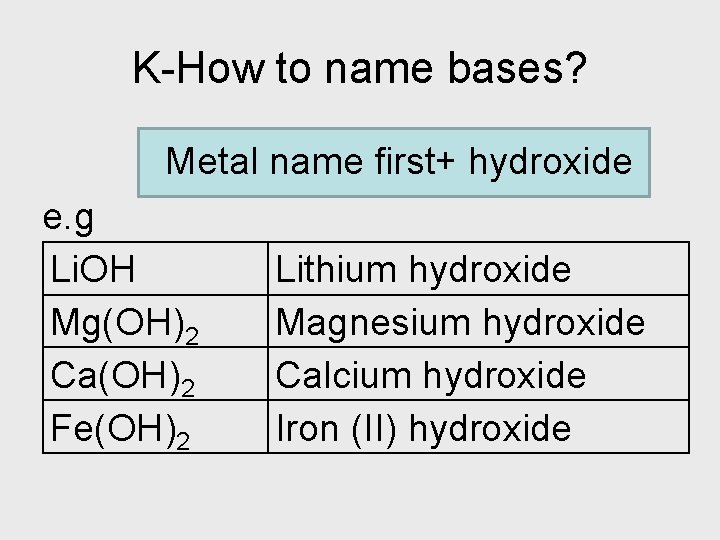
K-How to name bases? Metal name first+ hydroxide e. g Li. OH Mg(OH)2 Ca(OH)2 Fe(OH)2 Lithium hydroxide Magnesium hydroxide Calcium hydroxide Iron (II) hydroxide

L. Common Acid Properties Bases: • L 1. are bitter • L 2. feel slippery • L 3. are water-soluble • L 4. are good electricity conductors

M. Bases in everyday life M 1. Drain cleaners contains Na. OH M 2. Windex (NH 3) M 3. Baking soda (Na. HCO 3)

N. What would you do if? N 1. Your car battery acid (H 2 SO 4) is spilled?

SALTS

O. What is a salt? O 1. Salts are ionic compounds composed of a metal ion and a nonmetal ion O 2. A salt is formed when acid react with base E. g. Cu. SO 4

P. Applications of Salts P 1. Soaps and Detergents

P. Applications of Salts (cont. ) P 2. Fertilizer (e. g. Ammonium sulfate, magnesium sulfate)

Q. Salt and our Canadian’s weather Q 1. Why put salt on the sidewalk when it snows?

Q. Salt and our Canadian’s weather Q 1. Road salt helps reduce the freezing point of snow

R. A Cultural Look at Food Preservation Table salts

• What are the most 2 interesting things you find out about ACIDS today? • What are the most 2 interesting things you find out about BASES today? • What are the most 2 interesting things you find out about SALTS today?
- Slides: 32